Transcatheter Leadless Pacemaker Implantation in a Patient with Recurrent Complications Following Transvenous Pacemaker Systems
Tuomas Paana1, Wail Nammas1, Juhani Koistinen2 and Pasi P Karjalainen1*
1Heart Center, Satakunta Central Hospital, Finland
2Department of Cardiology, Vaasa Central Hospital, Finland
Submission: April 19, 2017; Published: June 01, 2017
*Corresponding author: Pasi P Karjalainen, Heart Center, Satakunta Central Hospital, Sairaalantie 3, FIN-28500, Pori, Finland, Tel: +358 2 6277755; Fax: +358 2 6277757; Email: pasi.karjalainen@satshp.fi
How to cite this article: Tuomas P, Wail N, Juhani K, Pasi P K. Transcatheter Leadless Pacemaker Implantation in a Patient with Recurrent Complications Following Transvenous Pacemaker Systems. J Cardiol & Cardiovasc Ther 2017; 5(4): 555668 . DOI:10.19080/JOCCT.2017.05.555668
Abstract
A 79-year-old man had a DDD permanent pacemaker inserted in September 2007 for symptomatic third-degree atrioventricular block. Over the following several years, he underwent repeated battery removal and reinsertion in a different site, due to recurrent subcutaneous pocket complications (mostly pocket infection), after transvenous pacemakers implanted in pectoral and abdominal (epicardial lead) pockets. Ultimately, he presented with failure of capture from an epicardial pacing lead. A leadless transcatheter pacemaker was implanted, via femoral vein access. Post-procedural electrical measurements revealed a pacing threshold of 0.38V at 0.24msec, and R wave sensitivity 15mV; they were stable at 1-month follow-up.
Keywords: Atrioventricular block; Leadless pacemaker
Case Report
A 79-year-old man had a DDD permanent pacemaker inserted (left subclavian) in September 2007 for symptomatic third-degree atrioventricular block. In 2005, He underwent primary percutaneous coronary intervention with stenting for acute myocardial infarction, followed by subacute stent thrombosis. In October 2008, he presented with signs of infected pacemaker pocket. He was hospitalized, received clindamycin- and penicillin-based antibiotic therapy; during the same hospitalization, he developed left axillary vein thrombosis for which he received enoxaparin. Culture of fluid aspirated from the pocket was negative. Three weeks later, he presented with recurrent pocket infection; the pacemaker was removed. In January 2009, he presented with asymptomatic trifascicular block, and intermittent atrioventricular block; he had a DDD pacemaker inserted (right subclavian). In April 2009, he presented again with infected pacemaker pocket; culture from the pocket discharge revealed coagulase-negative staphylococcus sensitive to vancomycin; the pacemaker was again removed. In October 2009, he complained of fatigue, and ECG revealed Mobitz II second-degree atrioventricular block (2:1 conduction), with a ventricular rate of 40bpm; a DDD pacemaker was implanted (left subclavian). In August 2010, he presented with swollen pacemaker pocket; yet, no signs or lab evidence of infection. Culture of fluid aspirated from the pocket was negative. In October 2010, he presented with a fistula of the pacemaker pocket, oozing discharge. The pacemaker was removed, and a temporary pacing lead was inserted via the right internal jugular vein, under parenteral antibiotic cover (vancomycin, cephalosporin). During the same hospitalization, an epicardial VVI pacemaker was implanted in a left abdominal pocket; pacing threshold was 0.5V at 0.4msec. In February 2014, he presented with a solid mass around the abdominal pacemaker pocket; yet, no signs of infection. Ultrasound revealed a seroma 9.5 x 11cm in size. In May 2014, he underwent exploration of the pacemaker pocket, seroma was removed, and the pacemaker was relocated medially; pacing threshold was 0.75V at 0.4msec. In January 2016, he complained of fatigue, and ECG revealed failure of capture, third-degree atrioventricular block, with a ventricular rate of 40bpm; pacing threshold was 5.5V at 1msec. In February 2016, he had a leadless transcatheter pacemaker system (Micra™ TPS, Medtronic, MN, USA) implanted (Figure 1A-1D).
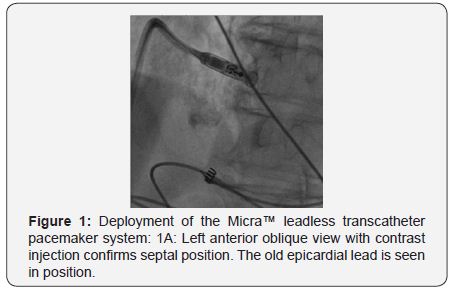
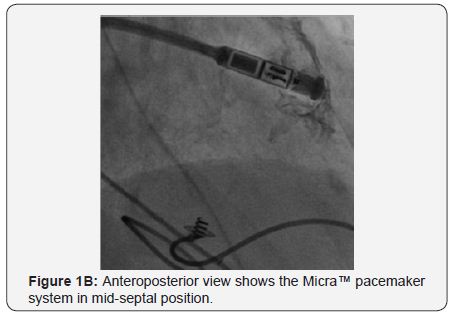
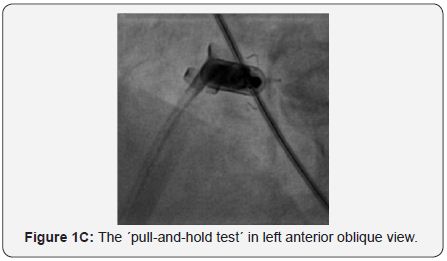

Discussion
Currently, transvenous pacemakers are the standard management for patients with an indication for permanent pacing. Most of the major complications of transvenous pacemakers are related to the transvenous lead/s or subcutaneous pocket [1]. Transcatheter leadless pacemakers demonstrated a safety profile similar to transvenous pacemakers while providing low and stable pacing thresholds [2-4]. In the current case report, the patient had recurrent pocket complications after transvenous pacemakers implanted in subcutaneous pectoral and abdominal pockets. The last battery (abdominal pocket) showed failure of capture 5 years after implantation; insertion of a new battery in another subcutaneous pocket portended a high probability of pocket complications. In such case, transcatheter leadless pacemaker systems would be a convenient solution. Implantation was successful at first attempt, with satisfactory electrical measurements both post-procedural, and at 1-month followup.
References
- Udo EO, Zuithoff NP, van Hemel NM, de Cock CC, Hendriks T, et al. (2012) Incidence and predictors of short and long-term complications in pacemaker therapy: the FOLLOWPACE study. Heart Rhythm 9(5): 728-735.
- Ritter P, Duray GZ, Steinwender C, Soejima K, Omar R, et al. (2015) Early performance of a miniaturized leadless cardiac pacemaker: the Micra Transcatheter Pacing Study. Eur Heart J 36(37): 2510-2519.
- Reynolds D, Duray GZ, Omar R, Soejima K, Neuzil P, et al. (2015) A Leadless Intracardiac Transcatheter Pacing System. N Engl J Med 374(6): 533-541.
- Bonner M, Eggen M, Haddad T, Sheldon T, Williams E (2015) Early Performance and Safety of the Micra Transcatheter Pacemaker in Pigs. Pacing Clin Electrophysiol 38(11): 1248-1259.






























