Successful Treatment by Mistletoe Viscum Album Extract of a Patient with Recurrent Triple Negative Breast Cancer who declined Chemotherapy
Parameswara Vinay K and Hancock Mark J*
Humanizing Medicine, Georgia
Submission: October 12, 2021; Published: November 19, 2021
*Corresponding author: Mark Hancock, Humanizing Medicine, 135 Maple St Building A, Decatur, Georgia
How to cite this article: Parameswara Vinay K, Hancock Mark J. Successful Treatment by Mistletoe Viscum Album Extract of a Patient with Recurrent Triple Negative Breast Cancer who declined Chemotherapy. J Complement Med Alt Healthcare. 2021; 11(3): 555815. DOI: 10.19080/JCMAH.2021.11.555815
Abstract
Triple negative breast cancer (TNBC) is an aggressive breast cancer with a high incidence of recurrence and poor prognosis even with the full spectrum of standard oncology treatments. Ever since its introduction about 100 years ago for cancer therapy, mistletoe viscum album extract (VAE) has been widely used in Europe as a popular complementary therapy for many cancers. We present a case of a patient diagnosed with stage 3c (T3N3bM0(i+)) TNBC who declined chemotherapy after a recurrence 2 years later. The patient received subcutaneous, intratumoral as well as intravenous VAE therapy for her recurrence. Her breast masses and lymph node sizes dramatically decreased under ultrasound examination within 2 months. After nearly three years since the tumor resolution, she is currently being managed on weekly subcutaneous VAE as well as intravenous VAE once in 6-8 weeks. She has excellent quality of life, no sign of tumor recurrence or other adverse side effects from her treatments. Though we believe the best outcomes in oncology are by integrating complementary and conventional care together, this case shows that VAE can have standalone effectiveness at least in some cases. Use of VAE to augment conventional treatments should be considered to bolster breast cancer care.
Keywords: Mistletoe; Viscum Album Extract; Breast Cancer; Triple Negative Breast Cancer
Introduction
Breast cancer continues to be the most common cancer diagnosed and the leading cause of death among women globally [1]. Breast cancers that do not express the estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) are known as triple negative breast cancers (TNBC). TNBC accounts for 10-15 % of all breast cancers subtypes and is very aggressive, with a high risk of multiple, distant metastasis and recurrence [2]. The median survival for patients with metastatic TNBC is only 13.3 months with treatment [3] and the lack of targeted specific therapy poses challenges to finding cost-effective regimens with minimal side effects that ensure a good quality of life (QOL). Stage 3c TNBC treated only with surgery has an expected median survival of about 30% [4]. Over the years, complementary and alternative medicines (CAM) have been playing more and more significant role in cancer therapy throughout the world. One such CAM is Mistletoe viscum album extract (VAE), which was first introduced for cancer therapy about 100 years ago. An increasing number of studies have reported improvement in QOL and anticancer activity of VAE [5-10]. Cancer trials with VAE are underway to study the overall survival and health related QOL in Europe [11]. Studies outside the USA have shown improvement in breast cancer as well with VAE [12-15].
Here, we report a case of a patient with stage 3c TNBC who declined chemotherapy after her recurrence. She was treated with subcutaneous (SC), Intratumoral (IT) and intravenous (IV) VAE.
Case Report
A 47-year-old woman with a 2-year past history of stage 2 TNBC of the left breast presented with a large 7 cm left breast mass and axillary lymph node enlargement. Her past history is significant for 14 pack years of tobacco, BMI fluctuating between 25 and 30 for past 20 years and she was not on any prescription medications. Her only pregnancy resulted in emergency Cesarian section and she exclusively breast fed her son for 8 months. After she turned 40 years, she underwent annual screening mammograms for 2 years only. As soon as she turned 45 years, she felt a lump in her left breast. Based on her symptoms and third mammogram, she underwent an excisional biopsy of her breast mass. The pathology report showed a 3 cm triple negative invasive ductal carcinoma, and she was diagnosed with stage 2a TNBC.
She refused further diagnostic, surgical and therapeutic advice from her oncology treatment team. Instead, she traveled to Mexico for a dietary and detoxification program offered at a CAM hospital. While she stayed in Mexico for a period of 28 days, she received daily 30-100 g of IV Vitamin C (IVC). She was also given 4 rounds of Autologous Dendritic cell therapy, Autologous bone marrow stem cell transplant, multiple rounds of Hyperthermia FAR infrared therapy and Hyperbaric oxygen chamber therapy as well as multiple daily herbal supplements along with other CAM treatments. Since her return, she has undergone periodic laboratory studies and regular mammograms under the care of a USA physician. She has been taking weekly IVC and daily CAM supplements. Over a period of 18 months, she remained symptom free and her Biocept circulating tumor cells had been between 10 and 13. Around her 47th birthday, her mammogram showed suspicious lesion on her left breast. Patient once again refused conventional chemotherapy advised by her oncology treatment team and this time, she opted to come to our clinic instead of CAM hospital in Mexico.
Patients Positron emission tomography computed tomography scan (PET/CT) showed a centrally necrotic 4.2 x 6.7 cm mass with SUV 7.4 in her left breast. Mediastinal and axillary lymph nodes were enlarged with focal intense FDG uptake. She started her IV infusions of VAE as well as IT VAE. As anticipated, a fever response was observed 24 hours after every IT and IV VAE infusions. She tolerated the procedure well. She received a total of 9 IT VAE over a period of about 2 months. The dosage was gradually increased from 10 mg to 200 mg. During this time period she received 7 SC 10-20 mg VAE injections as well as 9 IV VAE. The dosage of IV VAE increased from 60 to 100 mg. Over the course of 2 months patient started feeling better and her health QOL improved. Her Ultrasound examination revealed the mass on her left breast to have shrunk to 4.3 x 4.8 cm. (Table 1)
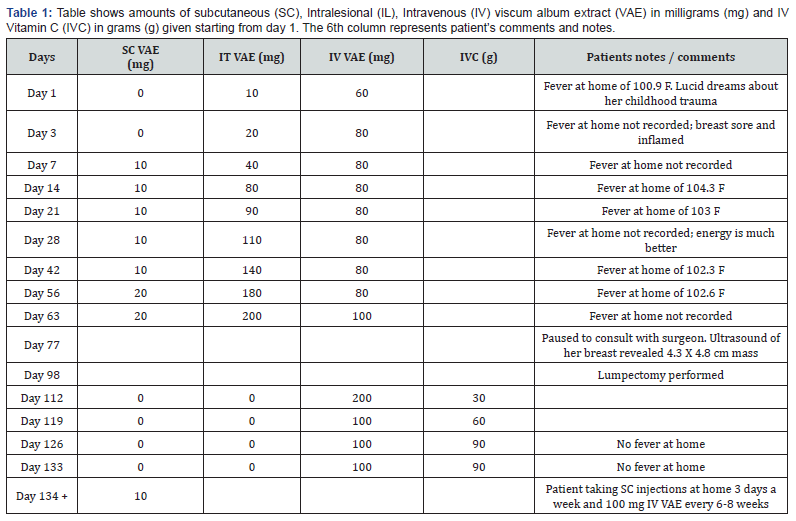
The VAE injections and infusions were paused for about 2 months in anticipation of surgery. A left breast lumpectomy was performed. The gross pathology report showed a well circumscribed cavity with necrotic center measuring 2.8 x 2.2 cm. Histopathology results showed breast tissue with abscess cavity, abundant necrosis, numerous histiocytes, dense fibrosis and fibroblast proliferation admixed with recent and old blood. Remaining breast tissue shows fibrocystic changes. There was no evidence of malignancy. Her initial tumor markers, CA 15-3 was 29 U/ml and CA 27.29 was 44 U/ml and her Biocept circulating tumor cells was 19. One year after her VAE was started, her carcinoma antigens, CA 15-3 were 9 U/ml and CA 27.29 to 21 U/ml, remaining stable 3 years later at 7 and 17 respectively Two months after the 9th dose of IV VAE, she resumed weekly IV VAE for about 4 weeks (Table 1). Since there was no tumor, IL VAE was not instituted. Along with her weekly IV VAE patient also received weekly IVC which was gradually increased from 30 g to 90 g. Patient continued SC VAE after the IV VAE were stopped.
Currently, about 3 years later, patient injects 10 mg SC VAE 3 days a week and once in 6-8 weeks she receives 100 mg of IV VAE. She undergoes periodic Ultrasound of her breast and axilla. Her lymph nodes continue to be between 1 and 2 cm in their largest diameter. Otherwise, she has been tumor free, symptom free and does not complain of any side effects.
Discussion
Triple negative breast cancer (TNBC) is a very aggressive subtype of breast cancer associated with a high recurrence rate and generally poor prognosis especially when patients don’t undergo chemotherapy. Chemotherapy has been the main treatment option and within the past decade repurposed as well as new drugs like polyadenosine diphosphate-ribose polymerase inhibitors are being tried [16-18]. Despite this, the treatment options for TNBC remain limited. When patients undergo aggressive chemotherapy for TNBC to improve overall survival, practical hurdles namely, medication and treatment costs, hospitalizations and treatment associated side effects and reduction in QOL can be frustrating. Not infrequently, patients seek out CAM and sometimes without disclosing that information to their oncology treatment team [19,20]. Unrestricted CAM usage could interact in unpredictable ways with conventional therapies. Very few of the thousands of CAM have been studied for safety and efficacy.
VAE was first used for cancer therapy over a 100 years ago as a part of Anthroposophic Medicine [21-24]. Gradually, over a century, numerous in vitro, in vivo and human studies have been published. Mistletoe lectins are the subject of most studies [25] and have both pro-apoptotic effects as well as a proliferative effect on natural killer (NK) and T Lymphocytes. Viscotoxins are another component that directly causes tumor cell death [26,27]. Other components of VAE have favorable effects on inflammation, endorphins and VEGF and thus impact the broader “terrain” of the patient with cancer.
Although some authors question the survival and QOL advantages to prescribing VAE [28,29] numerous studies and review articles indicate otherwise [30-33]. In breast cancer treatment, VAE has been particularly beneficial in improving QOL and overall survival [11-14,34,35,36]. Up to 75% of cancer patients use VAE alongside their therapy in Europe. VAE is most often used as a SC injected therapy that causes a small ~3cm area of erythema and swelling with each injection. There may be a slight elevation of body temperature of one degree F but usually no other side effects. Mistletoe Fever Induction Therapy (MFIT) is safe [37] and used routinely by several integrative hospitals in Europe in selected patients who are robust enough to have a high fever up to 1040F [38]. Specific doses and routes of high lectin mistletoe are used in MFIT. Hyperthermia and fever have a known anticancer effect as well as upregulating the immune system. Intratumoral injection of VAE is also performed in selected patients, usually in the setting of MFIT. The unveiling of the tumor during high fever while simultaneously creating a beacon for cancer killing NK and T-cells may be the explanation for several reported local tumor responses as well as abscopal effects, where other distant tumors also responded to this therapy [39,40].
Intratumoral injection of VAE is also performed in selected patients, usually in the setting of MFIT. The unveiling of the tumor during high fever while simultaneously creating a beacon for cancer killing NK and T-cells may be the explanation for several reported local tumor responses as well as abscopal effects [41,42] where other distant tumors also responded to this therapy [43].
Our patient had TNBC that recurred after 2 years. She declined any conventional chemotherapy offered by her oncology treatment team due to her personally held beliefs. She came to our clinic with the intention of using VAE. She responded appropriately to the VAE injections by developing a fever that lasted for about a day. She noticed improvement in her symptoms by the 3rd dose of VAE injections. Gradually, in about 2 months of VAE therapy, her breast mass shrunk dramatically, QOL improved, and her breast mass was cancer free. She is currently on maintenance VAE injections and has remained cancer free for about 3 years.
Conclusion
We report a case of recurrent TNBC in a patient who declined conventional medicine. It is not very rare to see patients who decline conventional treatments for cancer and request only to be treated with CAM- such cases present an ethical dilemma for clinicians, including the authors. We find that the best ethical solution is to form a bridge for these patients using CAM ideally alongside conventional therapy. With this case we aim to inform clinicians about VAE used widely to treat cancers in other countries especially Germany. In particular, we have shown, survival and cancer remission, as well as improvement in QOL in a patient with recurrent TNBC.
Conflicts of Interest
The authors declare there is no conflict of interest regarding the publication of this paper.
References
- Torre LA, Siegel RL, Ward EM, Jemal A (2016) Global Cancer Incidence and Mortality Rates and Trends-An Update. Cancer Epidemiol Biomarkers Prev 25(1): 16-27.
- Dent R, Trudeau M, Pritchard KI, Hanna WM, Kahn HK, et al. (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13(15 Pt 1): 4429-4434.
- Kassam F, Enright K, Dent R, Dranitsaris G, Myers J, et al. (2009) Survival outcomes for patients with metastatic triple-negative breast cancer: implications for clinical practice and trial design. Clin Breast Cancer 9(1): 29-33.
- https://breast.predict.nhs.uk/tool last accessed on May 8, 2021.
- Kienle GS, Berrino F, Büssing A, Portalupi E, Rosenzweig S, et.al (2003) Mistletoe in cancer -a systematic review on controlled clinical trials 8(3): 109-119.
- Kienle GS, Kiene H (2007) Complementary cancer therapy: a systematic review of prospective clinical trials on anthroposophic mistletoe extracts 12(3): 103-119.
- Melzer J, Iten F, Hostanska K, Saller R (2009) Efficacy and safety of mistletoe preparations (Viscum album) for patients with cancer diseases. Eur J Med Res 16(4): 217-226.
- Schad F, Thronicke A, Steele ML, Antje Merkle, Burkhard Matthes, et al. (2018) Overall survival of stage IV non-small cell lung cancer patients treated with Viscum album L. in addition to chemotherapy, a real-world observational multicenter analysis. PLoS One 13(8): e0203058.
- Tröger W, Galun D, Reif M, Schumann A, Stanković N, et al (2013) Viscum album [L.] extract therapy in patients with locally advanced or metastatic pancreatic cancer: a randomised clinical trial on overall survival. Eur J Cancer 49(18): 3788-3797.
- Loef M, Walach H (2020) Quality of life in cancer patients treated with mistletoe: a systematic review and meta-analysis. BMC Complement Med Ther 20(1): 227.
- Wode K, Hök Nordberg J, Kienle GS, Elander NO, Bernhardson BM, et al. (2020) Efficacy of mistletoe extract as a complement to standard treatment in advanced pancreatic cancer: study protocol for a multicentre, parallel group, double-blind, randomised, placebo-controlled clinical trial (MISTRAL). Trials 21(1): 783.
- Marvibaigi M, Supriyanto E, Amini N, Abdul Majid FA, Jaganathan SK (2014) Preclinical and clinical effects of mistletoe against breast cancer. Biomed Res Int 2014: 785479.
- Piao BK, Wang YX, Xie GR, Mansmann U, Matthes H, et al. (2004) Impact of complementary mistletoe extract treatment on quality of life in breast, ovarian and non-small cell lung cancer patients. A prospective randomized controlled clinical trial. Anticancer Res 24(1): 303-309.
- Grossarth-Maticek R, Ziegler R (2006) Prospective controlled cohort studies on long-term therapy of breast cancer patients with a mistletoe preparation (Iscador). Forsch Komplementmed 13(5): 285-292.
- Tröger W, Jezdić S, Zdrale Z, Tišma N, Hamre HJ (2009) Quality of life and neutropenia in patients with early stage breast cancer: a randomized pilot study comparing additional treatment with mistletoe extract to chemotherapy alone. Breast Cancer (Auckl) 6(3): 35-45.
- Caparica R, Lambertini M, de Azambuja E (2019) How I treat metastatic triple-negative breast cancer. ESMO Open 4(Suppl 2): e000504.
- Rangarao R, Smruti BK, Singh K, Gupta A, Batra S, et al. (2018) Practical consensus recommendations on management of triple-negative metastatic breast cancer. South Asian J Cancer 7(2): 127-131.
- Spini A, Donnini S, Pantziarka P, Crispino S, Ziche M (2020) Repurposing of drugs for triple negative breast cancer: an overview. Ecancermedical science 14: 1071.
- Buckner CA, Lafrenie RM, Dénommée JA, Caswell JM, Want DA (2018) Complementary and alternative medicine use in patients before and after a cancer diagnosis. Curr Oncol 25(4): e275-e281.
- Alsharif F (2021) Discovering the Use of Complementary and Alternative Medicine in Oncology Patients: A Systematic Literature Review. Evid Based Complement Alternat Med 2021: 6619243.
- Bussing A (2000) Mistletoe The genus Viscum. Hardwood Academic Publishers, Amsterdam, United Kingdom.
- Kienle GS, Albonico HU, Baars E, Hamre HJ, Zimmermann p, et al. (2013) Anthroposophic Medicine: An Integrative Medical System Originating in Europe. Glob Adv Health Med 2(6): 20-31.
- Bartelme RR (2020) Anthroposophic Medicine: A Short Monograph and Narrative Review-Foundations,Essential Characteristics, Scientific Basis, Safety, Effectiveness and Misconceptions. Glob Adv Health Med 9: 2164956120973634.
- Martin D (2020)100-Year Anniversary of Anthroposophic Medicine as an Integrative Medical System. Complement Med Res 27(6): 375-378.
- Nazaruk J, Orlikowski P (2016) Phytochemical profile and therapeutic potential of Viscum album L. Nat Prod Res 30(4): 373-385.
- Szurpnicka A, Kowalczuk A, Szterk A (2020) Biological activity of mistletoe: in vitro and in vivo studies and mechanisms of action. Arch Pharm Res 43(6): 593-629.
- Elluru S, Duong Van Huyen JP, Delignat S, Prost F, Bayry J, et al. (2006) Molecular mechanisms underlying the immunomodulatory effects of mistletoe (Viscum album ) extracts Iscador. Arzneimittelforschung 56(6A): 461-466
- Freuding M, Keinki C, Micke O, Buentzel J, Huebner J (2019) Mistletoe in oncological treatment: a systematic review : Part 1: survival and safety. J Cancer Res Clin Oncol 145(3): 695-707.
- Freuding M, Keinki C, Kutschan S, Micke O, Buentzel J, et al. (2019) Mistletoe in oncological treatment: a systematic review : Part 2: quality of life and toxicity of cancer treatment. J Cancer Res Clin Oncol 145(4): 927-939.
- Evans M, Bryant S, Huntley AL, Feder G (2016) Cancer Patients' Experiences of Using Mistletoe (Viscum album): A Qualitative Systematic Review and Synthesis. J Altern Complement Med 22(2): 134-144.
- Sunjic SB, Gasparovic AC, Vukovic T, Weiss T, Weiss ES, et al. (2015 Sep) Adjuvant Cancer Biotherapy by Viscum Album Extract Isorel: Overview of Evidence Based Medicine Findings. Coll Antropol 39(3): 701-708.
- Kienle GS, Kiene H (2010)Review article: Influence of Viscum album L (European mistletoe) extracts on quality of life in cancer patients: a systematic review of controlled clinical studies. Integr Cancer Ther 9(2): 142-157.
- Pelzer F, Tröger W, Nat Rer (2018) Complementary Treatment with Mistletoe Extracts During Chemotherapy: Safety, Neutropenia, Fever, and Quality of Life Assessed in a Randomized Study. J Altern Complement Med 24(9-10): 954-961.
- Beuth J, Schneider B, Schierholz JM (2008) Impact of complementary treatment of breast cancer patients with standardized mistletoe extract during aftercare: a controlled multicenter comparative epidemiological cohort study. Anticancer Res 28(1B): 523-527.
- Semiglazov VF, Stepula VV, Dudov A, Schnitker J, Mengs U (2006) Quality of life is improved in breast cancer patients by Standardised Mistletoe Extract PS76A2 during chemotherapy and follow-up: a randomised, placebo-controlled, double-blind, multicentre clinical trial. Anticancer Res 26(2B): 1519-1529.
- Oei SL, Thronicke A, Kröz M, von Trott P, Schad F, et al (2020) Impact of Oncological Therapy and Viscum album L Treatment on Cancer-Related Fatigue and Internal Coherence in Nonmetastasized Breast Cancer Patients. Integr Cancer Ther 19: 1534735420917211.
- Steele ML, Axtner J, Happe A, Kröz M, Matthes H, et al. (2015) Use and safety of intratumoral application of European mistletoe (Viscum album L) preparations in Oncology. Integr Cancer Ther 14(2): 140-148.
- Orange M, Lace A, Fonseca MP, von Laue BH, Geider S, et al. (2012) Durable Regression of Primary Cutaneous B-Cell Lymphoma Following Fever-inducing Mistletoe Treatment: Two Case Reports. Glob Adv Health Med 1(1): 18-25.
- Mallory M, Gogineni E, Jones GC, Greer L, Simone CB (2016) Therapeutic hyperthermia: The old, the new, and the upcoming. Crit Rev Oncol Hematol 97: 56-64.
- Crestani A, Benoit L, Touboul C, Pasquier J (2020) Hyperthermic intraperitoneal chemotherapy (HIPEC): Should we look closer at the microenvironment? Gynecol Oncol 159(1): 285-294.
- Orange M, Reuter U, Hobohm U (2016) Coley's Lessons Remembered: Augmenting Mistletoe Therapy. Integr Cancer Ther 15(4): 502-511
- Grass GD, Krishna N, Kim S (2016) The immune mechanisms of abscopal effect in radiation therapy. Curr Probl Cancer 40(1): 10-24.
- Büssing A (2006) Immune modulation using mistletoe (Viscum album) extracts Iscador. Arzneimittelforschung 56(6A): 508-515.






























