Organoids As a Tool to Fuel Basic Medical Research
Ru Wang1, Yihang Zhang1* and Jiazhen Wang1,2*
1Collaborative Innovation Center for Chinese Medicine and Respiratory Diseases Co-Constructed by Henan Province and Education Ministry of People’s Republic of China, Henan University of Chinese Medicine, China
2Academy of Chinese Medicine Science, Henan University of Chinese Medicine, China
Submission: February 07, 2023; Published: February 21, 2023
*Corresponding author: Yihang Zhang and Jiazhen Wang, Collaborative Innovation Center for Chinese Medicine and Respiratory Diseases Co-Constructed by Henan Province and Education Ministry of People’s Republic of China, Academy of Chinese Medicine Science, Henan University of Chinese Medicine, China
How to cite this article: Ru Wang, Yihang Zhang and Jiazhen Wang. Organoids As a Tool to Fuel Basic Medical Research. J Complement Med Alt 002 Healthcare. 2023; 11(5): 555823. DOI: 10.19080/JCMAH.2023.11.555823
Abstract
The use of animal models in biomedical research has been a long-standing challenge due to their limited ability to accurately depict human physiology and pathology. However, the development of organoids has changed this by allowing the replication of the structure and function of human organs in a controlled laboratory setting. Organoids are 3D cell cultures that possess the key features of specific organs, and contain a population of self-renewing stem cells that can differentiate into various cell types that mimic the organ-specific properties and spatial organization. Organoids can be derived from both adult and pluripotent stem cells [1]. Recent advancements in the field demonstrate that 3D organoids robustly exhibit self-organizing properties in both embryonic and adult mammal stem cells, exhibiting key structures and functional characteristics of organs such as the brain, heart, kidney, liver, lung, and retina. This article provides an overview of recent progress in organoid technology and its impact on advancing basic medicine.
Keywords: Organoids; Stem cells; Organoid culture; Disease modeling; Basic medicine
Introduction
In past studies, animal models and two-dimensional cell lines have been successfully applied in the biomedical field, however, the differences between animal and human species and the lack of hierarchy, dimension and diversity of two-dimensional cell lines are obstacles to further promoting the development of basic medicine. With the emergence of organoids, the morphogenesis, development and differentiation of stem cells are explained in detail. Organoids have broad prospects for biomedical research and drug research. In this review, we discuss the latest research progress of organoids and describe the main categories of organoids.
Heart organoids
The heart is the first organ to initiate formation during the embryonic stage of development, and its continued functioning is critical for the progression of an animal from early embryos to adulthood [1,2]. However, adult mammalian cardiomyocytes do not exhibit effective regenerative capabilities following injury [3]. Furthermore, both animal models and non-cardiomyocyte cell lines are insufficient in evaluating drug efficacy and cardiotoxicity [4,5]. Despite substantial investments of time and resources, 90% of newly developed drugs fail during the first phase of clinical trials, with cardiotoxicity being a common cause [6-8]. Thus, research on cardiac organoids has become increasingly important. The use of 3D culture techniques has led to the development of two artificial tissues that exhibit functional and structural characteristics that are reminiscent of the native heart.
The human-engineered heart tissue (hEHT) is a well-known organoid consisting of diverse heart cell types and biological components [9]. Compared to animal models, hEHT provides a more human-relevant disease modeling approach. Furthermore, hEHT can replicate the atria, ventricles, or bipolar structures of the heart, enabling improved disease modeling and cardiac regeneration, as well as more effective research on cardiomyocyte-based cardiac pumping [10].
The alternative to human-engineered heart tissue (hEHT) is the human heart organoid, generated from stem cells specific to the organ or pluripotent stem cells (PSCs). Unlike hEHT, the cardiac organoids are produced through the differentiation of stem cells. These cardiac organoids have the potential to grow and mature into cardiomyocytes over time [11]. Researchers use various components, such as cardiomyocytes that mimic the cardiac architecture, as well as endothelial layers and epicardium-lining, to replicate the cardiac architecture of the heart. This allows them to investigate potential mechanisms behind cardiac development impairment, such as cryo-damage, and congenital heart malformations [12].
Brain Organoids
The human brain is a complex organ composed of various cell types, including neurons, astrocytes, oligodendrocytes, and microglia [13]. The development and dysfunction of the human brain have been a major focus in the field of neurobiology. However, acquiring human brain tissue samples for research purposes can be challenging due to difficulties in obtaining samples and limitations in tissue culture methods, making it difficult to fully understand the molecular mechanisms behind brain diseases [14,15]. The advent of brain organoids offers a more promising platform for studying human brain development and diseases.
To date, various regions of the human brain have been studied using organoids, including the hippocampus [16], midbrain [17], hypothalamus [18], and cerebellum [19]. Brain organoids can serve as developmental models of the nervous system and provide insight into various aspects of human neurodevelopment. For example, brain organoids can be used to recreate cell types and structural features of the human brain and as a tool for investigating the mechanisms and phenotypes of brain diseases, potentially leading to improved diagnosis and treatment strategies. One such example is the discovery of primary microcephaly, a congenital genetic disorder, in brain organoids generated from pluripotent stem cells of a patient carrying a truncating mutation in the microcephaly gene CDK5RAP2 [20]. The compromised functioning of the CDK5RAP2 gene leads to altered division patterns of progenitor cells, resulting in premature nervous system development, stem cell depletion, and ultimately cerebellar malformations [21].
Liver Organoids
The liver is predominantly composed of epithelial cells, including hepatocytes and cholangiocytes, which perform essential metabolic and secretory functions for human physiology and homeostasis [22]. Historically, liver development research has relied primarily on animal models, which can exhibit significant biological differences from the human body. Recently, researchers have succeeded in maintaining the long-term culture of hepatocytes, preserving their morphological and functional characteristics [23]. Liver organoids serve as effective models for studying human prenatal development, tissue maintenance, and pathology.
Initially, liver organoids were used to model monogenic liver diseases, but recent studies have demonstrated their application in analyzing complex acquired liver diseases, such as primary liver cancer (PLC). Conventional studies of PLC have been limited by the use of two-dimensional cell culture and transgenic animal models, which cannot accurately simulate tumor tissue structure or are time-consuming and expensive [24]. However, a research group has successfully cultivated primary liver tumor organoids (also known as tumoroids) using tumor tissue specimens from 8 PLC patients, preserving the original tissue characteristics even after prolonged culture time. The transplantation of tumoroids into immunocompromised mice showed that they retained the metastatic potential of the original tumor [25]. With the ability to continuously differentiate and expand, liver organoids have the potential to serve as an alternative source of transplanted cells, resolving the challenge of donor shortages and limitations of immunosuppressive therapy in orthotopic liver transplantation [26]. These studies demonstrate the potential of liver organoids to shed light on the key features of liver-related diseases and provide deeper insight into disease initiation and progression.
Kidney Organoids
The kidney is a very complex tissue that already has more than 25 different cell types after birth. The functions of the kidney include fluid homeostasis, removal of nitrogenous waste from the body, and regulation of blood pressure [27]. The gradual development of kidney organoids started in early 2014, accompanied by the publication of a series of articles. According to Taguchi's article, a method for culturing kidney organoids has been identified for the first time. The article carefully describes that mesenchymal cells can be induced to become nephrons, the latter of which also contains glomeruli, and can be a source of Wnt signaling [28]. Kidney organoids can now mimic the cellular complexity of the kidney and have great potential for being used in studying human diseases in vitro.
Kidney organoids are mainly used to study the regenerative medicine of the kidney and the establishment of disease models. Recent advances have revealed the generation of nephron progenitors from human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs). Therefore, human induced pluripotent stem cells (hiPSC) can be easily differentiated from tissues of patients with kidney disease. After inducing and culturing kidney organoids, specific models of kidney disease patients can be established [29]. Furthermore, kidney organoids that are cultured using HPSCs can be combined with bioengineered kidneys, a strategy that will possibly lead to the replacement of current dialysis and kidney transplantation therapies.
Lung Organoids
The respiratory system, which is comprised of the lungs and trachea, originated from the endoderm of the foregut [30]. The main function of the lungs is to facilitate gas exchange. The gas exchange in the lungs is mainly carried out in the alveolar epithelium, which accounts for more than 99% of the inner surface area of the lung. The alveolar epithelium is mainly composed of alveolar epithelial type I cells (Alveolar type 1, AT1) and alveolar epithelial type II cells (Alveolar type 2, AT2). AT1 cells are flat and closely connected with the pulmonary capillary plexus. They are responsible for the gas-liquid exchange. AT2 cells are cuboid and they secrete a pulmonary surfactant, which reduces alveolar surface tension, in addition to maintaining the alveolar shape. The airways are mainly composed of goblet, ciliated, secretory, and basal cells.
The concept of mouse lung organoids was proposed by Aaron E. Freeman in 1980 [31]. Although cultured organoids are not suitable for human research because they cannot simulate the real internal environment, they also lay a solid foundation for the cultivation of organoids in vitro. In recent years, researchers successfully cultivated airway organoids that can be expanded for long periods without losing the main characteristics of epithelial cells in vivo [32]. This makes these organoids a powerful tool for modeling lung diseases and studying molecular mechanisms.
The latest study shows that researchers successfully grew lung cancer organoids using tissue samples from lung cancer patients, prior to identifying cancer-associated gene mismatches and oncogenic mutations in primary tumor tissue [32]. These studies accurately highlighted the histopathology and mutation status of primary lung cancer, also contributed to xenotransplantation and in vitro drug screening. On the other hand, fibrosis-limited disease features were also found in airway organoids from cystic fibrosis patients. Airway organoids provide new options for in vitro functional models of cystic fibrosis [32].
Gastrointestinal Organoids
The main functions of the gastrointestinal (GI) tract are digestion, absorption, excretion, and protection. The stomach and intestines are fundamentally responsible for digestion and absorption in the body [33]. Until the advent of GI organoids, cell lines and explanted tissues have been used for modeling. However, both approaches have significant limitations [34]. For instance, the immortalization of our intestinal cell lines is usually performed using malignant tissue specimens or by the introduction of oncogenes, and this limits their application in redevelopment, physiology, and regeneration research [35]. Intestine and stomach organoids in the gastrointestinal tract were first discovered in 2009 and 2010, respectively [36, 37].
Today, intestinal organoid technology, including classical organoid models, has great application prospects in medical research and the development of new therapies. For example, Toshiro Sato's research group analyzed the mutations of ulcerative colitis epithelial cells using organoids, which can also be used to analyze the molecular basis of the chronic inflammatory microenvironment [38]. Additionally, gastrointestinal organoids are also applicable in cancer research. Interestingly, cancer stem cells can also cultivate organoids because of their self-renewal and differentiation capabilities. The same Toshiro Sato research group established a colorectal tumor organoid library, which is made up of 55 different tumor organoid clones [39]. These can be further used to culture and compare the differences between precancerous lesion cells and tumor ones, in human patients. Second, intestinal organoids are also of great importance in regenerative medicine. In 2012, researchers demonstrated that colon organoids that were differentiated from adult stem cells could regenerate a damaged epithelium after transplantation into mice [40]. All these provide important strategies for the treatment of various diseases such as short bowel syndrome or intestinal failure.
Retinal Organoids
Vision is one of the senses that are of paramount importance in daily life activities. The retina is known as the window to the brain, and dysfunction of its neurons is the leading cause of blindness [41]. Using animal models to study the overall morphology, physiology, and molecular characteristics of the human retina is impractical. Therefore, there is a great need for in vitro models that are based on human stem cells to make studying the retina feasible (Table 1).
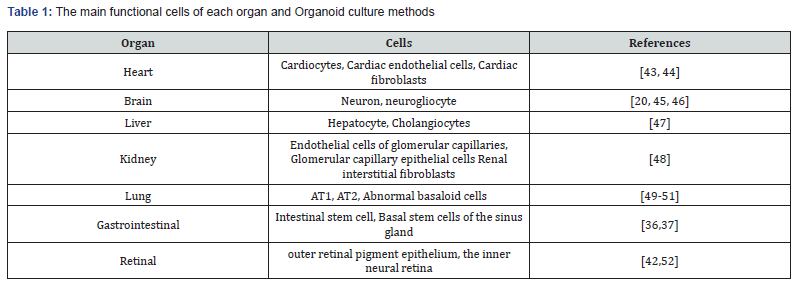
In 2011, Sasai's group cultured retinal organoids in vitro from mouse embryonic stem cells [42]. This is regarded as a milestone toward the cultivation of central nervous system organoids. Retinal organoids can be used as disease models as was the case when they were used for disease modeling in patients with mutations in the splicing factor PRPF31 (RP11) [43]. Moreover, researchers are continuously improving retinal organoid culture methods to obtain small molecular compounds for the treatment of the optic nervous system.
Discussion and Outlook
Organoids provide a powerful tool for researchers, as they can be used to study complex biological processes in a controlled, laboratory setting and can help to advance our understanding of human physiology and disease. Although organoid technology is still in its infancy, organoid technology has great potential application in bioregenerative medicine, precision medicine, and drug toxicity disciplines. There are still some challenges for organoids in clinical translational research, including limited maturity and complexity in mimicking actual organs, heterogeneity within the organoids, the need for standardized protocols, and high costs. However, the rapid technological advances in this field increase the hope that human organoid systems will offer unprecedented opportunities for improving human health.
Author Contributions
Conceptualization, J.W. and writing original draft preparation, R.W.; writing review, R.W.; editing review, R.W. and Y.Z.; visualization, R.W.; supervision Y.Z. and J.W. project administration, J.W.; funding acquisition, J.W.
References
- Schutgens F, Clevers H (2020) Human Organoids: Tools for Understanding Biology and Treating Annu Rev Pathol 15: 211-234.
- Pane LS, Moretti A, R Chien KR, Ludwig Laugwitz K, et al. (2013) Embryonic heart progenitors and cardiogenesis. Cold Spring Harb Perspect Med 3(10):
- Xin M, Olson EN, Bassel-Duby R (2013) Mending broken hearts: cardiac development as a basis for adult heart regeneration and Nat Rev Mol Cell Biol 14(8): 529-541.
- Gintant GA, Limberis JT, McDermott JS, Wegner CD, Cox BF, et al. (2001) The canine Purkinje fiber: an in vitro model system for acquired long QT syndrome and drug-induced J Cardiovasc Pharmacol 37(5): 607-618.
- Kramer J, Obejero-Paz CA, Myatt G, Kuryshev YA, Bruening-Wright A, et al. (2013) MICE models: superior to the HERG model in predicting Torsade de Pointes. Sci Rep 3:
- Hay M, Thomas DW, Craighead JL, Economides C, Rosenthal J (2014) Clinical development success rates for investigational drugs. Nat Biotechnol 32(1): 40-51.
- Liang P, Lan F, Lee AS, Gong T, Sanchez-Freire V, et (2013) Drug screening using a library of human induced pluripotent stem cell-derived cardiomyocytes reveals disease-specific patterns of cardiotoxicity. Circulation 127(16): 1677-1691.
- Frommeyer G, Eckardt L (2016) Drug-induced proarrhythmia: risk factors and electrophysiological Nat Rev Cardiol 13(1): 36-47.
- Zhao Y, Rafatian N, Feric NT, Cox BJ, Aschar-Sobb R, et al. (2019) A Platform for Generation of Chamber-Specific Cardiac Tissues and Disease Cell 176(4): 913-927.e18.
- Li RA, Keung W, Cashman TJ, Backeris PC, Johnson BV, et al. (2018) Bioengineering an electro-mechanically functional miniature ventricular heart chamber from human pluripotent stem Biomaterials 163: 116-127.
- Hofbauer P (2021) Cardioids reveal self-organizing principles of human cardiogenesis. Cell 184(12): 3299-3317.
- Lewis-Israeli YR, Wasserman AH, Gabalski MA, Volmert BD, Ming, et al. (2021) Self-assembling human heart organoids for the modeling of cardiac development and congenital heart Nat Commun 12(1): 5142.
- Cakir B, Xiang Y, Tanaka Y, Kural MH, Parent M, et al. (2019) Engineering of human brain organoids with a functional vascular-like system. Nat Methods 16(11): 1169-1175.
- We Z, Nam Nguyen H, Guo Z, Lalli MA, Wang X, et al. (2014) Synaptic dysregulation in a human iPS cell model of mental disorders. Nature 515(7527): 414-418.
- Brennand KJ, Simone A, Jou J, Gelboin-Burkhart C, Tran N, et al. (2011) Modelling schizophrenia using human induced pluripotent stem cells. Nature 473(7346): 221-225.
- Sakaguchi H, Kadoshima T, Soen M, Narii N, Yoshihito Ishida, et (2015) Generation of functional hippocampal neurons from self-organizing human embryonic stem cell-derived dorsomedial telencephalic tissue. Nat Commun 6: 8896.
- Monzel AS, Smits LM, Hemmer K, Hachi S, Moreno EL, et al. (2017) Derivation of Human Midbrain-Specific Organoids from Neuroepithelial Stem Stem Cell Reports 8(5): 1144-1154.
- Qian X, Nguyen HN, Song MM, Hadiono C, Ogden SC, et al. (2016) Brain-Region-Specific Organoids Using Mini-bioreactors for Modeling ZIKV Cell 165(5): 1238-1254.
- Muguruma K, Renner M, Martin CA, Wenzel D, Louise S Bicknell, et al. (2015) Self-organization of polarized cerebellar tissue in 3D culture of human pluripotent stem Cell Rep 10(4): 537-550.
- Lancaster MA, Renner M, Martin CA, Wenzel D, Bicknell LS, et (2013) Cerebral organoids model human brain development and microcephaly. Nature 501(7467): 373-739.
- Sidhaye J, Knoblich JA (2021) Brain organoids: an ensemble of bioassays to investigate human neurodevelopment and Cell Death Differ 28(1): 52-67.
- Prior N, Inacio P, Huch M, Liver organoids: from basic research to therapeutic applications (2019) Gut 68(12): 2228-2237.
- Hu H, Gehart H, Artegiani B, LÖpez-Iglesias C, Dekkers F, et al. (2018) Long-Term Expansion of Functional Mouse and Human Hepatocytes as 3D Cell 175(6): 1591-1606.e19.
- He S, Hu Bo, Chao Li, Ping Lin, Wei-Guo Tang, et (2018) PD Xliver: a database of liver cancer patient derived xenograft mouse models. BMC Cancer 18(1): 550.
- Huch M, Gehart H, van Boxtel R, Hamer K, Blokzijl F, et al (2015) Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell160(1-2): 299-312.
- Assy N, Adams PC, Myers P, Simon V, Ghent CN, et (2007) A randomised controlled trial of total immunosuppression withdrawal in stable liver transplant recipients. Gut, 2007. 56(2): 304-306.
- Little MH, A.N. Combes AN (2019) Kidney organoids: accurate models or fortunate Genes Dev 33(19-20): 1319-1345.
- Taguchi A, Kaku Y, Ohmori T, Sharmin S, Ogawa M, et al. (2014) Redefining the in vivo origin of metanephric nephron progenitors enables generation of complex kidney structures from pluripotent stem cells. Cell Stem Cell 14(1): 53-67.
- Takahashi, K., et al., Induction of pluripotent stem cells from adult human fibroblasts by defined Cell, 2007. 131(5): 861-72.
- Morrisey EE, L. Hogan BL (2010) Preparing for the first breath: genetic and cellular mechanisms in lung development. Dev Cell 18(1): 8-23.
- Yoshida,Y, Hilborn V, Freeman AE (1980) Fine structural identification of organoid mouse lung cells cultured on a pigskin In Vitro 16(11): 994-1006.
- Sachs N, Papaspyropoulos A, Zomer-van Ommen D, Heo I, Böttinger L et al. (2019) Long-term expanding human airway organoids for disease modeling. Embo j 38(4): e100300.
- Cheng LK, Grady GO, Du P, Egbuji JU, Windsor JA, et (2010) Gastrointestinal system. Wiley Interdiscip Rev Syst Biol Med 2(1): 65- 79.
- Pageot LP, N Perreault, N Basora, C Francoeur, P Magny, J F Beaulieu JF, et al. (2000) Human cell models to study small intestinal functions: recapitulation of the crypt-villus axis. Microsc Res Tech 49(4): 394-406.
- Paul EC, Hochman J, Quaroni A (1993) Conditionally immortalized intestinal epithelial cells: novel approach for study of differentiated Am J Physiol 265(1 Pt 1): 266- 278.
- Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N et (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459(7244): 262-265.
- Barker N, Huch M, Kujala P, van de Wetering M, Hugo J Snippert HJ, et al. (2010) Lgr5(+ve) stem cells drive self-renewal in the stomach and build long-lived gastric units in Cell Stem Cell 6(1): 25-36.
- Nanki K, Fujii M, Shimokawa M, Matano M, Nishikori S, et (2020) Somatic inflammatory gene mutations in human ulcerative colitis epithelium. Nature 577(7789): 254-259.
- Fujii M, Shimokawa M, Date S, Takano Ai,Mami Matano M, et al. (2016) A Colorectal Tumor Organoid Library Demonstrates Progressive Loss of Niche Factor Requirements during Cell Stem Cell 18(6): 827-838.
- Yui S, Nakamura T, Sato T, Yasuhiro Nemoto, Tomohiro Mizutani, et (2012) Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5⁺ stem cell. Nat Med 18(4): 618-623.
- Kruczek K, Swaroop A (2020) Pluripotent stem cell-derived retinal organoids for disease modeling and development of Stem Cells 38(10): 1206-1215.
- Eiraku M, Takata N, Ishibashi H, Kawada M, Sakakura E, et (2011) Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472(7341): 51-56.
- Buskin A, Zhu L, Chichagova V, Basu B, Mozaffari-Jovin S, et (2018) Disrupted alternative splicing for genes implicated in splicing and ciliogenesis causes PRPF31 retinitis pigmentosa. Nat Commun 9(1): 4234.
- Giacomelli E, Meraviglia V, Campostrini G, Cochrane A, Cao Xu, et al. (2020) Human-iPSC-Derived Cardiac Stromal Cells Enhance Maturation in 3D Cardiac Microtissues and Reveal Non-cardiomyocyte Contributions to Heart Disease. Cell Stem Cell, 26(6): 862-879.e11.
- Drakhlis L, Biswanath S, Clara-Milena F, Lupanow V, Teske J, et (2021) Human heart-forming organoids recapitulate early heart and foregut development. Nat Biotechnol 39(6): 737-746.
- Kadoshima T, Sakaguchi H, Nakano T, Soen M, Ando S, et al. (2013) Self-organization of axial polarity, inside-out layer pattern, and species- specific progenitor dynamics in human ES cell-derived neocortex. Proc Natl Acad Sci U S A 110(50): 20284-20289.
- Trujillo CA, Gao R, Negraes PD, Jing Gu, Justin Buchanan, et (2019) Complex Oscillatory Waves Emerging from Cortical Organoids Model Early Human Brain Network Development. Cell Stem Cell 25(4): 558-569.e7.
- Huch M, Gehart H, van Boxtel R, Hamer K, Francis Blokzijl F, et (2015) Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 160(1-2): 299-312.
- Takasato M, X Er P, Chiu HS, Maier B, Baillie GJ, et al. (2015) Kidney organoids from human iPS cells contain multiple lineages and model human Nature 526(7574): 564-568.
- Dye BR, Hil DRl, Ferguson MAH, Hwai Tsai Yu, Nagy MS, et al. (2015) In vitro generation of human pluripotent stem cell derived lung organoids Elife 4.
- Chen YW, Huang SX, Toste de Carvalho ALR, Siu-Hong H, Naimul Islam M, et (2017) A three-dimensional model of human lung development and disease from pluripotent stem cells. Nat Cell Biol 19(5): 542-549.
- Kuwahara A, Ozone C, Nakano T, Saito K, Eiraku M, et (2015) Generation of a ciliary margin-like stem cell niche from self-organizing human retinal tissue. Nat Commun 6: 6286.






























