Isolation and Characterization of Kerosene from Fire Debris Using Chromatographic Techniques
Shipra Rohatgi1* and Himanshu Khajuria2
1Amity institute of forensic sciences, Amity University Noida, India
2Amity Institute of Forensic Sciences, Amity University, India
Submission:August 23, 2019; Published: October 02, 2019
*Corresponding author:Shipra Rohatgi, Ex-Msc student ,Amity institute of forensic sciences, Amity University Noida, Uttar Pradesh, India
How to cite this article:Shipra R, Himanshu K. Isolation and Characterization of Kerosene from Fire Debris Using Chromatographic Techniques. J Forensic Sci & Criminal Inves. 2019; 12(5): 555846. DOI: 10.19080/JFSCI.2019.12.555846.
Abstract
Dowry Deaths and the malicious fire are very common in the society and are being achieved by making the use of accelerants. The common accelerants available in the modern era are petrol, diesel, kerosene among which kerosene being the cheaper is highly recommended for creating deliberate fires and committing dowry deaths. The ability to detect accelerants mainly petrol, kerosene and diesel is very critical in the case of dowry deaths and malicious fires. However, it is also difficult to identify very small quantities of the kerosene in different conditions of cloth pieces- Unburned, partially burned and completely burned. There is a need for the isolation and characterization of kerosene from the fire debris so that a quick result can be obtained with a great ease and accuracy and the culprit can be catch hold faster. This paper focuses on the extraction methods of kerosene from the different conditions of cotton cloth piece followed by the analysis of the samples using GC-FID. With the Development of this technique it would be possible to retrieve the data from the different prevailing forms of cloth pieces which in result will fasten the process. The GC analysis will give the outcome of the detection limit of the kerosene from different forms of cloth piece which will prove beneficial for the forensic cases.
Keywords: Kerosene; Isolation; Fire debris; Characterization; Gas chromatography; Dowry deaths; Petroleum products; kerosene lamp; Powering jet engines; Mosquito breeding; Criminal and civil court
Introduction
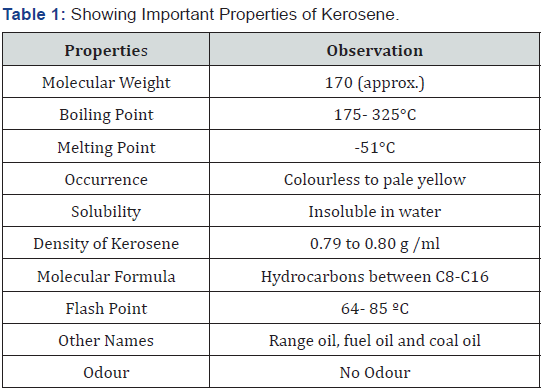
Petroleum products are substances or products produced after distillation process. The processing of crude oil is done in oil refinery; which consists of machines and other setup which are designed such that they cause physical and chemical changes in crude oil and convert it into various useful petroleum products. The process of production of petroleum products including; cracking and catalyzing with different gases separating at different temperatures from the liquids [1,2]. History of Petroleum industry states that the Petroleum Products were first produced by five businessmen in the month of February in 1938 in Beckley state [3,4]. First Oil Lake was drilled by E.L drake in 1859; in Pennsylvania. Kerosene contains a blend of different hydrocarbons produced by the process of distillation of crude oil. It is one of the important petroleum products. Kerosene is also known as paraffin oil and paraffin is commonly known as a fuel for jet engines. It is widely used as a as a fuel in domestic lightning (mainly kerosene lamp); cleaning solvent; etc. The use of kerosene in different areas of would have reduced tremendously; but still it is being commonly used as a fuel for heating; cooking and as a source of lightning lamp. Before the era of electricity; kerosene was majorly used for cooking; automobiles and lightning houses [4-7]. kerosene is a mixture of alkanes hydrocarbons and it consist of n- paraffin – 99 weight % that have carbon atoms ranging from 7 to 18 or Iso –paraffin that consist of 7 to 18 carbon atoms along with cyclo- paraffin containing 9 to 18 carbon atoms [5-7].
Applications of Kerosene
a) It is primarily used for heating lightning a home.
b) It is also widely used for powering jet engines aircraft.
c) It has many domestic uses.
d) It is also used to operate portable stoves for camping stoves.
e) Folk medicine reveals that Kerosene is known to be used for treating snake bites; to kill lice and to prevent mosquito breeding.
f) It is also used as lubricant; cleaning solvent etc.
g) Kerosene is also used by the performers (fire dancers; fire jugglers etc.) for entertainment. [4-8]
In India crime against women are increasing at a tremendous rate among which bride burning for dowry demand is one of the major crimes. Some of the other crimes against women involve eve teasing and acid throwing. According to the records of National Crime Records Bureau (NCRB); in year 2012 about 8;233 dowry death cases were reported in different states of India. While going through reports due to fire in kitchen and the number of pendency in courts has also increased at a faster rate [9,10]. Arson and dowry deaths have become a significant crime in today’s society and hence new approaches are required for quicker and more sensitive analysis of debris from suspected arson and dowry death cases. In year 1940; the analysis of the fire debris was conducted for the first time and after that different methods for separation and identification came into the field followed by modifications since today. Although various analytical methods have been suggested for this purpose. The most popular analytical method till date for the identification of fire residues is gas chromatography. Since normally there is little fire hand witness evidence in criminal and civil court actions [10-12].
Methodology
The analysis procedure for arson evidences consist of two stages: -In first stage the isolation of compound is done while in the second stage the separation of chromatographic analyte is done followed by its analysis and calculation of retention time and concentration of analyte [11-13]. The two hypotheses were made in order to achieve the objective of this study:
a. The first hypothesis was made that the Kerosene can be detected and characterized from the fire debris using Thin layer chromatography and GC- FID (Gas chromatography flame ionization detector).
b. Secondly; it was also hypothesized that the Kerosene can also be detected and characterized from the fire debris at different stages of burning i.e. From Unburned cloth piece; partially or semi – burned cloth piece and completely burned cloth piece using thin layer chromatography and GC- FID (Table 1).
Sampling
The samples for the analysis through gas chromatography were the cotton cloth piece which were soaked in kerosene and the sampling procedure was as follows:
a) Three (3) Cloth pieces of approximately 20cm x 15 cm were taken and dipped in 5ml of accelerant sample i.e. kerosene.
b) The clothes were allowed to absorb some accelerant (kerosene) and were then taken out.
c) One cloth after being dipped into the accelerant (kerosene) was taken into the conical flak for extraction.
d) The second cloth dipped in kerosene was taken and lit on fire with the help of a matchstick. The fire was
e) doused without allowing the cloth piece to burn completely. The partially burned cloth sample was then taken
f) in the conical flask for extraction.
g) The third cloth piece dipped in kerosene was taken and was lit on fire with the help of matchstick. The cloth was allowed to burn completely till the ash is formed. The ash of cloth piece was then collected and corked into a conical flask for extraction.
Extraction Process
The steps for the extraction of unburned; semi burned and partially burned cloth piece was as follows:
i. Conical flask containing the cloth samples for the extraction of the accelerant traces in solvent.
ii. The conical flasks were then corked and shaken properly and cautiously.
iii. The conical flasks were then left undisturbed for about 24 hours.
iv. Next day the diethyl ether extracts were then filtered to obtain a clear filtrate.
v. The extract was then reduced to its half volume by keeping it in sunlight for drying.
vi. The clear solutions were then taken into a 5 ml volume stoppered volumetric flasks and sealed with aluminum foil.
vii. The samples were then spotted on TLC plates followed by gas chromatography by injecting 5μl volume of samples.
Results
TLC –Thin layer chromatography
Solvent system
The TLC for the kerosene sample along with the standard kerosene was carried out in three different solvent systems
a. Chloroform: Acetone: Methanol (50:8:25)
b. n-hexane: n-heptane with 8 % formaldehyde (80:20)
c. Chloroform: Benzene: Acetic acid (10:4:2)
GC-FID (Gas chromatography -flame ionization detector)
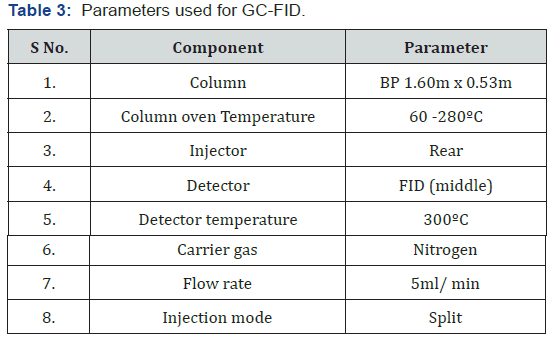
The detector i.e. FID consist of a flame which causes analyte components to ionize or loose electron. The ion cause an electric current to flow. This current may be amplified and displayed as peaks. The more the analyte component; the more is the current produced and hence a bigger peak. This detector can be used for the qualitative and quantitative analysis. Normally; hydrogen gas is used as a flame in the presence of oxygen in the detector. The parameters for carrying out the analysis for detection and characterization of Kerosene from different degree of burned clothes were as follows: The graph obtained after the analysis of different samples by performing GC-FID are as follows Table 2. The above Figure 1 shows the Chromatogram for the standard Kerosene. The chromatogram shows characteristics peaks for the Kerosene. This chromatogram of standard kerosene was used for the purpose of comparison with the samples of kerosene obtained in different cloth samples. The above Figure 2 shows the chromatogram for the Kerosene detected from unburned cloth piece using GC-FID (Gas chromatography flame ionization detector). The above Figure 3 shows the chromatogram for the Detected Kerosene in a partially burned cloth piece. The given Figure 4 shows the chromatogram for the detected Kerosene amount from completely burned cloth piece (Table 3).
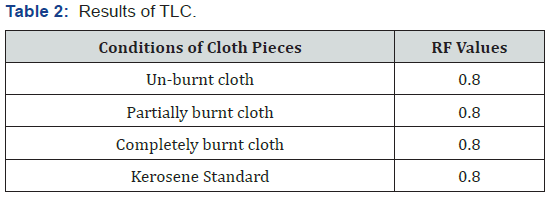



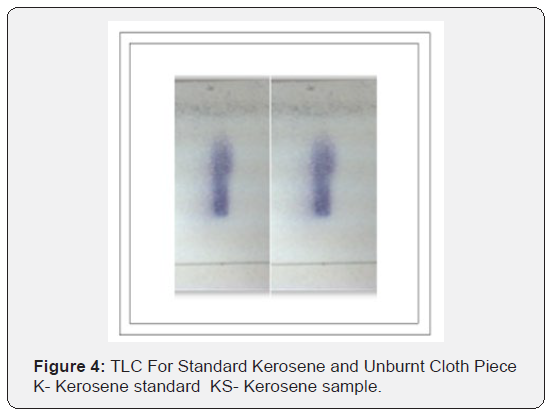
Conclusion
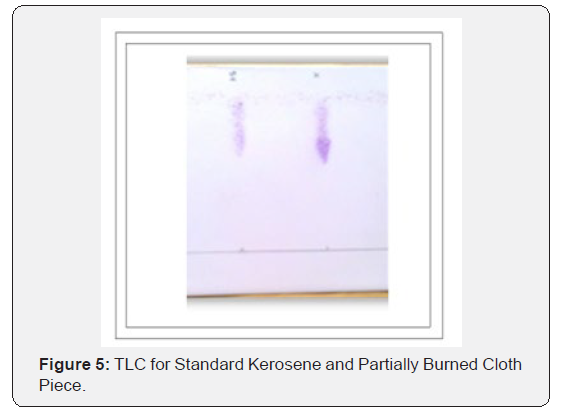


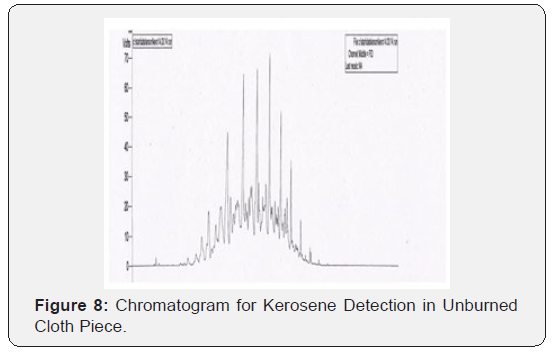
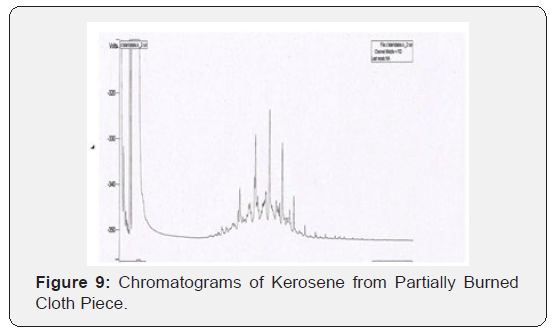
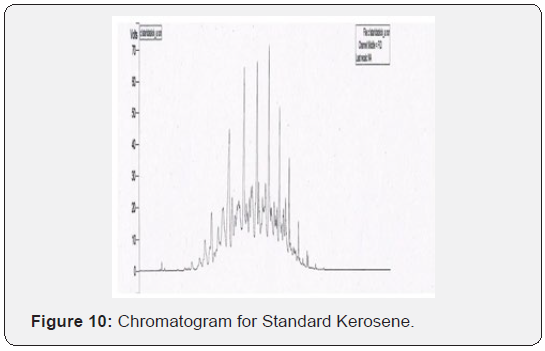
Through this research it has been tried to demonstrate the ability to isolate; detect and characterize the kerosene from fire debris. The chromatographic techniques were used to detect and characterize the kerosene from different cloth samples with different stage of burning. Based on objective of the current study we can hypothesize that Kerosene can be detected and characterized from the fire debris at different stages i.e. Un-burned; semi–burned and fully burned cloth piece using Chromatographic Techniques (TLC and GC- FID).By performing TLC (thin layer chromatography) in three different solvent systems it was found that the Retention factor (RF) value for standard kerosene was 0.80 that which matched with the kerosene samples obtained from different cloth piece and the result was found to be similar. Then the GC-FID was done for the further analysis of kerosene in the cloth samples and the chromatogram was obtained (Figure 5 & 6) The standard Kerosene chromatogram was matched with the kerosene samples obtained from different cloth pieces. After comparing the chromatograms obtained by GC-FID it can be concluded that in case of unburned material; partially burned fire debris sample; the Kerosene can be isolated using gas chromatography with flame ionization detector; therefore the hypothesis was validated .In case of completely burned fire debris and the extracts were devoid of traces of Kerosene as shown in (Figure 7). However; Kerosene could be positively identified as shown in (Figure 8).Therefore I would like to conclude with the help of (Figure 8) that the Kerosene can be isolated from unburned fire debris and can be easily characterized by comparing with the standard (Figure 8 & 9) It is also evident from (Figure 10) that kerosene can be isolated and extracted better from semi – burned fire debris than from completely burned fire debris .Kerosene is one of the most important petroleum distillate.
It has been greatly used in many criminal activities including culpable homicide. Kerosene being inexpensive and easily available as compared to the other petroleum products such as gasoline and diesel is used in the cases of Arson as well as Dowry deaths. According to many magazines and newspaper report the rate of kerosene for the use of killing the brides has been increased tremendously. Every year police reports about 2500 of bride burning of cases in which kerosene is the most often used fuel. Therefore by applying the similar method of analysis to the evidences recovered from the crime scene the detection and characterization of the Kerosene as well as different petroleum products can be done effectively and efficiently with a faster speed As a result of which the Arson and Dowry death cases can be solved easily and in a quick period of time so that the culprit can be punished as early as possible. In future this project can be furthered by performing quantitative analysis for the detection of Kerosene in debris burned for different time lengths using mass spectrometry or other such detectors [12-15].
Acknowledgement
The authors are grateful to the authorities of State forensic Science Laboratory; Govt. of NCT of Delhi; Madhuban Chowk; Sector14; Rohini; New Delhi-110085; India.
References
- Caldicott (2008) A Distillation, Cracking and Catalyzing; Refining petroleum. Watershed sentinel08:04.
- Shackne S (1960) Petroleum Geology of the United States. Oil for the World (2ndedn).
- Mosley L (1973) Oil in the Middle East. Power play (2ndedn).
- JHB (1994)The History of the British Petroleum company, The Anglo-Iranian Years, 1928-1954. Cambridge University p. 2.
- http://www.britannica.com
- http://dictionary.reference.com
- Clive M (2007) Oil Sands - Glossary. Oil Sands Royalty Guidelines. Government of Alberta.
- Masahiko Shibuya, Tsutomu Yoshida, Yasuyuki Komatsu, Noe Takehira (2004) Petroleum Products, CA2549922A1,Shell Oil Company, p. 8.
- Painter T (2010) Properties of kerosene.
- Nick SykesCo, David A Khan (2010)What is Kerosene and What are its Uses.
- Lakhani Avnita (2005)Bride Burning: The Elephant in the Room Is Out of Control. Rutgers University, USA.
- (1997) Kitchen fires Kill Indian Brides with Inadequate Dowry. UPI, New Delhi, India.
- (2010) Disposal of Cases by Courts. National Crime Records Bureau, India.
- KKLandes (1970) Petroleum Geology of the United States.In: S Shackne, ND Drake (Edt.), Oil for the World, (2nd),L. Mosley, Power Play: Oil in the Middle East.
- McWilliam IG,Dewar RA (1958) Flame Ionization Detector for Gas Chromatography. Nature 181(4611): 760.






























