Opioid-Free Anesthesia in Morbid Obese Patients: Review Article
Sameh Abdelkhalik Ahmed*
Department of Anesthesiology and Intensive Care, Faculty of Medicine, Tanta University, Egypt
Submission: July 09, 2020; Published: September 24, 2020
*Corresponding author: Dr. Sameh Abdelkhalik Ahmed, assistant Professor of Anesthesiology and Intensive Care, Faculty of Medicine, Tanta University, Egypt. Saied st, Tanta, Elgharbia Governate, Egypt, Tell: 00201002977048
How to cite this article: Sameh A A. Opioid-Free Anesthesia in Morbid Obese Patients: Review Article. J Anest & Inten care med. 2020; 11(1): 555803. DOI: 10.19080/JAICM.2020.11.555803
Abstart
Morbid obese patients are highly susceptible to postoperative respiratory depression and delayed recovery owing to the pathophysiological changes of the obesity and the increased sensitivity to opioids. Opioid-free anesthesia technique is a technique in which the opioids aren’t administrated throughout any route with the use of non-opioid analgesics to maintain analgesia, satisfaction, and hemodynamic stability of the patients. Many agents that can be used as non-opioid analgesics as clonidine, dexmedetomidine, ketamine, lidocaine, magnesium, non-steroidal anti-inflammatory agents, gabapentin, pregabalin, and dexamethasone. Each one of them has a different mechanism of analgesia through its effect on the pain pathway. Opioid-free anesthesia is indicated mainly in morbid obese patients with increased risk of obstructed breathing. It has many advantages especially the improvement of recovery profile, avoidance of respiratory depressant effect of opioids, and avoidance of opioid use and opioid-induced hyperalgesia. Hypotension and bradycardia are common complications of OFA technique especially with the use of alpha-2 adrenergic receptor agonists. It is contraindicated in patients with increased risk of hypotension as hypovolemic patients, patients with ischemic heart disease, and patients with autonomic neuropathy. The use of OFA technique requires the presence of monitoring of the depth of anesthesia and expert anesthesiologists. Mulier, described a technique for OFA that depends upon the use of an infusion called mulimex which is composed of premixed 1-3 of dexmedetomidine, lidocaine, ketamine, magnesium, deep neuromuscular block, and titrated MAC of desflurane. Many other regimens of OFA technique were described by many authors as that of Sultana. Despite the growing use of OFA technique in many bariatric centers, no definite protocol has an evidence base as each center have its own protocol. For introduction of OFA technique into the era of evidence-based medicine, many clinical trials evaluating different regimens and medications are required.
Keywords:Obesity, Opioids, Non-opioid analgesics, OFA, Recovery
Introduction
Opioids are widely used in anesthesia practice either for premedication, induction, and maintenance of anesthesia, or postoperative analgesia. They have many advantages especially hemodynamic stability and postoperative analgesia. However, certain drawbacks exist with the use of opioids especially respiratory depression and nausea and vomiting [1]. Besides, the excessive use of opioids may lead to opioid hyperalgesia, tolerance, or addiction [2]. So,many anesthesiologists tend to avoid their use either partially or totally and tend to use regional anesthesia techniques or other medications with limited side effects instead of them [3]. Morbid obese patients have multiple pathophysiological changes that can affect most of the body organs. It is usually associated with a picture of restrictive lung disease, obstructive sleep apnea syndrome, pulmonary hypertension, right to left shunting, and increased risk of arterial hypoxemia [4]. Furthermore, obesity is associated with multiple organ disorders as cardiovascular complications, metabolic syndrome, venous thromboembolism, fatty infiltration of the liver, and psychological disturbances [5].The pharmacokinetics and pharmacodynamics of opioids are both altered in obese patients [6].The blood flow to the adipose tissue is decreased in obese population and this will alter the volume of distribution of the drug that will affect the drug dose regimen. However, there is no available consensus that can be used for determining the body weight upon which the dose of the drug should be calculated [7]. The metabolism of opioids is increased in obese patients owing to an increase in glucuronidation. This leads to a high initial level of effective drug concentration with a fast decline of this high concentration [8]. The opioid potency in morbid obese population is highly variable. Certain studies suggested that in morbidly obese patients, the pain intensity is increased, and the opioid potency is decreased. This may be due to the change in the number and the potency of opioid receptors in obese patients [9]. Therefore, the use of opioids in morbid obese patients should be done under strict precautions owing to increased risk of postoperative pulmonary and cardiovascular complications and the increased sensitivity of the central nervous system to the depressant effect of opioids [10]. Opioid-free general anesthesia (OFA) was first introduced by Mulier. It may represent an excellent alternative as many studies revealed adequate control of pain with such technique [11,12].
Definition
Opioid-free anesthesia (OFA) is an anesthesia technique in which no perioperative opioids are administrated through either systematic, neuraxial, or intracavitary routes considering patient’s comfort, analgesia, or hemodynamic stability are not affected [11,13].
Indications
The risk of postoperative respiratory depression in obese population is the main reason for the use of opioid-free anesthesia techniques. Other reasons for its use include the risk of obstructed breathing, increased sedation and hypersomnolence, postoperative nausea and vomiting, muscle weakness, tolerance and addiction, negative inotropism, dizziness, and urinary retention [13]. Nowadays, opioid-free anesthesia (OFA) technique is widely used to improve wound healing [14], avoid the immunosuppressive effect of opioids [15], improve the oncological outcomes [16], avoid perioperative cognitive dysfunction [17], and avoid opioidinduced hyperalgesia [18]. Also, it is indicated in patients with addiction to opioids or those with an increased risk of developing chronic pain [19]. Furthermore, non-opioid analgesics should not be overlooked during the management of postoperative pain in major surgeries [20].
Contraindications
It is better avoided in patients with nodal rhythm, autonomic dysfunction, ischemic heart diseases, beta-blocker therapy, hypovolemia, and polytrauma. In such conditions, it can cause vasodilatation of the peripheral blood vessels that impair perfusion to the vital organs. Moreover, hypersensitivity to any of the used medications represents a contraindication to OFA [21].
Advantages
Opioid free anesthesia has many advantages especially avoiding opioid overdose and opioid-induced hyperalgesia. The most important advantage of OFA seems to be the potential improvement of recovery profile in obese patients. Also, it is an easy protocol that avoids the respiratory depressant effect of opioids and minimizing the risk in patients with obstructive sleep apnea syndrome. Furthermore, it guarantees an adequate depth of muscle relaxation that facilitates laparoscopic interventions and ensures an adequate level of analgesia and amnesia [22]. The incidence of awareness is rare if the bispectral index is below 60 [21]. Certain opinions suggested that OFA is associated with a lesser incidence of postoperative urine retention and shivering [23].
Disadvantages
The main disadvantage of opioid-free anesthesia is the potential risk of hypotension that may require vasopressor support. Hypotension and bradycardia commonly occur with the use of alpha-2 agonists. Besides, the risk of awareness remains possible especially in the absence of monitoring of the depth of anesthesia or lack of experienced anesthesiologists. Also, the use of ketamine may interfere with EEG monitoring. Additional small doses of short acting opioids may be required to abolish the stress response of tracheal intubation [21].
Requirements
Opioid free anesthesia technique is a skill that requires time for learning and practice. During the period of training, it is recommended not to stop the use of opioids suddenly, but a transition of limited opioid use is required. Anesthesiologists should be aware of the pharmacology of opioid substitutes. Moreover, monitoring of the depth of anesthesia is mandatory [24,25].
Pharmacology of non-opioid analgesics
The pain pathway is complex. Noxious stimuli are transmitted from the peripheral receptors through afferent fibers to the dorsal horn of the spinal cord, then, it is modulated by descending pathway, glia, and spinal interneurons. Sympathetic and parasympathetic interactions also occur. The pain transmission involves activation of many neuroreceptors as N-methyl-D-Aspartate (NMDA), gammaaminobutyric acid (GABA), enkephalins, and others. Opioid free anesthesia offers an alternative mechanism of suppression of the pain transmission [26].
The non-opioid analgesics include a variety of medications. It may include clonidine, dexmedetomidine, ketamine, lidocaine, magnesium, and ketorolac. Each of those agents has a specific mechanism of action by targeting a certain site in the pain pathway. Understanding the mechanism of action of each agent is very crucial in the of the combination of agents in opioid-sparing anesthesia [27].
Clonidine
Clonidine is an alpha-2 adrenergic receptor agonist agent that is used safely in general and regional anesthesia practice. Its affinity to alpha-2 receptors is 220 times its affinity to alpha-1 receptors [28]. It possesses sedative, analgesic, hypnotic, and antisympathetic action [29]. It acts mainly through the stimulation of alpha-2 receptors located in the terminal primary afferents, superficial lamina neurons of the spinal cord, and the locus ceruleus. So, it has a central and peripheral analgesic effect. Also, it may increase the effect of local anesthetics in peripheral nerve block despite the absence of its receptors on the nerve axons. This may be due to its effect on C and Aδ fibers [29,30]. The perioperative use of systemic clonidine was suggested by many clinical studies to reduce postoperative pain intensity, opioid consumption, and the incidence of nausea and vomiting. However, its use may increase the risk of hypotension and bradycardia. Its use as an adjuvant in regional anesthesia techniques improves the sensory and the motor blocking criteria and prolong the postoperative analgesia but increases the risk of perioperative hypotension [30]. The use of clonidine is largely replaced by dexmedetomidine.
Dexmedetomidine
Dexmedetomidine is a highly selective and specific alpha-2 adrenergic receptors agonist that has analgesic, sedative, sympatholytic, and amnestic properties. It is 8-10 times more potent than clonidine. It gained the approval of the FDA for intravenous use as a sedative and analgesic agent [31]. The mechanism of action is not fully obvious. Stimulation of alpha-2 receptors may exert its analgesic effect through the hyperpolarization of the nonadrenergic neurons that depress the firing in the locus ceruleus. Stimulation of central adrenergic receptors depresses the release of norepinephrine that causes hypnotic effect without affecting the respiratory functions. Moreover, the anti-nociceptive effect may be produced through the stimulation of alpha-2 receptors in the superficial dorsal horn neurons. Stimulation of the postsynaptic alpha-2 receptors may be responsible for the incidence of bradycardia [32]. The use of dexmedetomidine infusion in morbid obese patients was assessed by many clinical trials. They revealed that the use of dexmedetomidine has the same analgesic effect as the use of fentanyl with greater hemodynamic stability obtained with the use of dexmedetomidine. Dexmedetomidine has the advantage of having no respiratory depressant effect. Also, it has an amnestic effect. Dexmedetomidine does not affect the sleep pattern and maintains the airway patency [33]. Moreover, the use of dexmedetomidine decreased postoperative opioid use by 50% [34-36]. Dexmedetomidine can be used in morbid obese patients in a wide range of loading or maintenance doses (0.2-2 ug/kg/hr) [35]. The most common side effects associated with dexmedetomidine use are hypotension, bradycardia, sinus rhythm, and dry mouth. It should not be used in hypovolemic patients, uncontrolled diabetes mellitus, uncontrolled hypertension, or in patients with predominant vagal tone [37].
Ketamine
Ketamine, the dissociative anesthetic agent, is an antagonist of NMDA receptors that have anesthetic, sedative, analgesic, and amnestic effects. It has secondary advantages as bronchodilatation, maintenance of the sympathetic tone, non-respiratory depressant, and maintenance of the airway reflexes. Moreover, it may have neuroprotective and anti-inflammatory effects [38,39]. Certain studies suggested that fentanyl is capable of stimulating NMDA receptors leading to an increase in the sensitivity to pain and the use of low dose ketamine can abolish this effect [40]. Ketamine overuse may be associated with cardiovascular complications (tachycardia and hypertension) and psychological disorders (nightmares). This effect may be limited by the combination of it with alpha-2 agonists [41]. A small dose of ketamine (0.25-0.5 mg/kg/hr) was found to decrease the opioid requirements up to 50% [40].
Magnesium
Magnesium has an NMDA receptor antagonist effect through a mechanism different from this of ketamine. So, a combination of both ketamine and magnesium is better than the use of one agent [42]. Magnesium is used as an analgesic in a loading dose of 20-40 mg/kg and a maintenance dose of 2 mg/kg/hr [43]. The clinical study of Chan and Shetty evaluated the use of ketamine alone, magnesium alone, or combination of both in morbid obese patients undergoing laparoscopic bariatric surgeries and concluded that the requirement of rescue analgesia was significantly decreased with the use of a combination of both of them. The use of two adjuvants together appeared to improve pain control [44]. Side effects of magnesium administration include cardiovascular collapse, hypotonia, respiratory weakness, and renal impairment [41].
Lidocaine
Lidocaine is commonly used as a local anesthetic and antiarrhythmic agent. Furthermore, it possesses anti-inflammatory, immunomodulating, and anti-nociceptive effects [45]. Lidocaine is an important adjuvant in OFA during laparoscopic bariatric surgeries through its effect on inhibition of cytokines activity in the intestine [46]. Intravenous administration of lidocaine at a dose of 1.5 mg/kg and a maintenance dose of 2 mg/kg/hr in obese patients undergoing bariatric surgery was found to decrease intraoperative opioid consumption [47]. It is to be noted that the maximal dose of intravenous lidocaine to avoid local anesthetic toxicity is 5 mg/kg. Continuous infusion of lidocaine 2mg/kg may be the most suitable regimen to avoid lidocaine toxicity. Lidocaine toxicity can be manifested by arrhythmias, bradycardia, agitation, and confusion [48]. Moreover, the local infiltration of lidocaine and the intraperitoneal instillation in laparoscopic surgeries of obese patients was recommended by Mulier to be a part of OFA [11].
Non-steroidal anti-inflammatory drugs
Ketorolac, the non-steroidal anti-inflammatory drug (NSAID), is analgesic adjuvant that is commonly used as it improves postoperative analgesia without increasing the incidence of postoperative nausea and vomiting. Also, it has the advantages of no sedative effect, prolonged analgesic effect, and early restoration of the bowel function. However, it may increase the risk of gastric bleeding [46].
Gabapentin and pregabalin
Gabapentin is analogue of the (GABA) that acts mainly through inhibition of calcium release through binding to the delta subunit of alpha-2 receptors. Also, it interacts with NMDA receptors that leads to decreased release of neurotransmitters as glutamate and substance P [49]. Certain studies suggested that they can be used in morbid obese patients as they improved the postoperative analgesia without developing postoperative nausea and vomiting [49,50].
Dexamethasone
It is a glucocorticoid agonist that has anti-inflammatory action through inhibiting leukocyte infiltration, interfering with the release of inflammatory mediators, and suppressing the hormonal immune response. Also, it has an anti-edematous effect and antiemetic action [51]. Dexamethasone intravenous administration was suggested to decrease opioid consumption and the incidence of postoperative nausea and vomiting with improved patient rehabilitation [51,52]. Caution must be taken upon administration of dexamethasone to a diabetic patient, however, studies revealed that single dose of dexamethasone of 0.1 mg/kg was not associated with elevation of blood sugar of diabetic patients in the first 12 hours [52,53].
Techniques of opioid-free anesthesia
Opioid-free anesthesia (OFA) was first described in Europe by Mulier [11], then, multiple clinical studies reported its use and its benefit in morbidly obese patients [36,54,55].Mulier described an infusion called Mulimix which is composed of premixed 1-3 of the following agents [11]:
1. Sympatholytic, anesthetic, and analgesic agent to reduce the anesthetic requirements (clonidine or dexmedetomidine). Loading dose of dexmedetomidine: 0.5-1 mg/kg IBW followed by an infusion of 0.5-1 mg/kg IBW/h [56].
2. Non-opioid analgesic (low dose ketamine at a loading dose of 0.125 to 0.25 mg/kg followed by infusion of 0.125 to 0.25 mg/kg IBW/h) [38].
3. Co-anesthetic and sympatholytic agent (intravenous lidocaine 1.5 mg/kg IBW followed by infusion of 1.5-3 mg/kg IBW/h I.V) [57].
4. Intravenous infusion of magnesium as supplementary analgesic [58].
5. Deep neuromuscular block throughout the whole operative period that was adequately reversed.
6. Titrated minimum alveolar concentration of inhalational anesthetic (Desflurane 0.7-1.0 MAC) to maintain an adequate reading of bispectral index reading.
Additional medications that may be used to induce multimodal analgesia include dexamethasone, non-steroidal antiinflammatory analgesics (NSAID), paracetamol, or gabapentin. Loco-regional anesthesia as local wound infiltration and intraperitoneal instillation of local anesthetics are commonly used [21].
The Anesthetic technique of Mulier
The pre-prepared Mulimix should be started before the induction of anesthesia. It should be adjusted at a rate of 20ml/ hr. Induction of anesthesia is then carried out through propofol 2 mg/kg ideal body weight (IBW) along with an intubating dose of rocuronium (1mg/kg IBW). Anesthesia is then maintained by a titrated concentration of inhalational anesthetics (0.7-1.0 MAC of desflurane) and continuous infusion of rocuronium (0.5 mg/ kg IBW/hr) till the end of the surgery. The Mulimix infusion is continued at the loading rate throughout tracheal intubation, patient position, abdominal insufflation, and placement of laparoscopic ports. Then, its rate is decreased gradually to 5-10 ml/hr and stopped at the end of the surgery [21]. The neuromuscular block should be adequately reversed at the end of the surgery by either neostigmine (0.05 mg/kg IBW) and atropine (0.01 mg/kg IBW) or better by sugammadex 4 mg/kg IBW [59]. Ondansetron is administrated routinely as an anti-emetic agent. Also, a high dose of dexamethasone is administrated as both anti-emetic and analgesic adjuvant. Moreover, paracetamol 1-2 gm intravenous infusion, parecoxib 40 mg, and local anesthetic infiltration and intraperitoneal instillation are used. Magnesium is not used routinely. During the postoperative care in the postanesthesia care unit (PACU), Mulimix infusion may be restored at a low infusion rate (5ml/hr) in case of inadequate control of pain [21].
The Anesthetic technique of Sultana
Despite the high prevalence and use of Mulier’s technique of OFA, an alternative technique of opioid-sparing was developed by Sultana and consists of [13]: -
1. Preoperative administration of midazolam 2-3 mg intravenous and starting dexmedetomidine infusion at a rate of 5 ug/kg/hr.
2. Induction of anesthesia is carried out by propofol 2 mg/ kg IBW in addition to rocuronium 1 mg/kg IBW.
3. Maintenance of anesthesia is carried out by desflurane 0.5-1 MAC and rocuronium 0.5 mg/kg IBW/hr till the end of the surgery.
4. Dexmedetomidine infusion is decreased to 0.5-1 ug/kg/ hr once the laparoscopic trocars are placed.
5. Paracetamol infusion (1-2 gm) and non-steroidal anti-inflammatory agent (parecoxib 40 mg) are administrated intravenously. Local wound infiltration was performed at the surgical site.
6. Anti-emetic prophylaxis by giving both ondansetron and dexamethasone.
Other Anesthetic techniques
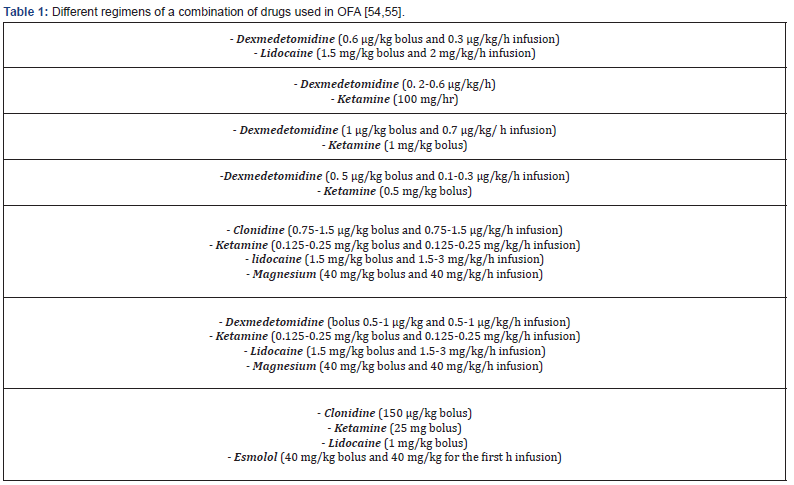
Mulier and Sultana protocols are not generally used all over the world. Each center may have its own protocol for the application of OFA practice. Many formulae that use different medications in a different dose regimen are available. Many drug combination formulae are illustrated in Table 1. Moreover, no- pharmacological methods as aromatherapy and hypnosis may be beneficial [60].
Role of loco-regional anesthesia
Local infiltration at the side port of laparoscope and intraperitoneal instillation of local anesthetics were described by Mulier technique [21].However, the retrospective study of Moncada et al. [61] did not find any significant change in the postoperative analgesia after sleeve gastrectomy with the use of bupivacaine local infiltration [61]. Moreover, the prospective randomized study of Ruiz-Tovar concluded that either epidural analgesia or local infiltration anesthesia had the same analgesic effect as intravenous analgesia [62].
Transversus abdominus plane block (TAPB) is used largely in abdominal surgeries and associated with better pain relief and decreased postoperative analgesic consumption [63]. Sinha et al. [64] suggested that the use of TAP block in morbid obese patients improved postoperative analgesia, reduced postoperative opioid consumption, enhance ambulation of the patients, and improved patient satisfaction [64]. However, clinical studies failed to prove that TAPB in morbid obese patients can decrease postoperative analgesic consumption [65,66]. This may be attributed to the technical difficulties and changed anatomical landmarks in obese patients [67].
Postoperative analgesia after OFA
Non-steroidal anti-inflammatory drugs (NSAID) should be given twice daily for at least 2 days. Moreover, paracetamol should be continued (1 g every 6 h) for several days. Once the patients can resume oral intake, gabapentin 150-300 mg can be administrated. In case of severe postoperative pain, continuous infusion of low doses of dexmedetomidine, lidocaine, and magnesium may be required. However, this requires the admission of the patient to a high dependency care unit for close monitoring [36].
Revision of literature
The high prevalence of opioid addiction in many countries may motivate the medical services to avoid the use of opioids as possible as they can. The risk factors of postoperative opioid addiction may include the poor control of postoperative pain, the use of large doses of opioids, and the prolonged use of opioids. This may allow the anesthesiologists to use opioid-free anesthesia. However, the opioid-free anesthesia technique is not applicable in all centers, all patients, or all types of surgical interventions. [68] The use of OFA seems to be safe and effective in the control of the postoperative pain and more comfortable to the patients than the conventional opioid-based anesthesia [69]. Opioid free anesthesia technique was adopted by Sydney Institution of Obesity Surgery to be the anesthetic technique of choice in bariatric surgeries whether sleep apnea exists or not. Also, OFA represents a part of the protocols of enhanced recovery after surgery in bariatric surgeries [70].
The practice of OFA was evaluated by many case reports before even its description by Mulier. The case study of Hofer et al [36] described morbid obese male patient (over 400 kg) with obstructive sleep apnea, moderate to severe restrictive lung disease, pulmonary hypertension, gastroesophageal reflux, and chronic hypoxemia scheduled for bariatric surgery under general anesthesia devoid of opioids which were replaced by dexmedetomidine infusion. The patient showed fewer opioid requirements on the second postoperative day with a decreased incidence of respiratory complications. The recovery profile and the postoperative analgesic requirements on the first postoperative day could not be evaluated as the patient was left on mechanical ventilation for the first night [36].
Also, the case series of Lam et al. [71] aimed to investigate the use of the multimodal analgesia protocol in morbid obese patients presented for laparoscopic bariatric surgery. The protocol consisted of premedication by pantoprazole, paracetamol, pregabalin, and midazolam, then, intraoperative dexmedetomidine (0.2 ug/kg/hr), ketamine (0.3 mg/kg as a loading dose followed by repeated boluses dose as required), parecoxib (40 mg intravenous bolus), tramadol (100mg), and local infiltration anesthesia were used. They concluded that OFA improved the postoperative analgesic profiles. However, this was an observational study with no control group and did not demonstrate the effect of opioid-free anesthesia on postoperative respiratory complications [71]. Moreover, a case study of Aronsohn et al. [72] described a case of morbid obese female patients with severe obstructive sleep apnea undergoing laparoscopic sleeve gastrectomy. Opioid-free total intravenous anesthesia with propofol and ketamine was used. The patient showed a rapid recovery profile with adequate control of the postoperative pain without opioids [72].
The benefits of opioid free anesthesia in different surgical interferences were evaluated by multiple clinical trials. Feld et al. [46] conducted a randomized clinical study on 30 morbid obese patients scheduled for gastric bypass surgery. The patients were classified to receive either fentanyl or opioid-free anesthesia (ketorolac, clonidine, lidocaine, ketamine, magnesium sulfate, and methylprednisolone). They suggested that OFA decreased postoperative sedation, facilitated the recovery, and decreased postoperative opioid usage [46]. Moreover, Mansour et al. [73] studied 28 morbid obese patients undergoing abdominal surgeries under either opioid-based general anesthesia (fentanyl infusion) or non-opioid based general anesthesia (ketamine infusion) and found that non-opioid based general anesthesia provided more pain relief than opioid-based technique. The hemodynamic parameters including heart rate, blood pressure, arterial oxygen saturation, and end-tidal carbon dioxide were indifferent between the two techniques [73].
The randomized controlled placebo trial of Bakhamees et al. [74] revealed that the intraoperative infusion of dexmedetomidine in morbid obese patients undergoing gastric bypass surgery significantly decreased the intraoperative fentanyl and propofol requirements, improved the recovery profile of the patients, and decreased the postoperative pain and analgesic requirements [74]. Also, Ziemann-Gimmel [55] in their randomized clinical trial suggested that opioid-free total intravenous anesthesia significantly decreased the incidence of postoperative nausea and vomiting in comparison to volatile anesthesia-opioid based anesthesia [55]. Moreover, the study of Singh et al. [75] demonstrated that intraoperative infusion of dexmedetomidine in obese patients undergoing bariatric surgery significantly improves the recovery, decrease postoperative pain, postoperative morphine consumption, and postoperative nausea and vomiting [75]. Similarly, the observational cohort trial of Vaughns et al. [76]evaluated the effect of intraoperative infusion of dexmedetomidine on the postoperative pain and analgesic requirement of adolescents undergoing bariatric surgery and concluded that dexmedetomidine significantly decreased the postoperative morphine consumption, but, it increased the incidence of intraoperative hypotension [76].
Furthermore, Bakan et al. [54] in their study compared remifentanil-based anesthesia to opioid-free anesthesia (propofol, dexmedetomidine, and lidocaine infusion) in patients presented for laparoscopic cholecystectomy. They concluded that OFA significantly decreased intraoperative hemodynamic instability, postoperative rescue analgesia consumption, postoperative pain score, and postoperative nausea and vomiting [54].
The POFA trial of Beloeil et al. [77] is a multicenter, prospective randomized, single-blinded study for assessment of the effect of opioid-free anesthesia on the postoperative conditions of the patients. Opioid free anesthesia was found to be beneficial to the patients. The study revealed that OFA has the benefits of reducing the postoperative opioid consumption and hence the postoperative opioid adverse events. Also, it significantly decreases the morbidity of the patients, the length of hospital stays, and the cost. Moreover, a retrospective levelled regression analysis study of Mulier et al. [78] included 9246 patients who were scheduled for laparoscopic bariatric surgeries and evaluated the effect of the continuous deep neuromuscular block and OFA. They suggested that deep neuromuscular block and OFA were associated with improved outcomes after bariatric surgeries and decreased the incidence of complications [78].
The effect on the recovery with the use of OFA is a matter of debate. Multiple studies revealed that length of recovery stay is insignificant with the use of opioid-based or opioidfree anesthesia. Opioid free anesthesia protocols are well known to decrease the effect on the respiratory system which may decrease the length of stay in the post-anesthesia care unit (PACU) and the incidence of respiratory complications. However, these studies are limited by the small sample size and the inconsistency between them. Multiple studies with a larger sample size and evaluating the duration of PACU stay and hospital stay are required [79,80].
The use of OFA in total intravenous anesthesia (TIVA) is not well established. The anesthetic requirements of desflurane are markedly decreased with the use of dexmedetomidine [81,82]. Furthermore, the propofol-based anesthesia technique relies mainly on the pharmacological interaction between propofol and short-acting opioids [83]. The prospective randomized doubleblinded clinical study of Elbakry et al. [82] suggested that opioidfree total intravenous anesthesia (propofol and dexmedetomidine) in patients undergoing bariatric surgeries provided better recovery profile and better postoperative analgesia as compared to inhalational anesthesia using desflurane [82].On the other hand, it is to be noted than many non-opioid analgesic agents as propofol, ketamine, or gabapentin may have potential addictive properties than can lead to long term complications. [84-87] Brandal et al. [80] revised the impact of the application of enhanced recovery and opioid-free anesthesia protocol in their institution and revealed that the intraoperative opioid consumption was decreased dramatically without a significant decrease in the opioid prescribed at discharge. This means that the reduction of intraoperative opioid use is the result of the effort of the institution and additional efforts are required in order to decrease all the postoperative opioid consumption [80].
Anyway, multimodal opioid-free anesthesia seems to be superior to the use of opioids provided that alternative analgesic plane is available and applicable [24]. However, the concept of OFA is practiced without a sufficient evidence base. Despite the growing number of centers using OFA, little data are available in the literature. Most of the published clinical trials about OFA represent the experience of the authors in their centers. Each center may have its own protocol for different surgical interventions using different medications in different doses [68,88].
The introduction of OFA techniques in the era of evidencebased medicine is not proven until this time. The logical thinking may lead us to believe that the avoidance of the use of opioids will decrease its related side effects. However, the change in medical practice is carried out only through obtaining a larger body of evidence through enough clinical trials, systematic review, and meta-analysis. Multiple available clinical studies evaluated the use of OFA techniques in morbid obese patients, however, they evaluated different regimens and different medications. We need multiple clinical studies evaluating the use of different drugs and different protocols of OFA in different surgical procedures. Its effect on the recovery profile, the drug’s interaction, the patient’s satisfaction should be evaluated. Moreover, the duration of recovery, the length of hospital stays, and the cost should be assessed. Indeed, we desperately need a protocol for opioid-free anesthesia practice in morbid obese patients.
Acknowledgment
I would like to acknowledge all the members of the Department of Anesthesia and intensive care, Faculty of Medicine, Tanta University.
References
- Kaiko R (1997) Pharmacokinetics and pharmacodynamics of controlled‐release opioids. Acta anaesthesiologicascandinavica41(1):166-174.
- Brush DE (2012) Complications of long-term opioid therapy for management of chronic pain: the paradox of opioid-induced hyperalgesiaJ Med Toxicol8(4):387-392.
- Schug SA, Raymann A (2011) Postoperative pain management of the obese patient. Best Pract Res Clin Anaesthesiol25(1):73-81.
- Heymsfield SB, Wadden TA (2017) Mechanisms, pathophysiology, and management of obesity. N Engl J Med 376(3):254-266.
- Sarandan M, Guragata-Balasa C, Papurica M, Duta C, Hordovan E, et al. (2011) Anesthesia in laparoscopic bariatric surgery (gastric sleeve)-preliminary experience. TMJ61(1-2):26-31.
- Lloret-Linares C, Lopes A, Declèves X, Serrie A, Mouly S, et al. (2013) Challenges in the optimisation of post-operative pain management with opioids in obese patients: a literature review. Obes Surg 23(9):1458-1475.
- Ingrande J, Lemmens H (2010) Dose adjustment of anaesthetics in the morbidly obese. Br J Anaesth105(suppl_1):i16-i23.
- Lloret-Linares C, Luo H, Rouquette A, Labat L, Poitou C, et al. (2017) The effect of morbid obesity on morphine glucuronidation. Pharmacol Res118:64-70.
- Burghardt PR, Rothberg AE, Dykhuis KE, Burant CF, Zubieta J-K (2015) Endogenous opioid mechanisms are implicated in obesity and weight loss in humans. J Clin Endocrinol Metab100(8):3193-201.
- Gallagher SF, Haines KL, Osterlund LG, Mullen M, Downs JB (2010) Postoperative hypoxemia: common, undetected, and unsuspected after bariatric surgery. J Surg Res 159(2):622-626.
- Mulier J, Wouters R, Dekock M(2014)Pourquoi et comment éviter les opioïdesenanesthésieambulatoire? Non-opioid surgical anaesthesia. JEPU Conference, Paris, UK.
- Urman RD, Böing EA, Khangulov V, Fain R, Nathanson BH, et al. (2019) Analysis of predictors of opioid-free analgesia for management of acute post-surgical pain in the United States. Curr Med Res Opin35(2):283-289.
- Sultana A (2015) Opioid free anesthesia and analgesia in the bariatric patient. Population8: 11.
- Martin JL, Koodie L, Krishnan AG, Charboneau R, Barke RA, et al. (2010) Chronic morphine administration delays wound healing by inhibiting immune cell recruitment to the wound site. Am J Pathol176(2): 786-799.
- Eldufani J(2018) Role of the multidrug-based approach to control chronic pain and cognitive impairment in people with chronic refractory pain: literature review. Papyru: Institutional Repository.
- Fodale V, DArrigo MG, Triolo S, Mondello S, La Torre D (2014) Anesthetic techniques and cancer recurrence after surgery. ScientificWorldJournal2014: 2014.
- Patil SK, Anitescu M (2012) Opioid‐Free Perioperative Analgesia for Hemicolectomy in a Patient with Opioid‐Induced Delirium: A Case Report and Review of the Analgesic Efficacy of the Alpha‐2 Agonist Agents. Pain Pract12(8): 656-662.
- Fletcher D, Martinez V (2014) Opioid-induced hyperalgesia in patients after surgery: a systematic review and a meta-analysis. Br J Anaesth112(6): 991-1004.
- Sultana A, Torres D, Schumann R (2017) Special indications for opioid free anaesthesia and analgesia, patient and procedure related: including obesity, sleep apnoea, chronic obstructive pulmonary disease, complex regional pain syndromes, opioid addiction and cancer surgery. Best Pract Res Clin Anaesthesiol31(4): 547-560.
- Fiore Jr JF, Olleik G, El-Kefraoui C, Verdolin B, Kouyoumdjian A, et al. (2019) Preventing opioid prescription after major surgery: a scoping review of opioid-free analgesia. Br J Anaesth123(5): 627-636.
- Mulier JP, dep Anaesthesiology C, Brugge-Oostende(2015) Opioid free anaesthesia (OFA) a Paradigm shift. AZ Sint.
- Forget P (2019) Opioid-free anaesthesia. Why and how? A contextual analysis. Anaesth Crit Care Pain Med 38(2): 169-172.
- Persyn J, Mulier J-P, Van Lancker P(2016)Comparison of opioid free anaesthesia with opioid anesthesia on postoperative shivering in morbidly obese patients scheduled for bariatric surgery. Euroanaesthesia.
- Kumar K, Kirksey MA, Duong S, Wu CL (2017) A review of opioid-sparing modalities in perioperative pain management: methods to decrease opioid use postoperatively. AnesthAnalg125(5): 1749-1760.
- Lavand’homme P, Estebe J-P (2018) Opioid-free anesthesia: a different regard to anesthesia practice. CurrOpinAnaesthesiol31(5): 556-561.
- Dubin AE, Patapoutian A (2010) Nociceptors: the sensors of the pain pathway. J Clin Invest 120(11): 3760-3772.
- Misiołek H, Zajączkowska R, Daszkiewicz A, Woroń J, Dobrogowski J, et al. (2018) Postoperative pain management-2018 consensus statement of the Section of Regional Anaesthesia and Pain Therapy of the Polish Society of Anaesthesiology and Intensive Therapy, the Polish Society of Regional Anaesthesia and Pain Therapy, the Polish Association for the Study of Pain and the National Consultant in Anaesthesiology and Intensive Therapy. Anaesthesiol Intensive Ther50(3): 179-199.
- Eisenach JC, De Kock M, Klimscha W (1996) α2-Adrenergic Agonists for Regional Anesthesia. A Clinical Review of Clonidine (1984-1995). Anesthesiology 85(3): 655-674.
- Richards M, Skues M, Jarvis A, Prys-Roberts C(1990) Total iv anaesthesia with propofol and alfentanil: dose requirements for propofol and the effect of premedication with clonidine. BJA65(2): 157-163.
- Fernandes HS, Santos SA, Ashmawi HA (2018) Clonidine in anesthesiology: a brief review. Biomed J Sci Tech ResBJSTR.MS.ID.001481.
- Panzer O, Moitra V, Sladen RN (2011)Pharmacology of sedative-analgesic agents: dexmedetomidine, remifentanil, ketamine, volatile anesthetics, and the role of peripheral mu antagonists. Anesthesiology clinics29(4): 587-605.
- Candiotti KA, Bergese SD, Bokesch PM, Feldman MA, Wisemandle W, et al. (2010) Monitored anesthesia care with dexmedetomidine: a prospective, randomized, double-blind, multicenter trial. AnesthAnalg110(1): 47-56.
- Mantz J, Josserand J, Hamada S (2011) Dexmedetomidine: new insights. Eur J Anaesthesiol28(1): 3-6.
- Feld JM, Hoffman WE, Stechert MM, Hoffman IW, Ananda RC (2006) Fentanyl or dexmedetomidine combined with desflurane for bariatric surgery. J Clin Anesth18(1): 24-28.
- Gaszyński T, Czarnik K, ŁazińskiŁ, Gaszyński W (2016) Dexmedetomidine for attenuating haemodynamic response to intubation stimuli in morbidly obese patients anaesthetised using low-opioid technique: comparison with fentanyl-based general anaesthesia. Anaesthesiol Intensive Ther48(5): 275-279.
- Hofer RE, Sprung J, Sarr MG, Wedel DJ (2005) Anesthesia for a patient with morbid obesity using dexmedetomidine without narcotics. Can J Anaesth52(2): 176-180.
- Afshani N(2010) Clinical application of dexmedetomidine. Southern African Journal of Anaesthesia Analgesia16(3): 50-56.
- Kurdi MS, Theerth KA, Deva RS (2014) Ketamine: current applications in anesthesia, pain, and critical care. Anesth Essays Res 8(3): 283-290.
- Kawasaki T, Ogata M, Kawasaki C, Ogata J-i, Inoue Y, et al. (1999) Ketamine suppresses proinflammatory cytokine production in human whole blood in vitro. AnesthAnalg89(3): 665.
- Laulin JP, Maurette P, Corcuff JB, Rivat C, Chauvin M, et al. (2002) The role of ketamine in preventing fentanyl-induced hyperalgesia and subsequent acute morphine tolerance. AnesthAnalg94(5): 1263-1269.
- Green SM, Clem KJ, Rothrock SG () Ketamine safety profile in the developing world: survey of practitioners. Acad Emerg Med 3(6): 598-604.
- Stessel B, Ovink JK, Theunissen HM, Kessels AG, Marcus MA, et al. (2013) Is s-ketamine with or without magnesium sulphate an alternative for postoperative pain treatment? Randomised study. Eur J Anaesthesiol30(2): 91-93.
- Sousa AM, Rosado GM, Neto Jde S, Guimarães GM, Ashmawi HA (2016) Magnesium sulfate improves postoperative analgesia in laparoscopic gynecologic surgeries: a double-blind randomized controlled trialJ Clin Anesth34: 379-384.
- Chan JW, Shetty P (2016)Abstract PR633: Does the Use of Ketamine or Magnesium Decrease Post-Operative Pain Scores in Laparoscopic Bariatric Surgery for Morbid Obesity? AnesthAnalg123(3S): 814-815.
- Beaussier M, Delbos A, Maurice-Szamburski A, Ecoffey C, Mercadal L (2018) Perioperative Use of Intravenous Lidocaine. Drugs78(12): 1229-1246.
- Feld JM, Laurito CE, Beckerman M, Vincent J, Hoffman WE (2003) Non-opioid analgesia improves pain relief and decreases sedation after gastric bypass surgery. Can J Anaesth50(4): 336-341.
- De Oliveira GS, Duncan K, Fitzgerald P, Nader A, Gould RW, et al. (2014) Systemic Lidocaine to Improve Quality of Recovery after Laparoscopic Bariatric Surgery: A Randomized Double-Blinded Placebo-Controlled Trial. Obes Surg 24(2): 212-218.
- Eipe N, Gupta S, Penning J(2016) Intravenous lidocaine for acute pain: an evidence-based clinical update. BJA Education16(9): 292-298.
- Eidy M, Fazel MR, Abdolrahimzadeh H, Moravveji AR, Kochaki E, et al. (2017) Effects of pregabalin and gabapentin on postoperative pain and opioid consumption after laparoscopic cholecystectomy. Korean J Anesthesiol70(4): 434-438.
- Alimian M, Imani F, Faiz SH, Pournajafian A, Navadegi SF, et al. (2012) Effect of oral pregabalin premedication on post-operative pain in laparoscopic gastric bypass surgery. Anesth Pain Med2(1): 12-16.
- De Oliveira GSJ, Castro-Alves LJS, Ahmad S, Kendall MC, McCarthy RJ (2013) Dexamethasone to Prevent Postoperative Nausea and Vomiting: An Updated Meta-Analysis of Randomized Controlled Trials. AnesthAnalg116(1): 58-74.
- De Oliveira Gildàsio S, Almeida Marcela D, BenzonHonorio T, McCarthy Robert J ()Perioperative Single Dose Systemic Dexamethasone for Postoperative Pain: A Meta-analysis of Randomized Controlled Trials. Anesthesiology115(3): 575-588.
- Polderman JAW, Farhang-Razi V, van Dieren S, Kranke P, DeVries JH, et al. (2019) Adverse side-effects of dexamethasone in surgical patientsCochrane systematic review. Anaesthesia74(7): 929-939.
- Bakan M, Umutoglu T, Topuz U, Uysal H, Bayram M, et al. (2015) Opioid-free total intravenous anesthesia with propofol, dexmedetomidine and lidocaine infusions for laparoscopic cholecystectomy: a prospective, randomized, double-blinded study. Braz J Anesthesiol65: 191-199.
- Ziemann-Gimmel P, Goldfarb AA, Koppman J, Marema RT (2014)Opioid-free total intravenous anaesthesia reduces postoperative nausea and vomiting in bariatric surgery beyond triple prophylaxis. BJA112(5): 906-911.
- Grewal A (2011) Dexmedetomidine: New avenues. J Anaesthesiol Clin Pharmacol27(3): 297-302.
- McCarthy GC, Megalla SA, Habib AS (2010) Impact of Intravenous Lidocaine Infusion on Postoperative Analgesia and Recovery from Surgery. Drugs70(9): 1149-1163.
- Albrecht E, Kirkham K, Liu S, Brull R (2013) Peri‐operative intravenous administration of magnesium sulphate and postoperative pain: a meta‐analysis. Anaesthesia68(1): 79-90.
- Schaller SJ, Fink H (2013) Sugammadex as a reversal agent for neuromuscular block: an evidence-based review. Core Evid 8: 57-67.
- Kim JT, Ren CJ, Fielding GA, Pitti A, Kasumi T, et al. (2007) Treatment with Lavender Aromatherapy in the Post-Anesthesia Care Unit reduces Opioid Requirements of Morbidly Obese Patients Undergoing Laparoscopic Adjustable Gastric Banding. Obes Surg 17(7): 920-925.
- Moncada R, Martinaitis L, Landecho M, Rotellar F, Sanchez-Justicia C, et al. (2016) Does Preincisional Infiltration with Bupivacaine Reduce Postoperative Pain in Laparoscopic Bariatric Surgery? Obes Surg 26(2): 282-288.
- Ruiz-Tovar J, Muñoz JL, Gonzalez J, Zubiaga L, García A, Jimenez M, et al. (2017) Postoperative pain after laparoscopic sleeve gastrectomy: comparison of three analgesic schemes (isolated intravenous analgesia, epidural analgesia associated with intravenous analgesia and port-sites infiltration with bupivacaine associated with intravenous analgesia). Surg Endosc31(1): 231-236.
- Brogi E, Kazan R, Cyr S, Giunta F, Hemmerling TM (2016) Transversus abdominal plane block for postoperative analgesia:a systematic review and meta-analysis of randomized-controlled trials. Can J Anaesth63(10): 1184-1196.
- Sinha A, Jayaraman L, Punhani D (2013) Efficacy of Ultrasound-Guided Transversus Abdominis Plane Block After Laparoscopic Bariatric Surgery: a Double Blind, Randomized, Controlled Study. Obes Surg 23(4): 548-553.
- Wassef M, Lee DY, Levine JL, Ross RE, Guend H, et al. (2013) Feasibility and analgesic efficacy of the transversus abdominis plane block after single-port laparoscopy in patients having bariatric surgery. J Pain Res 6: 837-841.
- Albrecht E, Kirkham KR, Endersby RVW, Chan VWS, Jackson T, et al. (2013) Ultrasound-Guided Transversus Abdominis Plane (TAP) Block for Laparoscopic Gastric-Bypass Surgery: A Prospective Randomized Controlled Double-Blinded Trial. Obes Surg 23(8): 1309-1314.
- Ripollés J, Mezquita SM, Abad A, Calvo J (2015) Analgesic efficacy of the ultrasound-guided blockade of the transversus abdominis plane - a systematic review. Rev Bras Anestesiol65: 255-280.
- Lirk P, Rathmell JP (2019) Opioid-free anaesthesia: Con: it is too early to adopt opioid-free anaesthesia today. Eur J Anaesthesiol36(4): 250-254.
- Hontoir S, Saxena S, Gatto P, Khalife M, Ben Aziz AM, et al. (2016) Opioid-free anesthesia:what about patient comfort? A prospective, randomized, controlled trial. Acta AnaesthesiolBelg67(4): 183-190.
- Thorell A, MacCormick AD, Awad S, Reynolds N, Roulin D, et al. (2016) Guidelines for Perioperative Care in Bariatric Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations. World J Surg 40(9): 2065-2083.
- Lam KK, Mui WL (2016) Multimodal analgesia model to achieve low postoperative opioid requirement following bariatric surgery. Hong Kong Med J22(5): 428-434.
- Aronsohn J, Orner G, Palleschi G, Gerasimov M (2019) Opioid-free total intravenous anesthesia with ketamine as part of an enhanced recovery protocol for bariatric surgery patients with sleep disordered breathing. J Clin Anesth52: 65-66.
- Mansour MA, Mahmoud AA, Geddawy M (2013) Nonopioid versus opioid based general anesthesia technique for bariatric surgery: A randomized double-blind study. Saudi J Anaesth7(4): 387-391.
- Bakhamees HS, El-Halafawy YM, El-Kerdawy HM, Gouda NM, Altemyatt S (2007) Effects of dexmedetomidine in morbidly obese patients undergoing laparoscopic gastric bypass. Middle East J Anaesthesiol19(3): 537-551.
- Singh PM, Panwar R, Borle A, Mulier JP, Sinha A, et al. (2017) Perioperative analgesic profile of dexmedetomidine infusions in morbidly obese undergoing bariatric surgery: a meta-analysis and trial sequential analysis. Surg ObesRelat Dis 13(8): 1434-1446.
- Vaughns JD, Martin C, Nelson J, Nadler E, Quezado ZM (2017) Dexmedetomidine as an adjuvant for perioperative pain management in adolescents undergoing bariatric surgery: An observational cohort study. J Pediatr Surg 52(11): 1787-1790.
- Beloeil H, Laviolle B, Menard C, Paugam-Burtz C, Garot M, et al. (2018) POFA trial study protocol: a multicentre, double-blind, randomised, controlled clinical trial comparing opioid-free versus opioid anaesthesia on postoperative opioid-related adverse events after major or intermediate non-cardiac surgery. BMJ Open 8(6): e020873.
- Mulier JP, Dillemans B (2019) Anaesthetic Factors Affecting Outcome After Bariatric Surgery, a Retrospective Levelled Regression Analysis. Obes Surg 29(6): 1841-1850.
- Hakim KYK, Wahba WZB (2019) Opioid-Free Total Intravenous Anesthesia Improves Postoperative Quality of Recovery after Ambulatory Gynecologic Laparoscopy. Anesth Essays Res 13(2): 199-203.
- Brandal D, Keller MS, Lee C, Grogan T, Fujimoto Y, et al. (2017) Impact of Enhanced Recovery After Surgery and Opioid-Free Anesthesia on Opioid Prescriptions at Discharge from the Hospital: A Historical-Prospective Study. AnesthAnalg125(5): 1784-1792.
- Khafagy HF, Ebied RS, Mohamed AH, El-said MH, El-haddad AM, et al. (2017)Effect of dexmedetomidine infusion on desflurane consumption and hemodynamics during BIS guided laparoscopic cholecystectomy: A randomized controlled pilot study. Egyptian Journal of Anaesthesia33(3): 227-231.
- Elbakry AE, Sultan WE, Ibrahim E (2018) A comparison between inhalational (Desflurane) and total intravenous anaesthesia (Propofol and dexmedetomidine) in improving postoperative recovery for morbidly obese patients undergoing laparoscopic sleeve gastrectomy: A double-blinded randomised controlled trial. J Clin Anesth45: 6-11.
- de Sousa GC, Cruz FF, Heil LB, Sobrinho CJS, Saddy F, et al. (2019) Intraoperative immunomodulatory effects of sevoflurane versus total intravenous anesthesia with propofol in bariatric surgery (the OBESITA trial): study protocol for a randomized controlled pilot trial. Trials20(1): 300.
- Samuelson ST, Bryson EO(2017) The impaired anesthesiologist: what you should know about substance abuse. Can J Anaesth64(2): 219-235.
- Wischmeyer PE, Johnson BR, Wilson JE, Dingmann C, Bachman HM, et al. (2007) A Survey of Propofol Abuse in Academic Anesthesia Programs. AnesthAnalg105(4): 1066-1071.
- Xiong M, Shiwalkar N, Reddy K, Shin P, Bekker A (2018) Neurobiology of Propofol Addiction and Supportive Evidence: What Is the New Development? Brain 8(2): 36.
- Johansen ME (2018) Gabapentinoid Use in the United States 2002 Through 2015. JAMA Intern Med178(2): 292-294.
- Doleman B, Sutton AJ, Sherwin M, Lund JN, Williams JP (2018) Baseline Morphine Consumption May Explain Between-Study Heterogeneity in Meta-analyses of Adjuvant Analgesics and Improve Precision and Accuracy of Effect Estimates. AnesthAnalg126(2): 648-660.






























