Predictive Values of Different ECG Algorithms in Differentiating Left Ventricular Outflow Tract from Right Ventricular Outflow Tract Premature Ventricular Beats Origin
Mostafa Abdelmonaem and Amira Nour*
Department of Cardiology, Ain Shams University Hospital, Egypt
Submission: November 08, 2019; Published: December 17, 2019
*Corresponding author: Amira Nour, Congenital and Structural Heart Disease Unit, Department of Cardiology, Ain Shams University Hospital, Abbassya, Cairo, Egypt
How to cite this article:Mostafa Abdelmonaem, Amira Nour. Predictive Values of Different ECG Algorithms in Differentiating Left Ventricular Outflow Tract from Right Ventricular Outflow Tract Premature Ventricular Beats Origin. J Cardiol & Cardiovasc Ther. 2019; 15(3): 555915. DOI: 10.19080/JOCCT.2019.15.555915
Abstract
Background:Premature ventricular beats (PVBs) are early myocardial electrical oscillations, which are not life threatening in patients with normal cardiac architecture, however causing annoying morbidity; radiofrequency catheter ablation became a curative tool for elimination of this arrhythmia.
Methods: This study was held on 80 symptomatic patients with PVBs of same morphology provided being of significant burden, these patients were eligible for PVB electrophysiological study and catheter ablation in EP unit, Ain Shams University. Detailed history, comprehensive general and local examination was fulfilled. Trans-thoracic echocardiographic study and Holter ECG monitoring was offered for the studied population. Different algorithms for PVB origin identification were utilized in all patients before electrophysiological localization and radiofrequency ablation was done.
Results: mean age of studied population was 39.9±12.97 years, encompassing 44 (59.4%) males, admitted to our centre for EP study and ablation. PVB origin was unleashed initially via various algorhythms, which matched precise intra-cardiac origin in 78 cases; patients with PVBs originating from LVOT showed earlier transition, narrowed QRS complex, higher V2 transition ratio and R wave duration index as compared to those with PVBs originating from RT side. With multivariate logistic regression, the only significant independent predictor for RVOT origin was R wave duration index utilizing a cut off value of ˂ 0.3 (P=0.0057).
Conclusion:Accurate localization of PVB origin is feasible using surface ECG with positive impact on acute procedural success as well as reducing procedural time.
Keywords:PVCS; LVOT; RVOT
Introduction
Premature ventricular ectopic beats (PVBs) are early ventricular depolarizations. Usually carrying benign course in patients with structurally free heart. However, with frequent PVBs many patients develop debilitating symptoms with impaired note of life carrying a risk of cardiomyopathy, syncope and sudden death [1,2].
Routine 12 lead surface ECG identified ventricular ectopics in 1% of apparently healthy population, and this percentage jumped to 40–75% after using extended ECG monitoring [3].
Ventricular outflow tracts are the most common site of origin in normal heart. They could manifest as isolated, clustered ectopics, or as sustained tachycardia. The main pathophysiological cause is triggered activity; nevertheless, scar related re-entry and abnormal automaticity were also accused. Medical therapy encompassing Beta blocker activity or calcium channel blockers usually show limited effectiveness in relieving patients’ complaints. Catheter ablation can be curative, unless limited by tough and unpredictable induction [4,5].
We investigated and compared different algorithms to reach a precise localization of PVB origin using surface ECG which is then emphasized using intra-cardiac mapping.
Methods
Study population
Eighty patients with very frequent (>10% PVBs burden documented on holter monitoring [6]) monomorphic PVBs, regardless the presence of structural heart disease, who were persistently symptomatic despite anti-arrhythmic medications encompassing beta blockers, were studied. Patients with atrial Tachy-arrhythmias, thyroid malfunction, hypertrophic cardiomyopathy, non-revascularized myocardial ischemia, and cardio-myopathic patients with worse NYHA class, were not included.
Methodology
Detailed history, precise examination, laboratory samples (electrolytes, thyroid profile) to rule out reversible causes, was done.
Trans-thoracic echocardiogram was done at 1st patient appointment to unleash the presence of structural heart disease, and infrequently post-procedural to exclude short term complications.
48 hours Holter monitoring was done to determine the absolute PVBs number, burden and to exclude other forms of arrhythmias.
12 lead ECG
PVBs site of origin was identified using various formulas as PVC transition in chest leads, lead V2 transition ratio (the percentage R-wave in VT: (R/R+S) VT divided by the percentage R-wave in sinus rhythm (SR): (R/R+S)SR), and R wave duration index (dividing the QRS complex duration by the longer R wave duration in lead V1 or V2). PVB duration, coupling interval and inferior leads axis [7-9].
Electro-physiologic study (EP) study and radiofrequency (RF) ablation
Patients received local anesthesia after holding medications for at least 6 half-lives. Anti-coagulation was achieved by parentral unfractionated heparin (bolus of 75 U/kg then 1000 U/ hour) during the procedure.
Electro-anatomical mapping was achieved using the CARTO 3 system (Biosense, Diamond Bar, CA, USA) or the EnsiteNavX® system (St Jude Medical, Inc, St Paul, MN) referenced to doctor wish and availability. Ablation catheters, (Thermocouple 4mm tip 7F for EnsiteNavX system and Thermocool 3.5mm 8F for CARTO 3 system) were utilized. plus a multi-electrode (quadripolar or decapolar) catheter positioned in the RV (apex or RVOT) for pacing and as a reference catheter if required.
Mapping techniques
Targeted activation map was created for the chamber of interest; ventricular navigation was achieved using catheter dragging technique over the endocardium. The earliest ventricular activation (red isochrones in CARTO 3 or white isochrones in NavX) with a local electrogram racing QRS onset by at least 30 msec was ablated. Voltage maps were created in patients with suspected scar tissue (areas with local voltage <0.5 mv).
In cases of PVB paucity, pharmacological induction with adrenaline (0.1mcg/kg/min) was used. If PVBs still very infrequent, pace mapping protocol was done at suspected sites within the ventricle of interest. Ablation was commenced at sites with excellent score of 12/12 when compared to surface ECG recording of clinical PVBs.
A written and informed consent was taken from all patients and the study was guaranteed by the Research and Ethics Committee of the cardiology department, Ain Shams University
Statistics
Data were analyzed using SPSS version 21 for Windows and graphics by MS Excel. Categorical data were expressed as frequencies and percentages, while continuous data were expressed as mean ± SD or median. Comparison between categorical variables was done using Chi square or Fisher’s exact test as appropriate. Comparison between continuous variables was done using t-test or Mann-Whitney test according to normality of distribution. Multivariate stepwise logistic regression analysis was used to identify predictors of PVB origin. Receiver operating characteristics (ROC) curve analysis was done to find the impact of different ECG parameters on PVB localization. Cutoff values were selected if area under the curve (AUC) was significantly different from 0.5. P value was considered significant if <0.05.
Results
80 cases with a mean age of 39.9±12.97 years, with male predominance, and suffering from frequent monomorphic PVBs, who were referred to the EP clinic at Ain Shams University hospitals, were included in the current study.
PVB origin in relation to different ECG algorithms
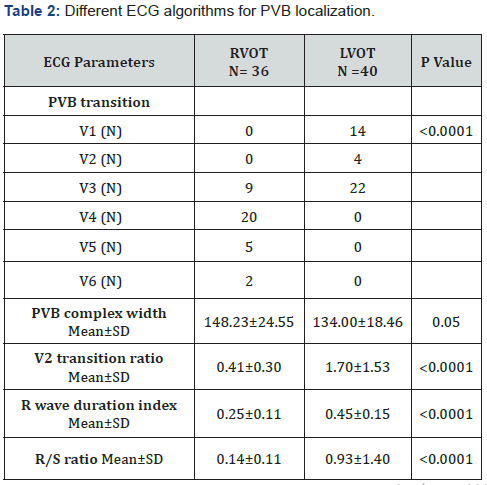
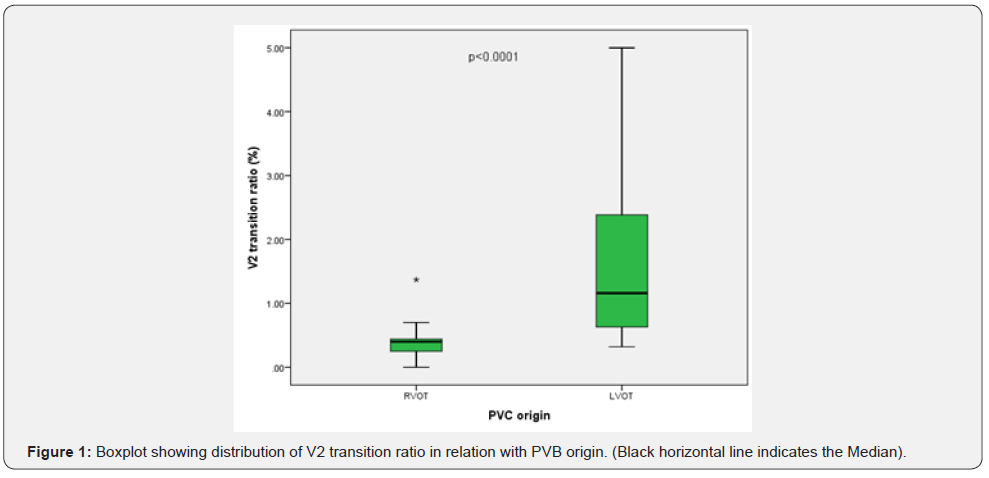
Initial localization of ectopic beats was done utilizing various algorithms. Based on surface ECG identification of origin matched EP localization in 78 cases, apart from 2 patients in the LVOT group who had initially inaccurate localization. Four cases had non outflow origin of PVBs evidenced by superior axis in limb leads (Table 1). Moreover, group of patients with LV origin had earlier transition, shorter complex width, higher V2 transition ratio and R wave duration index compared to RVOT group (Table 2).
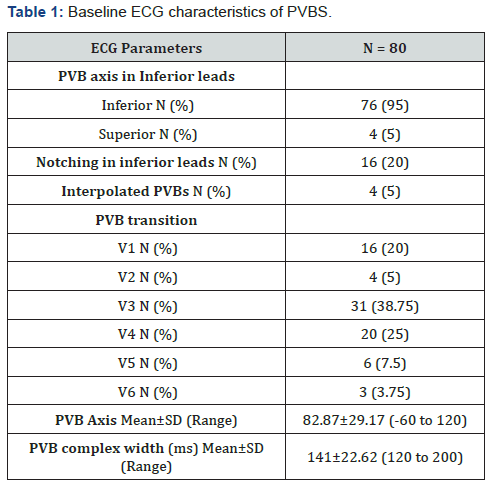
A statistically powerful bond was noticed between PVB transition and origin, (P value = 0.0001). From fourty patients in the LV group, fourteen patients had PVB transition at V1, four at V2 and twenty tow patients at lead V3.
In contrary, among patients in the RV group (n=36), nine had transition in V3, twenty at V4, five lead V5 and tow patient at lead V6.
PVB complex width was related to PVB origin, in which mean width for RVOT PVBs was 148.23ms versus 134 ms in LVOT PVBs with P value of 0.05, as shown in (Table 2), V2 transition ratio was calculated for outflow tract PVBs and was statistically significant with PVB origin, the mean ratio for RVOT origin was 0.41 while for LVOT origin it was 1.7, with P value = 0.004 (Table 2 & Figure 1).
R wave duration index had statistically significant relation with PVB origin, the mean index for RVOT origin was 0.25, however in LVOT and supra-aortic group was 0.45, with P value of 0.0001, as shown in (Table 2 & Figure 2).
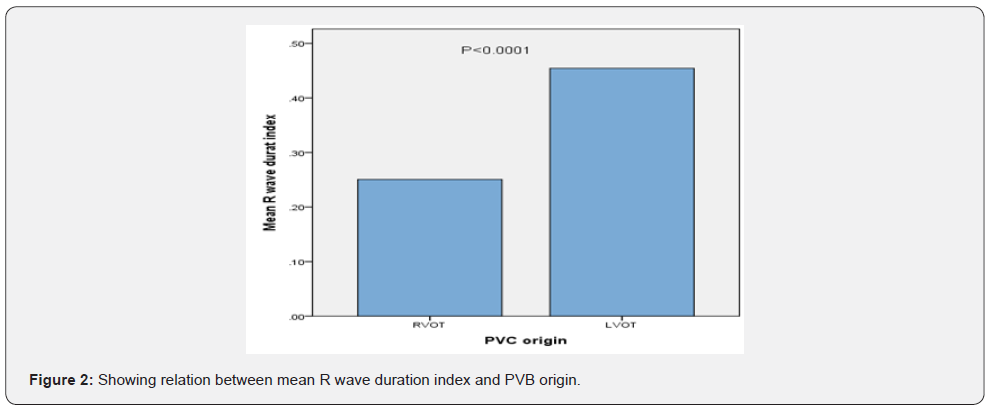
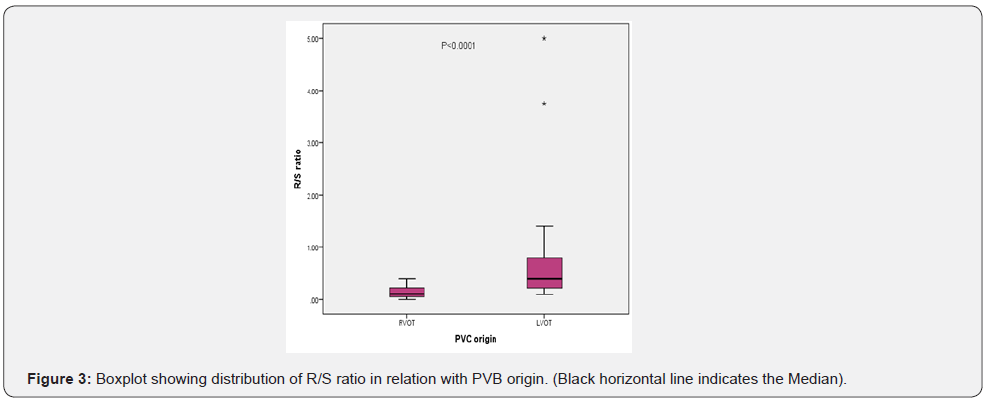
R/S ratio had statistically significant relation with PVB origin with mean ratio of 0.14 for RVOT PVBs and 0.93 for LVOT PVBs, with P value of 0.0001, as shown in (Table 2 & Figure 3).
Uni-variate analysis was done to verify the predictive value of different maneuvers, V2 transition ratio ≤0.58 predicted RVOT PVB origin reaching sensitivity of 88.24% and specificity of 87.50%, P value of ˂ 0.0001, R/S ratio ˂0.28 denoted RVOT ectopics origin with sensitivity of 64.71% and specificity of 94.12%, P value of ˂0.0001.
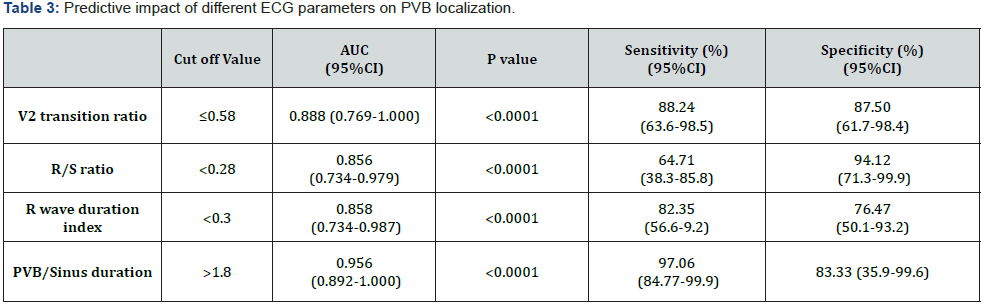
R wave duration index ˂0.3 predicted RV focus with sensitivity of 82.35% and specificity of 76.47%, P value of ˂0.0001. Identifying free wall from septal origin was done through calculating the ratio of PVB duration to its preceding sinus beat, ratio ˃1.8 supported free wall origin with sensitivity of 97.06% and specificity of 83.33%, P value of ˂0.0001 (Table 3 & Figure 4).

Multivariate logistic regression was done to predict RVOT focus using surface ECG through various algorithms, encountering PVB transition, R wave duration index, and R/S ratio, revealed that R wave duration index was the sole significant independent predictor of RVOT ectopics, with cut off value of ˂ 3 (P=0.0057).
Discussion
The most frequent non sustained arrhythmia seen by cardiologists is isolated ventricular ectopics. A big proportion of patients with frequent ectopic beats is suffering and often requires long-term anti-arrhythmic therapy. Unfortunately, pharmacological tools are rarely useful, and may be hazardous, to the extent of increasing mortality, mostly owing to their proarrhythmic nature. There is a high variability in the incidence of PVBs among different observational studies, ranging from 4.6% in healthy participants and up to 62% in patients with structural heart disease. Outflow tracts are the commonest origin, either beneath or just above the semi-lunar valves. Prognostic value of ectopics differs according to patient’s age, presence of structural heart disease and co-morbidities. Radio-frequency ablation is an accepted therapeutic tool to alleviate patient’s morbidity despite receiving pharmacological therapy including beta-blockers or asymptomatic patients with either high burden of ectopics or PVB induced cardiomyopathy. [10-13].
This study encountered 80 patients with monomorphic ventricular ectopics refractory to medical therapy including betablockers, calcium channel blockers, and class III anti-arrhythmics. The studied cohort went through initial noninvasive work-up including 12 lead surface ECG, trans-thoracic echocardiogram, lab profile and scarce use of CMR in patients with heart muscle disease, and then they underwent EP study with RF catheter ablation after precise ECG and electrophysiological localization. Pre-procedural identification of origin was fulfilled using surface ECG. PVB transition in chest leads was very helpful, as V1, V2 transition was associated absolutely with LV ectopics, the same for V4-V6 transition which predicted RV origin, thirty one patients transition at lead V3, 22 of them were mapped in LVOT and aortic sinuses. Inferior axis denoted exclusively outflow tract ectopics (n= 76), while superior axis was encountered in the remaining four cases with non outflow origin.
RR’ notching in lead AVL which was noticed in a single case, denoted Para-hissian origin, matching what stated by Zhang et al. [9] that RSR’ or RR’ pattern in lead AVL predicts ectopics originating from the His bundle region with 94.87% and 100% positive predictive value respectively [9].
One of the different algorithms for ectopic localization is the V2 transition ratio. In the current study, the mean transition ratio for RVOT ectopics was 0.41 and 1.7 for LVOT PVBs with cut off value of ≤0.58 to predict RVOT PVB origin reaching a sensitivity of 88.24% and specificity of 87.50%. In the study conducted by Betensky et al. [7] on 40 patients with outflow tract PVBs, the V2 transition ratio was higher for LVOT PVBs (1.27 ± 0.60 vs. 0.23 ±0.16) and a V2 transition ratio ≥0.60 was used as a cut of value of LVOT origin with 95% sensitivity and 100% specificity [7].
R wave duration index ˂0.3 supported RVOT origin with sensitivity of 82.35% and specificity of 76.47%, and R/S ratio with cut off value of ˂0.28 predicted RVOT PVB origin with sensitivity of 64.71% and specificity of 94.12%. which is supported by Zhang et al. [9] in his study on 39 patients with outflow tract ectopics, R wave duration index of less than 0.5 and the R/S wave amplitude index of less than 0.3 predicted RVOT PVBs with 94.87 sensitivity and 100% positive predictive value [9].
With the aid of multivariate stepwise logistic regression, we concluded that R wave duration index < 0.3 was the only significant independent predictor of RVOT origin (P=0.0057).
Differentiating septal from free wall origin of PVB was predicted using the ratio of PVB duration to preceding sinus beat duration, ratio ˃1.8 predicted free wall origin of PVB with sensitivity of 97.06% and specificity of 83.33%, which is comparable to Zhang et al. [9] finding, that a PVB-QRS width/ the preceding sinus beat width ≥1.9 denoted PVBs originating from the free wall with 94.44% sensitivity and 77.27% positive predictive value [9].
Conclusion
Radiofrequency ablation is an effective and practical tool to eradicate outflow tract ventricular ectopics regardless their origin and the presence or absence of structural heart disease. Accurate localization of PVB origin is feasible using surface ECG with positive impact on acute procedural success as well as reducing procedural time.
Conflict of Interest
Small study size and lack of facilities for epicardial mapping were the main limitations of our study.
References
- Sheldon SH, Gard JJ, Asirvatham SJ (2010) Premature Ventricular Contractions and Non-sustained Ventricular Tachycardia: Association with Sudden Cardiac Death, Risk Stratification, and Management Strategies. Indian pacing of electrophysiology J 10(8): 357-371.
- Niwano S, Wakisaka Y, Niwano H, Fukaya H, Kurokawa S, et al. (2009) Prognostic significance of frequent premature ventricular contractions originating from the ventricular outflow tract in patients with normal left ventricular function. Heart 95(15): 1230-1237.
- Kostis JB (1992) The prognostic significance of ventricular ectopic activity. Am J Cardiol 70(7): 807-808.
- Friedman PA, Asirvatham SJ, Grice S, Glikson M, Munger TM, et al. (2002) Noncontact mapping to guide ablation of right ventricular outflow tract tachycardia. J Am CollCardiol 39(11): 1808-1812.
- Fung JW, Chan HC, Chan JY, Chan WW, Kum LC, et al. (2003) Ablation of nonsustained or hemodynamically unstable ventricular arrhythmia originating from the right ventricular outflow tract guided by noncontact mapping. Pacing Clin Electrophysiol 26(8):1699-1705.
- Ban JE, Park HC, Nagamoto Y, Park JS, Nagamoto Y, Choi JI, et al. (2013) Electrocardiographic and electrophysiological characteristics of premature ventricular complexes associated with left ventricular dysfunction in patients without structural heart disease. Europace 15(5): 735-741.
- Betensky G, Robert EP, Marchlinski FE, Hutchinson MD, Garcia FC, et al. (2011) The V2 Transition Ratio A New Electrocardiographic Criterion for Distinguishing Left from Right Ventricular Outflow Tract Tachycardia Origin. JACC 57(22): 2255-2262.
- Ouyang F, Fotuhi P, Ho SY, Hebe J, Volkmer M, et al. (2002) Repetitive monomorphic ventricular tachycardia originating from the aortic sinus cusp: electrocardiographic characterization for guiding catheter ablation. J Am Coll Cardiol 39(3): 500-508.
- Zhang F, Chen M, Yang B, Ju W, Chen H, et al. (2009) Electrocardiographic algorithm to identify the optimal target ablation site for idiopathic right ventricular outflow tract ventricular premature contraction. Europace 11(9): 1214-1220.
- Ge B, Ji KT, Ye HG, Li J, Li YC, et al. (2012) Electrocardiogram features of premature ventricular contractions/ventricular tachycardia originating from the left ventricular outflow tract and the treatment outcome of radiofrequency catheter ablation. BMC Cardiovasc Disord 12: 112.
- Takemoto M, Yoshimura H, Ohba Y, Matsumoto Y, Yamamoto U, et al. (2005) Radiofrequency catheter ablation of premature ventricular complexes from right ventricular outflow tract improves left ventricular dilation and clinical status in patients without structural heart disease. J Am Coll Cardiol 45(8): 1259-1265.
- Hinkle LE Jr, Carver ST, Stevens M (1969) The frequency of asymptomatic disturbances of cardiac rhythm and conduction in middle-aged men. Am J Cardiol 24(5): 629-650.
- Rillig A, Lin T, Ouyang F, Kuck KH, Tilz RR, et al. (2015) Which Is the appropriate arrhythmia burden to offer RF ablation For RVOT tachycardia. JAFIB 7(4): 41-49.






























