Bacteriological and Histopathological Investigation of Pneumonia in Black Bengal Goat
Sukanta Kumar Sen1,3, Mohammed Rashed Chowdhury2*, ATM Mahbub-E-Elahi1 and Abu Bakr Siddique1
1Department of Microbiology and Immunology, Faculty of Veterinary & Animal Science, Bangladesh
2Department of Biochemistry & Chemistry, Faculty of Biotechnology & Genetic Engineering, Bangladesh
3Department of Livestock Services (DLS), Ministry of Fisheries & Livestock, Bangladesh
Submission: May 13, 2018; Published: July 16, 2018
*Corresponding author: Mohammed Rashed Chowdhury, Department of Biochemistry & Chemistry, Faculty of Biotechnology & Genetic Engineering, Sylhet Agricultural University, Sylhet-3100, Bangladesh, Email: mrchowdhury@sau.ac.bd
How to cite this article: Sukanta K S, Mohammed R C, ATM Mahbub E E, Abu B S. Bacteriological and Histopathological Investigation of Pneumonia in Black Bengal Goat. Dairy and Vet Sci J. 2018; 6(4): 555695. DOI: 10.19080/JDVS.2018.06.555695
Abstract
The study was designed to evaluate the isolation and identification of causative bacteria from caprine pneumonic lungs, antibiotics sensitivity test with isolated bacteria, gross and histopathological changes to determine the types of pneumonia. A total of 210 Black Bengal goat carcasses were subjected to gross examination and infected with pneumonia were collected for bacteriological and histopathologica study. Pneumonia was detected in 40 carcasses. Bacteriological examinations revealed the presence of ovine pathogens, such as Staphylococcus spp. (35%), Escherichia coli (25%), Pasteurella spp. (20%), Streptococcus spp. (15%), and Klebsiella spp. (5%). The antibiogram study indicated that the most effective antimicrobial agents against all the tested isolates were ciprofloxacin; ceftriaxone; and oxytetracycline; could be the choice of antibiotics for the treatment of pneumonia in Black Bengal goats. The gross lesions were recorded as hemorrhage (25%), congestion (20%), hemorrhage and congestion (30%), emphysema (15%), and hepatization (10%) of lungs. Based on the histopathological study, 30% of affected carcasses were bronchopneumonia, 25% with fibrinous bronchopneumonia, 20% with hemorrhagic pneumonia, 15% with interstitial pneumonia, and 10% with purulent bronchopneumonia. Both gross and histopathological lesions of above diseases were enunciated. However, other etiology may also have a considerable effect on the occurrence of pneumonia in Black Bengal goat of Bangladesh.
Keywords:Antibiogram; Black Bengal goat; Histopathology; Isolates; pneumonia; Livestock; Etiology; Mortality; Morbidity; Shipping fever; Ciprofloxacin; Ceftriaxone; Oxytetracycline; Bronchopneumonia; Capsules; Bacteria; Virus; Parasites; Fungi
Introduction
Black Bengal goats called as a “poor man’s cow” is the second important livestock in Bangladesh. Goat rearing is easy, less expensive, less laborious and highly profitable business [1], is seriously hampered due to various diseases. The diseases affecting the respiratory tract: Pesti Des Petits Ruminants (PPR), Contagious Caprine Pleuropneumonia (CCPP) and Pasteurellosis impose a substantial loss through high morbidity and mortality [2,3]. Furthermore, most of the infectious agents that because the respiratory disease is ubiquitous in nature and are normal inhabitants of the nasopharynx [2]. Unfortunately, a large number of goat and lamb population die due to pneumonia at the early stage of their lives including reduced growth rate, carcass condemnation and consequent substantial economic impact on animal husbandry because of the need of medicine and vaccination programs [3,4]. Pneumonia sometimes called goat “shipping fever” is one of the most common problem encountered today associated with expensive treatments. Therefore, early diagnosis and proper treatment are necessary for effective control of the diseases [5].
Pneumonia caused by the interaction of several pathogens (bacteria, virus, parasites, fungi), host defence, environmental factors [6], and stress [7,8]. Though viral, fungal and other causal agents are responsible for the goat pneumonia, the present study conducted to investigate pneumonia caused by bacterial pathogens. Some bacteria as like Pasteurella spp. and Staphylococcus spp. normally found in the respiratory tract of healthy goat, however, they sometimes cause pneumonia during the stress conditions or in association with other pathogens. Klebsiella spp. causing pneumonia, while it produces virulence factors such as smooth lipopolysaccharide, pili for adhesion to host cells, capsules (K antigen) that are antiphagocytic aid the bacterium in its competition with the host for iron uptake [9,10]. The respiratory problem in kids less than one year thought to be an association with Pasteurella spp. and Parainfluenza-3 [11]. For the prevention and control of goat pneumonia, isolation and characterization of causal agents and antibiogram study of isolates are essential. Thus, a present study was undertaken to isolate the pathogen responsible for pneumonia in Black Bengal goat with their antibiogram and histopathological study.
Materials and Methods
Sample Collection and Processing
A total of 210 lung samples was obtained from different slaughterhouses of Bangladesh subjected to gross examination for the presence of pneumonic lesions. No details of sex breed or husbandry conditions of goat were available. Lungs with microscopic pneumonic lesions were attained by excluding parasitic lesions. Following the gross inspection, all the samples of apparently affect lungs transferred to the Microbiology and Pathology laboratory for bacteriological and histopathological examination. Samples of affected areas aseptically collected and placed in sterile plates. The outer surface of the pneumonic lungs first seared with a heated spatula before cutting the inner surface of lungs. The lung swabs aseptically collected from the inner core of lungs by cotton bud and immediately placed into the Falcon tube containing 10ml nutrient broth for isolation of causative bacteria.
Isolation and Identification of Causative Bacteria
Primary culture was performed in both nutrient agar and broth media. For sub-culturing, suspected bacteria were inoculated separately into different bacteriological agar media under the aseptic condition and subsequently incubated at 370C for 24 hours. Pure culture was made as per the procedures described by Cheesbrough, Buxton & Fraser [12,13]. In order to identify the isolated bacteria, cultural, morphological, and biochemical characteristics were performed. The cultural characteristics or colonial morphology of the bacteria grown on the nutrient and blood agar media was recorded. Gram staining method was performed to study the morphology and staining characteristics of bacteria according to the technique described by Merchant and Packer. Motility test was executed to differentiate motile bacteria from the non-motile one. Hemolytic activity of the bacteria has done according to the procedure mentioned by Carter [14]. Biochemical tests, such as- sugar fermentation, catalase, methyl red, indole production, the Voges- Proskauer, citrate utilization, TSI (Triple sugar iron) agar slant reaction, lysine iron agar, oxidation fermentation was performed according to the standard methods [13]. The classification and specification of the organisms were done by following the scheme presented in Animal Microbiology [12]. The stock culture was maintained based on the procedures of Carter et al. [15].
Antimicrobial Sensitivity Test
A disc diffusion method [16,17] was used to determine the susceptibility of the bacterial isolates against antibiotic agents. For this, commercially available 11 different antibacterial discs (Oxoid Ltd., Baring-stoke Hampshire, England) were used. The names and concentrations of the antibacterial discs were mentioned in Table 3. The interpretation on antibiotic susceptibility was recorded according to the guidelines of Clinical and Laboratory Standard Institute.
Pathological study of pneumonic lungs
All the positive (infected) lung samples carefully examined for gross abnormalities. The gross tissue lesions were observed and recorded carefully, and representative part of tissue samples was fixed in Bouin’s fluid for further histopathological studies. Afterward, the preserved samples were dehydrated in alcohol, cleared in xylene, impregnated and embedded in paraffin wax, sectioned at seven (7)μm and finally stained with hematoxylin and eosin (H&E) for histopathological examination as described by Luna [18].
Milk Fat Depression
Isolation and Identification of Causative Bacteria
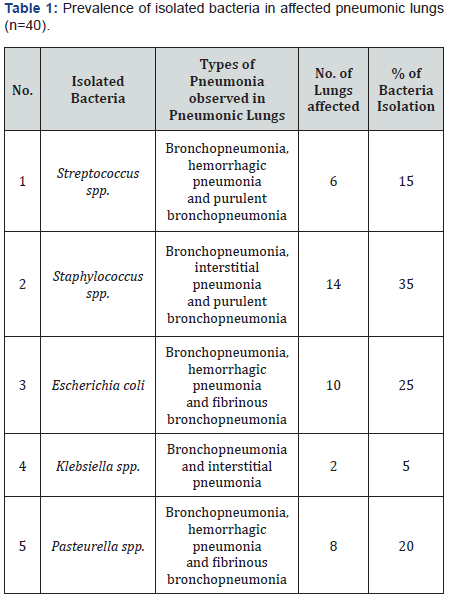
The prevalence of Klebsiella spp. 5%, Streptococcus spp. 15%, Pasteurella spp. 20%, Escherichia coli 25%, Staphylococcus spp. 35%. Klebsiella spp. was isolated from bronchopneumonia and interstitial pneumonia. Streptococcus spp. and Staphylococcus spp. Isolated from bronchopneumonia, hemorrhagic pneumonia, and purulent bronchopneumonia. Pasteurella spp. and Escherichia coli were isolated from hemorrhagic pneumonia, bronchopneumonia and fibrinous bronchopneumonia (Table
Biochemical Tests
Biochemical tests were conducted with all the isolates of causative bacteria by using five sugar media such as dextrose, maltose, lactose, sucrose, and mannitol. The detailed characterization of all the bacterial isolates is tabulated in Table 2.
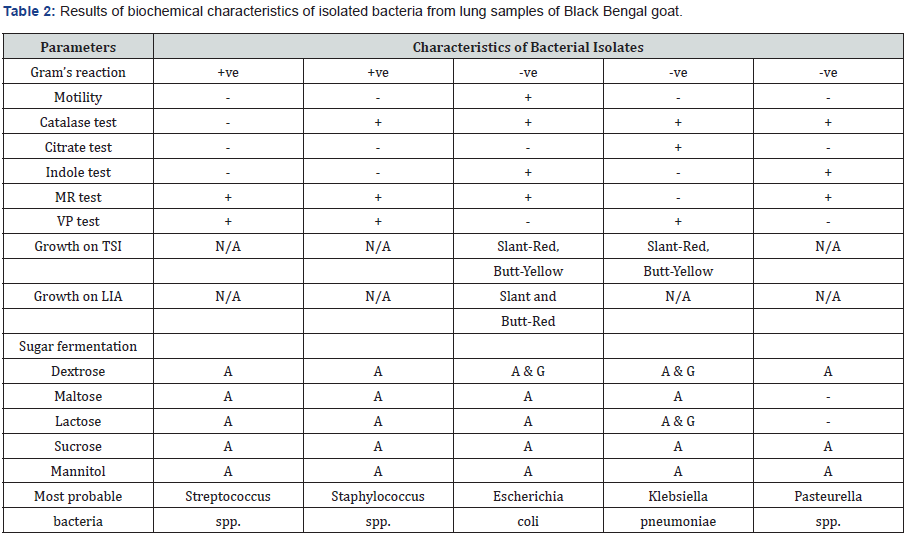
N/A: Not applicable; A: Acid production only but no gas production; (+): Positive reaction; A & G: Acid & gas production; (-): Negative reaction.
Antibiogram Study
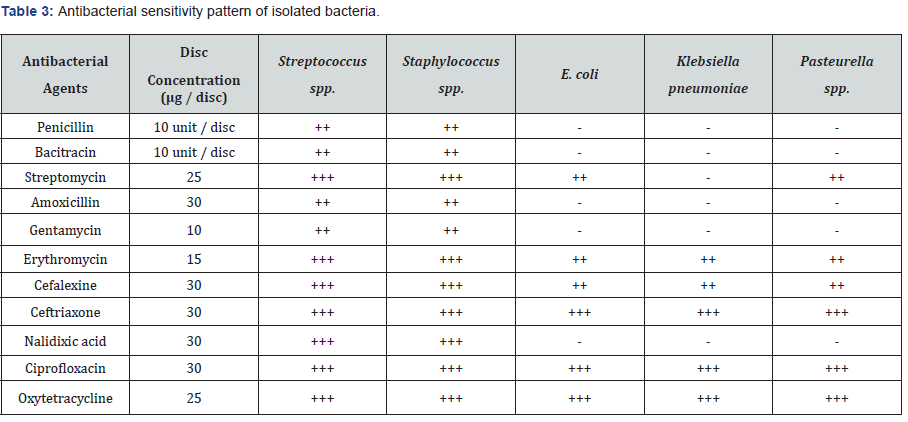
Antibiotic sensitivity pattern of different isolates was performed against eleven commonly used antibacterial agents. The antibiogram study was revealed that the isolates are highly sensitive to ciprofloxacin, oxytetracycline, and ceftriaxone, moderately sensitive to streptomycin, cefalexin, nalidixic acid, erythromycin, and resistant to gentamicin, bacitracin, penicillin, amoxicillin. The summary of antibiotic sensitivity pattern is shown in Table 3.
Histopathology of Pneumonic Lungs
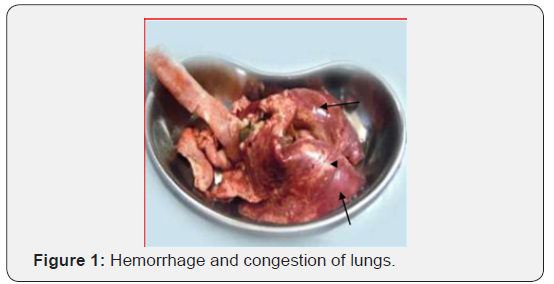
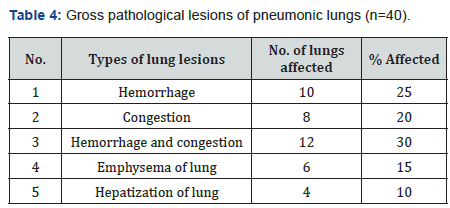
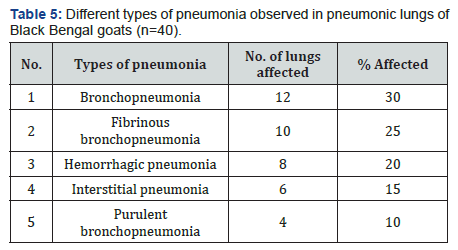
Grossly the lung lesions (Table 4) were categorized into following types: (a) hemorrhage 25% (b) Congestion 20% (c) hemorrhage and congestion 30% (d) emphysema 15% (e) hepatisation of lungs 10% (Figures 1 & 2). From histopathological study of pneumonic lung tissues of Black Bengal goat, five types of pneumonia were identified according to their histopathological changes (a) bronchopneumonia (30%), (b) fibrinous bronchopneumonia (25%), (c) hemorrhagic pneumonia (20%), (d) interstitial pneumonia (15%), and (e) purulent bronchopneumonia 10% (Table 5).

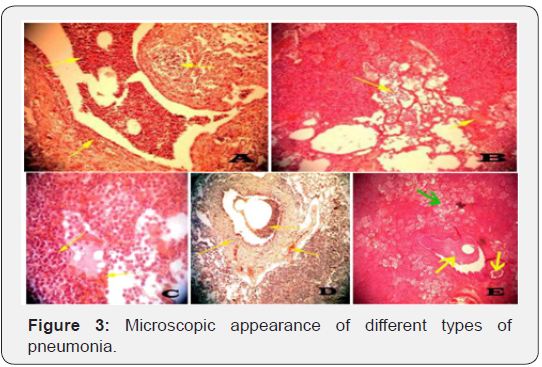
Lungs with bronchopneumonia were characterized by exudates in the alveoli, congestion of blood vessels, hemorrhages and sometimes hyperplasia of associated bronchial lymphoid tissues (Figure 3A). Lungs with fibrinous bronchopneumonia were characterized by inflammatory zone around the respiratory bronchiole with abundant fibrinous exudates and visible fibrin strand, interlobular septa, distended pleura and alveoli because of fibrinous exudation and neutrophilic exudation (Figure 3B). Hemorrhagic pneumonia was categorized by excessive haemorrhages within the bronchi, alveoli and interalveolar septa related to leukocytic infiltration (Figure 3C). While the lungs with interstitial pneumonia were considered by severe congestion of blood vessels, hemorrhage in the alveoli, inflammatory cells in the lumen of bronchus, reactive cells in and around the bronchial wall, and sometimes inter-alveolar septa were thickened due to the accumulation of mononuclear cells and proliferation of fibrous connective tissue (Figure 3D). Bronchopneumonia with endothelial damage marked the consolidation of the parenchyma with many neutrophils in the alveoli and severe desquamation of the bronchial epithelium with hyperplasia of the bronchial associated lymphoid tissues was categorized as purulent bronchopneumonia (Figure 3E).
Discussion
In our current study, five types of bacteria isolated and identified by biochemical and staining properties from pneumonic Black Bengal goats. These isolated organisms are common in pneumonic lung tissues as widely documented by several authors [9,19-24]. These bacteria are also common in pulmonary mixed infections since the respiratory pathways act as a reservoir for potentially pathogenic micro- organisms which develop into pneumonia following stress, decline of hygiene measures or climatic conditions [25], with obvious pneumonic lesions than when a single bacterium was incriminated [26]. Staphylococcus spp. was the major organism followed by Escherichia coli and Pasteurella spp. isolated from lung swabs [27,28]. Staphylococcus spp. resides in the upper respiratory tract and is involved in diseased processes only when stress conditions prevail [29]. Ferdausi et al. [30] reported the prevalence rate of Staphylococcus spp., Pasteurella spp., and Bacillus spp. were 36.67%, 11.67%, and 3.33%, respectively.
However, our current study could not find out any Bacillus spp. but the prevalence rate of Staphylococcus spp. and Pasteurella spp. Were almost similar with findings. Rashid et al. [31] also observed the similar data likewise Staphylococcus spp. (40%), Escherichia coli (25%), Pasteurella spp. (15%) and mixed infections (Escherichia coli and Staphylococcus spp. 20%) which were almost adjacent to the present findings, but mixed infections aren’t considered in our present work. Staphylococcus spp. and Streptococcus spp. isolated from bronchopneumonia, interstitial pneumonia, and purulent bronchopneumonia. As this bacterium is known as pyogenic bacteria thus, it might speculate that this purulent bronchopneumonia produced by these microbes. Lesions of purulent pneumonia were scattered in the lung parenchyma. Based on the distribution of lung lesions, the path of infection might be due to the hematogenous route. The antibiogram study revealed that the Streptococcus spp. and Staphylococcus spp. Were moderately sensitive to penicillin, bacitracin, amoxicillin, and gentamycin while Escherichia coli, Klebsiella spp. and Pasteurella spp. were resistant. Furthermore, Streptococcus spp. And Staphylococcus spp. were highly sensitive to streptomycin, erythromycin, cephalexin, ceftriaxone, nalidixic acid, ciprofloxacin, and oxytetracycline while Escherichia coli, Klebsiellapneumonia and Pasteurella spp. were moderately sensitive to streptomycin, erythromycin, cephalexin. All isolated bacteria were highly sensitive to ciprofloxacin, oxytetracycline, and ceftriaxone. The findings recorded in this study are in agreement with the results of Ghanem et al [9,21,32-37].
In consideration of the most effective antimicrobial agents against all the tested isolates, ciprofloxacin, oxytetracycline and ceftriaxone [38] are the drugs of choice in the treatment of pneumonia in Black Bengal goats. The variation of antibiotic sensitivity pattern of the respiratory bacterial isolates might be due to the presence of resistance genes. Indiscriminate use of antibiotics for treating the infected animals might also be responsible for acquiring antibiotic resistance. In this investigation, grossly the lung lesions categorized into hemorrhage 25%, congestion 20%, hemorrhages and congestion 30%, emphysematous lungs 15%, hepatization in lungs 10%. Pus and cysts in lungs recorded by Jubb et al. [39]. However, no such lesions recorded in the present investigation. The highest prevalence recorded for bronchopneumonia followed by fibrinous bronchopneumonia, hemorrhagic pneumonia, interstitial pneumonia, and purulent bronchopneumonia. Histopathological study of pneumonic lungs by Ferdausi et al. [30] reported pneumonia 6.67%, bronchopneumonia 3.33%, purulent bronchopneumonia 5%, and hemorrhagic pneumonia 3.33%. This variation might be due to some calculating factor as authors calculated out of total examined goat, but this investigation conducted within only affected lungs of a goat. Histopathology of bronchopneumonia and hemorrhagic pneumonia described in this present investigation corresponded to the lesions of other investigators [40-47].
Conclusion
The results of a bacteriological and histopathological study of pneumonic lung tissues indicated that the microbial factors might play an important role in the development of pneumonia in Black Bengal goat. However, there are some effective antimicrobial agents like ciprofloxacin; ceftriaxone; and oxytetracycline for the treatment of pneumonia. Nevertheless, some bacterial organisms such as Pasteurella spp., Streptococcus spp., Staphylococcus spp., Klebsiella spp [48-58]. And Escherichia coli responsible for the development of goat pneumonia. Further study for other etiology and molecular understanding of these bacteria required to characterize the causative agent of goat pneumonia.
References
- Kamaruddin KM (2003) Goat farming as a means of poverty alleviation Proceeding of the BSVER symposium. Goats farming in Bangladesh: Problems and prospects BSVER 2: 26-34.
- Sisay T, Zerihun A (2003) Diversity of Manheimia Haemolytica and P. trehalosi serotypes from apparently healthy sheep and abattoir specimens in the highlands of Wollo. North Eastern Ethiopia. Vet Res Commun 27(1): 3-14.
- Goodwin KA, Jackson R, Brown C, Davies PR, Morris RS, et al. (2004) Pneumonic lesions in lambs in New Zealand: patterns of prevalence and effects on production. NZ Vet J 52(4): 175-179.
- Jones GE, Field AC, Gilmour JS, Rae AG, Nettleton PF, et al. (1982) Effects of experimental chronic pneumonia on bodyweight, feed intake and carcass composition of lambs. Vet Rec 110(8): 168-173.
- Islam S, Ahad A, Chowdhury S, Barua SR (2006) Study on pneumonia in Black Bengal goat in selected areas of Bangladesh. Bangladesh J Vet Med 4: 137-140.
- Lacasta D, Ferrer LM, Ramos JJ, González JM, De las Heras M (2008) Influence of climatic factors on the development of pneumonia in lambs. Small Rumin Res 80(1-3): 28-32.
- Roy JHB (1990) Respiratory infections in the calf management of health. JHB Roy (edn), Butterworths, London, UK, 3: 132-153.
- Wikse SE, Baker JC (1996) The Bronchopneumonias. Large animal internal medicine. Diseases of Horses, cattle, Sheep and Goats. St. Louis, Mosby- Year Book (2nd edn), 5: 632-650.
- Ghanem MM, Yousif HM, Abd El-Ghany AH, Abd El-Raof YM, El-Attar HM (2015) Evaluation of pulmonary function tests with hematobiochemical alterations in Boer goats affected with Klebsiella pneumoniae. Benha Veterinary Medical Journal 1: 53-62.
- Izquierdo L, Merino S, Regue M, Rodriguez F, Tomas J (2003) Synthesis of a Klebsiella pneumoniae O antigen hetero-polysaccharide (O12) requires an ABC 2 transporter. J Bacteriol 185(1): 1634-1641.
- Radostits OM, Blood DC, Gay CC (2002) Veterinary Medicine: A textbook of the Diseases of Cattle, Sheep, Pigs, Goats and horses. (8th edn), Billiere Tindall 3: 250-257.
- Buxton A, Fraser G (1977) Animal Microbiology. Vol. I. Balackwell scientific publications. Oxford, London, UK, 47: 400-480.
- Vlaams Diergen Tijds, Cheesbrough M (1985) Medical laboratory manual for tropical countries 11(3): 400-480.
- Carter GR (1986) Essential of veterinary bacteriology and mycology, (3rd edn), Lea and Febiger Philadelphia USA 2: 90-95.
- Carter GR (1955) Studies on Pasteurella multocida: A haemagglutination test for the identification of serologic types. Am J Vet Res 16: 481-484.
- Catry B, Govaere JLJ, Devriese L, Laevens, H, Haesebrouck F, et al. (2002) Bovine enzootic bronchopneumonia prevalence of pathogens and its antimicrobial susceptibility.
- Hassan SO, Elsheikh HM (2012) Pneumonia in Goats in Sudan. Int J Anim Vet Adv 4: 144-145.
- Luna LG (1968) Manual of Histologic staining methods of the Armed Forces Institute of pathology, (3rd edn) McGraw Hill Book Co New York, USA, 2: 45-68.
- Ozyildiz Z, Tel OY, Yilmaz R, Ozsoy SY, Keskin O (2013) Pathological and microbiological investigations of pneumonic pasteurellosis in sheep. The Journal of the Faculty of Veterinary Medicine, University of Kafkas 19: 103-108.
- Azizi, S, Korani, FS, Oryan A (2013) Pneumonia in slaughtered sheep in south-western Iran: pathological characteristics and aerobic bacterial aetiology. Vet Ital 49(1): 109-118.
- Tijjani AN, Ameh JA, Gambo HI, Hassan SU, Sadiq MA, et al. (2012) Studies on the bacterial flora and pathologic lesions of caprine pneumonic lungs in Maiduguri. North-Eastern Nigeria. Afr J Microbiol Res 6(48): 7417-7422.
- Yener Z, Ilhan F, Ilhan Z, Saglam YS (2009) Immunohistochemical detection of Mannheimia (Pasteurella) haemolytica antigens in goats with natural pneumonia. Vet Res Commun 33(4): 305-313.
- Shiferaw G, Tariku S, Ayelet G, Abebe Z (2006) Contagious Caprine pleuropneumonia and Manheimia hemolytica associated acute respiratory disease of goats and sheep in Afar Region, Ethiopia. Rev Sci Tech 25(3): 1153-1163.
- Ozbey G, Muz A (2004) Isolation of aerobic bacterial agents from the lungs of sheep and goats with pneumonia and detection of Pasteurella multocida and Mannheimia haemolytica by polymerase chain reaction. Turk J Vet Anim Sci 28: 209-216.
- Moustafa AH (2004) Study of some aerobic bacterial causes of respiratory affection in slaughtered camels in Dakahlia Govt. Assiut Vet J 50: 95-105.
- Novert MH (2002) Bacteriological and mycological studies on lung infection in newly born calves. J Egypt Vet Med Assoc 62: 189-194.
- Yesuf M, Mazengia H, Chanie M (2012) Histopathological and Bacteriological Examination of Pneumonic Lungs of Small Ruminants Slaughtered at Gondar, Ethiopia. American-Eurasian J Sci Res 7: 226- 231.
- Haque MM, Ershaduzzaman M, Islam ME, Das T, Rahman MM (2007) Isolation and identification of etiological agents from diarrhoeic goats. Asian J Anim Vet Adv 2: 1-8.
- Megra T, Sisay T, Asseged B (2006) The Aerobic Bacterial flora of the Respiratory passageways of healthy goats in Dire Dawa Abattoir, Eastern Ethiopia. Rev Méd Vét 157: 84-87.
- Ferdausi T, Hossain MM, Baki AM (2008) Pathology of caprine lungs with isolation and identification of bacteria. MS Thesis, Department of Pathology, Faculty of Veterinary Science, BAU, Mymensingh 2: 35-46
- Rashid MM, Ferdoush MJ, Dipti M, Roy P, Rahman MM, et al. (2013) Bacteriological and pathological investigation of goat lungs in Mymensingh and determination of antibiotic sensitivity. Bangladesh J Vet Med 11: 159-166.
- Ghoneim HA, Hassan NI, Elhalawany HA, Nabih AM (2010) Mixed Infection of Bovine Viral Diarrhoea Virus, Mycoplasma Species and Mannheimia haemolytica in Calves Showed Chronic Pneumonia with Reference to the Histopathological Findings of the Affected Lungs. J Am Sci 6: 538-555.
- Montgomery A (2007) Incidence and sensitivity of major bovine respiratory disease pathogens in Europe. Vet Times 37: 21-24.
- Amaechi N, Ugbogu GA (2006) Antimicrobial susceptibility profile of Staphylococcus aureus isolates from ear and nostrils of farm animals at Michael Okpara University of Agriculture, Umudike Abia State, Nigeria. Nigerian J Anim Vet Adv 5: 882-885.
- Hill BD, Johnson RB (2005) Pasteurella multocida septicemia in two calves. Austral Vet J 69: 197-198.
- Afshari G, Atyabi N, Bayati M (2004) Isolation of Histophilus somni (Haemophilus somnus) from pneumonic lesions of slaughtered cattle in suburbs of Tehran. J Vet Res 59: 391-394.
- Hutt A, Goossens L (2001) Bacterial pathogens and their susceptibility towards antibiotics in connection with bovine respiratory infection. Der Prakisce Tierarzt 82: 936-947.
- Al-Doughaym AM, Mustapha KM, Mohammed GE (1999) An etiological study of pneumonia in camel (Camelus dromedaries) and in vitro antibacterial sensitivity pattern of the isolates. Pak J Biol Sci 2: 1102- 1105.
- Jubb KVF, Kennedy PC, Palmer N (1993) Pathology of Domestic Animals. (4th edn), 2: 634-638.
- Jones TC, Hunt RD, King NW (1997) Veterinary Pathology. (6th edn), Williams and Wilkins 3: 340-345.
- Alam KJ, Hossain MM, Bari ASM, Chowdhury EH, Hossain Akma, et al. (2001) Etiopathological investigation of systemic diseases in slaughtered Black Bengal Goats. Bangladesh Veterinary Journal 35: 53- 58.
- Akbor A, Haider MG, Ahmedullah F, Khan M, Hossain MJ, et al. (2007) Pathology of trachea and lungs of buffaloes. Bangladesh J Vet Med 5: 87-91.
- Adler K, Radeloff SIB, Greife H, Hellmann K (2007) Bacteriological and virological status in upper respiratory tract infections of cats. Berl Münch Tierärztl Wochenschr 120: 120-125.
- Ashok K, Vinod G, Rana R, Vaid RK, Vihan VS, et al. (2004) Incidence of caprine pneumonia in relation to climatic factors in semiarid zone. Indian J Small Rumin 10: 131-133.
- Bauer AW, Kirby WMM, Sherris JC, Turch M (1966) Antibiotic testing by Standardized single disc method. Am J Clin Pathol 45: 493-496.
- Clinical and Laboratory Standard Institute (2007) Performance standard for antimicrobial susceptibility testing: Seventeenth informational supplement pp. M100-S17.
- Cruickshank RD, Duguid JP, Marmion BP, Swain RHA (1975) Medical microbiology, (12th edn), Churchill Liviengstone Edinburg and New York 4: 469-471.
- Dabo SM, Taylor JD, Coufer AW (2007) Pasteurella multocida and bovine respiratory disease. Anim Health Res Rev 8: 129-150.
- Diker KS, Akan M, Haziroglu R (1994) Antimicrobial susceptibility of Pasteurella haemolytica and Pasteurella multocida isolated from pneumonic ovine lungs. Vet Rec 134: 597-598.
- Emikpe BO, Jarikre TA, Eyarefe OD (2013) Retrospective study of disease incidence and type of pneumonia in Nigerian small ruminants in Ibadan, Nigeria. Afr J Biomed Res 16: 107-113
- Food and Agricultural Organization (2007) Black Bengal promising goat genetic resource of Bangladesh 2: 78-80.
- Guler L, Baysal T, Gunduz K, Erganis O, Kaya O, et al. (1996) Biotyping and serotyping of Pasteurella haemolytica strains isolated from sheep and goats Ministry of food, Agriculture and Livestock. Department of Training, Extension and Publications Turkey 2: 5-12.
- Kanar NS, Paliwal OP, Ram K, Kumar AA, Kumar R (1998) Pneumonic pasteurellosis in goats. Indian J Com Microbiol Immunol Infect Dis 19: 99-101.
- Lancelot R, Lesnoff M, McDermott JJ (2002) Use of Akaike information criteria for model selection and inference an application to assess Guinean goats in Kolda, Senegal. Prev Vet Med 55: 217-240.
- Merchant LA, Packer RA (1967) Veterinary bacteriology and virology. (7th edn), The IowaUniversity Press, Ames, Iowa, USA 4: 211-306.
- Pinheiro RR, Gouveia AMG, Alves FSF, Haddad JPA (2000) Epidemiological aspects of goat production Ceara State, Brazil. Braz J Vet Anim Sci 52: 534-543.
- Sharma RK, Patil RD, Kishtwaria RS, Asrani RK (2011) An outbreak of pneumonic mannheimiosis in a livestock farm in sub-temperate region of India, Palampur. Haryana Vet 50: 89-91.
- Upadhyaya TN, Rahman T (1993) Prevalence and Pathology of caprine pneumonia in Assam. J Assam Vet Coun 3: 47-49.






























