Management of Patients with Gastrointestinal Hemorrhage While Receiving Warfarin: Vitamin K, Prothrombin Complex Concentrates and Others
Ozgur Karcioglu1*, Mandana Hosseinzadeh2, Ebru Yilmaz3, Selman Yeniocak4, Göksu Afacan5 and Huseyin Metin2
1Emergency Physician, University of Health Sciences, Dept. of Emergency Medicine, Istanbul Education and Research Hospital, Turkey
2Emergency Physician, Department of Emergency Medicine, Bezmialem University, Turkey
3Emergency Physician, Department of Emergency Medicine, Nizip Community Hospital, Turkey
4Emergency Physician, Department of Emergency Medicine, University of Health Sciences, Haseki Education and Research Hospital, Fatih, Turkey
5Emergency Physician, Department of Emergency Medicine, Biruni University, Turkey
Submission: February 18, 2020; Published: March 04, 2020
*Corresponding author: Ozgur Karcioglu, Emergency Physician, University of Health Sciences, Dept. of Emergency Medicine, Istanbul Education and Research Hospital, Turkey
How to cite this article: Karcioglu O, Hosseinzadeh M, Yilmaz E, Yeniocak S, Afacan G et al. “Management of Patients with Gastrointestinal Hemorrhage While Receiving Warfarin: Vitamin K, Prothrombin Complex Concentrates and Others” . What is the evidence?. J Anest & Inten Care Med. 2020; 10(3): 555792. DOI: 10.19080/JAICM.2020.10.555792
Abstract
Introduction: Prothrombin complex concentrates (PCC) has long been used to reverse vitamin K antagonists (VKA) induced coagulopathy rapidly and safely. This article is a review and critical analysis of the most recent literature to analyse treatment with PCC, focusing on gastrointestinal hemorrhage (GIH) in patients on warfarin.
Methods: Currently available literature on the use of PCC in GIH was identified by searches of electronic database. Utilization of PCC was addressed in the literature data found by searches of databases. The indications, efficacy and outcomes associated with the use of the product (three vs four-factors) were reviewed in the articles.
Results: In general, studies point out promising results with respect to PCC use to overcome the VKA–related coagulopathy in GIH. Comparison of different aspects of treatment with PCC and vitamin K, Fresh frozen plasma (FFP) are discussed. PCC is the treatment of choice for normalization of prothtombin time and INR values and resuscitation in emergency setting in patients diagnosed with GIH. Dosing principles are also mentioned.
Conclusions: PCC is the front-line agent to reverse the VKA–related coagulopathy. PCC should be considered for resuscitation in emergency and intensive care in case of severe hemodynamic compromise in GIH associated with high INR values.
Keywords: Prothrombin complex concentrates; Vitamin K antagonists; Warfarin; Coagulopathy; Gastrointestinal hemorrhage
Introduction
Anticoagulants, including vitamin K antagonists (VKA) are administered in a greeat majority of patients who are in need of prophylaxis and treatment of thromboembolic events allover the world. Many patients benefit from anticoagulant effects of VKA in acute and chronic venous and arterial thromboembolic diseases, although nowadays they have been partly replaced by newly generated direct oral anticoagulants. Treatment with VKA therapy necessitates great caution because of its narrow therapeutic window. These drugs also mandate frequent monitoring and dose adjustments. In brief, these agents are not free of adverse effects. The most important and deadly untoward effect is bleeding. The incidence of bleeding in anticoagulated patients is 15–20% in a year [1] and among these, around one-sixth are major bleeding complications (1.7–3.4%) [2]. Although there have been many different treatment modalities in the management of these patients, PCC, FFP and vitamin K are the most commonly used treatment modalities, sometimes in combinations, to mitigate the effects of chronic or acute overdose of VKA. This article is a review and critical analysis of the most recent literature to analyse treatment with Prothrombin Complex Concentrate (PCC) in gastrointestinal hemorrhage (GIH) in patients on warfarin.
Clinical implications of GIH and major bleeding while taking VKA
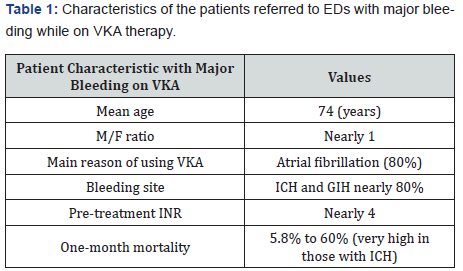
The bleeding events associated with VKA use typically involve the gastrointestinal system, central nervous system, and/ or soft tissues [2]. In 1996, Palerati et al. showed that GIH is the most common major bleeding complication of VKA therapy [3]. Recent publication by the Outcomes Registry for Better Informed Treatment of Atrial Fibrillation (ORBIT-AF) emphasized that GIH comprised more than one-third (38%) of major bleeding episodes in those on warfarin [4]. A number of studies reported that the annual incidence rate of major bleeding episodes in VKA treated patients is between 1% and 3% [5-7]. Every year, life-threatening bleeding occurs in approximately 0.25% of this group of patients [3]. In a Dutch study, Brekelmans et al. analyzed the patients presented with bleeding while on VKA therapy [8]. Mean age was 74 years, 54% were male and 79% received VKA for atrial fibrillation. Most patients presented with ICH (41%) or GI bleeding (36%) (Table 1). Mortality rates are quite variable, in accord with the severity of the patients in the sample. Brekelmans et al. reported a rate as 22% in 2017, while in other studies three out of every five patients have died [8]. Chai-Adisaksopha recorded a rate of 25% in their meta-analyic study in 2016 [9]. More optimistic numbers were given by Sarode in 2013 and Johansen in 2015, as 5.8-7.8%, respectively [10]. Some authors classified the bleeding episodes during VKA treeatment into four classes. A severe clinical presentation (mostly category 3) was observed in almost two-thirds of the patients [11]. Category 4 describes immediately fatal or near-fatal events on presentation and includes around 1% to 5% of the patients.
Assessment of bleeding severity
If >=1 of the following factors are identified in a given patient, the bleeding is classified as a major bleeding.
a) Bleeding in a Critical Site: Intracranial hemorrhage, pericardial tamponade, airway, including posterior epistaxis, hemothorax, intra-abdominal bleeding and retroperitoneal hemorrhage, extremity bleeds
b) Hemodynamic instability: mean arterial pressure<65 mm Hg in invasive monitoring; urine output <0.5 mL/kg/h
c) Overt bleeding with hemoglobin drop >=2 g/dL or administration of >=2 U of packed RBCs
Laboratory thresholds and features (INR and others)
High INR levels are commonly encountered in these patients with warfarin use. This laboratory measurement is thought to be closely related to the risk of GIH. Nearly two decades ago, researchers noted that GIH is three times more common in patients with an INR >3 than in those with INR is found between 2 and 3 [12].
Can we guide PCC and other treatments via laboratory tests?
Yes. Thromboelastometric assays (e.g., ROTEM™ Assays) use citrated whole blood (300μL per assay), which is recalcified and activated by tissue factor (extrinsic pathway), ellagic acid (intrinsic pathway), or ecarin (direct prothrombin activation). Extrinsically activated assays (e.g., EXTEM), intrinsically activated assays (e.g., INTEM, HEPTEM), and ecarin-activated assays have been devised to utilize in this context [13-15]. Since coagulation is triggered via the extrinsic pathway, initial thrombin generation and hence initial clotting mainly depend on the activity of the coagulation factors VII, X, V, II, and fibrinogen in EXTEM test. EXTEM CT can be used to guide FFP and PCC administration in patients suffering from bleeding due to vitamin K-dependent factor deficiency, e.g., due to warfarin therapy [13-15].
EXTEM assay uses [CaCl2 + recombinant tissue factor + polybrene] to evaluate the integrity of the extrinsic pathway. The test allows the clinician to yield objective comment on a probable deficiency of factors of the extrinsic pathway, for example, VKAs (coumadin/ warfarin) and also gives clues on indication for PCC administration.
The history and background of Prothrombin Complex Concentrate (PCC)
Nonactivated PCCs are either 3-factor-PCC, including factors II, IX, and X or 4F-PCC, containing factors II, IX, X, and clinically relevant amounts of factor VII [16]. These were initially developed for use in people with a congenital deficiency in vitamin K-dependent coagulation factors when purified specific coagulation factor is not available [17-19]. The agent is now used for the expedient reversal of untoward high levels of international normalized ratio (INR) [20]. Compared to the 3-factor PCC, the 4-factor PCC has higher concentrations of factor VII in addition to some anticoagulant proteins (Protein C, Protein C, Antithrombin, and heparin) [21].
Treatment principles
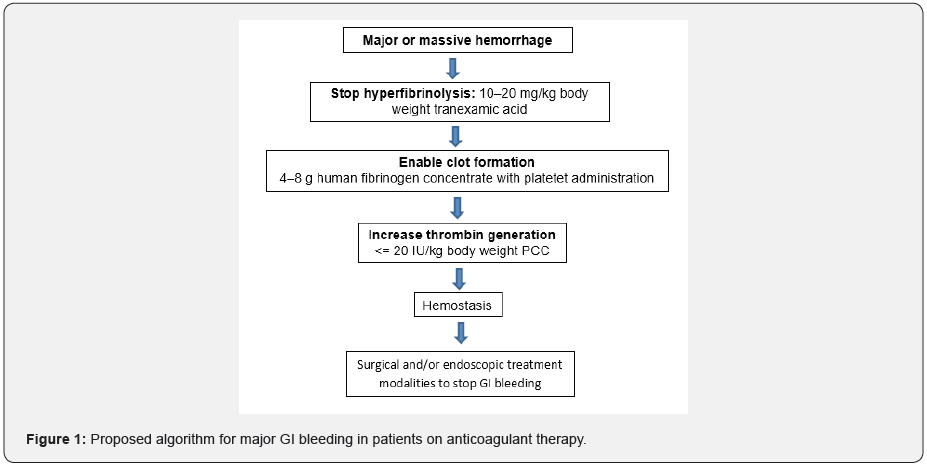
Patients with major bleeding while taking VKA therapy eventually diagnosed with acute overdose of VKA require rapid reversal of life-threatening coagulopathy. Among non-activated agents, four-factor PCC (4 F- PCC) was suggested as the backbone of the treatment strategy of VKA-induced major bleeding from any bodily region by the current guidelines [22,23]. PCC products are purified and concentrated solutions containing primarily the vitamin-K-dependent clotting factors. The preferred PCC is now the 4-factor PCC solution, which contains factors II, VII, IX, and X, as well as the anti-thrombotic factors proteins C and S [24]. For example, activated PCCs are indicated for treatment of hemophilia A or B with inhibitors. The clinical course of VKA-associated major bleeding events treated with PCC was categorized as 3 or 4 in terms of severity with above mentioned criteria, in 50% of patients [8]. Animal and human studies implicated that 4F-PCC demonstrated similar efficacy in relieving coagulopathy associated with the factor Xa inhibitors apixaban and rivaroxaban [25,26]. Indeed, PCC also represents an anticoagulation reversal strategy for patients treated with direct FXa inhibitors, as an alternative to dabigatran and andexanate. Karaca et al. compared the efficacy of PCC and FFP at lowering the INR level, decreasing active hemorrhages visible by endoscopy, and shortening the length of stay at the ED [27]. They noted that patients receiving PCC had INR levels reversed more quickly, less active bleeding on endoscopy, and shorter ED length of stay than in the FFP group. More recently, Refaai et al. focused on shortening the time period spent till the necessary procedure to stop bleeding in those with GIH using four-factor PCC and FFP in an UCLA study [28]. They reported that acute/severe GIH in need of urgent VKA reversal before an invasive procedure, 4F-PCC was linked with lesser infusion volumes, shorter infusion times, and reduced time to procedure, when compared to FFP. Lim et al. enrolled 934 patients with nonvariceal upper GIH in a prospective analysis, which identified the lone independent risk factor associated with all-cause mortality in high risk patients (Glasgow-Blatchford Score≥12) to be the time lapse between presentation and endoscopy [29]. This conclusion can not be extrapolated to low-risk patients (Glasgow-Blatchford Score<12). Only 4-factor PCCs are licensed for rapid warfarin reversal. The agent has the advantage of not requiring blood type testing. Also, it can be stored at room temperature (as lyophilized powder) PCC is dosed based on INR and body weight [9,30]. Administration of PCC was associated with effective haemostasis in 68% to 72% of patients with VKA-associated major bleeding [8,10]. This finding is in line with recommendations on PCC cited in current guidelines [22,31]. If a patient is actively bleeding or requires an urgent invasive procedure, infusion with a plasma-derived coagulation factor concentrate containing the four vitamin K-dependent factors, for example, PCC, plus IV vitamin K 1 (5–10mg), would be appropriate. FFP would be the least efficient replacement therapy due to the large transfusion volume (10–20mL/kg) required to partially and temporarily replenish deficient vitamin K-dependent factor levels [32] (Figure 1).
Alternative agents in the treatment of GIH
FFP is widely used to replace volume and coagulation factors in those with bleeding in order to reverse warfarin. FFP contains all coagulation factors, including fibrinogen. An initial dose of 15- 20ml/kg is deemed suitable [33]. It possesses the advantage of being at hand easily and a lower cost than most other treatment options. On the other hand, being a human product, it can trigger allergic reactions, transmit infections or cause a transfusion related lung injury (TRALI). An ABO compatibility test should be undertaken, which can take a long time to defrost. In addition, a high infusion volume (>1.5L) and a protracted infusion time could be needed to increase coagulation factor concentration [34]. These factors can limit usability of FFP in the emergency setting. Furthermore, high infusion volume can represent a drawback for patients with renal failure or cardiac insufficiency. Any deficiencies of “extrinsic” coagulation factors can be treated with direct supplementation of vitamin K. The agent can be given per oral or parenteral (IV), while 5 to 10 mg of IV vitamin K is preferred with its more rapid onset than oral administration in high-acuity situations [23]. However, the practitioner should keep in mind that it does not result in immediate correction of coagulopathy and thus must be accompanied by PCC or FFP in the management of major bleeds. In addition, a number of welldesigned multicentric controlled studies such as CRASH have pointed out that tranexamic acid can be used with considerable success in traumatic hemorrhages including most fatal ones, i.e., intracranial hemorrhages and shocky states without a defined source of bleeding. In these grave circumstances, tranexamic acid is recommended to be administered within first 3 hours following trauma [35]. However, tranexamic acid has not proven to decrease blood loss or improve clinical outcomes in patients presenting with GI hemorrhage to date [36].
Comparison of PCC with FFP
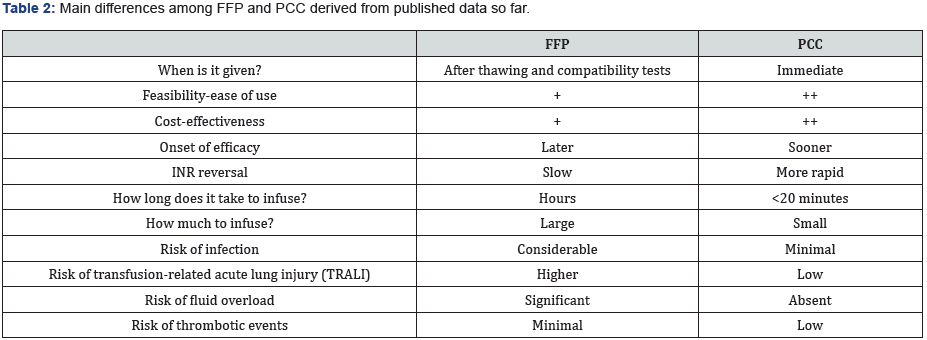
FFP indicates fresh frozen plasma; PCC, Prothrombin Complex Concentrate
Adverse effects and safety issues
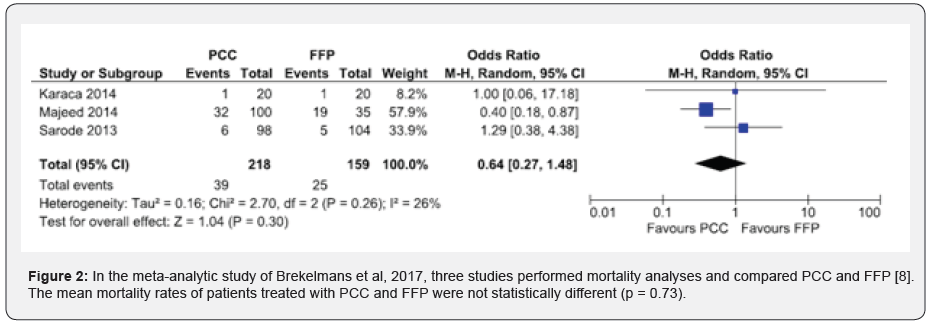
The safety of PCC has been studied by many researchers in the recent decades. Thrombotic events are recognized as the main adverse effects of the treatment and have been postulated to increase in patients treated with higher doses PCC [38]. In many studies, the thrombotic complication rates were reported to fall in the range between 4% and 6.2% [8-10,39,40]. In a metaanalysis, 9 studies (incl. 2262 patents) reported on thrombotic complications. These complications were noted in 0-18% (mean 2.5%) of PCC and in 6.4% of those receiving FFP (BMPA 8). The thrombotic complication rate did not differ between the treatment arms (PCC or FFP) (p = 0.54). The authors concluded that 4-factor PCC is a safe option in reversal of VKA bleeding events in terms of adverse effects.
Dosing issues
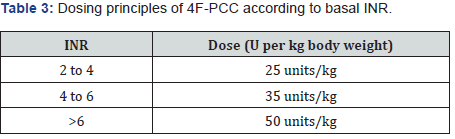
One of the main disadvantages of PCC formulations used by different brands may be its non-standardized nature. Different sources of PCC are standardized based on factor IX levels. A main concern is their compositional differences whose effect on the outcomes are not established clearly [41]. Some sources of PCC supply a low level of factor VII, which are called 3 F-PCC. These 3 F-PCC are thought to be less efficacious in the treatment of VKAinduced coagulopathy [42]. One of the main dosing principles for PCC is using basal PCC as a guide for treatment. Table 3 depicts this rule for three different INR groups. Maximum dose for warfarin reversal is 5,000 units (calculated for 100 kg body weight). PCC is generally administered with vitamin K 10 mg IV [30,43].
Conclusion
PCC compounds has long been used to reverse coagulopathy following use of VKA. In selected cases with major bleeding, vitamin K can be used in conjunction with more rapidly-acting reversal agents such as PCCs. PCC has practical and theoretical advantages to FFP, as it can be used more rapidly and it does not warrant to be thawed or cross-match of blood groups. In patients bleeding on VKA, PCC dosage is established based on patient weight and target INR.
References
- R Sarode, K Matevosyan, R Bhagat, C Rutherford, C Madden, et al. (2012) Rapid warfarin reversal: A 3-factor prothrombin complex concentrate and recombinant factor VIIa cocktail for intracerebral hemorrhage. J Neurosurg 116(3): 491-497.
- S Schulman, RJ Beyth, C Kearon, MN Levine (2008) Hemorrhagic complications of anticoagulant and thrombolytic treatment: American College of Chest Physicians evidence-based clinical practice guidelines (8th Edition). Chest 133(6):257-298.
- G Palareti, N Leali, S Coccheri, Poggi M, Manotti C, et al. (1996) Bleeding complications of oral anticoagulant treatment: An inception-cohort, prospective collaborative study (ISCOAT). Lancet 348(9025): 423-428.
- BA Steinberg, DN Simon, LThomas, Ansell J, Fonarow GC et al. (2017) Management of Major Bleeding in Patients with Atrial Fibrillation Treated with Non-Vitamin K Antagonist Oral Anticoagulants Compared With Warfarin in Clinical Practice (from Phase II of the Outcomes Registry for Better Informed Treatment of Atrial Fibrillation [ORBIT-AF II]). Am J Cardiol 119(10):1590-1595.
- Leissinger CA, Blatt PM, Hoots WK, Ewenstein B (2008) Role of prothrombin complex concentrates in reversing warfarin anticoagulation: a review of the literature. Am J Hematol 83(2): 137-143.
- Jackson SL, Peterson GM, Vial JH, Daud R, Ang SY (2001) Outcomes in the management of atrial fibrillation: clinical trial results can apply in practice. Intern Med J 31(6): 329-336.
- Dentali F, Riva N, Crowther M, Turpie AG, Lip GY, et al. (2012) Efficacy and safety of the novel oral anticoagulants in atrial fibrillation: a systematic review and meta-analysis of the literature. Circulation 126: 2381-2391.
- Brekelmans MPA, Ginkel KV, Daams JG, Hutten BA, Middeldorp S, et al. (2017) Benefits and harms of 4-factor prothrombin complex concentrate for reversal of vitamin K antagonist associated bleeding: a systematic review and meta-analysis. J Thromb Thrombolysis 44(1): 118-129.
- Chai-Adisaksopha C, Hillis C, Siegal DM, Movilla R, Heddle N, et al. (2016) Prothrombin complex concentrates versus fresh frozen plasma for warfarin reversal. A systematic review and meta-analysis. Thromb Haemost 116(5): 879-890.
- Sarode R, Milling TJ Jr, Refaai MA, Mangione A, Schneider A, et al. (2013) Efficacy and safety of a 4-factor prothrombin complex concentrate in patients on vitamin K antagonists presenting with major bleeding: a randomized, plasma-controlled, phase IIIb study. Circulation 128(11): 1234-1243.
- Brekelmans MPA, Abdoellakhan RA, Scheres LJJ, Biedermann JS, Hutten BA, et al. (2017) Clinical outcome of patients with a vitamin K antagonist-associated bleeding treated with prothrombin complex concentrate. Res Pract Thromb Haemost 2(1): 77-84.
- CS Landefeld, RJ Beyth (1993) Anticoagulant-related bleeding: Clinical epidemiology, prediction, and prevention. Am J Med (95)3: 315-328.
- Schmidt DE, Holmström M, Majeed A, Näslin D, Wallén H, et al. (2015) Detection of elevated INR by thromboelastometry and thromboelastography in warfarin treated patients and healthy controls. Thromb Res 135(5): 1007-1011.
- Blasi A, Muñoz G, de Soto I, Mellado R, Taura P, et al. (2015) Reliability of thromboelastometry for detecting the safe coagulation threshold in patients taking acenocoumarol after elective heart valve replacement. Thromb Res 136(3): 669-672.
- Inaba K, Rizoli S, Veigas PV, Callum J, Davenport R, et al. (2015) 2014 Consensus conference on viscoelastic test-based transfusion guidelines for early trauma resuscitation: Report of the panel. J Trauma Acute Care Surg 78(6): 1220-1229.
- Ageno W, Gallus AS, Wittkowsky A, Crowther M, Hylek EM, et al. (2012) Oral anticoagulant therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 141(2): 44-88.
- CSL Behring (2013) Kcentra_ Highlights of Prescribing Information.
- (2013) Octapharma, Octaplex Summary of Product Characteristics.
- CSL Behring (2016) Beriplex P/N Summary of Product Characteristics.
- Hellstern P, Halbmayer W-M, Köhler M, Seitz R, Müller-Berghaus G (1999) Prothrombin complex concentrates: indications, contraindications, and risks: a task force summary. Thromb Res 95(4): 3-6.
- Grottke O, Levy JH (2015) Prothrombin complex concentrates in trauma and perioperative bleeding. Anesthesiology 122(4): 923-31.
- Keeling D, Baglin T, Tait C, Watson H, Perry D et al. (2011) Guidelines on oral anticoagulation with warfarin –fourth edition. Br J Haematol 154: 311-324.
- Holbrook A, Schulman S, Witt DM, Vandvik PO, Fish J, et al. (2012) Evidence-based management of anticoagulant therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141(2): 152-184.
- Hörer T, DuBose JJ, Rasmussen TE, White JM (2020) Endovascular Resuscitation and Trauma Management; Bleeding and Haemodynamic Control.
- Martin AC, Le Bonniec B, Fischer AM, Marchand-Leroux C, Gaussem P, et al. (2013) Evaluation of recombinant activated factor VII, prothrombin complex concentrate, and fibrinogen concentrate to reverse apixaban in a rabbit model of bleeding and thrombosis. Int J Cardiol 168(4): 4228-4233.
- Marlu R, Hodaj E, Paris A, Albaladejo P, Crackowski JL, et al. (2012) Effect of non-specific reversal agents on anticoagulant activity of dabigatran and rivaroxaban: a randomized crossover ex-vivo study in healthy volunteers. Thromb Haemost 108(2): 217-224.
- Karaca MA, Erbil B, Ozmen MM (2014) Use and effectiveness of prothrombin complex concentrates vs fresh frozen plasma in gastrointestinal hemorrhage due to warfarin usage in the ED. Am J Emerg Med 32(6): 660-664.
- Refaai MA, Kothari TH, Straub S, et al. (2017) Four-Factor Prothrombin Complex Concentrate Reduces Time to Procedure in Vitamin K Antagonist-Treated Patients Experiencing Gastrointestinal Bleeding: A Post Hoc Analysis of Two Randomized Controlled Trials. Emerg Med Int 2017: 8024356.
- LG Lim, KY Ho, YH Chan, Teoh PL, Khor CJ, et al. (2011) Urgent endoscopy is associated with lower mortality in high-risk but not low-risk nonvariceal upper gastrointestinal bleeding. Endoscopy 43(4): 300-306.
- Tomaselli GF, Mahaffey KW, Cuker A, Dobesh PP, Doherty JU, et al. (2017) 2017 ACC Expert Consensus Decision Pathway on Management of Bleeding in Patients on Oral Anticoagulants: A Report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. J Am Coll Cardiol 70(24): 3042-3067.
- Hemphill JC 3rd, Greenberg SM, Anderson CS, Becker K, Bendok BR, et al. (2015) Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 46(7): 2032-2060.
- Eby CS (2016) Bleeding and Vitamin K Deficiency. In: Teruya J. Management of Bleeding Patients.
- Hunt BJ, Allard S, Keeling D, Norfolk D, Stanworth SJ, et al. (2015) A practical guideline for the haematological management of major haemorrhage. Br J Haematol 170(6): 788-803.
- Di Fusco SA, Lucà F, Benvenuto M, Iorio A, Fiscella D, et al. (2018) Major bleeding with old and novel oral anticoagulants: How to manage it. Focus on reversal agents. Int J Cardiol 268: 75-79.
- Stansfield R, Morris D, Jesulola E (2020) The Use of Tranexamic Acid (TXA) for the Management of Hemorrhage in Trauma Patients in the Prehospital Environment: Literature Review and Descriptive Analysis of Principal Themes. Shock 53(3): 277-283.
- Smith SR, Murray D, Pockney PG, Bendinelli C, Draganic BD, et al. (2018) Tranexamic Acid for Lower GI Hemorrhage: A Randomized Placebo-Controlled Clinical Trial. Dis Colon Rectum 61(1): 99-106.
- Johansen M, Wikkelso A, Lunde J, Wetterslev J, Afshari A (2015) Prothrombin complex concentrate for reversal of vitamin K antagonist treatment in bleeding and non-bleeding patients. Cochrane Database Syst Rev 7: CD010555.
- Dager WE (2011) Using prothrombin complex concentrates to rapidly reverse oral anticoagulant effects. Ann Pharmacother 45(7-8): 1016-1020.
- Joseph R, Burner J, Yates S, Strickland A, Tharpe W, et al. (2015) Thromboembolic outcomes after use of a four-factor Prothrombin complex concentrate for vitamin K antagonist reversal in a real-world setting. Transfusion 56(4): 799-807.
- Sorensen B, Spahn DR, Innerhofer P, Spannagl M, Rossaint R (2011) Clinical review: Prothrombin complex concentrates–evaluation of safety and thrombogenicity. Crit Care 15(1):201.
- Sadeghi N, Kahn D, Sayed D, Hoppenstadt D, Jeske W, et al. (2014) Compositional differences in commercially available prothrombin complex concentrates. Clin Appl Thromb Hemost 20(3): 256-269
- Voils SA, Baird B (2012) Systematic review: 3-factor versus 4-factor prothrombin complex concentrate for warfarin reversal: does it matter? Thromb Res 30: 833-840.
- Frontera JA, Lewin JJ 3rd, Rabinstein AA, Aisiku IP, Alexandrov AW et al. (2016) Guideline for Reversal of Antithrombotics in Intracranial Hemorrhage: A Statement for Healthcare Professionals from the Neurocritical Care Society and Society of Critical Care Medicine. Neurocrit Care 24(1): 6-46.






























