The Effect of the Liquid Nitrogen Level on the Temperature in a Semen Storage Tank
A Ahmadzadeh1, A Hale1, B Shafii2 and J Dalton3*
1 Department of Animal, Veterinary and Food Sciences, University of Idaho, Moscow 83844, USA
1 Statistical Programs, College of Agricultural and Life Sciences, University of Idaho, Moscow 83844, USA
1 Department of Animal, Veterinary and Food Sciences, University of Idaho, Caldwell 83605, USA
Submission: January 17, 2022; Published: January 27, 2022
*Corresponding author: Joseph C Dalton, College of Agricultural and Life Sciences, Animal, Veterinary and Food Sciences, Caldwell, Idaho 83605, USA Dairy
How to cite this article: A Ahmadzadeh, A Hale, B Shafii, J Dalton. The Effect of the Liquid Nitrogen Level on the Temperature in a Semen Storage Tank. Dairy and Vet Sci J. 2022; 15(2): 555906. DOI: 10.19080/JDVS.2022.15.555906
Abstract
The temperature in a liquid nitrogen storage tank is critical to maintain the viability of bovine semen stored within the tank. The objective was to determine the relationship of the temperature gradient in the neck of a tank to the level of liquid nitrogen in the tank. Using an electronic thermometer, temperature was measured at three locations: a) 2 cm below the top of the neck, b) 7 cm below the top of the neck (and below the frost line), and c) 2 cm above the level of liquid nitrogen. Liquid nitrogen volume in the tank was incrementally decreased from 18 L to 6 L. The experiment was repeated twice, and temperatures at each location and nitrogen level were recorded four times. Data on the effect of liquid nitrogen level, location, and two-way interaction on the temperature gradient in the neck of the tank were analyzed using a general linear mixed model procedure. There was an effect of location and volume by location interaction on tank temperature (P < 0.01). Mean tank temperature was greater (P < 0.01) 2 cm below the top of the neck as compared to 7 cm below the top of the neck (and below the frost line) (6.4 ± 0.1 °C vs -38.3 ± 0.3 °C). The effect of liquid nitrogen volume on tank temperature was not consistent across locations. As liquid nitrogen levels decreased, the temperature remained above 0 °C and did not change at 2 cm below the top of the neck. However, the temperature increased (from -41 ± 1 to -36.7 ± 1 °C;P < 0.05) at 7 cm below the top of the neck (below the frost line) as the liquid nitrogen level decreased. At 2 cm above the level of liquid nitrogen, the temperature increased (from -187 ± 0.3 to -185 ± 0.3 °C;P < 0.05) as liquid nitrogen levels decreased. Based on the covariance parameters estimated, the temperature below the frost line in the neck of the tank demonstrated the most variability. Data provide evidence of an increase in temperature at 7 cm below the top of the neck (below the frost line) as the liquid nitrogen level decreases. Therefore, removal of semen straws should be done efficiently, to preserve semen viability via minimization of exposure to increased temperatures in the neck of the tank.
Keywords: Liquid nitrogen; Semen storage tank; Temperature gradient
Introduction
Commercial bovine artificial insemination (AI) stud and custom semen collection businesses provide a highly fertile product to their customers. When semen is purchased and transferred to the producer’s or professional AI technician’s liquid nitrogen tank, the maintenance of male fertility is in the hands of the producer, farm employees, and AI technicians. According to guidelines provided by bovine stud companies, special consideration should be given to semen storage and care and maintenance of the liquid nitrogen tank to ensure the maximal potential fertility within straws of frozen semen. During the last 25 years countless hours have been spent developing, refining, and implementing estrous and ovulation synchronization programs, while little effort has been focused on semen storage and handling. Semen is a significant investment, not only in cost of purchase, but in potential revenue from genetic progress. Therefore, proper maintenance and care of the liquid nitrogen storage tank is important to protect that investment. Indeed, failure to monitor the liquid nitrogen level in semen storage tanks, resulting in loss of semen, has been reported previously [1].
Training manuals for AI schools routinely include a brief discussion focused on limiting semen exposure, including understanding the temperature gradient in the neck of the tank [2,3], and the importance of working below the frost line in the neck of the tank to fertility. As it is easy to see the frost line, this recommendation has become a semen handling rule of thumb. Kaproth [4] argued the potential impractical nature of the frost line rule of thumb, as he reported temperatures below the frost line varied from -40 to -120 °C in different tank models. No mention, however, was made of the corresponding liquid nitrogen levels in these tanks, although temperatures in the neck of a tank from another experiment were reported to be affected by liquid nitrogen level (15 vs 26 cm liquid nitrogen) [4]. Saacke [2] reported a -140 °C temperature below the frost line, providing evidence a static high level of liquid nitrogen was likely present in the tank at the time of temperature measurements. The paucity of data regarding the temperature gradient change in the neck of a tank as the liquid nitrogen level decreases provides an incomplete and confusing picture especially when coupled with the variation in reported neck temperatures [2,4], and the realization sperm injury occurs at temperatures as low as -80 °C [5-7]. Consequently, the objective of this study was to determine the relationship of the temperature gradient in the neck of a semen storage tank to the level of liquid nitrogen in the tank.
Materials and Methods
The experiment was conducted in the Reproductive Physiology Laboratory of the Department of Animal, Veterinary, and Food Sciences at the University of Idaho. The temperature of the laboratory during the experiment was approximately 22 °C.
A liquid nitrogen storage tank with a capacity of 30 L was used (Union Carbide-Linde, Type LR-30, USA). No semen was stored in this tank for the duration of the experiment. The tank neck diameter was 7 cm, and the length of the neck tube was 12 cm, which is similar to tanks currently in use on dairies by herdsmen-inseminators and professional AI technicians. The tank was filled with 18 L of liquid nitrogen (equivalent to 22 cm) at the initiation of the experiment. A hand-held thermocouple thermometer equipped with an air(gas) probe capable of a 6 second response time (TM602; Palmer Wahl Instrumentation Group, Asheville, NC, USA) was used to acquire temperature measurements at three locations: 1) 2 cm below the top of the neck (above the frost line), 2) 7 cm below the top of the neck (below the frost line and the recommended location for semen straw removal), 3) 2 cm above the level of liquid nitrogen (Figure 1).
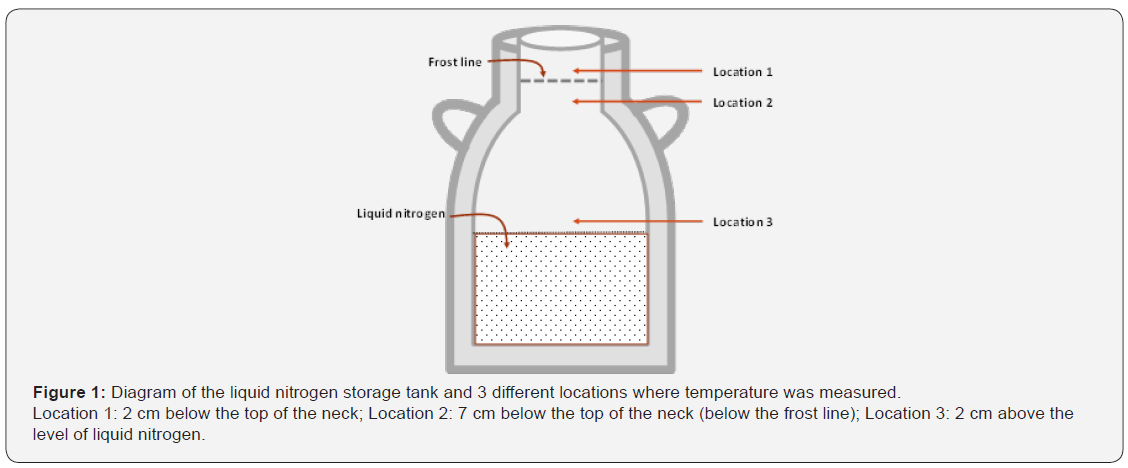
Liquid nitrogen level in the tank was incrementally decreased from 22 to 7 cm (the volume of liquid nitrogen incrementally decreased from 18 L to 6 L, respectively) by manually removing liquid nitrogen with a stainless steel dipper. Temperatures were measured four times at each location, as previously described, with 22, 20, 19, 18, 16, 12, 10, and 7 cm of liquid nitrogen in the tank. The elapsed time between each temperature measurement was at least 2 hours, allowing for an adequate wash-out period and to avoid potential carry-over effects. Once the first round of the experiment was completed, the same tank was again filled with liquid nitrogen, and the experiment was repeated for the second time, at the same levels of liquid nitrogen and replications.
Data on the effects of liquid nitrogen level, location and two-way interaction on the tank’s temperature were analyzed using a general linear mixed model procedure GLIMMIX in SAS [8]. The model included the effects of location, volume, and volume by location. Parameter estimates for temperature variability and least square mean temperatures at all tank locations were computed. Significance was declared at P < 0.05.
Results and Discussion
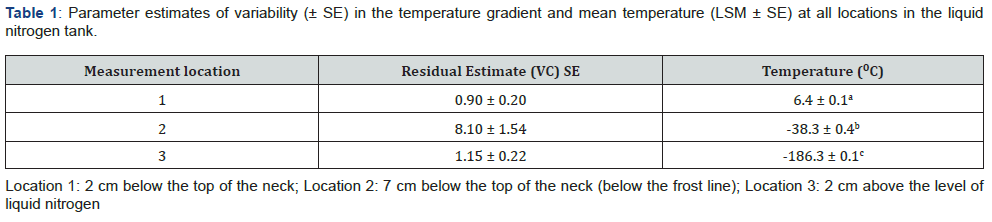
The temperature in semen storage tanks is critical to maintaining the viability of semen stored within the tanks. The variability in temperature across the three locations where temperatures were measured was not consistent. Based on the estimated variance, location 2 (7 cm below the top of the neck, and below the frost line) exhibited the greatest variability (Table 1). There was an effect of location and location by volume on tank temperature (P < 0.01).
Mean tank temperature was greater (P < 0.01) 2 cm below the top of the neck as compared to 7 cm below the top of the neck (and below the frost line) (6.4 ± 0.1 °C vs -38.3 ± 0.4 °C) (Table 1). Our data for location 2 temperature (below the frost line) is very close to the upper range (-40 °C) reported by Kaproth [4]. As nitrogen levels decreased, the temperature remained above 0 °C and did not change at 2 cm below the top of the neck (location 1). In contrast, in location 2, as liquid nitrogen volume decreased, the temperature increased (from -41.1 ± 1 to -36 .7± 1 °C; P < 0.05) at 7 cm below the top of the neck (Figure 2). These results agree with a previous report [4] that a warmer temperature in the neck region was associated with a lower tank liquid nitrogen level. Although the mean temperature in location 3 (2 cm above the level of liquid nitrogen) was -186.3 ± 0.1 (Table 1), as liquid nitrogen volume decreased, the temperature increased (from -187 ± 0.3 to -185 ± 0.3 °C; P < 0.05).
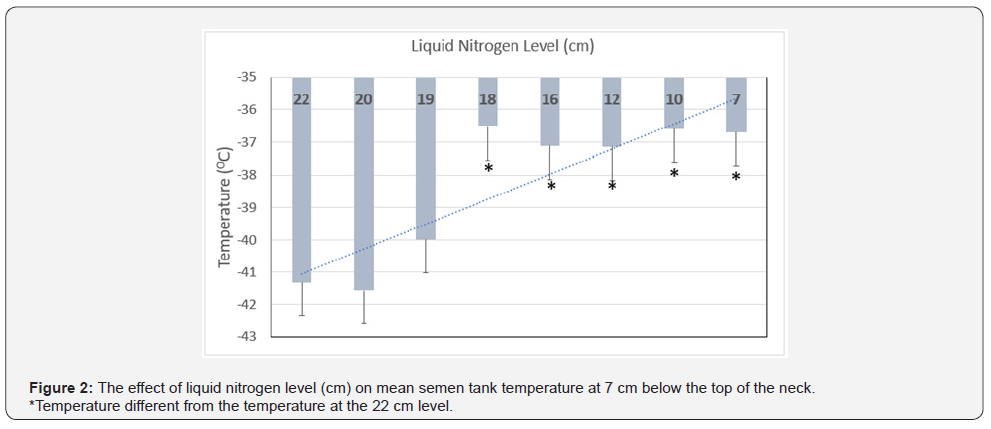
The frost line represents an important landmark as just below this location is the most commonly recommended area of semen straw removal. The mean temperature in both locations 1 and 2 were greater than previously reported [2], likely due to moderate to low liquid nitrogen levels in the present study as compared to Saacke [2]. The current study provides evidence the temperature in the neck of a semen storage tank, especially in the area below the frost line exhibits wide variation and is influenced by liquid nitrogen level. Berndtson et al. [9] reported the liquid nitrogen level in a tank can dramatically affect the temperature of straws repeatedly raised and lowered in the tank. In a classic experiment, elevation of a cane (containing a goblet with 5 semen straws) into the neck of the tank for 1 minute resulted in a straw temperature increase of 16 °C (from -196 to -180 °C) when the tank was full of liquid nitrogen. An increase in straw temperature was minimized as the goblet contained liquid nitrogen at the start of the exposure. Furthermore, the straw temperature reached -196 °C almost immediately upon return (within 1 minute), even after five repetitions [9]. In the same study, when the liquid nitrogen level in the tank was low (approximately 14 cm), the temperature of straws increased 73 °C (from -196 to -123 °C) during the first minute, and straw temperatures cooled only 30 to 40 °C two minutes after returning to the tank. Therefore, greater temperatures were reached during the second, third, fourth, and fifth repetitions. Greater than 10 minutes were required for the straw temperature to reach -196 °C upon return after the fifth repetition.
The fluctuation in semen straw temperature, as shown by Berndtson et al. [9] can also be envisioned by the results from the present study when a tank’s liquid nitrogen level is low. The ice pattern within a frozen solution such as bovine semen extender may change considerably (termed recrystallization) as it is warmed [10]. Stroud [11] asserted larger, extracellular ice crystals are associated with damage to sperm cell membranes and organelles, the severity of which is dependent upon how high the temperature rises, the duration of exposure above -130 °C, and the return to storage below -130 °C. Rapatz [10] provided further evidence that the danger zone for recrystallization appeared to be at temperatures above -80 °C in solutions containing glycerol (e.g., bovine semen extender).
When a 0.5 mL straw of semen was raised into the neck of a tank using forceps, the time necessary for the temperature to drop from -196 to -100 °C was 10 to 15 seconds [9]. Saacke [3] reported a similar result for the same temperature range. Therefore, to provide a margin of safety and avoid recrystallization injury to sperm in straws not removed from the same goblet or canister, the recommendation is for a cane (or canister containing canes) of frozen semen to be limited to eight seconds in the neck of a tank (below the frost line) when retrieving straws [3,9,11]. As originally described by Saacke [2] removal of a straw from a cane may expose other straws on the cane to injurious high temperatures depending on the time required to remove the straw. Working above the frost line, or in ambient air, will likely be particularly damaging to sperm even when applying the “eight second rule,” as the straw temperature will rapidly enter the danger zone [3,9,11]. Damage to sperm cannot be corrected by returning semen straws to liquid nitrogen (-196 °C). Consequently, damage to sperm in straws not removed during improper retrieval of other straws is additive [2,9,11].
Conclusion
As the liquid nitrogen level decreased in the current study, the temperature increased and exhibited significant variability 7 cm below the top of the tank (below the frost line, at the site of semen straw removal). This warmer temperature in the neck region of the tank increases the risk of semen exposure during semen handling, thereby potentially decreasing sperm viability. Producers and AI technicians are most likely to preserve semen fertility via minimization of straw exposure to increased temperature by 1) understanding the effect of liquid nitrogen level on neck temperature, 2) positioning the canister and canes containing semen straws below the frost line, 3) using tweezers to remove the desired straw and maintain cane position in the lower neck region, and 4) working expeditiously (adhering to the eight second rule) during straw retrieval.
Acknowledgment
This research was supported by a grant to Joseph C. Dalton and Amin Ahmadzadeh from Select Sires Inc., Plain City, OH. The authors also thank Idaho Dairymen’s Association for partial support of the project and Ms. Elery Martin for assistance with Figure 1.
Conflicts of Interest
No potential conflict of interest was reported by the authors.
References
- Senger PL, Becker WC, Hillers JK (1980) Quality of semen stored in on-the-farm semen tanks. Journal of Dairy Science 63(4): 646-649.
- Saacke RG (1974) Concepts in semen packaging and use. In: Proc 8th Conference on AI in Beef Cattle, pp. 11-19.
- Saacke RG (1978) Factors affecting spermatozoan viability from collection to use. In: Proc. National Association of Animal. Breeders 7th Tech. Conference on Artificial Insemination and Reproduction Columbia, MO, pp. 3-9.
- Kaproth M (2006) Forensics for the bull stud and semen processing lab: Interpreting tank, straw and semen quality clues. In: Proc. National. Association Animal Breeders 21st Technical Conference on Artificial Insemination and Reproduction, Columbia, MO, pp. 54-63.
- Etgen WM, Ludwick TM, Rickard HE, Hess EA, Ely F (1957) Use of mechanical refrigeration in preservation of bull semen. Journal of Dairy Science 40: 774-778.
- Bean BH, Pickett BW, Martig RC (1963) Influence of freezing methods, extenders and storage temperatures on motility and pH of frozen bovine semen. Journal of Dairy Science 46(2): 145-149.
- DeJarnette JM (1999) Factors affecting the quality of frozen semen after thawing. In: Proc. Society for Theriogenology Annual Conference, Nashville, TN, pp. 267-279.
- SAS Institute (2015) SAS Version 9.4. SAS Institute Inc. Cary, NC.
- Berndtson WE, Pickett BW, Rugg CD (1976) Procedures for field handling of bovine semen in plastic straws. In: Proc. Nat’l. Association Animal Breeders 6th Technical Conference on Artificial Insemination and Reproduction., Columbia, MO, pp. 51-60.
- Rapatz GL (1966) What happens when semen is frozen. In: Proc. National Association of Animal Breeders 1st Technical Conference on Artificial Insemination and Reproduction, Columbia, MO, pp. 54-63.
- Stroud B (2016) Consequences of mishandling frozen semen and embryos. In: Proc. Appl Reprod Strat in Beef Cattle, Des Moines, IA, pp. 119-130.
- Saacke RG, Lineweaver JA, Aalseth EP (1978) Procedures for handling frozen semen. In: Proc. 12th Conference on AI in Beef Cattle, pp. 46-61.






























