Acute Kidney Injury After Paediatric Cardiac Surgery with Extracorporeal Circulation: Incidence and Risk Factors? Prospective Observational Study in A Cardiac Centre In A Developing Country
Ba EB1*, Gaye I1, Diedhiou M2, Diagne PA1, Leye PA1, Ndiaye PI1, Sene MV3, Sene EB3, Diop U3, Diao EM3, Diene SC3 and Kane O1
1Faculty of Medicine of the Cheikh Anta Diop University, Senegal
2Faculty of Medicine of the Gaston Berger University of Saint Louis, Senegal
3Fann University Hospital Centre, Senegal
Submission: October 14, 2022; Published: January 25, 2023
*Corresponding author: Elhadji Boubacar BA, Faculty of Medicine of the Cheikh Anta Diop University, Senegal
How to cite this article: Ba EB, Gaye I, Diedhiou M, Diagne PA, Leye PA, et al. Acute Kidney Injury After Paediatric Cardiac Surgery with Extracorporeal Circulation: Incidence and Risk Factors? Prospective Observational Study in A Cardiac Centre In A Developing Country. J Anest & Inten care med. 2023; 12(4): 555841. DOI 10.19080/JAICM.2023.12.555841
Abstract
Introduction: Acute kidney injury is a clinico-biological syndrome that ranges in severity from Kidney attack to failure. The aim of this study was to determine the incidence and risk factors of AKI after paediatric cardiac surgery with extracorporeal circulation.
Methods: Prospective observational study, conducted over a period of 4 months at the cardiac centre of the CHU of Fann. The diagnosis of AKI was based on the modified RIFLE criteria for paediatrics.
Results: Our work involved 40 patients with a male predominance (sex ratio 1.8) and an average age of 7.2 years. We noted 02 types of surgery: congenital surgery (80%), valvular surgery (20%). All patients underwent scheduled surgery with a mean preoperative GFR of 130.2 ml/Kg/1.73 m2. The mean duration of extracorporeal circulation was 107.7 min. Postoperatively, 82.5% were placed on sympathomimetics, 12.5% had severe left ventricular systolic dysfunction, 27.5% had severe right ventricular systolic dysfunction and one quarter of the patients received a postoperative transfusion. The incidence of AKI was 30% with: 20% at risk, 7.5% with damage and only one patient in failure. Right ventricular systolic dysfunction and postoperative transfusion were the main risk factors. Mortality in our series was 10% and the dependence on AKI was significant (p=0.038).
Conclusion: The epidemiology of AKI after paediatric cardiac surgery depends on the definition used. The incidence is highly variable and depends on the epidemiological, intraoperative and postoperative characteristics of the patients.
Keywords: acute kidney injury, cardiac surgery, paediatric, extracorporeal circulation, incidence
Abbreviations: FIB: Fluid input-output balance, GFR: Glomerular filtration rate, ECC: Extracorporeal circulation, LVEF: Left ventricular ejection fraction, TAPSE: Tricuspid annular plane systolic excursion RIFLE: Risk, Injury, Failure, Loss of kidney function, and End-stage, kidney disease BES: Input-Output Balance Sheet, AKI: Acute kidney injury, KDIGO: Kidney disease improving global outcomes, ACEI: Angiotensin Converting Enzyme Inhibitor, IAC: Inter-atrial communication, VIC: Ventricular septal defect, AVC: Atrioventricular canal, T4F: Tetralogy of Fallot, APVR: Abnormal pulmonary venous return, MVR: Mitral valve replacement, PT: Tricuspid plasty, PM: Mitral plasty, RT: Tricuspid valve replacement, PDI type 5: Phospodiesterase type 5 inhibitors, SFAR : French Society of Anaesthesia and Intensive Care, SRLF: French-speaking resuscitation, society, FER: Formalised expert recommendations
Introduction
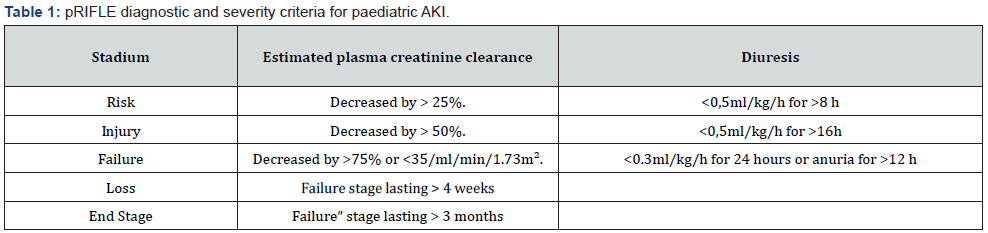
Acute kidney injury is a clinico-biological syndrome with multiple aetiologies and a range of severity from kidney attack to failure. Its identification and diagnosis of severity in children is based on the use of the paediatric modified RIFLE classification (pRIFLE) (Table 1): estimated creatinine clearance decreased by at least 25%, or diuresis < 0.5 ml/kg/h for 8 hours [1]. In this definition, estimated clearance is calculated according to the paediatric Schwartz formula [2]. AKI is a frequent and serious complication following cardiac surgery with extracorporeal circulation (ECC) [3]. The aim of this study was to determine the incidence and risk factors of AKI after paediatric cardiac surgery with extracorporeal circulation.
Patients and Methods
This was a prospective observational, descriptive and analytical study of 40 patients, conducted over a period of 04 months, from 09/11/2021 to 09/03/2022 in the cardiac center of the University Hospital of Fann in Senegal. In our work, we included paediatric patients (< 18 years) who underwent cardiac surgery with extracorporeal circulation during this period.
Exclusion criteria were: Patients who died on the operating table or at D0 (first 24 hours) and patients with incomplete preoperative or postoperative workup.
The data collection form had three sections (preoperative, intraoperative and postoperative) filled in by the anaesthetist.
All patients had an anaesthetic consultation (>2 weeks before surgery), a pre-anaesthetic visit (the day before), a recent ultrasound evaluation and a biological check-up.
All patients were operated on under general anaesthesia and tracheal intubation.
The following parameters were assessed intraoperatively in all patients: priming of the bypass graft, duration of extracorporeal circulation, duration of aortic clamping, lactate and pH at the end of bypass grafting, fluid input-output balance (FIB), modified ultrafiltration, number of doses of cardioplegia, intraoperative transfusion, use of sympathomimetics, average hourly diuresis, resumption of cardiac activity.
The parameters evaluated immediately postoperatively (first day of hospitalization in the intensive care unit or D0) were : pH on admission, extubating delay, ultrasound evaluation (LVEF, TAPSE), biological assessment (Hb, creatinine, GFR), transfusion, use of sympathomimetics, cardiac activity (stimulated or not), fluid BES on D0, average hourly bleeding on D0, use of diuretics on D0, resumption of surgery on D0, hourly diuresis over the first 24 hours, creatinine, change in GFR, length of hospitalization, mortality.
GFR was estimated using the Schwartz paediatric formula and the diagnosis of AKI was made according to the 2015 SFAR and SRLF FER based on the presence of at least one of these two criteria: GFR decrease of more than 25%, diuresis < 0.5 ml/Kg/H for more than 8 hours.
The statistical analysis of the data and the P values of the different crosses were obtained using the Sphinx Plus software. The Excel table was used to format the tables and graphs.
Descriptive Results
Epidemiological data
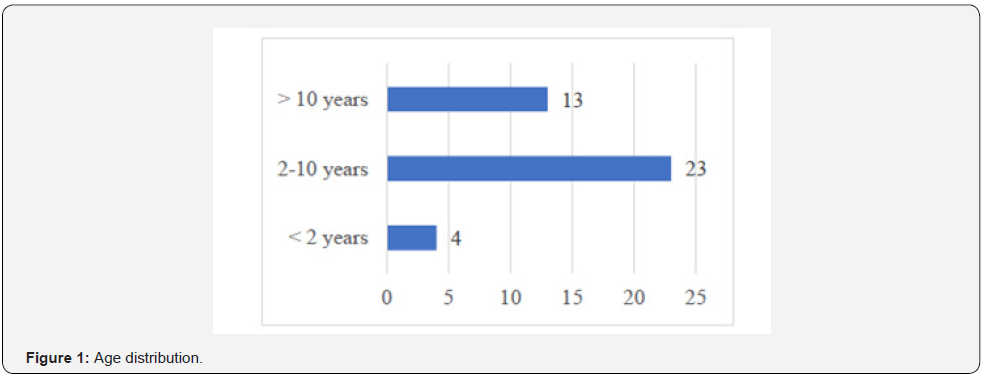
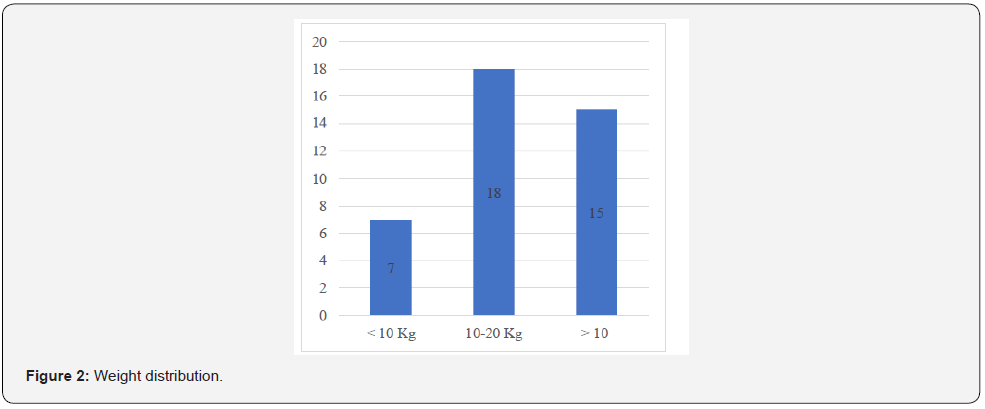
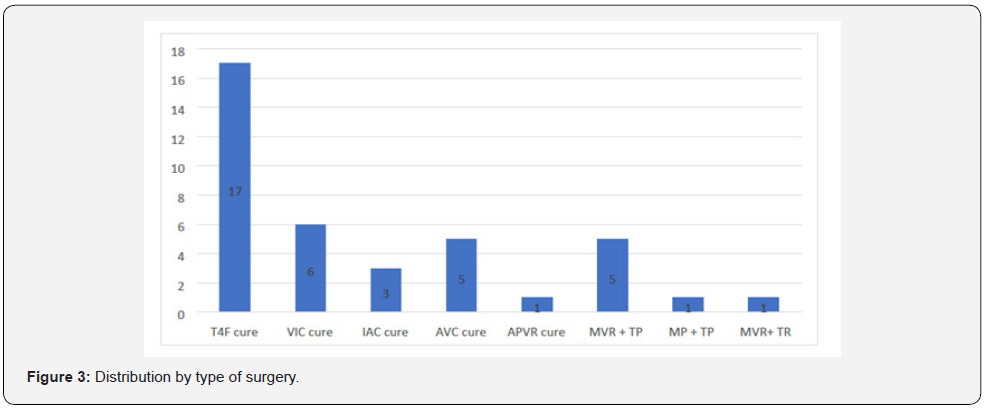
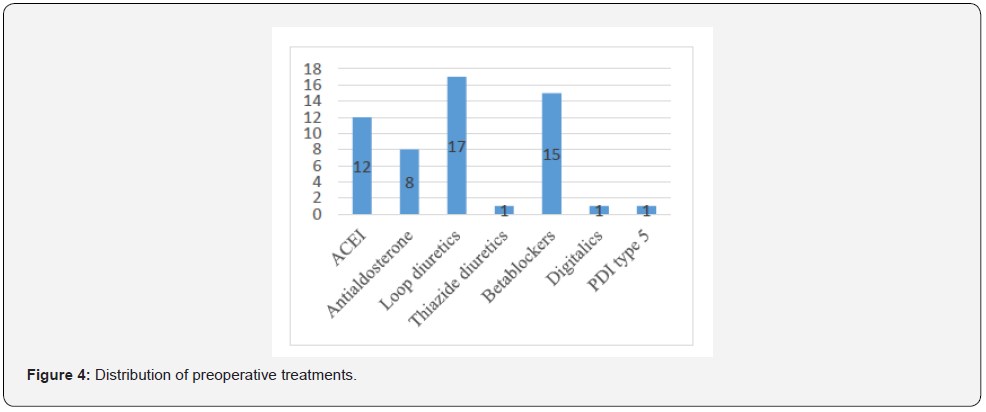
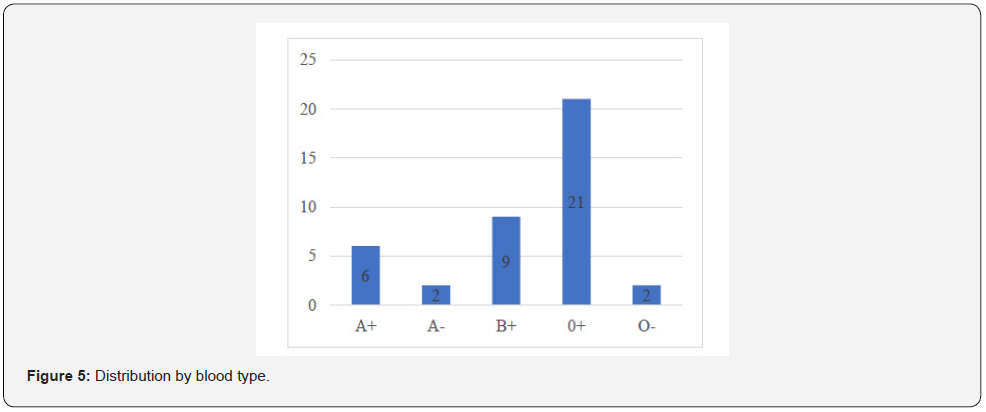
We included 53 paediatric patients operated on during this period, and subsequently excluded 13 patients according to the above criteria. A total of 40 patients were included, with a predominance of males and a sex ratio of 1.8. The mean age was 7.2 years +/- 4.4 and the median age 7 years with extremes of 6 months and 16 years. The majority of patients, 57.5%, were between 2 and 10 years of age (see Figure 1). The mean weight was 18.3 kg +/- 9.3 (5-43 kg) and 82.5% of the patients weighed > 10 kg (Figure 2). The mean height was 115.9 cm +/- 27.5 cm (65-171 cm) and 35% had a height < 100 cm. There were only 03 patients with comorbidity including 02 cases of sickle cell disease and 01 case of asthma. Two types of surgery were performed in our series, with a predominance of congenital surgery (80%) and valve surgery (20%) (Figure 3). All patients were operated on as scheduled surgery and none of them had acute heart failure on the day of surgery. The majority of the patients had a cardiac treatment (85%), of which 17.5% had at least three ongoing medications (Figure 4). None of the patients had LV systolic dysfunction preoperatively given the left ventricular ejection fraction (LVEF): mean 70.1% +/- 8.1 (55%-89%). There was no severe right ventricular dysfunction as assessed by TAPSE: mean 18.6 mm +/- 3.4 (12 mm; 26 mm). The 02 most represented blood groups were OO positive with 52.5%, and BB positive 22.5% (Figure 5). Only 25% of the patients had a haemoglobin level below 12 g/dl with a mean of 14.28 +/- 2.9 and extremes of 9.2 and 20.5 g/dl. Mean creatinine was 5.5 mg/L +/- 1.6 and extremes of 2.1 and 10 mg/L. The mean GFR was 130.2 +/- 47.3 (58.3 - 267.2) ml/Kg/1.73 m2 and 09 patients or 22.5% had a GFR less than 100 ml/Kg/1.73 m2.
Intraoperative data
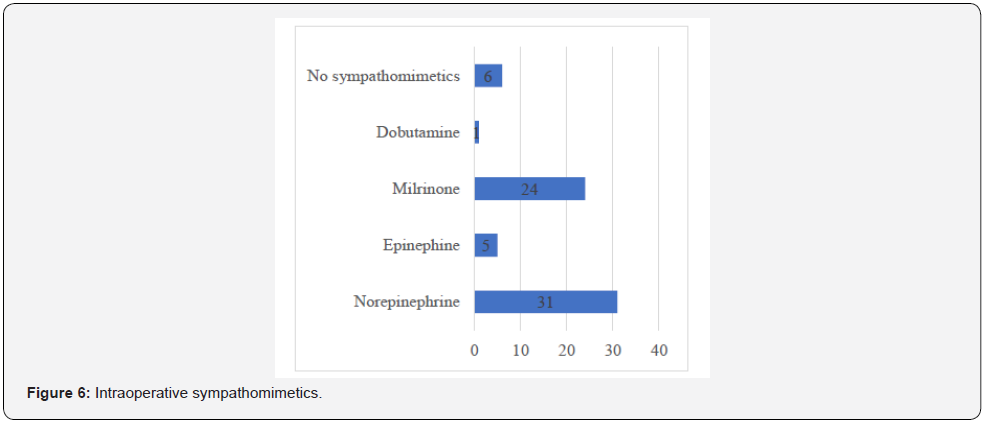
The priming of the extracorporeal circulation (CEC) was done with solutes only in 45% of cases (crystalloids alone or associated with colloids) and solutes with labile blood products in 55% of cases. The mean duration of the ECC was 107.7 +/- 59.1 min (34- 372) and 12 patients (30%) had a duration greater than 120 min. The mean duration of aortic clamping was 76.1 +/- 52.8 min (15- 338) and only 07 patients (17.5%) had a duration greater than 100 min. The input-output fluid balance was positive in 22 patients (55%) and negative in 18 patients (45%). The majority of patients had received a single dose of cardioplegia (60%) compared to 40% of patients who received 2 or more doses. Modified ultrafiltration in addition to hemofiltration was performed in 07 patients (17.5%). The gasometry at the end of the CEC showed 02 cases (5%) of severe acidosis with a pH lower than 7.20 and 20 cases (50%) of hyperlactatemia (lactates higher than 2 mmol/L. Concerning the resumption of electrical activity after aortic decompression, only 03 patients (7.5%) had benefited from an internal electric shock on ventricular fibrillation and only one had required electro systolic training afterwards. The majority of patients had received a labile blood product transfusion, i.e. 27 patients (67.5%). Only 05 patients did not receive sympathomimetics intraoperatively (15%) (Figure 6). The average intraoperative hourly diuresis was greater than 1ml/Kg/H in 90% of patients.
Postoperative data
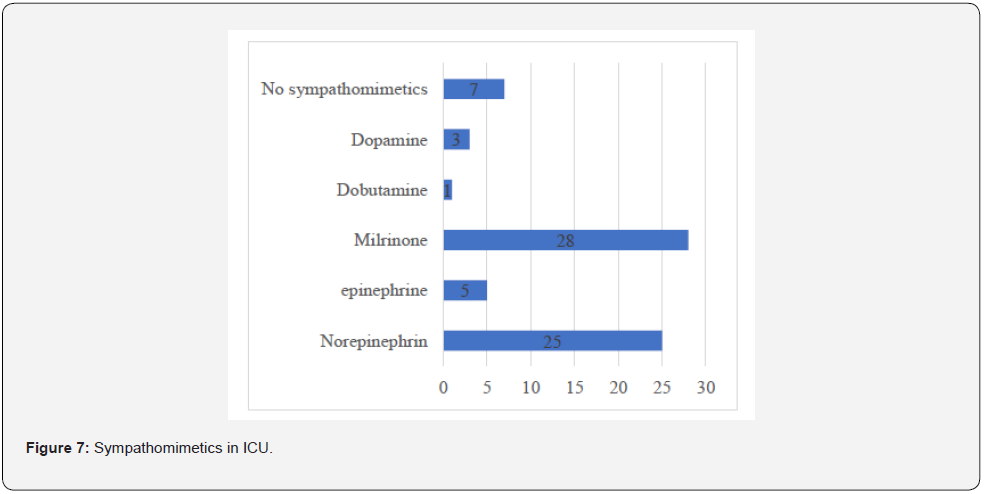
Extubating was in the majority of cases (87.5%) within the first 24 hours (JO) of which 67.5% (27/40) within the first 6 hours. Gasometry on admission to the intensive care unit revealed 07 cases of severe metabolic acidosis (17.5%). The majority of patients (82.5%) were put on sympathomimetics (Figure 7), 12.5% (05 patients) had severe systolic dysfunction (LVEF greater than 40%) and 11 patients (27.5%) had severe LV systolic dysfunction. The average hourly bleeding over 24 hours was greater than 1 ml/Kg/H in only 07 patients, i.e. 17.5%, and only 25% received a postoperative transfusion. The input output fluid balance for the first 24 hours was positive in 80% of cases and negative in 20%. The majority of patients in our series (60%) required diuretics to promote diuresis. There were only 03 cases of revision surgery at D0 (7.5%). The mean postoperative GFR was 132.9 +/- 71.4 ml/ min/1.73 m2 with extremes of 15.4 and 346.4. The incidence of acute kidney injury was 30% (12/40 patients) with: 20% at risk stage, 7.5% at damage stage and only one patient (2.5%) at renal dysfunction stage. No patient reached the severe stage requiring dialysis. The diuresis over the 24 hours postoperatively was only decisive for the diagnosis in one patient, who at the same time had a 78% drop in his GFR, and was therefore classified as dysfunctional. Regarding the short-term evolution of the patients in the intensive care unit, the average length of hospitalisation was 5 days (1/30) and the mortality rate was 10% (4/40 patients).
Analytical Results: Statistical Correlations
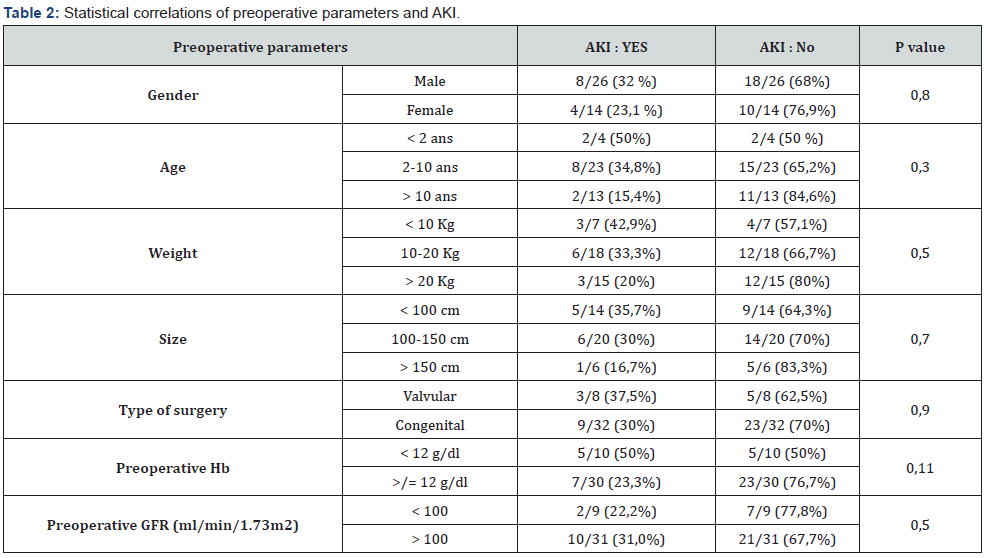
Preoperative parameters (Table 2)
Gender did not appear to be a discriminant for the occurrence of AKI with 30.8% for males versus 23% for females (p = 0.8). There was a decrease in the frequency of AKI with increasing age. That’s 50% of those under 02 years of age, 34.8% for those 2-10 years of age, and 15.4% for those over 10 years of age, however the correlation did not appear to be significant (p = 0.3).
The frequency of AKI appeared to decrease with increasing weight and height without significant statistical correlation with p at 0.5 and 0.7 respectively. There was a 2.1-fold increase in the incidence of AKI in patients with a haemoglobin level of <12g/dL, with little significant relationship (p=0.11). Other preoperative parameters did not appear to have a significant influence on the occurrence of AKI - see Table 2.
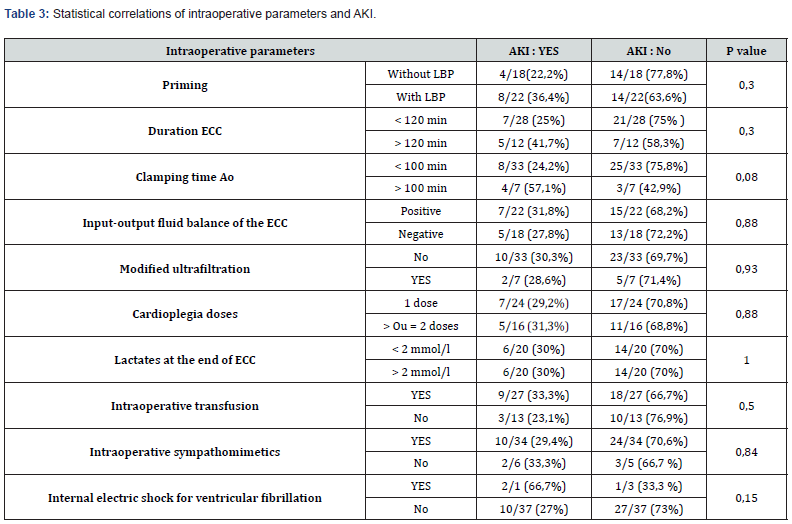
Intraoperative parameters (Table 3)
AKI was noted in 1.6 times more patients primed with labile blood products than with the rest of the patients who received only clear fluids (crystalloids or crystalloids + colloids), although p was not significant (p=0.3). CEC duration > 120 min was 1.6 times more associated with AKI than duration < 120 min (p=0.3). AKI was 2.3 times less common in patients with aortic clamp time < 100 min than in other patients, although the association was only marginally significant (p=0.08). Internal electrical cardioversion for ventricular fibrillation after aortic decompression was associated with a 2.47-fold greater risk of AKI than other patients, although the relationship was not significant (p=0.15). Transfused patients had 1.4 times more AKI than the rest of the patients (p=0.5). The use of sympathomimetics intraoperatively did not appear to have an impact on the occurrence of AKI (p= 0.55). There was no significant dependence on other intraoperative parameters, see table 3.
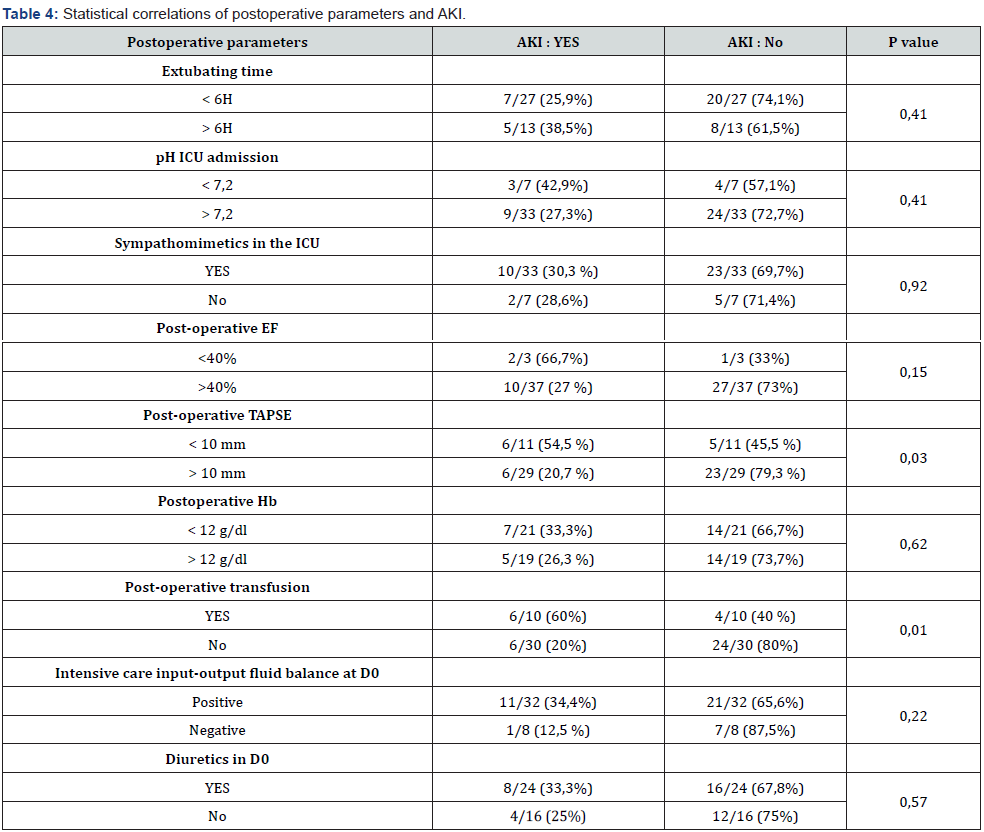
Postoperative parameters (Table 4)
Patients admitted to the resuscitation service with severe acidosis (pH < 7.2) had 1.6 times more AKI than others with a nonsignificant p value (p = 0.3). The incidence of AKI was 2.5 times higher in patients with a postoperative LVEF <40%, however the P value was insignificant (p=0.15). A postoperative TAPSE < 10 mm was associated with 2.6 times more cases of AKI and there was a significant dependence with a p = 0.03. Patients extubated beyond 6 hours were more likely to develop AKI (38.5% versus 25.9%), with no significant dependence (p = 0.34). Patients transfused postoperatively had a 3-fold higher incidence of AKI than nontransfused patients and the correlation was highly significant at p = 0.01. Of the patients who developed AKI, only one had a negative input-output fluid balance at D0 (first 24 hours), however the relationship appeared to be only marginally significant (p = 0.22). The use of diuretics at D0 did not appear to influence the occurrence of AKI (p = 0.57).
Duration of hospitalization and AKI
The average length of hospital stay was twice as high in the group of patients with AKI (7.58 days/3.61 days) and the static correlation was significant at p=0.05.
Mortality and AKI
The mortality in our series was 10% (4/40). It was 3 times higher in patients with AKI than in the rest of the patients. The association between AKI and death in our population was significant with a p = 0.038.
Discussion
Limitations
i. Small size of our study population, which may impact on statistical indices.
ii. Lack of a very recent renal assessment preoperatively (morning or day before surgery) and repeatedly postoperatively to obtain more accurate results.
Comments
The epidemiology of AKI after cardiac surgery depends on the definition used. Indeed, in recent years this was based on the 2012 KDIGO guidelines, which were applicable to adult and paediatric patients. However, they currently seem difficult to apply to children. Indeed, the RIFLE and AKIN criteria on which the KDIGO recommendations are based do not take into account the size of the patients and are based on plasma creatinine values, which poses a problem in children whose muscle mass is much lower than that of adults. Therefore, a paediatric version of RIFLE (pRIFLE) based on estimated plasma creatinine clearance and diuresis was proposed by Akcan et al. [4].
We used the pRIFLE to diagnose AKI and classify patients according to severity. Numerous studies, including that of Dianel et al. [5], have found that this test is the most sensitive for detecting AKI and it has been recommended in the 2015 RFE of the SFAR and SRLF [1]. The incidence of AKI in postoperative paediatric cardiac surgery is highly variable and was 30% in our work. Toth et al. [6] obtained a similar result with 31%, however the prevalence was lower at 14.1% in the study by Lee et al. [7] and much higher in the results of Tanyildiz et al. [8] and Elella et al. [9] with 61.3% and 90% respectively. It should be noted that in these studies, although the diagnostic tool was the same, there was a notable difference in the epidemiological data. The mean age was 7.2 years in our series, far higher than that found with Lee (16 months) [7], Tanyildiz (36.6 months) [8], and Elella (11 months) [9]. The types of surgery were also different: Congenital malformation surgery for Lee [7] and Tanyildiz [7], paediatric cardiac surgery (congenital and valvular) in our work and in Toth’s, congenital heart surgery with ECMO in Elella’s study [9].
Concerning the classification of the severity of AKI, still using the pRIFLE, on an incidence of 30%, we noted a predominance of the risk stage (20%), followed by the lesion stage (7.5%), then 2.5% for failure, without any case of recourse to dialysis. Some studies have found the same trend: Lee et al. [7] on a prevalence of 14.1%, there were 12.3% risk, 1.8% injury and 0 cases of failure; Tanyildiz et al. [8] on 61.3% incidence, 25.5% were for the risk stage, 20.4% injury, 15.3% failure. However, other studies have shown a different distribution: For Toth and al [6], out of an incidence of 31.9%, failure was predominant with 18.7%, followed by the risk stage (11.5%), 1.7% for the lesion stage and finally 6.4% had required dialysis. Elella et al. [9] in a series of more severe patients (cardiac surgery with ECMO) noted a prevalence of 90%, 57% in the failure stage with a dialysis rate of 49%.
Gender was not a determinant of the occurrence of AKI in our study (p= 0.8). This trend has been confirmed in many other studies [7,8,10]. The lower the age, the higher the prevalence of AKI in our series, although the dependence was not significant (p = 0.3). Lee et al. [7] did not find an effect of younger age in the incidence of AKI (p = 0.13), contrary to the work of Tanyilzid [8] and Hirano D. [10]. The lower the weight and height, the higher the percentage of AKI occurrence. However, these two factors did not have a significant dependence on AKI in our series, which is consistent with the series of Tanyilzid [8]. Lee et al. [7], on the contrary, found low weight to be a significant risk factor (p= 0.024). The type of surgery was not an important factor in postoperative AKI in our series (p = 0.9), which is confirmed by the study of Goldberg E. [11] with a p = 0.68, even if the latter was more of an adult population. A low haemoglobin level < 12g/dl was associated with a 2.1-fold increase in the risk of acute kidney injury, but the dependence was only marginally significant (p = 0.11). Low haemoglobin was also identified as an important factor in the study by SunKyung P. et al. [12].
None of the preoperative treatments in our patients appeared to have an effect on the occurrence of acute kidney injury, whereas ACE inhibitors have been widely cited as a risk factor in the literature [13-15]. A long duration of ECC has been found in many studies [8,10,11] to be a factor increasing the risk of AKI in contrast to our study (p = 0.3) and that of Lee et al. [7]. An aortic clamping time > 100 min was associated with 2.3 times greater risk of AKI in our series with a low statistical dependence (p= 0.08), unlike data from other work [8]. The use of vasoactive agents did not appear to play a role in postoperative AKI, contrary to many other data in the literature [8,13]. The incidence of AKI in our study was relatively higher in the group of patients with a delay in extubating beyond 6 hours. This is consistent with the identification of prolonged postoperative mechanical ventilation as a risk factor in other work [7,8]. Rosner et al [13] identified haemodynamic instability and the use of vasoactive agents as important risk factors for AKI. In our work, concerning postoperative haemodynamic, it was mainly right ventricular systolic dysfunction that was decisive with p = 0.03, alteration of ventricular systolic function had only a weakly significant association (p = 0.15). Postoperative transfusion was the most important risk factor for the occurrence of AKI in our series (p = 0.01), this is supported by Furnary et al. [16] who found a higher risk of AKI with a higher number of units of packed red blood cells. A positive input-output fluid balance on the first postoperative day was associated with 2.7 times more cases of AKI in our study, with a not very significant dependence (p=0.22).
However, it should be noted that postoperative volume overload has been accepted in many studies [8,13] as a determining factor in the occurrence of AKI in postoperative cardiac surgery. We found a significant association between the length of hospitalization of patients and AKI occurring on the first postoperative day with p= 0.05, as shown in the literature [7,8]. Mortality was 10% in our study and there appeared to be a significant dependence on the acute kidney injury presented by our patients with a p=0.03. The relationship between AKI in postoperative paediatric cardiac surgery and mortality has been described in many other studies [8,17].
Conclusion
The epidemiology of AKI after paediatric cardiac surgery depends on the definition used. It is currently accepted that the modified RIFLE classification (pRIFLE) is the most sensitive test for making the diagnosis. The incidence is highly variable and depends on the epidemiological characteristics, the type of surgery, the intraoperative and postoperative data of the patients. It was 30% in our study, with a predominance of the risk stage (2/3 of cases), without any cases in the severe stage requiring dialysis. Impaired right ventricular systolic function and postoperative transfusion were the main risk factors identified. Other factors were of lesser impact: preoperative haemoglobin < 12g/dl, aortic clamping time > 100 min, intraoperative internal electric shock, left ventricular ejection fraction < 40% postoperatively, and volume overload on day 1 in the ICU.
References
- 2015 SFAR-SRLF joint RFE.
- Schwartz GJ, Brion LP, Spitzer A (1987) The use of plasma creatinine concentration for estimating glomerular filtration rate in infants, children, and adolescents. Pediatr Clin North Am 34(3): 571-590.
- Mangano CM, Diamondstone LS, Ramsay JG, Aggarwal A, Herskowitz A, et al. (1998) Renal dysfunction after myocardial revascularization: risk factors, adverse outcomes, and hospital resource utilization. Ann Intern Med 128(3): 194-203.
- Akcan-Arikan A, Zappitelli M, Loftis LL, Wasburn KK, Jefferson LS, et al. (2007) Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int 71(10): 1028-1035.
- Lex DJ, Tóth R, Cserép Z, Alexander SI, Breuer T, et al. (2014) A comparison of the systems for the identification of postoperative acute kidney injury in pediatric cardiac patients. Ann Thorac Surg 97(1): 202-210.
- Tóth R, Breuer T, Cserép Z, Lex D, Fazekas L, et al. (2012) Acute kidney injury is associated with higher morbidity and resource use in paediatric patients undergoing cardiac surgery. Ann Thorac Surg 93(6): 1984-1990.
- Lee SH, Kim SJ, Kim HJ, Son JS, Lee R, et al. (2017) Acute kidney injury following cardiopulmonary bypass in children-risk factors and outcomes. Circ J 81(10): 1522e7.
- Tanyildiz M, Ekim M, Kendirli T, Tutar E, Eyileten Z, et al. (2017) Acute kidney injury in congenital cardiac surgery: pediatric risk-injury-failure-loss-end-stage renal disease and Acute Kidney Injury Network. Pediatr Int 59(12): 1251e60.
- Elella RA, Habib E, Mokrusova P, Joseph P, Aldalaty H, et al. (2017) Incidence and outcome of acute kidney injury by the pRIFLE criteria for children receiving extracorporeal membrane oxygenation after heart surgery. Ann Saudi Med 37(3): 201-206.
- Hirano D, Ito A, Yamada A, Kakegawa D, Miwa S, et al. (2017) Independent Risk Factors and 2-Year Outcomes of Acute Kidney Injury after Surgery for Congenital Heart Disease. Am J Nephrol 46(3): 204-209.
- Eliza Goldberg (2021) Risk factors for acute renal failure in postoperative cardiac surgery: ancillary study of the Cardiox study. Doctoral thesis in medicine, N°2021-59, Amiens Faculty of Medicine, University of Picardie Jules Verne.
- Park SK, Hur M, Kim E, Kim WH, Park JB, et al. (2016) Risk factors for acute kidney injury after congenital heart surgery in infants and children: a retrospective observational study. PLoS One 11(11): e0166328.
- Rosner MH, Okusa MD (2006) Acute kidney injury associated with cardiac surgery. Clin J Am Soc Nephrol 1(1): 19-32.
- Pannu N, Nadim MK (2008) An overview of drug-induced acute kidney injury. Crit Care Med 36(Suppl): 5216-5223.
- Bentley ML, Corwin HI, Dasta J (2010) Drug-induced acute kidney injury in the critically ill adult: recognition and preventive strategies. Crit Care Med 38(Suppl 6): 5169-5174.
- Furnary AP, Wu Y, Hiratzka LF, Grunkemeier GL, Page US 3rd (2007) Aprotinin does not increase the risk of renal failure in cardiac surgery patients. Circulation 116(Suppl I]: I127-I133.
- Blinder JJ, Goldstein SL, Lee VV, Baycroft A, Fraser CD, et al. (2012) Congenital heart surgery in infants: effects of acute kidney injury on outcomes. J Thorac Cardiovasc Surg 143(2): 368-374.






























