Effect of Selenium and Vitamin E on Development and Viability of Preimplanted Mouse Embryo
Hirotada T1, Miah AG2* and Salma U2
1Faculty of Agriculture, Shinshu University, Japan
2Faculty of Veterinary and Animal Science, Hajee Mohammad Danesh Science & Technology University
Submission: February 06, 2017; Published: June 02, 2017
*Corresponding author: Miah AG, Department of Genetics and Animal Breeding, Faculty of Veterinary and Animal Science Hajee Mohammad Danesh Science & Technology University, Dinajpur-5200, Bangladesh, India. Tel: +8801784973090; Email: agmiah2009@gmail.com
How to cite this article: Hirotada T, Miah A, Salma U. Effect of Selenium and Vitamin E on Development and Viability of Preimplanted Mouse Embryo. Dairy and Vet Sci J. 2017; 2(5): 555599. DOI: 10.19080/JDVS.2017.02.555599
Abstract
In vitro culture results higher level of reactive oxygen species (ROS) oxygen than in vivo environments that cause lipid peroxidation of cellular membranes. Selenium (Se) and Vitamin E (Vit-E) are the important antioxidants that protect mammalian cells against lipid peroxidation. Therefore, the present study was conducted to investigate whether Se or Vit-E and Se+Vit-E overcome the the undesirable oxidative stress produced by hydrogen peroxide (H2O2) and enhance the development of pre implanted mouse embryo. Co-incubating the embryos with 60nM Se and/or 100nM Vit-E were increased (P<0.05) the blastocyst development rate.
The addition of H2O2 reduced the development of mouse embryo, but the addition of Vit-E, Se and Se+Vit-E reduced the detrimental effect of H2O2 and influenced the higher rate of development to blastocysts, compared to CZB alone (P<0.05). The incorporation and oxidation of 14C-glucose in the blastocysts developed by the medium supplemented with Se and/or Vit-E in the presence or absence of H2O2 were significantly higher (P<0.05) than that of the control. Moreover, Vit-E is more effective than Se and Se+Vit-E in reversing ROS-induced mouse embryo toxicity. Therefore, Vit-E may be supplemented in the CZB medium for better development and viability of pre implanted mouse embryo.
Keywords: Mouse embryo; Selenium; Vitamin E
Abbrevations Ros: Reactive Oxygen Species; -Oh: Hydroxyl Radical; O2-: Superoxide Anion Radical; Se: Selenium; Gpx: Glutathione Peroxidase; Cpm :Counts Per Minutes
Introduction
In vitro embryo culture suffers from excessive developmental failure. Its inefficiency is linked with the generation of reactive oxygen species (ROS), such as H2O2, hydroxyl radical (-OH) and superoxide anion radical (O2-), appears as the by-products of cell metabolism [1]. Superoxide may also spontaneously break down into oxygen and H2O2. As ROS are highly reactive molecules, their accumulation can lead to damage and breakage of DNA strands. There are many evidences have been found that ROS compromises embryo development in many species [2-6].
Selenium (Se), an essential trace element for mammals, is an integral part of anti- oxidant system [7]. Se dependent glutathione peroxidase (GPx) has an important role in free radical protective mechanisms. Vitamin E (Vit-E), the predominant lipid-soluble antioxidant in animal cells, protects cells from oxygen radical damage in vivo [8,9] and in vitro [10,11].Our previous study investigated that Se and Vit-E, as the integral parts of antioxidant systems which play important roles for the in vitro maturation, fertilization and culture of porcine oocyte [12]. However, there is a very limited studies were conducted with the effect of Se and Vit-E on the development of preimplanted mouse embryo. Therefore, the present study was conducted to investigate whether Se, Vit-E or Se+Vit-Eovercome the undesirable oxidative stress produced by hydrogen peroxide (H2O2) and enhance the development of preimplanted mouse embryo.
Materials and Methods
Chemicals and reagents
The basic embryo culture medium used in this study was CZB [13] , which contains 1mM glutamine, 0.1 mM EDTA and 5mg/ml BSA, and considered as control. A part of CZB was added with 30% H2O2 to get a final concentration of 0.0003% that considered as negative control. According to the experimental layout, CZB (with or without H2O2) was supplemented with30, 60, 90Nm Se (sodium selenite; Sigma-Aldrich, St. Louis, MO, USA), 50, 100 and150nM of Vit-E (α-tocopherolacetate) and their combination (Se+Vit-E). Selenium, Vit-E and H2O2 were equilibrated in culture medium under CO2 incubator for 8 hours before the start of culture. Vit-E was dissolved in ethanol, and an emulsion was formed by vortex mixing before adding it to the embryo. PMSG and HCG used in this study were obtained from Sankyo Chemical Industries Ltd., Tokyo, Japan. The copulation plug was checked 24 hours later. Radioactive 14C (U)-glucose was purchased from American Radio labeled Chemicals (St. Louis, Mo, USA). All other chemicals were of analytical grade and purchased from Nacalai Tesque (Kyoto, Japan) unless otherwise indicated.
Collection of embryos
Embryos were obtained from 7-8 weeks old female ICR mice. They were offered feed with a balanced standard diet and ad libitum clean drinking water. Animals were kept in polycarbonate cage with wood shavings under a 12h light: 12h dark regimen (light on at 6:00), at a temperature of 20±1 °C in accordance with the "Guideline for Regulation of Animal Experimentation, Faculty of Agriculture, Shinshu University.” Female mice were induced to superovulation with PMSG (5 1U, i.p.) followed 48h later by hCG (51U, i.p.) and met with male mice. Zygotes of 1-cell stages were collected at 2 5 hours HCG post injection by flushing out from the fallopian tubes. The embryos were subsequently incubated in CZB medium.
Experimental layout
In fact, there are four experiments were conducted under this study In experiment 1, effect of CZB supplanted with different levels of Se (0, 30, 60 and 90nM) and Vit-E (0, 50, 100 and 150nM) on the development of mouse embryo from 1-cell stage to the blastocyst were evaluated. The experiment 11 were conducted to investigate the effects of Se (60nM), Vit-E (100nM) and their combination (60nM Se+100nM Vit-E) on the development from 2-cell to blastocyst stages of embryo in the presence (1mM) or absence of H2O2 in CZB medium. Where, the experiment 111 was conducted to the effect evaluate Se (60nM), Vit-E (100nM) and their combination (60nM Se+100nM Vit-E) on the accumulation of ammonia (NH4) in presence or absence of H2O2 due to metabolism of embryo during the development up to blastocyst stages. 1ncorporation and oxidation of 14C-glucose at blastocysts developed by the supplementation of Se (60nM), Vit-E (100nM) and their combination (60nM Se+100nM Vit-E) in the presence or absence of H2O2 were evaluated by conducting the experiment IV. These experiments were conducted five times repeated.
Culture of embryos
The collected zygotes of 1-cell stages were transferred to the culture dishes for washing, and grown in-vitro to the developmental stages up to blastocyst. Embryos were cultured according to the standard techniques, in groups of 10 zygotes were placed into 35mm-diameter culture dishes (Nunc Co., Denmark) containing 30|il of each CZB medium under a layer of paraffin oil and equilibrated overnight in an atmosphere of 5% CO2 in air at 37 °C. The pH of all media was 7.4 after equilibration. The developing embryos seemed to be normal in their morphology, with almost no fragmentation.
Ammonia determination
During incubation period embryos which developed in the presence of Se and/or Vit-E with or without H2O2, the ammonia concentrations in the medium were assessed by using the Bertholot-indophenol method as described in our previous study [14] . To determine the ammonia concentration in the medium, 100μ1 of the culture medium was removed every 2-4h and frozen at -40 °C until measurement. The procedure was carried out five times for the analysis. A calibration curve in the range 0.0003� of ammonia was run with each experiment. The mean coefficient for determination of the calibration curve of five experiments was 0.994.
Incorporation and oxidation of 14C-glucose
The experiment was initiated with 14C-glucose 18.5kBq/0.1mol (specific activity 9.69MkBq/mol, Moravek Biochemicals, 1nc., USA). Each of the ten blastocysts which developed in the presence of Se and/ or Vit-E with or without H2O2 was transferred in a microtube of 50μ1 CZB medium drop containing 14C-glucose then overlaid with mineral oil. On the other hand, 1ml of 2.5Mm NaOH solution was transferred into a 1.5ml micro tube as a trap for the evolved 14 CO2. Both microtubes of NaOH and 14C-glucosewith embryos were confined into a scintillation vial using a rubber stopper. The scintillation vials were incubated for 5h in an incubator at 37 °C. After incubation period, the metabolic reactions of embryos were stopped with an injection of 100μ1 of 10% perchloric acid (PCA) kept at room temperature for 24h. The acid insoluble materials were carefully washed by millipore filtration (8.0|iM white SCWP, 47mm; Millipore Corporation, Bedford, MA, USA) with 5% PCA and the filter papers were kept overnight under a lamp. After drying, the filter papers were transferred into scintillation vials. The NaOH solution was transferred into a new scintillation vial by washing 3-4 times with cocktail (0.5% PPO+0.03% POPOP solution in toluene). All the scintillation vials with 5ml of cocktail were set in a Liquid Scintillation Counter (LS-6500, Beckman Instruments, Inc. USA) to determine the levels of radio activity [15]. This experiment was conducted ten times to improve its accuracy. The values of incorporation and oxidation were expressed directly as counts per minutes (CPM).
Statistical analysis
Data obtained from this stud were analyzed by one-way ANOVA using the GLM procedure of SAS (SAS Institute, Cary, NC). The data expressed as percentage were tested by Chi-square test. Data were presented as mean ±SEM of at least 5 replicates and differences were considered significant at the level of P<0.05.
Results
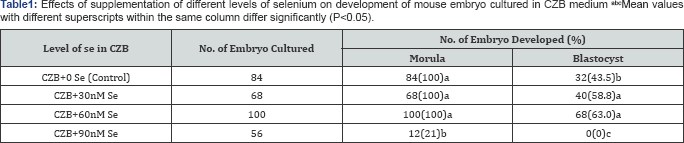
The effects of supplementation of different levels of Se (0, 30, 60 and 90nM) in CZB medium on the development of mouse embryo are presented in (Table 1). The results revealed that 30 and 60nM of Se was more effective (P<0.05) for mouse embryo development than that of the control and 90nM of Se, where the level showed detrimental effect on the development of embryo as blastocyst. On the other hand, (Table 2) demonstrated that the supplementation of 100nM of Vit-E was the most effective (P<0.05) for embryonic development than that of the 0, (control), 50 and 150nM. The lowest (P<0.05) percentage of blastocyst was observed when zygotes were cultured in CZB supplemented with 150nM of Vit-E.

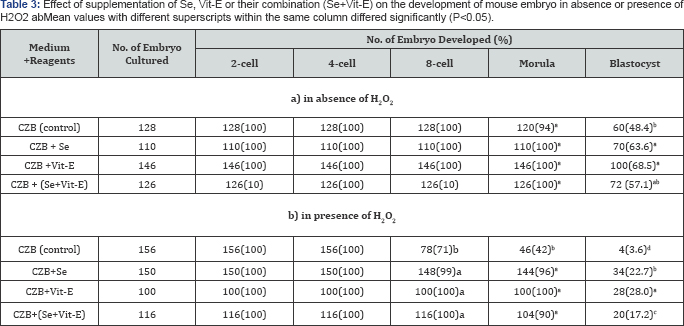
Detrimental effects of H2O2 and reducing or protecting ability of Se, Vit-E or their combination from the effects of H2O2 on the embryonic development were shown in (Table 3). The results demonstrated that the development of mouse embryos was reduced by the detrimental effect of H2O2 especially in CZB, the occurrence after 8 cell markedly decreased under H2O2 (P<0.05). However, the supplementation of Se, Vit-E and Se+Vit-E were able to reduce the detrimental effect of H2O2 and enhanced (P<0.05) the development of mouse embryo to be blastocyst. Highest percentage of blastocysts was obtained when the zygotes were cultured in the CZB medium supplemented with Vit-E in the presence or absence of H2O2.
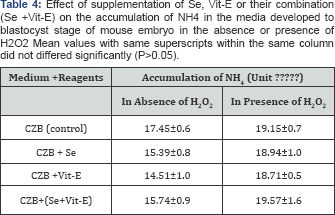
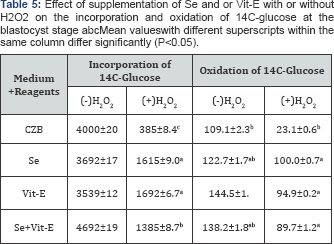
During the development of mouse embryos up to the blastocyst stage, accumulation of metabolic NH4+ in the CZB medium supplemented with Se, Vit-E and Se+Vit-E in the presence or absence of H2O2 is showed in (Table 4). The results revealed that the lowest (P<0.05) accumulation of metabolic NH4+ was observed in the CZB medium supplemented with Vit-E in the presence or absence of H2O2. The effects of supplementation of Se, V1t-E and Se+Vit-E in the CZB medium with or without H2O2 on incorporation and oxidation of 14C-glucose at the blastocyst stage are shown in (Table 5). The incorporation of 14C-glucose at the blastocyst stage cultured with Se, Vit-E and Se+Vit-E in the CZB medium were not significantly differed (P>0.05), but higher rate of incorporation (P<0.05) occurred by the supplementation of Se or Vit-E in the presence of H2O2. On the other hand, in the presence of H2O2, the oxidation of 14C-glucose by the blastocysts were higher (P<0.05) when zygotes were cultured in the CZB medium supplanted with Vit-E or Se alone or Se+Vit-E than the basic CZB medium. There was a slightly higher rate of oxidation of 14C-glucoseoccured in the blastocyst stage cultured with Se, Vit-E and Se+Vit-E.
Discussion
A number of intrinsic and extrinsic factors have been shown to influence in vitro survival of the embryos to the blastocyst stage in extended culture. Previous studies suggest that in vitro extrinsic factors such as prolonged culture conditions and the autocrine and paracrine activities of the embryos may also contribute to the failure of optimal embryo development. Among the factors that might affect in vitro development of embryos is the balance between oxidative stress, and the ability of the embryos to neutralize their effects [16] reported a sustained increase in oxygen, glucose and pyruvate uptake during in vitro embryo development. The embryos were dependent on oxidative phosphorylation for energy (ATP) production at all stages of pre-elongation development, with perhaps a shift in dependence towards glycolysis in conjunction with compaction. This enhancement in oxidative metabolism of the embryo could be linked to the detected increase in ROS, which are characterized by the presence of an unpaired electron [17] and free-radical intermediaries [18].
Free radicals are generated from leakage of high-energy electrons as they proceed down the electron transport chain. The free radicals have many harmful effects including DNA damage [19]. Many embryos under oxidative stress step into a transient cell cycle arrest which is activated by DNA damage response before apoptosis [20]. Legge & Sellens [21] were suggested that the 2-cell block in mouse embryo is at least in part, of free radical damage incurred by embryos during collection and culture, and that medium supplementation with the radical scavenger, reduced glutathione, can improve embryo development in vitro reported that enhanced oxidative metabolism of embryos may be associated with increased ROS levels detected. The gradual increase in ROS levels from the 2-cell embryo up to the late morula stage could depend on the metabolic change undergone by the embryo during its development. 1t is necessary to prevent ROS as much as possible during culture embryos. However, it is unclear as to which embryos may be adversely affected and to what extent.
The present study showed that Se, Vit-E and SE+Vit-E increased blastocyst formation compared to control. Especially, it suggested that the formation of mouse blastocysts cultured in the presence of 60nM/ml Se and 100nM/ml Vit-E were significantly higher than control. The trace element Se is a component of antioxidative seleno enzymes, Glutathione Peroxidase (GPx) and ThioredoxinReductase (ThxRed) that decrease oxidative stress. Se, as sodium selenite, has been reported as a co-factor for glutathione peroxidase and other proteins and used as an anti-oxidant in medium [22]. 1n cell culture system, sodium selenite protected cell from oxidative damage, free radicals and obstructed lipid peroxide products [23,24]. Se played a role in the antioxidant defense system in the formation of mouse blastocyst, which was essential for the catalytic activity of glutathione peroxidase. Glutathione, a thiol tripeptide component in all cell types has an important role in the transportation of amino acid, synthesis of the protein and DNA, and reduction of disulfide bonds [25].
In the present study, Se and Vit-E were used as combined supplements in the CZB medium for culture of mouse embryos, but this combination showed lower influence in the development of mouse blastocyst than Vit-E alone. Alpha-tocopherol (Vitamin E) is well known as an ROS scavenger in in vivo and in vitro conditions [26-28] and is the most important antioxidant present in ovarian tissue and follicular fluid. The antioxidant activity of a-tocopherol in preventing free-radical-induced tissue damage is accepted by most investigators and is believed to be the primary free radical scavenger and to inhibit lipid peroxidation in the mammalian cell membrane [26-29].The present study also demonstrated that the supplementation of Vit-E (specially, 100nM) played an important role in the development of mouse embryo. The results are in agreement with the results of our previous study and the study of [30] they also reported that the optimal concentration of Vit-E in embryo culture is 100nM.
There is a method to observe the effect of mild oxidative stress with retardation of embryo development in the medium supplemented with 1-5mM H2O2. The H2O2 used in this experiment was 1mM, and performed detrimental effects on the development embryo after 8 cells [31]. Reported that effects of H2O2 on blastocyst formation became more severe during the treatment of later stages of development. Embryos may also have different sensitivities to ROS at different developmental stages [32]. The exogenous oxidant H2O2 leads to over production of ROS, which may induce multiple cellular damages, including lipid peroxidation, nuclear DNA strand breaks, and mitochondrial alteration, consequently disturbing the development of pr implanted embryos in vitro [33-35]. Most of the embryos under oxidative stress step into a transient cell cycle arrest in vitro, which is activated by DNA damage response before apoptosis.
Vit-E(α-tocopherol)isapredominantlipid-solubleantioxidant that has been considered as a primary free radical scavenger in biological membranes [36-38]. a-tocopherolscavenges peroxyl radicals from polyunsaturated fatty acid in membrane phospholipids or lipoproteins that do not spread the radical chain, thereby protecting against lipid peroxidation [39]. Our previous study reported that supplementation of a-tocopherol maintains the development of mouse embryo and pig oocyte quality, fertilization rates and embryo development. Mouse preimplantation embryos can be cultured in a simple defined medium. Under such conditions energy substrates in the medium represent a major source of carbon for anabolism. Glucose is incorporated into macromolecules during in-vitro culture of cleaving mouse embryos and blastocysts [40]. 1n particular, both acid-soluble glycogen and desmoglycogen are rapidly synthesized from glucose presumably to act as a source of energy at implantation [41]. Overall metabolism, as assessed by oxygen consumption is low during the cleavage stages of development before rising sharply at the blastocyst stage. Moreover, metabolic activity has been shown to relate to developmental potential.
In this study, the accumulation of ammonia in the culture was measured regardless of the presence or absence of H2O2, the lower accumulation of ammonia in the blastocyst stage is better than the higher accumulation that occur incidence to the blastocyst [42] showed that high levels of ROS in culture media are associated with low rates of embryo development and blastocyst formation [43]. Suggested two possible mechanisms for the inhibitory effects of NH4+. Perturbation of intracellular pH requires the involvement of Na/K ATPase to transport NH4+ across membranes. Alternatively, may interact directly with enzymes, participating in a series of futile cycles which detoxify NH4+ and result in consumption of ATP. Thus, by whichever mechanism, inclusion of NH4+in culture media will divert ATP from growth to maintenance.
Azizimoghadam reported that pentose phosphate pathway activity of total glucose metabolism was increased at the compacted morula stage and was highest at the blastocyst stage [44]. 1n this study, incorporation and oxidation of 14C-glucose in the blastocyst was significantly influenced when cultured with Vit-E and Se in the presence of H2O2. The incorporation and oxidation of 14C-glucose at the blastocyst tended to resemble the development of embryo. The present study appeared that incorporation and oxidation of 14C- glucose was good for showing the activity of embryos.
Generation of ROS induced by glucose utilization was assumed to be caused by the activation of NADPH oxidase, an enzyme that catalyzes the oxidation of NADPH, generates NADP that serves as a coenzyme of the oxidative arm of the pentose phosphate pathway (PPP) [45]. The gradual increase in ROS levels from the 2-cell embryo up to the late morula stage could depend on the metabolic change undergone by the embryo during its development. 1t is necessary to prevent ROS as much as possible during culture embryos.
Conclusion
In conclusion, our results showed that culture of mouse zygotes in the CZB medium supplemented with 60nM Se and/ or 100nM Vit-E improves the developmental rate of mouse blastocyst formation in the absence or presence of free radicals or their sources. Therefore, the present study reveals that selenium and vitamin E improves mice blastocyst viability by minimizing the level of free radicals that might occur during development of blastocyst in vitro and which may be useful for assisted reproductive techniques.
References
- Durackova Z (2010) Some current insights into oxidative stress. Physiol Res 59(4): 459-469.
- Arias ME, Sanchez R, Felmer R (2012) Evaluation of different culture systems with low oxygen tension on the development, quality and oxidative stress-related genes of bovine embryos produced in vitro. Zygote 20(3): 209-217.
- Takenaka M, Horiuchi T, Yanagimachi R (2007) Effects of light on development of mammalian zygotes. Proc Natl Acad Sci 104(36): 14289-14293.
- Leoni GG, Rosati I, Succu S, Bogliolo L, Bebbere D, et al. (2007) A low oxygen atmosphere during IVF accelerates the kinetic of formation of in vitro produced ovine blastocysts. Reprod Domest Anim 42(3): 299304.
- Ciray HN, Aksoy T, Yaramanci K, Karayaka I, Bahceci M (2009) In vitro culture under physiologic oxygen concentration improves blastocyst yield and quality: a prospective randomized survey on sibling oocytes. Fertil Steril 91(4): 1459-1461.
- Kasterstein E, Strassburger D, Komarovsky D, Bern O, Komsky A, et al. (2013) The effect of two distinct levels of oxygen concentration on embryo development in a sibling oocyte study. J Assist Reprod Genet 30(8): 1073-1079.
- Hendry DG, Schuetzle D (1976) Reactions of hydroperoxy radicals. Comparison of reactivity with organic peroxy radicals. J Org Chem 41(19): 3179-3182.
- Chow CK (1979) Nutrtional influence on cellular antioxidant defense systems. Am J ClinNutr 32(5): 1006-1081.
- Miller JK, Brzezinska-Slebodzinska E, Madsen FC (1993) Oxidative stress, antioxidants and animal function. J Dairy Sci 76(9): 2812-2823.
- Tsujii H, Muranaka M, Hamano K (2002) Culture of in vitro mouse embryos with vitamine E improves development. Journal of reproduction and development 48(1): 25-29.
- Thiyagarajan B, Valivittan K (2009) Ameliorating effect of vitamin E on in vitro development of preimplantation buffalo embryos. J Assist Reprod Genet 26(4): 217-225.
- Tareq KM, Akter QS, Khandoker MA, Tsujii H (2012) Selenium and vitamin E improve the in vitro maturation, fertilization and culture to blastocyst of porcine oocytes. J Reprod Dev 58(6): 621-628.
- Chatot CL, Lewis JL, Torres I, Ziomek CA (1990) Development of 1-cell embryos from different strains of mice in CZB medium 42(3): 432-440.
- Tareq KMA, Obata R, Miah AG, Hamano K, Tsujii H (2005) Ammonia Concentration in Porcine Ovarian Developing Follicles. J Mammalian Ova Res 22(3): 185-189.
- TsujiiH, Nakamura Y (1999) Utilization of [U-14C] Glucose by Preimplantation Rat Embryo Cultured In Vitro. Journal of Mammalian Ova Research 16(3): 94-97
- Thompson JG, Partridge RJ, Houghton FD, Cox CI, Leese HJ (1996) Oxygen uptake and carbohydrate metabolism by in vitro derived bovine embryos. J Reprod Fertil 106(2): 299-306.
- Noda Y, Matsumoto H, Umaoka Y, Tatsumi K, Kishi J, et al. (1991) Involvement of superoxide radicals in the mouse two-cell block. Mol Reprod Dev 28(4): 356-360.
- Burton GJ, Hempstock J, Jauniaux E (2003) Oxygen, early embryonic metabolism and free radical-mediated embryopathies. Reprod Biomed Online 6(1): 84-96.
- Sturmey RG, Hawkhead JA, Barker EA, Leese HJ (2009) DNA damage and metabolic activity in the preimplantation embryo. Hum Reprod 24(1): 81-91.
- Liu L, Trimarchi JR, Keefe DL (2000) Involvement of mitochondria in oxidative stress-induced cell death in mouse zygotes. Biology of Reproduction 62(6): 1745-1753.
- Legge M, Sellens MH (1991) Free radical scavengers ameliorate the 2-cell block in mouse embryo culture. Hum Reprod 6(6): 867-871.
- Meister A, Anderson ME (1983) Glutathione. Annual Reviews Biochem 52: 711-760.
- Ebert R, Ulmer MS, Zeck S, Meissner-Weigl J, Schneider D, et al. (2006) Selenium supplementation restores the antioxidative capacity and prevents cell damage in bone marrow stromal cells in vitro. Stem Cells 24(5): 1226-1235.
- Tatemot H, Muto N, Sunagawa I, Shinjo A, Nakada T (2004) Protection of porcine oocytes against cell damage caused by oxidative stress during in vitro maturation: role of superoxide dismutase activity in porcine follicular fluid. BiolReprod 71(4): 1150-1157.
- Meister A, ME Anderson (1983) Glutathione. Annu Rev Biochem 52: 711-760.
- Olson SE, Seidel G (2000) Culture of in vitro-produced bovine embryos with vitamin E improves development in vitro and after transfer to recipients. Biol Reprod 62(2): 248-252.
- Liebler DC (1993) The role of metabolism in the antioxidant function of vitamin E. Crit Rev Toxicol 23(2): 147-169.
- Sies H, Stahl W (1995) Vitamins E and C, beta-carotene, and other carotenoids as antioxidants. Am J ClinNutr 62(6): 1315S-1321S.
- Chow CK (1991) Vitamin E and oxidative stress. Free Radical Biology and Medicine 11(2): 215-232.
- Jeong YW, Park SW, Hossein MS, Kim S, Kim JH, et al. (2006) Antiapoptotic and embryotrophic effects of alpha-tocopherol and L-ascorbic acid on porcine embryos derived from in vitro fertilization and somatic cell nuclear transfer. Theriogenology 66(9): 2104-2112.
- Bain NT, Madan P, Betts DH (2011) The early embryo response to intracellular reactive oxygen species is developmentally regulated. Reprod Fertil Dev 23(4): 561-575.
- Morales H, Tilquin P, Rees JF, Massip A, Dessy F, et al. (1999) Pyruvate prevents peroxide-induced injury of in vitro preimplantation bovine embryos. Mol Reprod Dev 52(2): 149-157.
- Liu L, Trimarchi JR, Keefe DL (2000) Involvement of mitochondria in oxidative stress-induced cell death in mouse zygotes. Biology of Reproduction 62(6): 1745-1753.
- Zhang C, Liu C, Li D, Yao N, Yuan X, et al. (2010) Intracellular redox imbalance and extracellular amino acid metabolic abnormality contribute to arsenic-induced developmental retardation in mouse preimplantation embryos. J Cell Physiol 222(2): 444-455.
- Kitagawa Y, Suzuki K, Yoneda A, Watanabe T (2004) Effects of oxygen concentration and antioxidants on the in vitro developmental ability, production of reactive oxygen species (ROS), and DNA fragmentation in porcine embryos. Theriogenology 62(7): 1186-1197.
- Abedelahi A, Salehnia M, Allameh A, Davoodi D (2010) Sodium selenite improves the in vitro follicular development by reducing the reactive oxygen species level and increasing the total antioxidant capacity and glutathione peroxide activity. Hum Reprod 25(4): 977-985.
- Olson S, Seidel G (2000) Culture of in vitro-produced bovine embryos with vitamin E improves development in vitro and after transfer to recipients. Biol Reprod 62(2): 248-252.
- Liebler DC (1993) The role of metabolism in the antioxidant function of vitamin E. Crit Rev Toxicol 23(2): 147-169.
- Grub S, Boelsterli UA, Trendelenburg C, Trommer WE, Wolf A (2003) Lipid peroxidation-independent mechanisms of vitamin e-mediated protection against cyclosporine a-induced hepatocellular toxicity and apoptosis. Toxicol Mech Methods 13(3): 187-197.
- Brinster RL (1969) Incorporation of carbon from glucose and pyruvate into the preimplantation mouse embryo. Exp Cell Res 58(1): 153 -158.
- Pike IL (1981) Comparative studies of embryo metabolism in early pregnancy. J Reprod Fert Suppl 29: 203-213
- Bedaiwy MA, Falcone T, Mohamed MS, Aleem AAN, Sharma RK, et al. (2004) Differential growth of human embryos in vitro: role of reactive oxygen species. Fertil Steril 82(3): 593-600.
- Schneider M, Marison IW, von Stockar U (1996) The importance of ammonia in mammalian cell culture. J Biotechnol 46(3): 161-185.
- Azizimoghadam A (2012) Metabolism of energy substrates of in vitro and in vivo derived embryos from ewes synchronized and super ovulated with norgestomet and porcine follicle stimulating hormone. J Anim Sci Biotechnol 3(1): 37.
- Iwata H, Akamatsu S, Minami N, Yamada M (1998) Effects of antioxidants on the development of bovine 1VM/1VF embryos in various concentrations of glucose. Theriogenology 50(3): 365-375.






























