Endomyocardial Fibrosis Secondary to Filariasis - A Vanishing Syndrome
Tarun Kumar1 and Satyam Rajvanshi2*
1PGIMER & Dr. R.M.L. Hospital, India
2Rajvansh Hospital & Institute of Medical Sciences, India
Submission: August 21, 2017; Published: August 30, 2017
*Corresponding author: Satyam Rajvanshi, DM (Cardiology), Rajvansh Hospital & Institute of Medical Sciences, Muzaffarnagar -251001, UP, India, Tel: +917042828380; Email: drrajvanshi.cardiology@gmail.com
How to cite this article: Tarun Kumar, Satyam R. Endomyocardial Fibrosis Secondary to Filariasis - A Vanishing Syndromet. J Cardiol & Cardiovasc Ther 2017; 7(5): 555723. DOI: 10.19080/JOCCT.2017.07.555723
Abstract
A 70-year-old gentleman presented with 8 months history of low grade fever and dyspnoea on exertion which worsened gradually. It was not associated with history of cough, weight loss and pedal edema. There was no prior significant chronic illness including bronchial asthma. Laboratory work-up revealed raised total leukocyte count with hypereosinophilia. Chest radiograph, lower limb venous Doppler, and computed tomography pulmonary angiogram were normal. Genetic testing for chronic eosinophilic leukemia was negative. However, IgG and IgM antibodies to filaria were raised. Echocardiogram showed right ventricular apical obliteration due to thrombus. Patient was started on Diethylcarbamazine and oral anticoagulants. He responded very favourably to treatment with improvement in symptoms, normalization of eosinophilia, and complete resolution of right ventricular apical thrombus over a period of 2 months.
Keywords: Endomyocardial; Fibrosis; Filaria; Tropical; Syndrome
Introduction
Helminth-induced eosinophilia has been linked to tropical endomyocardial fibrosis. Filaria and schistosomiasis are most frequently found to induce chronic eosinophilia. Rarely, it may lead to endomyocardial fibrosis (EMF) [1]. EMF is thought to be vanishing disease in India with occasional reported cases mainly from Kerala in India [2]. Our patient was a native resident of Uttarakhand in India which is 28°31' degree north of equator. This is a rare presentation from outside the usual geographic spectrum of filariasis and EMF. Recognition of this form of presentation of filariasis in non-endemic areas is crucial for timely management of this treatable illness.
Case Report
A 70-year-old male patient, resident of Uttarakhand, India, was referred to our hospital with a diagnosis of right ventricular apical clot and suspected pulmonary thromboembolism. Patient had a history of low grade fever and gradually progressive dyspnoea on exertion for last 8 months. No history of cough, weight loss, loss of appetite, abdominal distension, or pedal edema. There was no history of bronchial asthma, known allergy, or recent travel. His general and systemic examination was within normal limit.
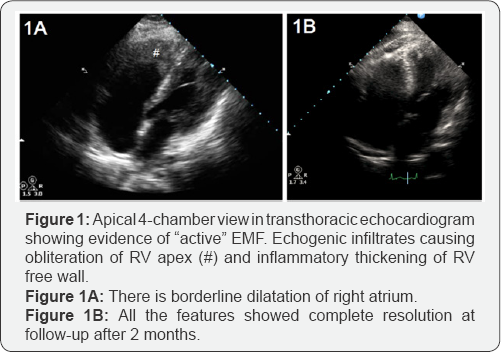
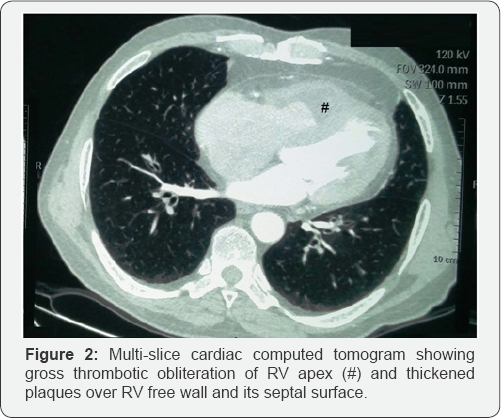
Hematological work-up revealed marked eosinophilic leucocytosis, with total leucocyte count of 11,400/cumm, 43% eosinophils, and absolute eosinophil count of 4902/cumm. Chest radiograph was normal with no pulmonary infiltrates. Electrocardiogram was suggestive of sinus tachycardia with incomplete right bundle branch block. Transthoracic echocardiogram revealed right ventricular (RV) dysfunction with obliteration of RV apex possibly with large thrombus (3x3cm) and mild tricuspid regurgitation with normal pulmonary artery pressure (Figure 1A). Left ventricle showed normal chamber dimensions with normal left ventricular systolic function and grade II diastolic dysfunction. Cardiac Computed tomography (CT) showed similar RV apical obliteration with mild RV dilation (Figure 1B). CT pulmonary angiogram was normal. Full array of differential diagnosis for etiology of eosinophila was investigated. Stool examination for parasite was negative. Polymerase chain reaction (PCR) of FIP1L1-PDGFRA gene rearrangement (For Chronic eosinophilic leukemia) was negative. Patient was not from endemic areas for filarial infection but surprisingly, IgG and IgM antibody assay for filaria was strongly positive. On the basis of investigations, a diagnosis of endomyocardial fibrosis (stage 2), secondary to hypereosinophilia related to filarial infection was made. Patient was started on diethylcarbamazine (100mg thrice a day for 14 days) and oral vitamin K antagonists for anticoagulation (INR range 2-3). He showed dramatic response to treatment, and his symptoms improved rapidly. At follow-up after 2 months, patient was asymptomatic; leucocyte count (10500/cumm), eosinophil differential count (2%), and absolute eosinophil count (208/cumm), were normalized; and apical RV thrombus and tricuspid regurgitation were completely resolved (Figure 2). Anticoagulation was stopped, and normal functional state and echocardiogram was found at repeat visit after 6 months.
Discussion
Eosinophilic cardiac disease is a relatively rare condition that was first described in 1936 by Wilhelm Loffler, who called it 'fibroplastic parietal endocarditis with blood eosinophilia' [3]. In fact, several types of cardiac damage may be encountered in the context of eosinophilia, from acute myocarditis to EMF. All result from toxicity of infiltrating eosinophils into cardiac tissue.
According to Gottdiener et al. [4] three phases are classically described. The first stage is due to eosinophilic infiltration into the tissues. The second stage is represented by thrombus formation. The third stage corresponds to fibrotic scarring. The pathologist, Jack NP Davies [5] identified EMF in Uganda in 1948. Since then, reports of EMF have come from other parts of tropical Africa, South Asia, and South America, regions that are within 15 degrees of the equator. Several hypotheses exist regarding etiopathogenesis of EMF indicating the role of filarial infections, helminths, eosinophilia, and malnutrition.
Initially, EMF was identified by pathologist, Jack NP Davies [5], from Uganda, in 1948. Subsequently, EMF cases were reported from regions that are within 15 degrees of the equator -tropical parts of Africa, South America, and South Asia. Aetiopathogenesis of EMF includes several hypothesis, indicating significant role of eosinophilia, malnutrition, filarial and helminthic infections.
EMF may affect either of the ventricles, but right ventricle is predominantly affected [6]. It usually involves subvalvular region, inflow tract, and apex, of one or both ventricles. It may involve papillary muscle of atrioventricular valves, tethering of which leads to mitral and tricuspid regurgitation [7]. Medical management of EMF consists of ameliorating acute diseases as well as preventing and treating heart failure, arrhythmias and thromboembolism. However medical treatment of EMF is often difficult and not particularly effective. When EMF has reached the fibrotic stage, surgery offers symptomatic improvement and is treatment of choice [8].
Contrary to uncertain or varied aetiology associated with EMF, we succeeded in finding the etiology for hypereosinophilia leading to EMF. In contrast to poor prognosis associated with EMF, our patient responded dramatically to anti filarial treatment, leading to resolution of symptoms, and normalization of hypereosinophilia and echocardiogram, all pointing to complete recovery.
Learning Points
a) The syndrome of tropical endomyocardial fibrosis (EMF) is uncommonly encountered in non-endemic areas. Etiopathogenesis is still poorly understood. Healthcare professionals should be aware of the variable presentation for timely diagnosis.
b) Eosinophilia requires extensive work-up to find its possible etiology.
c) Chronic form of EMF has poor long term prognosis. Evidence of prior Filarial infection maybe one of the treatable causes of eosinophilia and its dreaded sequelae.
References
- Rashwan MA, Ayman M, Ashour S, Hassanin MM, Zeina AA (1995) Endomyocardial fibrosis in Egypt: an illustrated review. Br Heart J 73(3): 284-289.
- Kutty VR, Abraham S, Kartha CC (1996) Geographical distribution of endomyocardial fibrosis in South Kerala. Int J Epidemiol 25(6): 1202-1207.
- Loffler W (1936) Endocarditis parietalis fibroplastica mit blu- teosinophilie. Ein eigenartiges Krankheitsbild. Schweiz Med Wochenschr 66: 817-820.
- Gottdiener JS, Maron BJ, Schooley RT, Harley JB, Roberts WC (1983) Two-dimensional echocardiographic assessment of the idiopathic hypereosinophilic syndrome. Anatomic basis of mitral regurgitation and peripheral embolization. Circulation 67(3): 572-578.
- Davies JPN (1948) Endomyocardial fibrosis in Uganda. East Afr Med J 25: 10-16.
- Chopra P, Narula J, Talwar KK, Kumar V, Bhatia ML (1990) Histomorphologic characteristics of endomyocardial fibrosis: an endomyocardial biopsy study. Hum Pathol 21(6): 613-616.
- Vasquez EF, Bautista CL, Navarette BA, Moreno IC, Guerrero AE, et al. (2003) Chronic thromboembolic pulmonary hypertension associated with Endomyocardial fibrosis of the right ventricle. Arch Bronchoneumol 39(8): 370-372.
- Moreas F, Lapa C, Hazin S, Tenorio E, Gomes C, et al. (1999) Surgery for endomyocardial fibrosis revisited. Eur J Cardiothoracic Surg 15(3): 309-312.






























