- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Veterinary Drug Residue: The Risk, Public Health Significance and its Management
Mohammed Ture*, Tsegaw Fentie and Bushura Regassa
Department of veterinary medicine and animal science, University of Gonder, Ethiopia
Submission: July 3, 2019; Published: July 26, 2019
*Corresponding author: Mohammed Ture, Department of veterinary medicine and animal science, University of Gonder, Ethiopia Dairy and
How to cite this article: Mohammed Ture, Tsegaw Fentie, Bushura Regassa. Veterinary Drug Residue: The Risk, Public Health Significance and its Management College of Veterinary Medicine and Animal Science. Dairy and Vet Sci J. 2019; 13(2): 555856. DOI: 10.19080/JDVS.2019.13.555856
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Abstract
Veterinary drugs are any substances applied to or administered to animals for their therapeutic, prophylactic and diagnostic purposes or modification of physiological function or behavior. They are used throughout the world and more than half of all medicines are prescribed, dispensed or sold improperly. In Ethiopia, also different studies revealed the improper utilization of drugs is common. The use of veterinary drugs in food-producing animals has the potential to generate residues in animal derived products and poses a health hazard to the consumer. The most likely reason for drug residues might be due to improper drug usage and failure to keep the withdrawal period. The residual amount ingested is in small amounts and not necessarily toxic. The major public health significances of drug residue are development of antimicrobial drug resistance, hypersensitivity reaction, carcinogenicity, mutagenicity, teratogenicity, and disruption of intestinal normal flora. The aim of this paper is to review about risk of occurrence of veterinary drug residue, public health effects and management. Even though, veterinary drugs have a great importance in treating, preventing and diagnosing diseases, it has major public health hazards. To avoid this it is important to use these drugs rationally, the safety levels of food must be strictly observed, drug products should be used in accordance with the labeled directions and public awareness should be created on the public health significance of drug residue.
Keywords: Antimicrobial; Drug; Residue; Risk; Veterinary drug
Abbrevations: ABZ: Albendazole; ADI: Acceptable Daily Intake; AMR: Antimicrobial Resistance; APCI: Atmospheric Pressure Chemical Ionization; APEC: Asian-Pacific Economic Cooperation; BZDs: Benzimidazoles; CFR: Code of Federal Regulation; DES: Diethylstilbestrol; DNA: Deoxyribonucleic acid; EC: European Community; EFSA: European Food Safety Authority; ELDU: Extra-label drug use; ELISA: Enzyme linked-immunosorbent assay; ELU: Extra-label use; ESI: Electrospray Ionization; EU: European Union; FAO: Food and Agricultural Organization; FDA: FOOD and Drug Administration; FDACVM: Food and Drug administration Center for Veterinary Medicine; FEB: Febantel; HPLC: High-performance liquid chromatography; IgE: Immunoglobulin E; LC-MS/ MS: Liquid chromatography-mass spectrometry/mass spectrometry; MBZ: Mebendazole; MRL: Maximum Residue Level; NAP: National Academies Press; NOEL: No observed effect level; RNA: Ribonucleic acid; UEMOA: West African Economic and Monetary Union; WHO: World health organization
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Introduction
Veterinary drug” means any substance or mixture of substances which is used, or is manufactured, sold or represented as suitable for use, in (1) the diagnosis, treatment, mitigation or prevention of disease or abnormal physical or mental state or the symptoms thereof in an animal; or (2) restoring, correcting or modifying any physical, mental or organic function in an animal [1]. The use of veterinary drugs in livestock production is inevitable as they are essential for treatment of diseases (therapeutic), prevention of diseases (prophylaxis), modification of physiological functions (such as tranquilizers, anesthetic drugs), improvement of growth and productivity (growth promoters) as well as for ensuring food safety [2]. The veterinary drugs are used throughout the world and they comprise a broad variety of classes of chemical compounds including vaccines, antimicrobials, antiparasitics and β-agonists [3]. Antimicrobials are the most important and most frequently used group of veterinary drugs [4]. Antimicrobials are medicine (natural, synthetic or semi-synthetic origin) that inhibits the growth of or destroys microorganisms when applied at low concentrations without causing host damage [5]. Among the antimicrobials that are commonly used in livestock production are tetracyclines, amprolium, penicillin, streptomycin, sulphonamides, tylosin, aminoglycosides, β-lactams, macrolides and lincosamides, quinolones and sulfonamides [6]. While that of antiparasitic agents include anthelmintics or coccidiostats, stilbenes, amphenicols, nitrofurans, nitroimidazoles, carbamates, pyrethroids and sedatives [5].
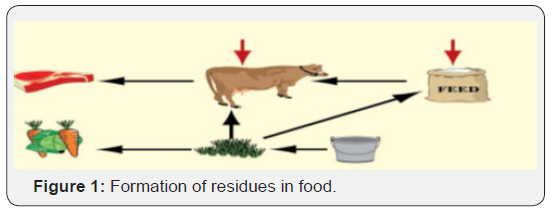
A residue, defined in the simplest terms, results when a drug or pesticide is deliberately applied to a food-producing animal or plant. Residues of veterinary drugs include the parent compounds and/or their metabolites in any edible portion of the animal product and include residues of associated impurities of the veterinary drug concerned [7]. Residual amounts of antimicrobials or their toxic metabolites found in meat, organs or other products such as milk and egg of food producing animals is called veterinary drug residues [8]. Consumption of such food products poses a major health risk due to the failure of treatment following the development of resistant microorganisms [9]. Many livestock producers treat their animals by themselves. Even if they use the same drugs as veterinarians, they have little understanding of the conditions and quantities to administer or the waiting periods. The uncontrolled use of anti-infectious agents can lead to residues in animal products, especially when users fail to respect waiting periods. The risks of residues in foodstuffs of animal origin could be reflected into several forms [10]. The immediate effect of antimicrobial residue is allergenicity and toxicity in human through the food chain [11]. The long-term health adverse effects such as increased likelihood include disruption of normal human flora in the intestine (microbiological effects), carcinogenicity, and teratogenicity [12]. Other drug residue problems are the development of antibiotic-resistant microbes and drug misuse [13]. The objective of this paper is to review: The risk of occurrence of veterinary drug residue, public health effects and management (Figure 1).
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Historical Background
A whole series of known or new foodborne biological and chemical hazards are threatening health [14]. In the European Union (EU), following a string of health crises, the food safety mechanism has evolved towards a risk analysis approach. This shift to the concept of ‘farm to fork’ risk management [15] led to the establishment of food safety agencies at the European level. The risks of residues from veterinary medicinal products used in livestock production were taken on board in the 1980s, most notably through European harmonization of the regulations on medicinal products for veterinary use. Over the past decade, the EU has improved its regulatory framework to better supervise, assess, monitor and control food production under the ‘Food Law’. More recently, the use of anti-infective in livestock and its contribution to the development of antimicrobial resistance has attracted considerable attention [16].
In Africa - particularly West Africa - only microbial pathogens, pesticide residues and aflatoxins have been the subject of measures to protect the safety of food for human consumption. These hazards were perceived as the greatest threat to public health. In April 2007, the eight UEMOA countries (Benin, Burkina- Faso, Cote d’Ivoire, Guinea-Bissau, Mali, Niger, Senegal and Togo) adopted regulation 07/2007/CM/UEMOA concerning plant, animal and food safety in the UEMOA area [17]. More recently, in 2010 and 2011, two training sessions were held in Benin to familiarize these countries with the theoretical framework for health risk analysis [18]. Yet, there have been very few studies on antimicrobial residues affecting food safety [19]. However, in developing countries, failure to respect waiting periods [20] leads to high exposure to antimicrobial residues [21].
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Veterinary Drug and their Use in Food Animals
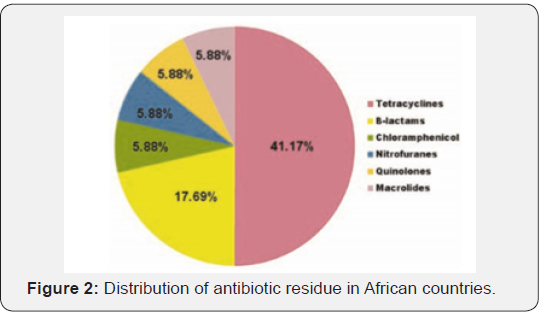
Drug in animals can be used as therapeutic, prophylactic and growth promotion. Therapeutic use refers to the treatment of established infections whereas prophylaxis is the use of drugs either in individual or groups to prevent the development of infections. Growth promoters (GPs) are any antimicrobial agents administered at low or sub therapeutic dose to destroy or inhibits growth of microbe which reduce the yield of food animals. The use of antimicrobials as feed supplements can promote the growth of food animals and also enhance feed efficiency. The uses of GPs are resulting in meat of better quality with less fat and increased protein contests [22]. The use of drugs in food animals is fundamental to animal health and well-being and to the economics of the industry. There are five major classes of drugs used in food animals: (1) topical antiseptics, bactericides, and fungicides used to treat surface skin or hoof infections, cuts, and abrasions; (2) ionophores, which alter rumen microorganisms to provide more favorable and efficient energy substrates from bacterial conversion of feed and to impart some degree of protection against some parasites; (3) steroid anabolic growth promoters (for meat production) and peptide production enhancers (bovine somatotropin for increased milk production in dairy cows); (4) antiparasite drugs; and (5) antibiotics as used to control overt and occult diseases, and to promote growth [23] (Figure 2).
Authorized Veterinary Antimicrobials
The medicinal products containing antimicrobials authorized for veterinary use are those that have passed the marketing authorization process of the competent national authority. After an evaluation of the scientific data proving the efficacy of the product and its safety for humans, animals and the environment the Competent Authority authorizes its importation, distribution and use. No medicinal product may be marketed unless it has first been authorized by the Competent Authority. However, there are huge shortcomings in the implementation because the technical evaluation of a marketing application is limited to an administrative procedure alone especially in most African countries [24].
Prohibited Veterinary Antimicrobials
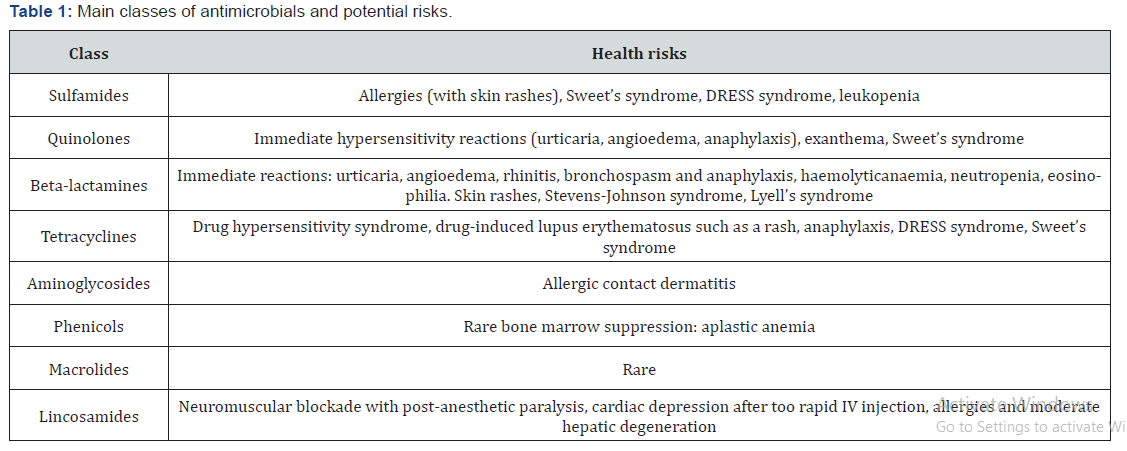
Prohibited antimicrobials are substances for which it is not possible to determine the Maximum Residue Level (MRL). Chloramphenicol, dimetridazole, ipronidazole, nitroimidazoles, furazolidone, nitrofurazone, and fluoroquinolones are prohibited for extra-label use in food-producing animals [24]. Chloramphenicol is a broad-spectrum antimicrobial against Gram-positive and Gram-negative bacteria. It was not possible to determine an MRL based on the available data. The inability to set a threshold value and shortcomings in the marketing authorization application led to chloramphenicol being classified in 1994 as a prohibited substance for use in food-producing animals. Dapsone, which is used to treat leprosy in humans, is not authorized for use in food-producing animals in Europe because of insufficient toxicology data, making it impossible to determine the acceptable daily intake (ADI) [25]. In the year 1995 European Union (EU) prohibited the use of nitrofurans for the treatment of bacterial diseases in livestock production, due to concerns about the carcinogenicity of their residues in edible tissue [26]. In subsequent years Australia, USA, Philippines, Thailand and Brazil also prohibited the use of nitrofurans in food animals [27] (Table 1).
Origin of Residue
Veterinary drugs are generally used in farm animals for therapeutic and prophylactic purposes and they include a large number of different types of compounds which can be administered in the feed or in the drinking water. The great majority of residues found in edible tissues of animals have their source at the farm of origin. In some cases, the residues may proceed from contaminated animal feedstuffs. By far the most common cause of residues is the failure to observe the proper withholding period following treatment [28].
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Risk of Drug Residue for the Public Health
Human health risk can result from the presence of residues of veterinary drugs and/or their metabolites in edible organs and tissues of treated animals, in particular residues in concentrations exceeding the MRL established by Council Regulation 2377/90 [29]. Occurrences of veterinary drug residues pose the broad range of health consequences in the consumers. The residues of antibacterial may present pharmacological, toxicological, microbiological and immunopathological health risks for humans [30].
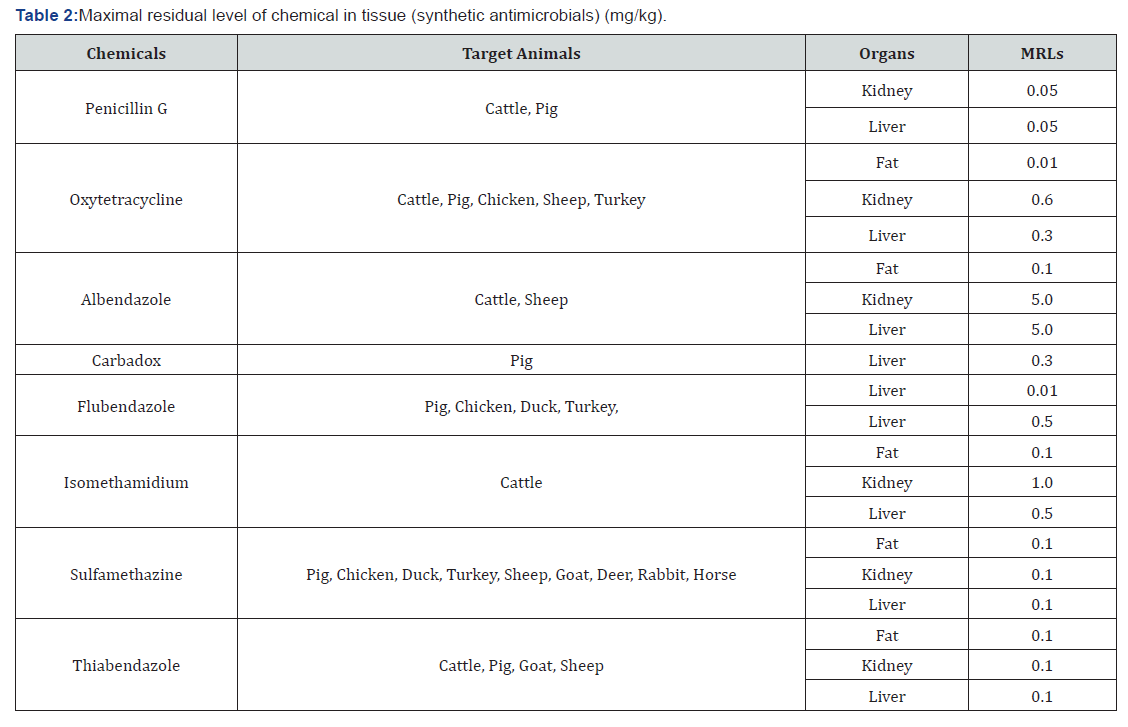
Anthelmintics, such as benzimidazoles and probenzimidazoles, are veterinary drugs used against endoparasites for the prevention of animal infestations caused by nematodes, cestodes and trematodes in food producing animals. Among the most popular benzimidazoles are Albendazole (ABZ) and Mebendazole (MBZ) [31]. Benzimidazoles (BZDs) such as albendazole (ABZ), fenbendazole and thiabendazole are a kind of broad-spectrum veterinary drugs for prevention and treatment of helminthic parasites in domestic animals. When BZD drugs were fed to domestic animals, they were metabolized and then converted into other compounds in vivo. Thus, these BZDs and their metabolites can be left inedible animal foods or exist in the environment for a period of time. The harmful BZDs and their metabolites residues in some foods lead to a series of toxic effects such as congenic malformations, teratogenicity, diarrhea, pulmonary edemas, polyploidy, and necrotic lymphoadenopathy [32]. Febantel (FEB) is a probenzimidazole with which is further metabolized in vivo to Fenbendazole, a benzimidazole anthelmintic also [31] (Table 2).
Risk Factors for Drug Residue Occurrence
Disease status: The disease status of an animal can affect the pharmacokinetics of drugs administered, which can influence the potential for residues. This can occur either when the disease affects the metabolic system (and consequently drug metabolism), or when the presence of infection and/or inflammation causes the drug to accumulate in affected tissues. For example, cattle with acutely inflamed mastitis quarters, apramycin penetrates these areas of the body, and concentrations of the drug have been observed at ten times over the level recorded from cows without mastitis [33].
Extra-label drug use: Extra-label drug use (ELDU) refers to the use of an approved drug in a manner that is not in accordance with the approved label directions. It occurs when a drug only approved for human use is used in animals, when a drug approved for one species of animal is used in another, when a drug is used to treat a condition for which it was not approved, or the use of drugs at levels in excess of recommended dosages. For instances, the use of enrofloxacin solution as a topical ear medication (Only approved for use as an injection) are the common ELDU in veterinary medicine [34].
Improper Withdrawal Time: Improper withdrawal time is another risk factor; the withdrawal time is the time required for the residue of toxicological concern to reach safe concentration as defined by tolerance. Based on the drug product, dosage form, and route of administration it may vary from few hours to days or weeks. It is the interval from the time an animal is removed from medication until permitted time of slaughter for the production of safe foodstuffs.
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Safety Evaluation and Detection Methods of Drug Residues
Safety Evaluation
Acceptable daily intake: Acceptable daily intake (ADI) is the amount of substance that can be ingested daily over a lifetime without appreciable health risk. The evaluation of the safety of residues is based on the determination of the ADI on which in turn maximum residues limits (MRL) is based. The ADI is determined by consecutive estimate of a safe ingestion level by the human population on the lowest no effect level of toxicological safety studies [35]. If the drug is not a carcinogen, the no observed effect level (NOEL) of the most sensitive effect in the most sensitive species divided by a safety factor is used to determine an ADI for drug residues. The FDA will calculate the safe concentration for each edible tissue using the ADI, the weight in kg of an average adult (60 kg), and the amount of the product eaten per day in grams as follows; Safe concentration = [ADI (μg/kg/day) x 60 kg] /[Grams consumed/ day].
Maximum residue level: A tolerance level (or maximum residue levels, MRLs) is the maximum allowable level or concentration of a chemical in feed or food at a specified time of slaughter or harvesting, processing, storage and marketing up to the time of consumption by animal or human [36]. The MRL in various foodstuffs (muscle, liver, kidney, fat, milk and eggs) is determined to minimize the risk of consumer exposure, considering dietary intake. Such considerations as food technology, good farming practices and the use of veterinary medicinal products may also be considered when setting the MRL [37].
Calculating Withdrawal Period: The withdrawal period is determined when the tolerance limit on the residue concentration is at or below the permissible concentration. Withdrawal times are determined in edible, target tissues. Most commonly, they are liver or kidneys as they are primary organs of elimination and typically display a residue for the longest time. During withdrawal studies, the target organ is determined and animals are sampled at various times after drug administration is stopped. For those drugs for which only a kidney or liver tolerances has been established, if a violative residue is found in the target organ, the whole carcass would need to be discarded. On the other hand, for the drugs for which a muscle tolerance has been established, even if a violative residue is found in the kidney or liver a violative residue is not found in the muscle, the carcass would not need to be discarded [38].
Detection Methods
Screening Test: Screening of food products from animal origin for the presence of antimicrobial residues started soon after the introduction of antibacterial therapy in veterinary medicine. Initially it mainly concerned process monitoring in the dairy industry to prevent problems in fermentative dairy production, but from the early 1970s regulatory residue screening in slaughter animals also became more commonly introduced. An efficient screening method needs to be low-cost and high-throughput, able to effectively identify potential noncompliant samples from a large set of negative samples [39].
Advantage of these methods is that they have a wide detection spectrum; they are simple to carry out and cheap; and can be used for the screening of a large number of samples; [40] Possibility of automatization; Reduced time to obtain the result; Good sensitivity and specificity and Detection capability with an error probability (b) < 5% [41]. This method includes a large variety of detection methods, ranging from physico-chemical analysis or immunological detection to microbiological method [42].
Immunological Detection
The immunological methods are based on the interaction of antigen-antibody which is very specific for a particular residue. The most usual technique consists in the enzyme linkedimmunosorbent assay (ELISA) and the detection system is usually based on enzyme-labeled reagents. There are different formats for antigen quantification like the double antibody or sandwich ELISA tests and direct competitive ELISA tests [43]. ELISA kits are allowing the analysis of a large number of samples per kit, do not require sophisticated instrumentation, the results are available in a few hours and are quite specific and sensitive. It has good performance for the analysis of antibiotic residues in meat like tylosin and tetracycline [44], chloramphenicol [45], nitroimidazoles [46] and sulphonamides [47] and also for sedatives [48].
Microbiological Detection
Microbial inhibitions assays are very cost-effective and they have the potential to cover the entire antibiotic spectrum within one test. There are two main test formats: the tube test and the (multi-) plate test. A tube (or vial, or ampoule) test consists of a growth medium inoculated with (spores of) a sensitive test bacterium, supplemented with a pH or redox indicator. At the appropriate temperature, the bacteria start to grow and produce acid, which will cause a color change. The presence of antimicrobial residues will prevent or delay bacterial growth, and thus is indicated by the absence or delay of the color change. This format is commonly applied in routine screening of milk, but it is also increasingly used for analysis of other matrices [49]. A plate test consists of a layer of inoculated nutrient agar, with samples applied on top of the layer, or in wells in the agar. Bacterial growth will turn the agar into an opaque layer, which yields a clear growth-inhibited area around the sample if it contains antimicrobial substances
Biosensors
Different types of biosensors have been developed in recent years as an alternative approach to screen veterinary drugs in meat. In general, these sensors usually contain an antibody as a recognition element that interacts with the analyte. The resulting biochemical signal is measured optically or converted into an electronic signal that is further processed in appropriate equipments [50]. Biosensors can be able to detect simultaneously multiple veterinary drugs residues in a sample at a time [51]. In general, these sensors are valid for control laboratories because they can detect multiple residues in one sample and can thus allow the analysis of a large number of residues and samples [52].
Identification and Confirmation
The next step after initial screening consists in the unambiguous identification and confirmation of the veterinary drug residues in foods of animal origin. The full procedure and the methodologies for confirmatory analysis are costly in time, equipment’s and chemicals. In addition, they require trained personnel with high expertise [53]. Different analytical techniques are available for such purpose. When the target analyte is clearly identified and quantified above the decision limit for a forbidden substance or exceeding the maximum residue limit (MRL) in the case of substances having a MRL, the sample is considered as noncompliant (unfit for human consumption). Identification is easier for a limited number of target analytes and matrices of constant composition [54]. Some examples of the available confirmatory methodologies are as follows: The use of HPLC-electrospray ionization (ESI) tandem mass spectrometry [55] or liquid chromatography-mass spectrometry with atmospheric pressure chemical ionisation (APCI) [56].
ESI technique facilitates the analysis of small to relatively large and hydrophobic to hydrophilic molecules and is thus very adequate for the analysis of veterinary drug residues [57] even though it is more sensible to matrix effects than APCI ionization [58]. ESI and APCI interfaces are the sources of choice to promote the ionization of antibiotics and both complement each other well with regards to polarity and molecular mass of analytes [59].
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Public Health Significance of Veterinary Drug Residues
Short Term and Direct Effect
Drugs used in food animals can affect the public health because of their secretion in edible animal tissues in trace amounts usually called residues. For example, oxytetracycline [60] and enrofloxacin residues [61] have been found above the maximum residual level in chicken tissues. Similarly, diclofenac residues were reported to be the cause of vulture population decline in Pakistan [62].
Allergic Reactions: Drug hypersensitivity is defined as an immune mediated response to a drug agent in a sensitized patient, and drug allergy is restricted to a reaction mediated by IgE. An allergic or hypersensitive effect following administration of a drug (i.e., drug allergy is quite similar to that typified by allergic response to protein, carbohydrate, and lipid macromolecules. Allergic reactions to drugs may include anaphylaxis, serum sickness, cutaneous reaction, a delayed hypersensitivity response to drugs appear to be more commonly associated with the antibiotics, especially of penicillin [63]. Certain macrolides may also in exceptional be responsible for liver injuries, caused by a specific allergic response to macrolide modified hepatic cells [64
Long term and Indirect Effect
Mutagenic Effects: The term mutagen is used to describe chemical or physical agents that can cause a mutation in a DNA molecule or damage the genetic component of a cell or organisms. Several chemicals, including alkalizing agents and analogous of DNA bases, have been shown to elicit mutagenic activity [65] that may have adversely affected human fertility [66]. Carcinogenic Effects: The term carcinogenic refers to any substance or an agent capable of altering the genetic makeup of an organism so that they multiply and become rancorous while carcinogen refers to any substance that promotes carcinogenesis, the formation of cancer or having carcinogenic activity. Carcinogenic residues functions by covalently binding intracellular components including DNA, RNA, proteins, glycogen, phospholipids and glutathione [67]. The ban of Diethylstilbestrol (DES), a hormone-like compound used for food producing animals, was as a result its strong carcinogenic effect. Teratogenic Effect: The teratogen applies to chemical agents that produce a toxic effect on embryo or fetus during a critical phase of gestation. Of the anthelmintic, benzimidazole is embryo toxic and teratogenic when given during early stage of pregnancy because of the anthelminthic activity of the drug [67]. Disruption of Normal Intestinal Flora: The normal Intestinal Flora is essential to human health. Not only does the symbiosis exist to contribute to nutrient absorption [68] it also obstructs and inhibits pathogen invasion, as well as aids in the development and optimal functioning of the host immune system [69]. The bacteria that usually live in the intestine act as a barrier to prevent incoming pathogenic bacteria from becoming established and causing disease [70] by producing antimicrobial substances (such as bacteriocins), altering luminal pH, and directly competing against pathogens for nutrients. In addition, commensal bacteria promote angiogenesis and the development of the intestinal epithelium [71]. Antibiotics might reduce total numbers of these bacteria or selectively kill some important species when consumed in food which contain their residues [70]. Development of Antimicrobial Resistance: Indiscriminate use of veterinary drugs, mainly antimicrobials, anthelmintics, and acaricides in food animals also play a major role in the development of antimicrobial resistance (AMR) which has put the public health at risk [72]. This problem is further worsened by irrational use through free access to prescription drugs and their administration at sub-therapeutic concentrations for a long period of time. Such conditions favor the selection and spread of antimicrobial resistant strains in animals, environment and humans [73]. The consequences of antimicrobial resistance in bacteria causing human infections include increased number of infections, frequency of treatment failures and severity of infection, and finally increased costs to society associated with disease. Increased severity of infection includes prolonged duration of illness and increased frequency of bloodstream infections, hospitalization, and mortality [74].
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
The Extent of Drug Residue in Ethiopia
Globally, more than half of all medicines are prescribed, dispensed or sold improperly. This is more wasteful, expensive and dangerous, both to the health of the individual patient and to the population as a whole that magnifies the problem of misuse of anthelmintic agents [75]. In many African countries, antibiotics may be used indiscriminately for the treatment of bacterial diseases or they may be used as feed additives for domestic animals and birds [76]. The ongoing threat of antibiotic contamination is one of the biggest challenges to public health that is faced by the human population worldwide [77]. Such residues are spreading rapidly, irrespective of geographical, economical, or legal differences between countries.
In Ethiopia, as the study conducted from March 2016 to June 2016 in University of Gondar veterinary clinic revealed, anthelmintic drugs are quite commonly but improperly utilized in the clinic. Three group of anthelmintics namely benzimidazoles (Albendazole, fenbendazole, mebendazole and triclabendazole), imidazothiazole (tetramisole and levamisole) and macrocyclic lactone (Ivermectin) were used. Utilization of limited group of drugs for a long period may favor the development of resistance which is risk factor for drug residues [78]. Though the primary purpose of veterinary drugs is to safeguard the health and welfare of animals [79], 44.3% anthelmintics were prescribed irrationally to treat diseases that were tentatively diagnosed as nonparasitic cases and 92.1% of anthelmintics were utilized to treat diseases that were tentatively diagnosed without getting correct laboratory supported diagnosis. This may be due to inadequate recognition of the disease, unavailability of diagnostic aids for confirmatory tests, and absence of a right drug and to make the treatment broader anthelmintics can be given in combination with other drugs [78].
There also other study conducted in this country in 2007 indicated that the proportion of tetracycline levels in beef; the study focused on the Addis Ababa, Debre Zeit and Nazareth slaughterhouses. Out of the total 384 samples analyzed for tetracycline residue 71.3% had detectable oxytetracycline levels. Among the meat samples collected from the Addis Ababa, Debre Zeit and Nazareth slaughterhouses, 93.8%, 37.5% and 82.1% tested positive for oxytetracycline respectively. Agricultural pesticides are important chemicals that are used to mitigate crop damage or loss and improve productivity. However, pesticides may cause negative environmental and human health effects depending on their specific distribution and use [80]. Its residue has become a major food safety hazard; synergy toxic made it a much higher risk. The toxicity of organic phosphorus, organochlorine, carbamate and other pesticides is mainly manifested as neurotoxicity [81].
Ethiopia is confronted with a number of problems associated with unsafe handling of pesticide distribution and use. Most pesticides used in Ethiopia are imported by international manufacturing companies represented by local agents [82]. Currently, pesticide use practices are changing as a result of the government plan to intensify and diversify agriculture by promoting high value export crops such as flowers and vegetables. For instance, more than 212 types of pesticides with different active ingredients are being used to cultivate roses in Ethiopia. But also, small holders growing vegetables are facing challenges because they are usually resource-poor but also risk averse and under these conditions it is challenging to decide when, how, how much and which pesticide to apply among the hundreds available on the market [80].
Herbicides are widely used in agricultural crops to control weed. Their introduction in the food chain via the environment can be considered a risk for human health due to the toxicity of the most of these compounds. In addition, herbicides are relatively long-lived in the environment, and can be accumulated by means of food chain amplification. Due to their extensive use in cultivation of crops (e.g. soybean, wheat, maize) and relatively stable nature in environments, the residues of herbicides were frequently detected in soil, cereal grain and water. To ensure human food safety, the United State (US), and the European Union (EU) have set maximum residue limits (MRLs) for some herbicide residues in soybean, corn and wheat in the range 0.01-2mg/kg, depending on the particular grain matrix and herbicide, but without the MRL for most herbicides [83].
Use of synthetic acaricides is the primary method of tick control. Synthetic insecticides particularly organophosphates, carbamates, pyrethroids and neonicotinoids have been extensively used by farmers for protecting medicinal and aromatic plants. Consequently, toxic residue of pesticides in raw material posed serious concerns of risk to human health. Therefore, an integrated management including cultural practices, plant-derived products and biological control has been experimented on limited scale [84].
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Management of Veterinary Drug Residue
Legislation and Regulations toward Drug Residue
The European Union has strictly regulated the use of veterinary drugs in food animal species. Some of these drugs can be permitted only in specific circumstances (therapeutic purposes) but under strict control and administration by a veterinarian [85]. The use of substances having hormonal or thyreostatic action as well as b-agonists is controlled by official inspection and analytical services following Commission Directive 96/23/EC on measures to monitor certain substances and residues in live animals and animal products. This Directive contributed to a sensible reduction in the number of growths promoting reported cases. However, laboratories in charge of residues control usually face a large number of samples with great varieties of residues to search in short periods of time making it rather difficult. The availability of simple and useful screening techniques is really necessary for an effective control [86].
Establishment of a legislative framework and of an institutional structure is the first step in the assessment and management of drug-related risk. From this point of view, according to pending European legislation the use of veterinary drugs must be based on risk evaluation. The risk due to the use of veterinary drugs is “any risk for animal or public health relating to the quality, safety and efficacy of the veterinary medicinal product and any risk of undesirable effect on the environment”. Risk management is a task of both private and public veterinary services that are involved in the prevention and control of all hazards arising from the use of veterinary drugs. A major tool for veterinarians to prevent and control drug-borne risk is “pharmacovigilance” [87]. Pharmacovigilance is the post-marketing surveillance of veterinary drug and vaccine safety used for prevention, diagnosis and therapy and consists of the report of any adverse effects of a drug by veterinarians, pharmacists, farmers and other health care professionals, in the improvement of knowledge about the pharmacological action of a drug and hence, in the evaluation of the risk/benefit balance of a drug [88].
The main tasks of pharmacovigilance can be summarized as follows: a. Control of clinical safety of veterinary medicinal products; b. Control of potential reaction in man linked to user safety; c. Evaluation of decreased efficacy or lack of expected activity of a veterinary medicinal product; d. Control of maximum residue levels (MRL) of veterinary drugs in food products of animal origin; e. Assessment of risks for the environment related to the use of veterinary drugs; f. Control of the development of drug resistance, with particular concern to antibiotic resistance [89].
Control and Preventive Measures
The control of parasitic helminths in domestic animals relies largely on the use of anthelmintic drugs. But inappropriate and indiscriminate use of anthelmintic leads to the emergence of anthelmintic resistance, treatment failure and increase in mortality and morbidity [90]. Most failures during anthelmintic therapy may occur when the parasite is unknown and anthelmintic drugs are administered empirically. To avoid these problems, it is important to apply confirmatory diagnosis and selection of the right anthelmintic [91]. Maximum Residue Limits (MRLs) in certain products of animal origin, including meat and milk have been established by the European Union. The need for more intensive residue controls becomes stronger considering several studies which indicate that benzimidazoles are not degraded after microwave and oven-baking, storage at -18 ℃ for three to eight months and after cooking. However, no major losses for residues of ABZ, MBZ or FBZ, after roasting of meat and liver (40min at 190 ℃) or shallow frying (muscle 8-12 min, liver 14-19min) in a domestic kitchen [91]. Consequently, conventional cooking hardly protects consumers against the ingestion of residues of anthelmintic veterinary drugs in these foods. Accordingly, the estimation of residues intake through certain food items consumption becomes a necessity to ensure that the Acceptable Daily Intakes (ADIs) of the drugs are not exceeded [92-95].
Irrational use of drugs in veterinary medicine as well as the need for control of their use becomes even bigger problem when used on food producing animals. In this case, there is the possibility that minimal quantities of drugs and their metabolites (residues) which remain in edible tissues or in animal products (meat, milk, eggs, honey) induce certain harmful effects in humans as potential consumers of such food [92]. When drugs are used to improve the productivity of food animals that are intended for human consumption, then there is possibility for producing adverse effects on humans. To prevent this risk, it is necessary to use drugs rationally, i.e., to use them only when they are really indicated, in the right way, at the right time, in the right dose and respecting withdrawal period. The residue control strategy is based on a twostep approach: (1) the detection of residues using sensitive tests with a low rate of false negatives; (2) followed by confirmation, requiring quantification against the MRL and identification with a low rate of false positives. Hence, the residue prevention strategy is based on preventing entry of violative residues in food of animal origin intended for human consumption by proper drug use guide developed for use by both veterinarians and food animal producers include the following: a. Herd health management; Drug residues are best avoided by implementing management practice and herd health program that keep animals healthy and producing efficiently. b. Use of approved drugs. c. Establishment of valid veterinarian-client-patient relationship; the use of prescription drug and the ELU necessitate a veterinary-client patient relationship. d. Proper drug administration and identification of treated animals; before administering or dispensing drugs one has to know the drugs approved for all classes of cattle on the farm and be familiar with approved dosage, route of administration, and withholding time. e. Proper maintenance of treatment records and identification of treated animals. f. Creating awareness of proper drug use, and methods to avoid marketing adulterated products principally educational, total residue avoidance program is based upon the objective of improving the livestock producer’s management and quality control of marketing animals with emphasis on avoidance of drug residues.
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References
Conclusion and Recommendation
Although the veterinary drugs have played a great role in control and prevention of disease in animals and promote the growth of food animals, its use is associated with problems such as development of resistance and residue effects in food animals. These adverse effects are generally due to irrational use of drugs such as misuse, extensive use, failure to keep strict adherence of withdrawal and withholding time of drugs. The development of resistant microorganisms in animals and the presence of drug residue in food of animal origin have significant effect on public health. Globally, more than half of all medicines are prescribed, dispensed or sold improperly. Many livestock producers treat their animals by themselves. The uncontrolled use of anti-infectious agents can lead to residues in animal products. The risks of residues in foodstuffs of animal origin could be reflected into several forms of adverse effects. The great majority of residues found in edible tissues of animals originated in farms but, some cases may proceed from contaminated animal feedstuffs. By far the most common cause of residues is the failure to observe the proper withholding period following treatment. In general, when various types of veterinary drugs; antimicrobials, antiparasitic and β-agonists, food additives, Industrial and agricultural products; pesticide, acaricide, herbicides etc. are used in food producing animals intended for human consumption and in the environment indiscriminately and they pose a great public health effect. Therefore, strict control measures to promote rational veterinary drug use have crucial importance on global economy and public health. Based on above conclusions the following recommendations are forwarded: a. The government should regulate irrational and unauthorized use of drugs and, implement residue control strategy such as management practice and herd health program that keep animals healthy and producing efficiently to avoid drug residues, b. Improperly prescribed, dispensed and sold drug should be regulated, c. Proper maintenance of treatment records and identification of treated animals should be implemented, d. The withdrawal time should be appropriately protected, e. Creating awareness of farmers, consumers and health professionals about drug residues and its public health significance.
- Review Article
- Abstract
- Introduction
- Historical Background
- Veterinary Drug and their Use in Food Animals
- Risk of Drug Residue for the Public Health
- Safety Evaluation and Detection Methods of Drug Residues
- Public Health Significance of Veterinary Drug Residues
- The Extent of Drug Residue in Ethiopia
- Management of Veterinary Drug Residue
- Conclusion and Recommendation
- References






























