Does the Use of Local Anesthesia with Conscious Sedation Rather Than General Anesthesia Improve the Outcomes of Transfemoral Transcatheter Aortic Valve Replacement?
Imane El Hajjaji1,2*, Jean-philippe Collet3, Philippe Cluzel4, Ralouka Makri5 and Pascal Leprince1
1Department of Cardio vascular and Thoracic Surgery, Canada
2University of Western Ontario, Canada
3Department of Interventional Cardiology, Canada
4Department of Vascular Interventional Radiology, Canada
5Department of Anesthesia and Intensive Care LaPitie-Salpetriere University Hospital, France
Submission: November 20, 2016; Published: December 14, 2016
*Corresponding author: Imane El Hajjaji, Department of Cardiovascular and Thoracic Surgery, University of Western Ontario, 291 Windermere Rd. Unit332, London, Ontario, Canada.
How to cite this article: Imane E H, Jean-philippe C, Philippe C, Ralouka M, Pascal L. Does the Use of Local Anesthesia with Conscious Sedation Rather Than General Anesthesia Improve the Outcomes of Transfemoral Transcatheter Aortic Valve Replacement?. J Cardiol & Cardiovasc Ther 2016; 2(3): 555589. DOI: 10.19080/JOCCT.2016.02.555589
Mini Abstract
According to the results of this study, TF-TAVR under LACS seems to be as safe and effective as it is under GA.LACS could reduce infectious complications and median hospital stay after TF-TAVR.
Abstract
Background: Transcatheter aortic valve replacement is a proven treatment for patients with aortic stenos is who are considered at high-risk or with contraindications for surgical aortic valve replacement. General anesthesia (GA) and local anesthesia with conscious sedation (LACS) have been described for this procedure. However, there is no randomized study that looked for the benefit of using LACS for this procedure. The anesthetic management during TAVR is still controversial, and some centers are still performing it mainly under GA. This study aimed to look for benefits of LACS over GA regarding the outcomes of transfemoral transcatheter aortic valve replacement (TF-TAVR).
Methods: We analyzed data of our patients’ cohort, who underwent TF-TAVR between February 2010 and October 2011 at La Pitie-Salpetriere Hospital, those patients were part of the French Aortic National Core Valve and Edwards 2(FRANCE2). The procedure was performed either under GA or LACS. Two TAVR systems were used. Device success, safety and efficacy endpoints, prosthetic valve performance, complications and hospital stay were compared between LACS and GA groups.
Results: From 78 consecutive patients who underwent TF-TAVR, 40 received LACS and 38 received GA. Device success rate was 90.0% in the LACS group and 92.1% in the GA group (p=1.00). Thirty-day mortality rate was 14.1% overall, with no significant difference between groups (p=0.29). There were no significant differences in safety and efficacy endpoints, the prosthetic valve performance, therapy-specific and prosthetic valve associated complications between the two groups. Infectious complications rate was significantly lower (p=0.04) and the median hospital stay was significantly shorter (p=0.04) in LACS group.
Conclusion: In our experience, TF-TAVR performed under LACS seems to be as safe and effective as it is under GA. LACS could reduce infectious complications and median hospital stay after the procedure.
Keywords: Aorticstenosis; TAVR; General anesthesia; Local anesthesia; SAVR
Abbreviations: AR: Aortic Regurgitation; AVA: Aortic Valve Area; AVB: Atrioventricular Block; AVR: Aortic Valve Replacement; CABG: Coronary Artery Bypass Graft; COPD: Chronic Obstructive Pulmonary Disease; Euro SCORE: European System for Cardiac Operative Risk Evaluation; GA: General Anesthesia; HF: Heart Failure; LACS: Local Anesthesia with Conscious Sedation; LV: Left Ventricular; LVEF: Left Ventricular Ejection Fraction; MI: Myocardial Infraction; MR: Mitral Regurgitation; SAVR: Surgical Aortic Valve Replacement; TAVR: Transcatheter Aortic Valve Replacement; TF-TAVR: Transfemoral Transcatheter Aortic Valve Replacement; VARC: Valve Academic Research Consortium
Introduction
Transcatheter aortic valve replacement (TAVR) has emerged as a credible alternative therapy for patients with aortic stenosis who are considered at high-risk or with contraindications for surgical aortic valve replacement (SAVR) [1]. Centers still disagree about the preferred anesthetic technique for TF-TAVR [2]. Most teams perform the procedure under general anesthesia (GA), although local anesthesia with conscious sedation (LACS) may suffice for the transfemoral approach [1].
The subanalysis of the French Aortic National Core Valve and Edwards 2 Registry (FRANCE2) [3] compared clinical outcomes and safety of TAVR Under GA versus local anesthesia, and didn’t demonstrated a clinical benefit of the local anesthesia over the GA. However, the lack of finding a benefit of performing the procedure under local anesthesia could be due to the nationwide nature of the registry and to the fact that many outcomes were self-reported and not independently adjudicated. The purpose of this study was to report the analysis of the clinical outcomes of transfemoral transcatheter aortic valve replacement (TFTAVR) under LACS compared to GA performed in our Centre, the patients included here were also part of FRANCE 2 registry [3]. The collect of clinical endpoints was more extensive in the present study.
DisMethodscussion
Study participants
We analyzed data of our cohort of patients who underwent TF-TAVR with either the Medtronic Core valve System or the Edwards Sapien valve, who were part of the FRANCE 2 registry [4], from February 2010, to October 2011. From 102 consecutive patients with severe Aorticstenosis who underwent TAVR at La Pitie-Salpetriere University Hospital, 78 patients had TF-TAVR. All consecutive patients referred to our center for the management of symptomatic severe native aortic stenosis with high risk for open-heart surgery as expressed by a logistic European System for Cardiac Operative Risk Evaluation (Euro SCORE)≥20% or with specific surgical contraindications as described previously [1,5] and who had a TF-TAVR were included. All these patients met the eligibility criteria defined for TAVR [1]. The decision to perform the procedure was made by a multidisciplinary team involving an interventional cardiologist, a cardiothoracic surgeon, a cardiologist, cardiac anesthesiologist, an echocardiographist and an imaging specialist. Written informed consent was obtained from each patient enrolled. The study protocol was approved by the local ethics committee.
Pre-procedural evaluation
All patients had trans thoracic echocardiography or transesophageal echocardiography (TEE) when required, Doppler ultrasound of the supra-aortic arteries, coronary angiography, and multi slice computed tomography of the aortic root and aortoiliofemoral system. Pulmonary function test was performed if there was a history of respiratory disease. Assessing the feasibility of TF-TAVR was mainly based on the results of multi slice computed tomography. For all patients, aortic annulus measurement was performed by the multi slices canner. Patients were excluded if this diameter was <18or>25mm, or if they had contraindications to the transfemoral approach [6]. A preanesthetic consultation was performed at least 48 hours before the procedure.
Trans catheter aortic valve replacement procedure
All TF-TAVRs were performed in a hybrid operating room. Detailed technical aspects of the TAVR procedure have been previously described [1,7]. An interventional cardiologist and a cardiac surgeon actively took part in the procedure. Aortic annul us dimensions determined by the multi slice computer tomography guided the choice of the device. Four types of prosthesis were used (Edwards SAPIEN valve [ESV, Edwards Life sciences Inc. CA, USA] and Medtronic Core valve Revalving system [MCRSTM, Core Valve Inc. Irvine, CA, USA] were initially used, gradually replaced by subsequent generations SAPIENXT and Core valve Accutrack). The procedure was guided by fluoroscopy and angiography. Immediately after TAVR, autography, and TEE when ever available, were performed to assess the presence of aortic regurgitation, the patency of the coronary arteries and to rule out complications [1].
Anesthesia
The procedure was performed either under GA or LACS. The choice of the type of anesthesia used for every patient was left to the discretion of the physician (s) involved. During the initial phase of the study, our practice of performing TF-TAVR was to operate mainly under GA then gradually we shifted to LACS. Anesthesia personnel involved in the procedure were a cardiac anesthetists and an experienced nurse anesthetist.
Detailed technical aspects of anesthetic and per operative management of patient sunder going the TAVR procedure have been previously reported [8,9]. GA was induced with an intravenous bolus of etomidate (0.25-0.4mg/kg) or propofol (1.2-2.5mg/kg) associated to either remifentanil (1μg/Kg), alfentanil (50-100μg/Kg), sufentanil (0.5-2μg/Kg) or fentanyl (2-50μg/Kg). The choice of anesthetic drug was at the discretion of the anesthesiologist. It was facilitated by atracurium (0.3- 0.6mg/Kg). Anesthesia was maintained with either a continuous infusion of propofol or in etomidate or an inhaled agent (desflurane, sevoflurane, or isoflurane) and the opioids used for induction. For LACS, ilioinguinal/Iliohypogastric blocks and infiltration with a mixture of lidocaine and ropivacaine were performed by the cardiac anesthesiologist approximately 30 minutes before the onset of the procedure. All patients under went sedation with remifentanil (target-controlled infusion of 1-3ng/mL adjusted to obtain a Ramsay score of 2-3). If maximal dose of remifentanil was insufficient, supplementation with a target-controlled infusion of propofol was performed.
Data collection and definition of evaluation criteria
Clinical and par clinical data have been collected partially prospectively taken from our center’s registry which is part of FRANCE 2 registry, and completed retrospectively from the health records for more extensive endpoints collect. We assessed base line patient characteristics, procedural characteristics and anesthesia-related information. Post-procedural outcomes, defined according to the Valve Academic Research Consortium criteria (VARC) [10], were collected over a follow-up of 30-days and were compared between the two groups: LACS versus GA.
Statistical Analysis
All statistical analyses were performed with the use of IBMSPSS Statistics version 20 (SPSS, Chicago, IL, USA). Continuous variables were presented as means ± standard deviations when normally distributed and as medians (25th-75thpercentiles) when not normally distributed. They were tested for differences with the Student’s t-test or non-parametric tests respectively. Categorical variables, were expressed as percentages, and were compared with the use of the Pearson’s chi-square or the Fisher’s exact test, as appropriate. All statistical tests were 2 sided and a p value<0.05 was considered significant.
Results
Baseline Patient characteristics and Procedural characteristics
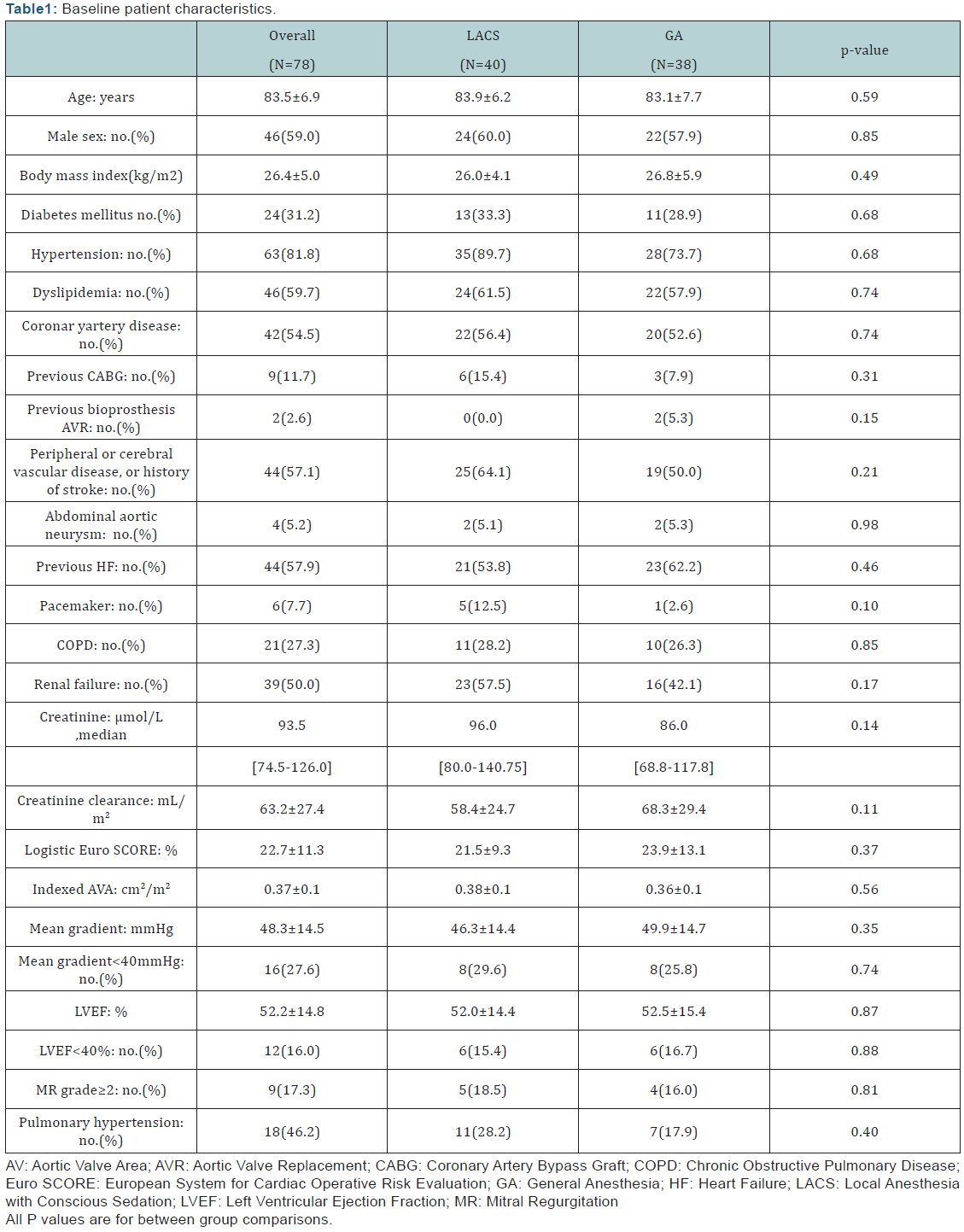

The procedural characteristics and device success rate are shown in (Table2). Overall, device success was achieved in 91.0% of patients, and was similar in the two groups (p=1.00). Three cases had device failures in GA group, they were due to a prosthetic aortic regurgitation grade ≥ 3 in 2 patients who had asymmetric aortic annulus and to an incomplete opening of the prosthesis secondary to calcareous fragments at the native valve in another patient. In LACS group, four cases didn’t meet device success criteria, two of them had prosthetic aortic regurgitation grade ≥ 3 due to prosthesis malposition, one case had left ventricular wall perforation and unfortunately had a peril-procedural death, and in one case the iliac artery was impassable and required conversion to on-pump SAVR.
Safety and efficacy endpoint sand Prosthetic valve performance
All P values are for between-group comparisons.
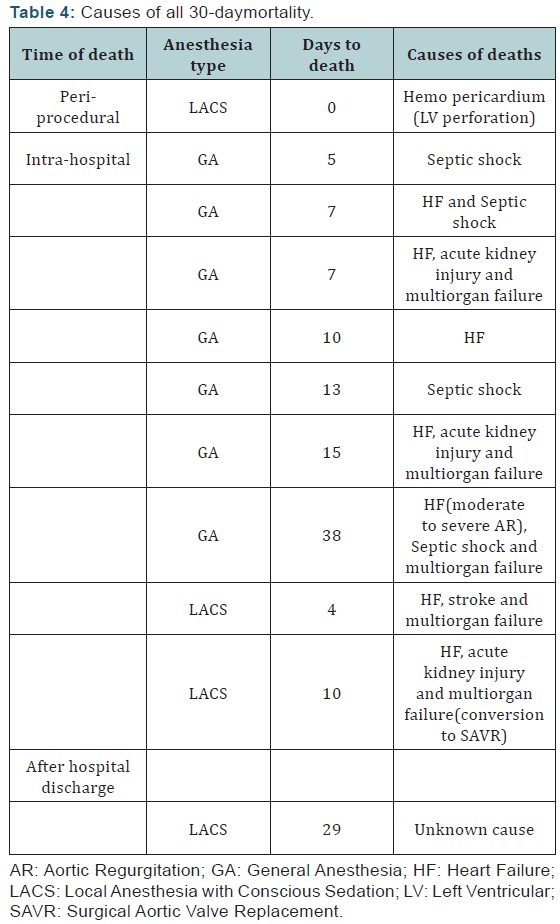
There were no significant differences in terms of efficacy and safety endpoints between the two groups (Table 3). The Valve Academic Research Consortium combined safety endpoint (all-cause mortality, major stroke, peril-procedural myocardial infarction, life-threatening bleeding, and kidney injury RIFLE stage3) was comparable for the two groups (10.0%[n=4] in LACS group vs.21.1% [n=8] in GA group, p=0.22). All-cause30- daymortality rate (including intra-hospital mortality) was 14.1% (n=11) for the whole of the series, 10.0% (n=4) in LACS group vs. 18.4% (n=7) in GA group, (p=0.29). Overall 10.3% (n=8) wereduetocardiovascularcauses.Thedistributionofthe30- daymortality according to its timing related to the procedure is illustrated in (Figure 1).
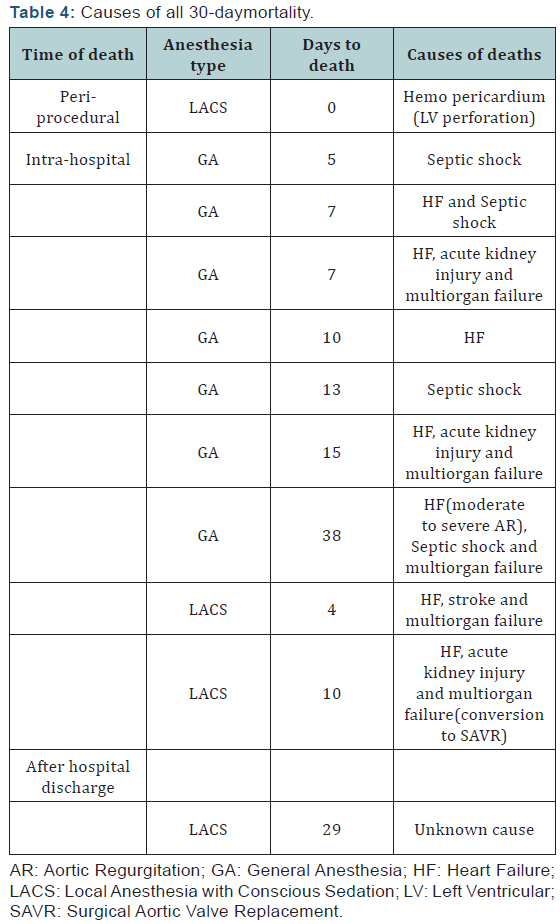
The details of all-cause 30-daymortality are shown in (Table 4). One death occurred on the day 38 post-procedure in GA group and was included in the 30-daymortality, as it occurred during the index hospitalization. A single peri-procedural death occurred in LACS group, due to a perforation of the left ventricular wall. In GA group, all 30-day deaths were intrahospital, but none was peri-procedural. One peril-procedural myocardial infarction occurred in LACS group, diagnosed after the procedure. This case had conversion to on-pump SAVR. One stroke occurred in LACS group. The prevalence of bleeding, vascular complications and acute kidney injury was the same between the 2 groups (Table 3). No deaths were directly related to a vascular access site complication.
Regarding the prosthetic valve performance, results were similar between the 2 groups (Table 3). Overall significant prosthetic aortic valve regurgitations (grade≥2), occurred in14 of 73 patients (19.2%), without significant difference between the two groups (p=0.78).
Other Complications
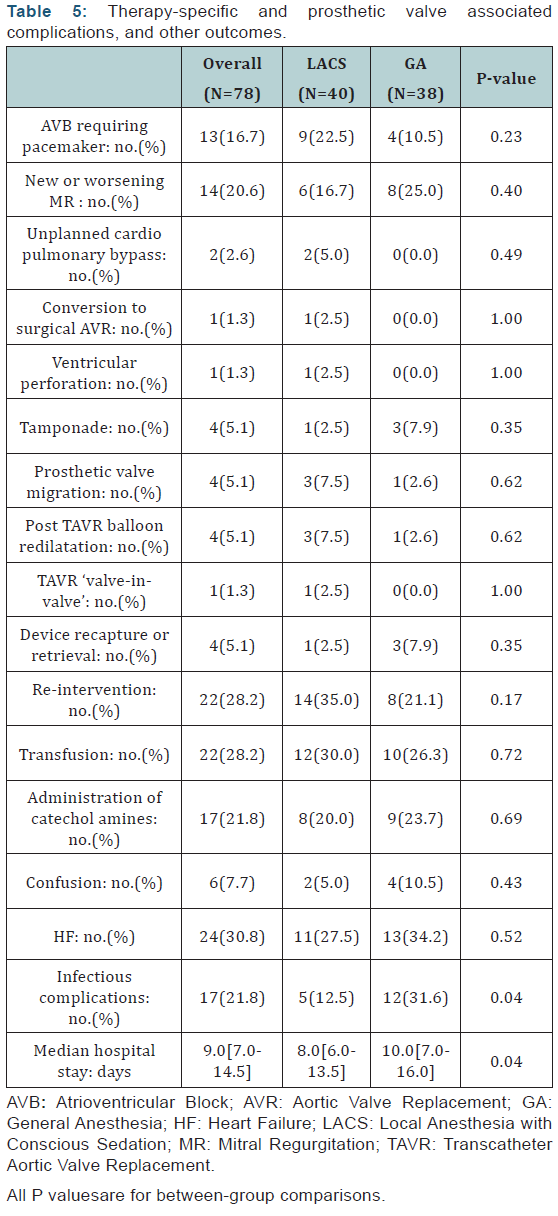
Prosthetic valve ‘associated’ complications and therapyspecific complications were similar between LACS and GA groups (Table 5). The occurrence of Atrioventricular bloc requiring a permanent pacemaker implantation didn’t differ significantly between the two groups. Conversion to an ‘open’ SAVR was performed in1case in LACS group. Requirement of any cardiac or vascular surgery after the index procedure was similar between the 2 groups, in 14(35.0%) patients in LACS group and 8 patients (21.1%) in GA group, p=0.17. Reasons for those subsequent surgeries were in LACS group: pacemaker in 9 patients, migration of the bio prosthesis in 1case, trans apical TAVR after failure of the femoral access in 1case, surgical repair of left ventricular wall ruptures in1case, insertion of extracorporeal membrane oxygenation in1case and wound infection of the scarp in1case. In GA group indications of re-interventions were: pacemaker implantation in 4cases, pseudoaneurysm in1case, surgical drainage of pericardial effusion in 2cases and wound infection of the scarp in1case.
Some anesthesia and intensive care related aspects are also presented in (Table 5). Infectious complications rate was significantly lower in patients receiving LACS compared to those receiving GA (5patients [12.5%] vs.12 patients [31.6%],p=0.04). Bronchopulmonary infections were the predominant infectious complications in GA group (8 cases of 12). In univariate analysis, we found that a history of chronic obstructive pulmonary disease was associated with the occurrence of bronchopulmonary infection (p=0.019) in GA group, where as this association was not found in the LACS group (p=1.00). The median of hospital stays was shorter in the LACS group (p=0.04).
Discussion
The choice of anesthesia type for TF-TAVR is still controversial. In FRANCE 2 registry TAVR were performed with the use of local anesthesia for 40.8% of TAVR with femoral approach [11]. In the subanalysis of the French aortic national Core valve and Edwards 2 registry that compared clinical outcomes and safety of TAVR under GA versus local anesthesia, no benefit of LACS over GA was demonstrated. But the lack of finding any benefit could be due to the nationwide nature of the registry, some bias could be due to the self-report of results individually by each participating center. The aim of this study was to analyze and compare early outcomes of F-TAVR between patients consecutively undergoing LACS versus GA groups. This study has the advantage of being well balanced in terms of the size of each group (LACS and GA groups), in contrast to other similar studies [12,13].
Our results suggest that TF-TAVR is feasible under LACS as well as it is under GA. The 2 groups were equivalent in terms of device success rate and prosthetic valve performance, without any increase in prosthetic valve associated complications or therapy-specific complications in LACS group at 30-day follow up (Tables 2,3,5). The distribution of all-cause 30-daymortality depend in go the time of death (Figure1) did not differ significantly between the 2 groups (p=0.10).
The previously reported studies considered that the TEE was useful for the correct positioning of the prosthesis [12]. In our study, in principle the patients in the LACS group didn’t have TEE and it was used in all the patients in the GA group. Thus, some of the expected disadvantages of the LACS were related to the lack of the use of TEE and its potential consequences on the accuracy of the bio prosthesis positioning. But nevertheless prosthetic valve performance in LACS group was not less good than in the GA group (Table3).
TAVR under LACS seems to be as safe and effective as TAVR under GA. GA certainly allows more comfortable working for the interventional team compared to the LACS by facilitating the procedure as the patient is completely relaxed and motionless. In our experience the use of LACS wasn’t associated to any increase of vascular complications or peril-procedural bleeding complications (Table 3).
Were ported also in this study severe surgical complications of TF-TAVR, and their rate seemed to below.
Conclusion
In the present study, the 2groups were equivalent in terms of the use of catecholamine’s (Table 5); unlike others studies where the use of catecholamine’s was higher in the GA group compared to the LACS group [12]. Our result could be explained by our method of induction of GA, which was slowly and carefully titrated to achieve the targeted effect. Post-procedural confusion was not less frequent as expected in LACS patients (Table 5). This similarity between the two groups might be due to the use of sedative agents and opioids even at low doses in LACS group.
Some authors believe that LACS has the advantage of simplified monitoring that shortens anesthesia preparation and could avoid nosocomial infections [12]. In this analysis, infectious complications rate was significantly lower in LACS group. Finally, LACS was associated with a shorter median hospital stay compared with GA (Table 5). This is comparable with reported data [12,14].Study limitations
This study reflects single-center experience, with limited number of patients. The study was observation a land nonrandomized, so we can’t conclude that the differences observed between TF-TAVR under LACS versus GA were due only to the type of anesthesia used. GA was used mainly during the initial phase of the study and was shifted progressively to LACS, so we can’t exclude that an increasing experience with time bias could have an effect on the results.
This study showed that TF-TAVR is feasible under LACS as well as it is under GA, with similar device success rate and prosthetic valve performance between the two groups. TAVR under LACS seems to be as safe and effective as the TAVR under GA. Thirty-day mortality didn’t differ between the groups. TAVR under LACS seems to be associated with a lower rate of in factious complications and a shorter median hospital stay compared to GA group. The LACS was considered by the heart center’s team as the preferred type of anesthesia for patients undergoing TF-TAVR.
Conflicts of Interest Statement
Dr. Pascal Leprince is a proctor for Medtronic Core Valve and a consultant for Edwards Life sciences.
Acknowledgement
We are grateful to Sadia Mohammedi, Clinical Research Associate at LaPitie-Salpetriere university hospital, for her input during end points collect.
References
- Vahanian A, Alfieri O, Al-Attar N, Antunes M, Bax J, et al. (2008) Transcatheter valve implantation for patients with aortic stenosis: a position statement from the European Association of Cardio-Thoracic Surgery (EACTS) and the European Society of Cardiology (ESC), in collaboration with the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J 29(11): 1463-1470.
- Fassl J, Seeberger MD, Augoustides JG (2011) Transcatheter Aortic Valve Implantation: Is General Anesthesia Superior to Conscious Sedation? J Cardiothorac Vasc Anesth 25(3): 576-577.
- Oguri A, Yamamoto M, Mouillet G, Gilard M, Laskar M, et al. (2014) FRANCE 2 Registry Investigators. Clinical outcomes and safety of transfemoral aortic valve implantation under general versus local anesthesia: subanalysis of the French Aortic National CoreValve and Edwards 2 Registry. Circ Cardiovasc Interv 7(4): 602-610.
- Gilard M, Eltchaninoff H, Iung B, Donzeau-Gouge P, Chevreul K, et al. (2012) Registry of transcatheter aortic-valve implantation in high-risk patients. N Engl J Med 366(18): 1705-1715.
- Stahli BE, Bunzli R, Grunenfelder J, Buhler I, Felix C, et al. (2011) Transcatheter Aortic Valve Implantation (TAVI) Outcome According to Standardized Endpoint Definitions by the Valve Academic Research Consortium (VARC). J Invasive Cardiol 23(8): 307-312.
- Attias D, Himbert D, Ducrocq G, Detaint D, Al-Attar N, et al. (2010) Immediate and mid-term results of transfemoral aortic valve implantation using either the Edwards Sapien transcatheter heart valve or the Medtronic CoreValve System in high-risk patients with aortic stenosis. Arch Cardiovasc Dis 103(4): 236-245
- Leon MB, Smith CR, Mack M, Miller DC, Moses JW, et al. (2010) Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med 363:1597-1607.
- Covello RD, Maj G, Landoni G, Maisano F, Michev I, et al. (2009) Anesthetic Management of Percutaneous Aortic Valve Implantation: Focus on Challenges Encountered and Proposed Solutions. J Cardiothorac Vasc Anesth 23(3): 280-285.
- Guinot PG, Depoix JP, Etchegoyen L, Benbara A, Provenchere S, et al. (2010) Anesthesia and perioperative management of patients undergoing transcatheter aortic valve implantation: analysis of 90 consecutive patients with focus on perioperative complications. J Cardiothorac Vasc Anesth 24(5): 752-761.
- Leon MB, Piazza N, Nikolsky E, Blackstone EH, Cutlip DE, et al. (2011) Standardized endpoint definitions for Transcatheter Aortic Valve Implantation clinical trials: A consensus report from the Valve Academic Research Consortium. J Am Coll Cardiol 57:253-269.
- Gilard M, Eltchaninoff H, Iung B, Donzeau-Gouge P, Chevreul K, et al. (2012) Registry of Transcatheter Aortic-valve Implantation in High-riks Patients. N Engl J Med 366(18): 1705-1715.
- Dehedin B, Guinot PG, Ibrahim H, Allou N, Provenchere S, et al. (2011) Anesthesia and Perioperative Management of Patients Who Undergo Transfemoral Transcatheter Aortic Valve Implantation. J Cardiothorac Vasc Anesth 25(6): 1036-1043.
- Yamamoto M, Meguro K, Mouillet G, Bergoend E, Monin JL, et al. (2013) Effect of local anesthetic management with conscious sedation in patients undergoing transcatheter aortic valve implantation. Am J Cardiol 111(1): 94-99.
- Motloch LJ, Rottlaender D, Reda S, Larbig R, Bruns M, et al. (2012) Local versus general anesthesia for transfemoral aortic valve implantation. Clin Res Cardiol 101(1): 45-53.






























