Intrathecal Dexmedetomidine as an Adjuvant to Bupivacaine Compared to Intravenous Route in Knee Arthroscopies
Amr M Hilal Abdou* and Eman M Kamal Abo Seif
Department of Anesthesia, Intensive Care and Pain Management, Ain Shams University, Egypt
Submission: March 28, 2018; Published: April1 16, 2018
*Corresponding author: Amr M Hilal Abdou, Department of Anesthesia, Intensive Care, and Pain Management, Ain Shams University, Cairo, Egypt, Tel: 01001479037; Email: amr_hilal@med.asu.edu.eg
How to cite this article: Amr M Hilal Abdou, Eman M Kamal Abo Seif . Intrathecal Dexmedetomidine as an Adjuvant to Bupivacaine Compared to Intravenous Route in Knee Arthroscopies. J Anest & Inten Care Med. 2018; 6(3): 555690. DOI: 10.19080/JAICM.2018.06.555690
Abstract
Objective: To evaluate the clinical efficacy of intrathecal Dexmedetomidine (DXM) in comparison with intravenous injection regarding analgesia and sedation during knee arthroscopies.
Background: Spinal block is the preferred mode of anesthesia in knee arthroscopies because of its rapid onset, superior block but lack of postoperative analgesia and relative short duration. Many adjuvants are used to potentiate its effect.
Patients and methods: A total of sixty healthy adult consented patients of both sexes undergoing knee arthroscopy were randomized to receive intrathecal 15mg in 3ml hyperbaric bupivacaine with 5^g in 0.5ml DXM (Group A) or receiving intravenous bolus dose of DXM 0.5 ^g/kg after standard spinal anesthesia (Group B). Assessment of sensory, motor and pain scales were done besides the hemodynamic monitoring, also the time of first rescue analgesic dose were recorded.
Results: Intrathecal DXM potentiate the onset of spinal block and reach the target level of block more rapidly than intravenous route P≤0.05, there were no differences regarding the hemodynamic monitoring.
Conclusion: This study confirmed that intrathecal DXM with dose of 5μg in 0.5 ml with spinal block might help in reaching the desired level of block more rapidly during knee arthroscopy procedures without Perioperative recorded complications with advantage of rapid recovery from the spinal block.
Keywords: Dexmedetomidine; Spinal; Intrathecal
Introduction
Knee arthroscopy procedures is one of the most commonly performed operations nowadays, it is usually performed as a day case procedure which could be performed under local, neuroaxial or general anesthesia. Spinal block is still the first choice because of its rapid onset, superior blockade, lower risk of infection as from catheter in situ, less failure rates and cost-effectiveness compared to epidural block, but has the drawbacks of shorter duration of block and lack of postoperative analgesia [1]. In recent years, use of intrathecal adjuvants has gained popularity with the aim to prolong the duration of block, better success rate, patient satisfaction, decreased resource utilization compared with general anesthesia and faster recovery [2] The quality of the spinal anesthesia has been reported to be improved by the addition of opioids (such as morphine, fentanyl and sufentanil) and other drugs [such as dexmedetomidine (DXM), clonidine, magnesium sulfate (Mg), neostigmine, ketamine and midazolam], but there is no drug to inhibit nociception is without associated adverse effects [3]. DXM was studied as an intrathecal adjunct to bupivacaine spinal block in different surgical procedures. These studies showed that intrathecal addition of DXM to bupivacaine produced shorter onset and longer duration of sensory, motor block, enhanced postoperative analgesia and sedation without serious side effects in different surgical procedures [4] another studies examined the benefits of intravenous DXM as an adjuvant to conventional spinal block in different surgical procedures [6] in this study we will compare the effect of intravenous DXM versus intrathecal route as an adjuvant to the conventional bupivacaine on spinal block in patients undergoing knee arthroscopy procedures.
Methodology
Following approval from our faculty ethical committee and obtaining written informed consent, we enrolled in this prospective randomized comparative study 60 healthy ASA I-II scheduled for knee arthroscopy either diagnostic or therapeutic procedures under spinal anesthesia after enrollment any patient with failed or unsatisfactory block will be excluded from the study. Patients with moderate to severe co-morbidities, allergic to amide local anesthetics, presence of any neurological problems, refusing or failed neuroaxial blockade were excluded from this study. On the operation day; routine preoperative investigations, standard monitoring were applied and the patients are prepared for conventional spinal anesthesia in sitting position. All patients were randomly assigned using computer generated randomized code into two groups; Group A (IT) received 15mg in 3ml hyperbaric bupivacaine intrathecally with 5 μg in 0.5ml DXM, The second group B (IV) received intravenous bolus dose of DXM 0.5 μg /kg over 10 minutes after Intrathecal injection of 15mg in 3ml hyperbaric bupivacaine, both groups will be maintained on intravenous infusion of normal saline.
This study is double blinded and evaluation was done by a separate investigator, vital signs including heart rate (HR), Mean blood pressure (MAP), oxygen saturation (SPO2) were recorded every 5 minutes till the end of surgery and in PACU, any abnormal event as hypotension (MAP less than 50mmHg), HR less than 50 beats/min or hypoxia (SPO2) less than 92% was recorded and treated appropriately. Assessment of level sensory block done by pinprick test and recorded from the peak sensory level every 10 minutes intraoperative and every 30 minutes post-procedure till sensory level regression (S1), Assessment of motor block was done using modified Bromage scale (Table 1) recording of time to reach Bromage 3 block and regression to Bromage 0 done after the operation. Pain assessment is crucial as it is one of the major concerns for patient's satisfaction; we used the visual analogue pain scale (VAS) between 0-10 (0= no pain, 10 = the most severe pain), Duration of complete analgesia was defined as the time from intrathecal injection to VAS score > 0. Duration of effective analgesia was defined as the time to VAS<4, at which point patient will receive Diclofenac75 mg intramuscular injection and Paracetamol 1gram IV infusion were given to achieve VAS < 4. The time of first analgesic request and the total analgesic consumption for 12 hours was recorded. Assessment of sedation intraoperatively and in PACU was done by the using Ramsay sedation scale at 15min interval during surgery and every 30 min interval in PACU. The scale, from 1 to 6, describes a patient as follows:

Awake -1- Anxious or restless or both -2- Cooperative, orientated and tranquil -3- Responding to commands, Asleep-4- Brisk response to stimulus-5- Sluggish response to stimulus-6 -No response to stimulus.
End points of the study was failure of spinal block or conversion into general anesthesia and hospital stay
Statistical Analysis
Sixty patients' data were collected, revised, coded and entered to the Statistical Package for Social Science (IBM SPSS) version 23. The quantitative data were presented as mean, standard deviations and ranges when their distribution found parametric and median with inter-quartile range (IQR) when found non parametric. The comparison between two paired groups with quantitative data and parametric distribution were done by using Paired t-test while data with non parametric distribution were done by using Wilcoxon-Rank test. Spearman correlation coefficients were used to assess the correlation between two quantitative parameters in the same group. The confidence interval was set to 95% and the margin of error accepted was set to 5%. So, the p-value was considered significant at the p < 0.05 and highly significant at the p < 0.01.
Results
During the period of January 2016 to January 2018 this study was performed at Ain Shams University Hospital, Cairo, Egypt. We enrolled 60 healthy patients candidates for knee arthroscopy procedures, these patients were collected into 2 groups; Group A (IT) and Group B (IV); median age 48 years, 48 males and 12 females, no significant adverse events were recognized between both groups. Regarding the comparison according to baseline hemodynamic parameters HR and MAP there was no statistically difference. When recording the MAP variable intraoperative and post procedure, we noticed a difference between both groups as MAP showed significant drop in Group B (IV) as described in Table 2. HR significantly dropped in Group B (IV) with p= 0.029 starting 5 minutes after the bolus infusion of DXM and lasted for 60 minutes as described in Table 3. Concerning the characteristics of spinal block; Group A (IT) reached T6 block and peak level faster than Group B (IV) with significant difference P= 0.028 and <0.001 respectively, regression time in sensory and motor block (Bromage 0) was prolonged in Group A (IT) with P <0.001 (Table 4). Also, as regard regression to Bromage 0, group A showed fastest regression after approximately 165 minutes, followed by group B after 205 minutes in average. Group A patients were the first to ask for analgesia after average 183 minutes, while group B patients asked for analgesia after average of 280 minutes which remarks a significant difference P <0.001 which could reflect the significant difference in VAS score and mean VAS score in favor to group A P <0.001.Finally with respect to the sedation the study showed highest sedation score in group B (IV) of average 2.54 compared to group A (IT) score 2.25 with highly significant value P <0.001.
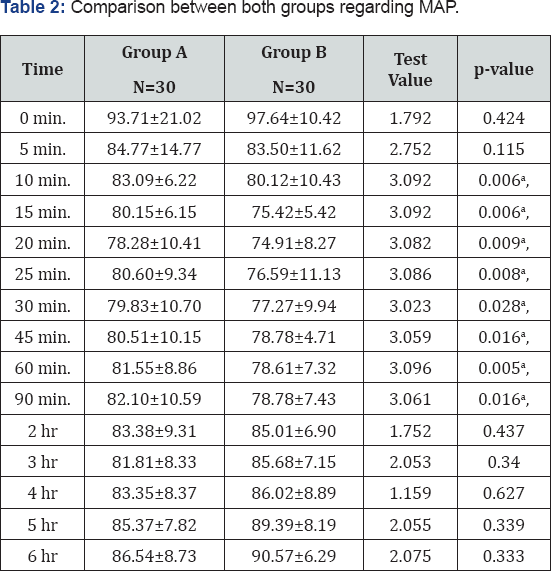
a: Significant difference between group A and group B.


T6: 6th Thoracic dermatome
Min: minutes; hr: hours
a: Significant difference between group A and group B.
Discussion
Day case procedures and its benefits had gained popularity nowadays, stabilizing the financial constrains with respect to patient satisfaction is a major challenge to the anesthesiologist. Knee procedures are commonly performed these days on fast track basis in order to minimize the hospital stay and avoid hospital acquired complications. The choice of spinal anesthesia for the procedures has several advantages as spared spontaneous respiration, low cost, reduced risk of pulmonary aspiration, facilitated surgery via provision of relaxation in the intestines and abdominal wall, elimination of the need for intubation, minimal disruption of blood chemistry, reduced surgical hemorrhage, and earlier return of intestinal motility. However, it has the drawbacks of shorter duration of block and lack of postoperative analgesia [6] in recent years, use of intrathecal adjuvants has gained popularity with the aim of prolonging the duration of block, better success rate, patient satisfaction, decreased resource utilization compared with general anesthesia and faster recovery. Various additives have been used in order to prolong the duration of spinal anesthesia e.g clonidine and ketamine. Agents such as opioids and neostigmine had also been used [7]. Clonidine, an α2-agonist, has been used extensively by intrathecal route and intravenous as an adjuvant to spinal anesthesia [8]. Dexmedetomidine is a novel selective α2 adrenoceptor agonist primarily used for IV sedation. The off-label use of DXM as a local anesthetic adjuvant has been increasingly reported in spinal anesthesia that may cause synergistic effects shown in many studies that examined different doses of intrathecal DXM and bupivacaine in spinal anesthesia as mentioned by Kanazi et al whom added 3μg DXM to 12mg Bupivacaine in spinal anesthesia in TURP surgeries [9], Another study done by Al Ghanem and his colleagues [10] evaluated the effects of intrathecal 10mg isobaric bupivacaine with 5μg DXM. Another study made on lower limb surgeries by Mahendru studied intrathecal 12.5mg bupivacaine plus 5μg DXM response. All these studies concluded that DXM could be an attractive alternative as an adjuvant to spinal anesthesia in alternative surgical procedures [11]. Interestingly, many studies showed that even single dose IV DXM improved spinal anesthesia [12] which has been compared by Kaya and his colleagues as a single preoperative dose of DXM 0.5μg/kg before spinal anesthesia with bupivacaine 0.5% 15mg to midazolam and placebo in lower limb surgeries. Based on the above trials, DXM intrathecal or intravenous has been used safely in patients undergoing surgical procedures under regional anesthesia to prolong the duration of the block, with the use of intrathecal DXM, safety profile should be considered. A number of studies conducted in rats, rabbits, and sheep reported that intrathecal DXM showed no neurological deficit at a dose range of 2.5-100 μg in rat model of perinatal excitotoxic brain injury, and provided potent neuroprotection mediated via the α2A- adrenoreceptors [13]. In human studies, 3-15 μg of DXM showed a prolonged duration of sensory and motor block without any neurological effects [10].
Our results revealed that intrathecal DXM was superior as an adjuvant to bupivacaine in spinal block. Group A had earlier sensory onset to T6 dermatome, higher peak sensory level, prolonged sensory regression time to S1 dermatome, lower mean 12 hours VAS scale, longer time to first analgesic request, less total analgesic consumption, and less overall side effects than group B, Although the mean onset time to reach Bromage 3 level of motor block was comparable between groups A and B, The regression time to Bromage 0 was prolonged in the intrathecal group. However, intravenous DXM was superior in providing better intra-operative sedation level. In our study, intrathecal DXM, enhanced the anesthetic and analgesic properties of bupivacaine and reduced the analgesic requirement more than IV DXM did. It appears that as an adjuvant, neuraxial administration is the appropriate route to DXM, because the analgesic effect of α2 agonists mostly occurs at spinal level, and dexmedetomidine's high lipophilicity facilitates rapid absorption into the cerebrospinal fluid and binding to the spinal cord α2-adrenoreceptor [14]. In our study, the mean arterial pressures, as well as heart rate were found to be significantly lower with IV rather than intrathecal DXM. Intrathecal DXM did not potentiate the effect of bupivacaine on blood pressure which was agreed by another study due to the central effects of DXM [15], finally regarding the side effects our results showed that the most frequent effects were bradycardia and hypotension. Both side effects were reported in both groups A & B but they were more frequent with the intravenous group similar to other study by Niu and his colleagues [4].
Conclusion
Both intrathecal and intravenous Dexmedetomidine were safe adjuvants to bupivacaine during spinal anesthesia in knee arthroscopies, the intrathecal route provided more stable hemodynamics, greater augmentation to sensory and motor block, better post operative analgesia and lesser overall side effects.
Conflict of Interest
Nothing to disclose.
Authors Contribution
We hereby declare that the article has not been published or submitted to or accepted for publication in any form in any other journal. I vouch that the authorship of this manuscript will not be contested by anyone whose names are not listed. On acceptance the article will become the copyright of Journal. The manuscript has been read and approved by all the authors.
References
- Shukla D, Verma A, Agarwal A, Pandey HD, Tyagi C (2011) Comparative study of intrathecal dexmedetomidine with intrathecal magnesium sulfate used as adjuvants to bupivacaine. J Anaesthesiol Clin Pharmacol 27(4): 495-499.
- Chatrath V, Attri JP, Kaur G, Khetarpal R, Bansal P (2014) Comparative evaluation of bupivacaine alone versus bupivacaine and dexmedeto- midine for spinal anesthesia in infraumbilical surgeries. Ain-Shams Journal of Anesthesiology 8(1): 83-88.
- Coskuner I, Tekin M, Kati I, Yagmur C, Elcicek K (2007) Effects of dex- medeto-midine on the duration of anesthesia and wakefulness in bupivacaine epidural block. Eur J Anaesthesiol 24(6): 535-540.
- Niu XY, Ding XB, Guo T, Chen MH, Fu SK, et al. (2013) Effect of Intravenous and Intrathecal Dexmedetomidine in Spinal Anesthesia: A Meta-Analysis. CNS Neurosci Ther 19(11): 897-904.
- Upadhyay SP, Samanth U, Tellicherry Sf, Mallick P (2015) Role of Intravenous dexmedetomidine in Prolonging Postoperative Analgesia and Quality of Block Following Spinal Anesthesia. A Systemic Review and Update. J Pain Relief 4: 2.
- Khan ZP, Munday IT, Jones RM, Thornton C, Mant TG, et al. (1999) Effects of dexmedetomidine on isoflurane requirements in healthy volunteers. 1: Pharmacodynamic and pharmacokinetic interactions. Br J Anaesth 83(3): 372-380.
- Bernard CM (2001) Epidural and spinal anesthesia. In: Barash PG (Ed.), Clinical anesthesia. Lippincott Williams & Wilkins, Philadelphia, pp. 64S-668.
- van Tuijl I, van Klei WA, van der Werff DB, Kalkman CJ (2006) The effect of addition of intrathecal clonidine to hyperbaric bupivacaine on postoperative pain and morphine requirements after Caesarean section: A randomized controlled trial. Br J Anaesth 97(3): 365-370.
- Kanazi GE, Aouad MT, Jabbour Khoury SI, Al Jazzar MD, Alameddine MM, et al. (2006) Effect of low dose dexmedetomidine or clonidine on thecharacteristics of bupivacaine spinal block. Acta Anesthesiol Scand 50(2): 222-227.
- Al Ghanem Subhi, Islam Massad, Mahmoud Al Mustafa, Khaled Al Za- ben, Ibrahim Y, et al. (2009) Effect of Adding Dexmedetomidine versus Fentanyl to Intrathecal Bupivacaine on Spinal Block Characteristics in Gynecological Procedures. Am J Applied Sci 6(5): 882-887.
- Mahendru V, Tewari A, Katyal S, Grewal A, Singh M R, et al. (2013) A comparison of intrathecal dexmedetomidine, clonidine, and fentanyl as adjuvants to hyperbaric bupivacaine for lower limb surgery: A double blind controlled study. J Anaesthesiol Clin Pharmacol 29(4): 496502.
- Kaya FN, Yavascaoglu B, Turker G, Yildirim A, Gurbet A, et al. (2010) Intravenous dexmedetomidine, but not midazolam, prolongs bupivacaine spinal anesthesia. Can J Anaesth 57(1): 39-45.
- Lo WC, Harris Jand Clarke RW (2003) Endogenous opioids support the spinal inhibitory action of an alpha 2-adrenoceptor agonist in the decerebrated, spinalised rabbit. Neurosci Lett 340(2): 95-98.
- Ishii H, Kohno T, Yamakura T, Ikoma Mand Baba H (2008) Action of dexmedetomidine on the substantia gelatinosa neurons of the rat spinal cord. Eur J Neurosci 27(12): 3182-3190.
- Talke P, Chen R, Thomas B, Aggarwall A, Gottlieb A, et al. (2000) The hemodynamic and adrenergic effects of perioperative dexmedetomidine infusion after vascular surgery. Anesth Analg 90(4): 834-839.






























