Serological Evidence of PPR Virus in Chadian Camels
Bidjeh Kebkiba1*, Abakar Oumar2, Ouagal Mahamat1, Tchari Doungous1 and Assane Kembé 1
1Livestock Research Institute for Development (LRID), Chad
2Faculty of Exact and Applied Sciences, University of N’Djamena, Chad
Submission: March 31, 2019; Published: April 17, 2019
*Corresponding author: Bidjeh Kebkiba, Livestock Research Institute for Development (LRID), Farcha street PO Box. 433, N’Djamena- Chad, Chad
How to cite this article: Bidjeh K, Abakar O, Ouagal M, Tchari D, Assane K. Serological Evidence of PPR Virus in Chadian Camels. Dairy and Vet Sci J.2019; 11(2): 555809. DOI: 10.19080/JDVS.2019.11.555809
Abstract
Peste des petits ruminants are an acute fever disease of goats and sheep. It is characterized in these animals by mucopurulent nasal and ocular discharge, erosive and necrotic stomatitis, enteritis and pneumonia. It is widespread, virulent and devastating, with significant impacts on the economy, food security and livelihoods of marginalized pastoralists. This study provides information on the serological prevalence of PPR virus in 510 camel. Most of the samples for this study were obtained at the N’Djamena abattoir. Sera used to detect PPR virus antibodies were not necessarily representative of the target population, although they came from a larger number of provinces. The test used for the analysis of these sera is the competition ELISA (c-ELISA) developed by CIRAD and marketed by IDVET. Examination of the 510 sera collected from camels revealed that the serological prevalence of PPR virus was 25.88%. This seroprevalence of PPR virus varied by province, age group, and geographic area. The study showed a much higher seroprevalence in animals ranging in age from 6 to 10 years than in animals of other age groups. In view of these results, an additional study is to be undertaken on the epidemiological surveillance in the camel population throughout the national territory to detect clinical cases of this disease in camels. In addition, it would be desirable to carry out a serological survey of this disease nationally on random representative sampling in order to find out the true seroprevalence rate of peste des petits ruminants in Chadian dromedaries.
Keywords: Serology; c-ELISA; Antibodies; Camels; Viruses; Peste des petits ruminants; Chad
Introduction
The Republic of Chad is a Sahelian country with a pastoral vocation, it bases its economy largely on the agricultural sector (40% of the national GDP). Chadian livestock contributes 37% of GDP to the agricultural sector. Apart from the oil sector, it is the largest export contributor, ranging from 30% to 50%. It supports 40% of the rural population and employs 80% of the working population, most of whom are women [1]. According to the last general census of livestock [2], Republic of Chad has about 94 million head of cattle all species including 6.4 million (6.8%) camelins. The Republic of Chad is one of the Sahelian countries where there is a significant increase in the camel population [3]. These species, very resistant to drought, once concentrated in the Saharan and Sahelian zone of the country. But in recent years, with climate change, they are even found in the Sudanian zone especially during the dry season, in search of pasture and water.
In Chad, the rearing system is 83.33% transhumant and associative (camels, cattle, sheep and goats). However, certain constraints limit the development of this sector. These include transboundary diseases such as PPR. In fact, PPR is an infectious and contagious disease of domestic or wild small ruminants. It affects domestic or wild small ruminants and camels. Cattle and pigs form a subclinical form of the disease but do not excrete the virus [4]. Buffaloes and pigs may be infected with the virus, but do not show clinical signs [5-7]. PPR is characterized in small ruminants by lacrimation, serous and mucopurulent discharge, oral lesions, diarrhea, pneumonia and often death. In camels, these signs are not clinically visible. This may be why surveillance agents do not report suspicions of the disease in these animals.
According to Roger, Khalafalla [8-10], in camels, PPR is clinically characterized by the sudden death of apparently healthy animals, bloody and yellowish diarrhea, abortion in pregnant females. Decrease in milk production, weight loss and decrease in water consumption sometimes up to 10-14 days. The morbidity rate is 90% and the mortality varies between 0 and 50% depending on the environment. The receptivity of camels to the PPR virus has been demonstrated in some African countries such as: Nigeria [11-13], Sudan [14], Libya [15] and Tanzania [16]. In Africa, the disease is recorded in several countries and is an impediment to the development of small ruminant livestock [17]. In Chad, the existence of PPR in small ruminants has been confirmed by serological surveys [18,19] and subsequent isolation of the pathogen [20]. However, in camels no information is available so far. The objective of this study is therefore to detect the antibodies directed against the PPR virus in camels. Due to the lack of financial means, the study focused on camels intended for the slaughterhouse of N’Djamena.
Material and Methods
Animals
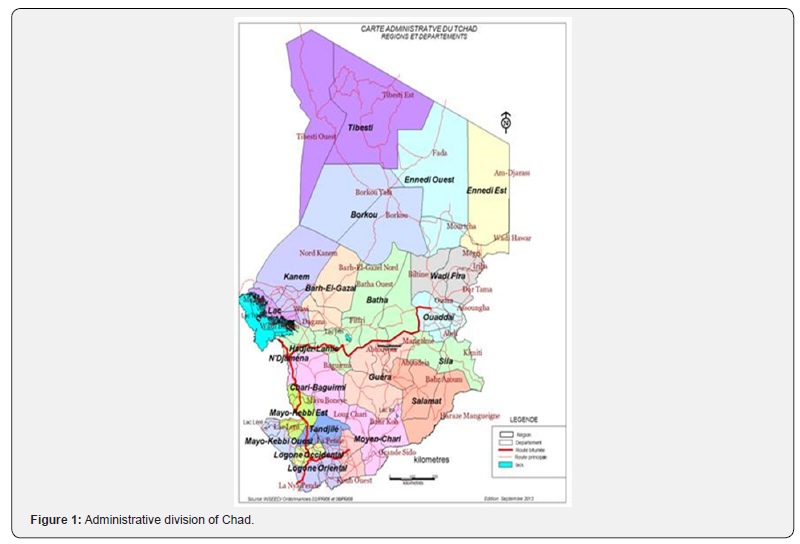
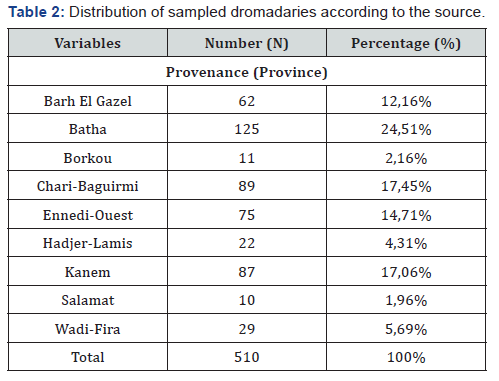
A total of 510 camels of which 313 females (61.37%) and 197 males (38.62%) were sampled. Beforehand, a survey of breeders and traders made it possible to know the origin of the dromedaries. According to the results of this survey, the animals come from Barh El Gazel, Batha, Borkou, Chari-Baguirmi, Ennedi- West, Hadjer-Lamis, Kanem, Salamat and Wadi-Fira (Figure 1). The animals sold in the markets of N’Djamena and brought to the slaughterhouse come mostly from the provinces of Batha and Chari Baguirmi.
Serums
The samples were taken at the abattoir of Farcha for 2 months (from September 20th to November 20th). The blood is taken from live dromedaries in the jugular vein in venoject dry tubes using a needle mounted on a tube holder. The sampling was done very early in the morning in the slaughterhouse car park before the animals passed on the slaughter line. The tubes filled with blood are then left in the shade for 1h 30 min for the blood to clot and then transported to the laboratory for the collection of sera. After extraction of the clot, the contents of the tubes are centrifuged at 3000rpm for 5min; the supernatant which constitutes the serum is then transferred into cryotubes and stored at -20 °C. for later use. A total of 406 camel sera were collected at the Farcha cold-storage facility (N’Djamena) and 104 sera collected from camels in the Ennedi West and Wadi-Fira Provinces, giving a total of 510 sera. The information relating to the animal taken is marked on the tubes using an indelible marker and those relating to the animals are also noted on the sheets specially prepared for this purpose.
Serum analysis in the laboratory
The sera collected are analyzed in the IRED virology laboratory by the competitive ELISA test (c-ELISA) for the detection of antibodies against the PPR virus. The said test was developed by CIRAD / EMVT [21] but marketed by IDvet IDScreen® PPR Competition provided free of charge by the International Atomic Energy Agency (IAEA) as part of a TCPProject # CHD2015005. The protocol used is that recommended by the manufacturer whose description is as follows:
Description and principle of the c-ELISA test
The plates were sensitized with the purified recombinant nucleoprotein (NP) of the PPR virus. The samples to be tested and the control sera are diluted to 1/2 and then distributed in the pre-sensitized cups. The anti-NP PPR antibodies, if present in the sera, form an antibody-antigen complex. An anti-NP peroxidase labeled PPR conjugate (po) is then dispensed into the wells. The latter binds to the antigen remained free thereby forming an antigen-antibody-conjugate-po complex. After removing the excess of the conjugate by washing, the reaction is revealed by a substrate solution (TMB). The resulting staining is related to the number of specific antibodies present in the samples to be tested.
a. In the absence of specific antibodies in the samples, a blue color appears which then becomes yellow after addition of the stop solution.
b. In the presence of specific antibodies in the samples, no staining appears. The reading is carried out using a PR4100 Bio RAD ELISA reader at a wavelength of 450nm. The reader is connected to a computer and a printer.
Test Procedure
Before use, all reagents are brought to a temperature of 21 °C (± 5 °C) and then homogenized using the vortex. The different steps of the procedure are as follows:
I. Distribute
a. 25μl of the Dilution Buffer 13 in each well;
b. 50μl Positive control serum in wells A1 and B1;
c. 50μl of diluent 13 used as Negative Control in wells C1 and D1;
d. 50μl of each sample to be tested in the remaining cups.
II. Incubate overnight at 21 °C (± 5 °C)
III. Empty the wells and wash them 3 times with 300μl of the Wash Solution. At this stage, it is necessary to avoid the drying of the cups between the washing phases.
IV. Prepare the 1X Conjugate by diluting the 10X Conjugate 1 / 10th in the Dilution Buffer.
V. Empty the wells and wash them 3 times with 300μl of the Wash Solution. Avoid drying the cups between the washings.
VI. Add 100μl of the Developer Solution to each well.
VII. Incubate at 21 °C (± 5 °C) for 15 min (± 2) min. Protected from light (dark).
VIII. Dispense 100μl Stop Solution into each well to stop the reaction.
IX. Read the 450nm wavelength response from the ELISA reader and record the optical densities (OD) on the computer connected to the reader
Validation of the Test
The test is validated if:
a. The average value of the negative control optical densities (DOCN) is greater than 0.700. (DOCN> 0.700);
b. The mean value of the optical densities of the positive controls (DOCP) is less than 30% of the DOCN. (DOCP / DOCN <0.3).
Interpretation
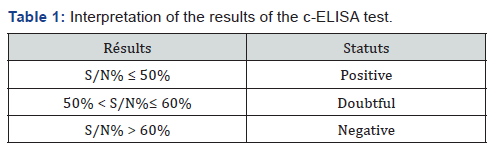
For the calculation of the percentage of the competition (S / N%), the average of the OD obtained in the wells containing diluent 13 is used as a negative control: S / N% = sample / DOCN x 100. Samples with S / N% less than or equal to 50% are considered positive; greater than 50% and less than or equal to 60% are considered doubtful and more than 60% are considered negative (Tables 1-3).
Statistical Analysis
All collected data and serum analysis results were entered into a relational database designed on Access. The Excel 2010, R and Quantum GIS software’s, my practical online mediatory, were used for the statistical analysis of the data.
Results
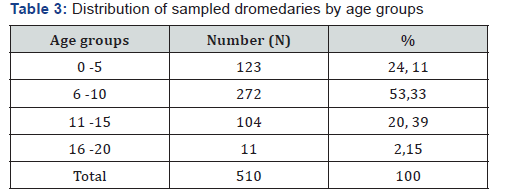
The sample size is 510 camels of which 313 females are 61.37% and 197 males are 38.63% (Table 4). The animals sold in the markets of N’Djamena and brought to the slaughterhouse come for the most part from the provinces of Batha and Chari Baguirmi 24.51% and 17, 45% respectively. Of the dromedaries sampled, 272 (53.33%) are aged between 6 and 10 years; 123 (24.12%) aged 0-5 years; 104 (20.39%) aged 11 and 15 and 11 (2.16%) aged between 16 and 20 years (Table 3). Regarding the serological results, out of the 510 sera tested, 132 or 25, 88% were positive vis-à-vis the PPR virus and 378 (74.12%) negative (Figure 2).
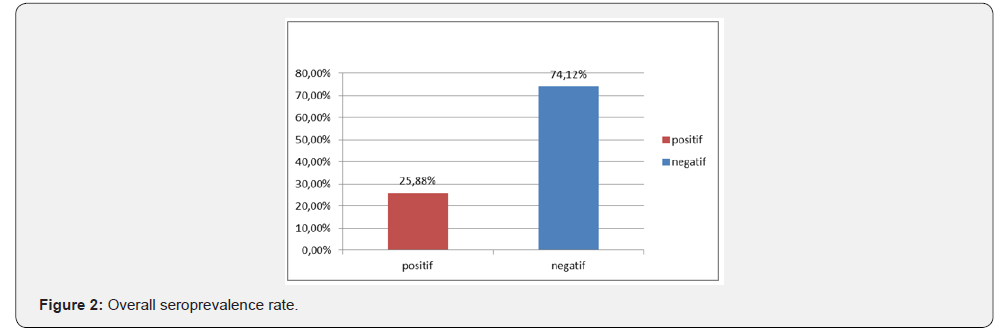
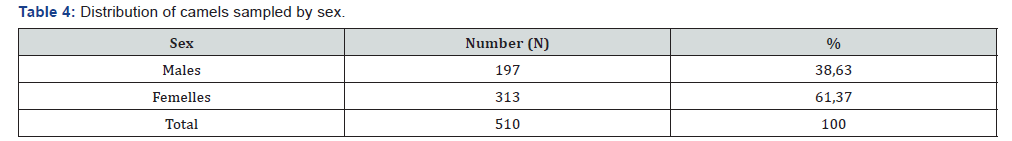
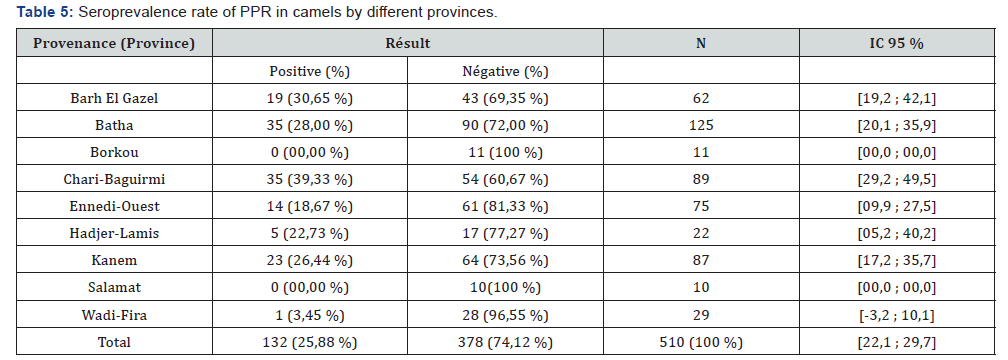
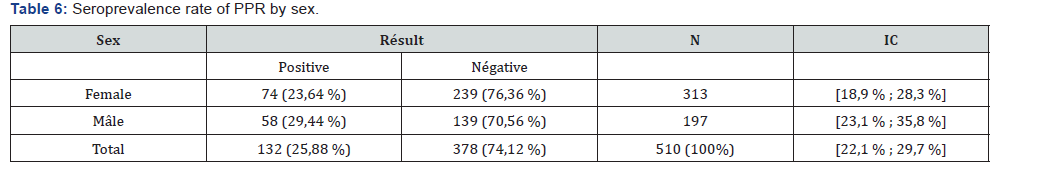
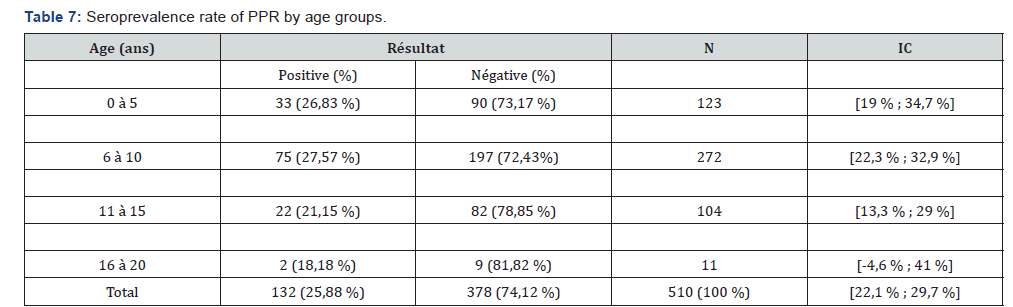
Table 4 presents the seroprevalence rate of PPR in the different provinces. Of the 9 provinces surveyed, seven (7) are affected by PPR against two (2) provinces with a seroprevalence rate of 0%. The province where the seroprevalence rate is higher is Chari-Baguirmi (39.33%), followed by Barh El Gazel Province (30.65%). On the other hand, the lowest seroprevalence rate is observed in the provinces of Borkou, Salamat (00.00%) and Wadi-Fira (3.45%). Table 5 shows the seroprevalence rate by sex. From this table it can be noted that the seroprevalence rate is relatively higher in males (29.44%) than in females (23.64%). Table 6 shows seroprevalence rate in camels as a function of age. From this table it can be noted that the highest seroprevalence rate (27.57%) is obtained in camels whose age is between 6 and 10 years. In animals aged between 0 and 5 years, the seroprevalence rate is 26.83%. The lowest seroprevalence rate is recorded in animals aged between 16 and 20 years (18.18%) (Table 7).
Discussion
PPR is one of the most serious viral infections affecting domestic and wild small ruminants, as well as camels, with increasing incidence and geographical distribution. Despite a very important socio-economic impact, this disease has received little public attention since its discovery, and this has to a large extent led to its wide dissemination. Since 1992, camels are suspected to be possible hosts of the PPR virus. Serological surveys conducted in different countries, Sudan, Egypt, Libya revealed their seropositivity but without clinical manifestation of the disease and isolation of the virus. The seroprevalence rate of 25.88% obtained in this study confirms the circulation of the PPR virus in the Chadian camel population. The study showed a higher prevalence rate in males than in females. These results are different from those obtained by Swai [16] in Tanzania, showing a seroprevalence rate of 2.7% in females and 2.0% in males, but are similar to those obtained by Woma [13] in Nigeria.
Although the seroprevalence of PPR is known in small ruminants in Chad [18-19,22-23], this study showed for the first time a Seroprevalence of PPR in Chadian dromedaries. The anti-PPR antibodies observed in this study could only come from a natural infection, as there is no documented evidence that camels were vaccinated against PPR Virus in Chad. Camels are believed to be responsible for the spread of the PPR virus. The serological results obtained in this study are superior to those obtained by Ismail [24] in Egypt (4.2%), Daneji [11] in Nigeria (4%), Roger [9] in Ethiopia (7.8%) and Swai [16] in Tanzania (2.6%). Chari-Baguirmi is the province with the highest seroprevalence rate (39.33%). This Province is a meeting point for transhumant animals. It should also be noted that this region is the area where there has been more suspicion of PPR in small ruminants [22]. The low rates of seroprevalence obtained in the Provinces of Wadi-Fira, Borkou and Salamat can be explained by the low number of serums taken since few camels sold on the markets of N’Djamena are those coming from these Provinces. The study showed a slightly higher prevalence rate in males than in females. These results are different from those obtained by Swai [16] in Tanzania, showing a seroprevalence rate of 2.7% in males and 2.0% in females, but are similar to those obtained by Woma [13] in Nigeria.
The seroprevalence rate is higher in animals between 6 and 10 years of age than in other age groups. A low rate of seroprevalence is observed in camels whose age varies between 16 and 20 years. These results are similar to those obtained in Nigeria [13] and in Tanzania where the seroprevalence rate among camels aged between 5 and 10 is high compared to other age groups [16].
Conclusion
Although clinical cases of PPR were not observed in the field, the results of this study showed a PPR seroprevalence rate of 25.88% in Chadian dromedaries, which confirms the receptivity of these animals to PPR virus. In view of the results obtained, further research should be considered in order to clarify the role of dromedaries in the epidemiology of the PPR virus in Chad. However, special attention should be paid to this species in the strategy of control and eradication of peste des petits ruminants. Also, an additional study is to be undertaken on the epidemiological surveillance in the camel population throughout the national territory to detect clinical cases of this disease in camels. In addition, it would be desirable to carry out a serological survey of this disease nationally on random representative sampling in order to find out the true seroprevalence rate of peste des petits ruminants in Chadian dromedaries. In order to demonstrate the real role of camels in the spread of the PPR virus, an experimental reproduction study of this disease in small ruminants and dromedaries in the resort is necessary.
Acknowledgements
The authors sincerely thank the International Atomic Energy Agency (IAEA) for providing the ELISA-PPR Kit free of charge. Our thanks also go to all those who, from near or far, contributed to the realization of this study.
Conflict of Interest
There is no conflict of interest






























