Intraoperative Serum Lactate Concentration and Central Venous Oxygen Saturation as Early Predictors for Early Graft Function during Liver Transplant
Sayed E1*, Selman E2, EL Kholy A1, Khalil M1 and Lotfy M2
1National Liver Institute, Department of Anaesthesia and Intensive Care, Menoufia University, Egypt
2Faculty of Medicine, Department of Anaesthesia and Intensive Care, Menoufia University, Egypt
Submission: December 16, 2017; Published: January 22, 2018
*Corresponding author: Eman Sayed Ibrahim, National Liver Institute, Department of anaesthesia, Menoufia University, 5th Abdullah Ismael -Nasser EL thawra, EL haram, Giza, Egypt, Tel: +202 01282271464; 0123548537; Email: emansayed825@gmail.com
How to cite this article: Sayed E, EL Kholy A, Selman E, Khalil M and Lotfy M. Intraoperative Serum Lactate Concentration and Central Venous Oxygen Saturation as Early Predictors for Early Graft Function during Liver Transplant. J Anest & Inten Care Med. 2018; 5(1) : 555655. DOI: 10.19080/JAICM.2018.05.555655
Abstract
Background: Initial poor graft function (IPGF) following living donor liver transplantation (LDLT) causes complications, result in poor prognosis. We evaluated that if intraoperative changes in blood lactate level and central venous oxygen saturation (ScvO2) after hepatic allograft reperfusion can reflect IPGF following LT.
Methods: 40(LDLT) recipients were studied. Patient State Index (PSI), monitored anaesthesia depth with Desflurane (Des) % and fentanyl altered accordingly. Transoesophageal Doppler (TED), invasive mean arterial blood pressure (MABP, mmHg) and heart rate (HR, beat/min) were monitoring; TED was used for fluid optimization. Intraoperative serum lactate and daily collection of serum and plasma samples to for routine biochemistry analysis, liver function test. Venous blood gases were collected from central venous catheter.
Results: IPGF occurred in 4 of the 40 patients (10%). Both serum lactate and ScvO2 following reperfusion in the non-IPGF group was markedly lower than that in the IPGF group (p<0.001). Lactate clearance was positive in non-IPGF group in contrast to IPGF group in which lactate clearance was in a negative value, (p<0.001). Receiver operating characteristics curve (ROC) analysis resulted in an area under the curve of 0.65 for serum lactate and 0.46 for ScvO2, with a sensitivity and specificity of 83 % and 50 % for serum lactate and 94% and 25 % for SvO2. The optimum cut-off values for lactate and ScvO2 predicting IPGF were 31.55% and 66.5 respectively.
Conclusions: Both changes in intraoperative blood lactate and ScvO2 after hepatic allograft reperfusion served as good predictor of initial graft function during LT.
Keywords: Lactate; Central venous oxygen saturation; Predictors; Graft function
Introduction
Adult living donor Liver transplantation (ALDLT) is currently considered to be the ultimate form of therapy for most patients with end-stage liver diseases. Liver transplantation represents a complex surgical procedure, in which several factors determine postoperative recipient and graft survivals. Three risk categories may be considered: namely, those related to the patient, to the organ to be transplanted and to the surgical procedure [1]. Early graft function after liver transplantation (LT) is an important prognostic parameter for the individual outcome. Initial poor graft function (IPGF) has been described as a borderline dysfunction with the potential to recover. IPGF is a multifactorial event, which is related to different risk factors, such as marginal donors, severe ischemia-reperfusion injuries, acute rejection episodes or vascular complications [2]. The diagnostic standard for IPGF has not been set yet, and there are different opinions among some reported definitions. As, Silberhumer GR et al. [3] proposed four grades of initial graft function over the first postoperative 5 days as:
a. Good function, AST maximal 1000 UI/L and spontaneous prothrombin time >50%;
b. Fair function, AST 1000-2500 UI/L, clotting factor support < 2 days;
c. IPF, AST >2500 UI/L, clotting factor support >2 days; and
d. PNF; Retransplantation required within 7 days. We designed this study to evaluate if intraoperative changes in blood lactate level and (ScvO2) after hepatic allograft reperfusion can reflect IPGF following LT.
Patients and Methods
Approval was obtained from the local ethics and research committees of the Anaesthesia and Intensive Care Department of National Liver Institute, Menoufia University, Egypt (IRB 0100/2014). This observational analytical prospective cohort study of 40 ALDLT recipients from both sexes. A written informed consent was taken from each patient. Patients aged 18 to 60 yrs. had model of end stage liver disease (MELD) between 12 and 20 we enrolled in this , patients with extra hepatic malignancy, severe renal impairment, cardiovascular disorders and patients with severe pulmonary disorders were exclude. preoperative assessment and all patients were monitored by standard routine monitoring by the monitor of DatexOhmeda (GE, USA) which includes ECG, noninvasive blood pressure (NIBP), continuous invasive blood pressure (IBP) and arterial blood gas analysis, pulse oximetry (SaO2), capnography to monitor end tidal CO2, fractional inspired oxygen concentration (FiO2) ,neuromuscular blockade monitoring via accleromyography, NMT module, Dragger, USA), core temperature (nasopharynx probe) ,continuous Central venous pressure (CVP), trans-esophageal Doppler TED (CardioQ) parameter (Deltex Medical, Chichester, UK, FDA approved) for cardiovascular function (Cardiac output) and perioperative fluid optimization, Patient State Index (PSI), SEDLine (Masimo, Irvine, CA) monitored anaesthesia depth (2550) with Desflurane (Des) % and fentanyl altered accordingly and rational Thromboelastometry. (ROTEM)(Pentapharm GmbH, Munich, Germany).
Adequate preoxygenation was done before the administration of any pharmacological agents. General anaesthesia was induced by rapid sequence induction with cricoid pressure using sleeping dose of Propofol titrated (approximately 20 mg every 10 seconds, 1-1.5 mg/kg) against the patient's response to both patient state index anaesthesia depth monitoring (SED-line, Masimo, Irvin, USA)and clinical signs. Rocuronium 0.9mg/kg to facilitate endotracheal intubation under neuromuscular nerve monitoring. Anaesthesia was maintained with Desflurane (Baxter, Erlangen, Germany) in O2/Air mixture (FIO2 =0.4), fentanyl, and rocuronium to keep the patient state index anaesthesia depth monitoring between 25-50. Lactate and Scvo2 data would be collected at the following times: The day before operation.
(T0); Base line data, one hour after induction.
(T1); Dissection phase, 30 min after clamping of the portal vein
(T2); Anhepatic phase, 10min after reperfusion of the portal vein
(T3); Reperfusion phase, 2 hours post reperfusion
(T4); Postoperative data collection include standard coagulation test: (International Normalized Ratio INR, PTT, PC), liver function tests (AST, ALT (U/L), Bilirubin Total and Direct (mg/dl)), renal function tests (Urea and creatinene) (mg/dl), Lactate levels (mg/dl) and they would be collected postoperatively at the first post- operative dayPOD1
(T),(T6) POD3 and
(T7); pod5.
Lactate clearance calculated from the formula:
Lactate clearance= (lactate at anhepatic stage (T2) - (lactate at 2 hours post reperfusion stage (T4) divided by (lactate at anhepatic stage (T2) x100%.
Sample Size and Power of The Study
Patients were admitted to our National Liver Institute from 2013-2015 meeting the inclusion and exclusion criteria. Forty patients were involved in our study. In the present study a was set to 0.05, and maximum accepted 20% with minimum power of study 80% of mean of predicted lactate.
Statistical Analysis of the Collected Data
Results were collected, tabulated and statistically analyzed by an IBM compatible personal computer with SPSS statistical package version 20 (SPSS Inc. Realesed 2011. IBM SPSS statistics for windows, version 20.0, Armnok, NY: IBM Corp.). Descriptive statistics, was expressed in: Number (No), percentage (%) mean (x) and standard deviation (SD). Analytic statistics, student's t-test is a test of significance used for comparison of quantitative variables between two groups of normally distributed data, while Mann Whitney’s test was used for comparison of quantitative variables between two groups of not normally distributed data. Wilcoxon test was used to compare between 2 consecutive means in the same group of not normally distributed data. Spearman correlation was used to study the correlation between two quantitative variables. p< 0.05 was considered statistically significant.
Results
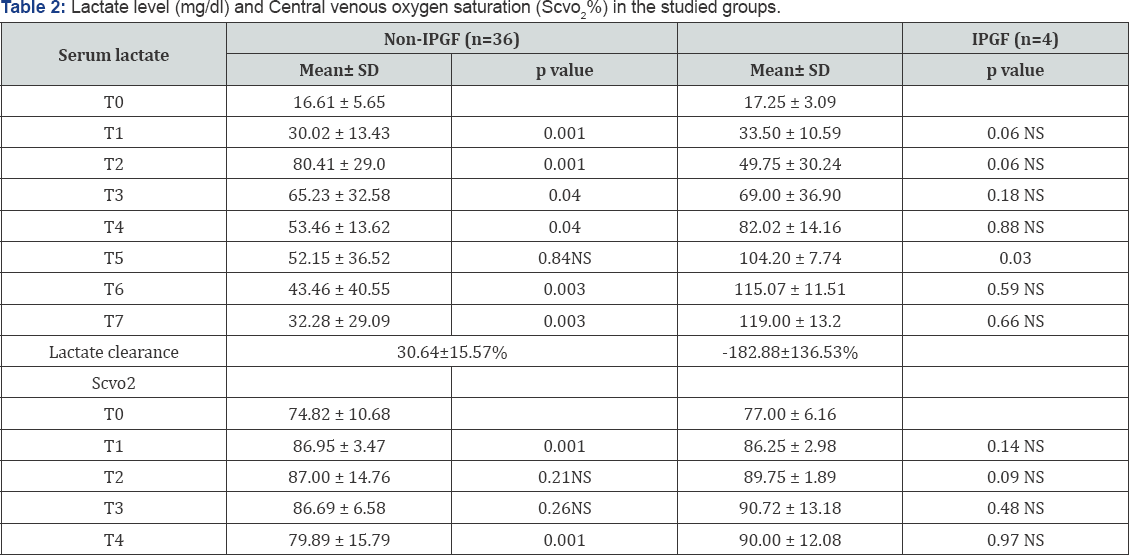
The two studied groups, patients with functioning graft group (non-IPGF) (n=36) and patients with initial poor graft function group (IPGF) (n=4). Patients' characteristics of both groups were comparable. Mean (MELD) values were (15.05±2.85 vs. 17.50±1.91) in non-IPGF group and IPGF group respectively, p=0.10.Sex distributions were comparable in the two studied groups.Concerning with the intraoperative date, there was statistically significant increase in mean operative duration (11.37±0.75 vs. 12.75±2.77 hrs. p= 0.01) in non-IPGF & IPGF group respectively. There were statistically significant increase in mean an hepatic, cold and warm ischemia times in IPGF group, The mean of graft body weight ratio (GBWR) was (1.02±0.15 vs. 0.97±0.06, p=0.40) in non-IPGF & IPGF group respectively, Table 1. In non-IPGF group, mean preoperative lactate was (T0 16.61±5.65) mg/dl.

Demographic data were presented as mean ± SD, tested by Mannwhiteny test, while sex difference tested by chi square test p>0.05.
Body mass index: BMI; Model End Stage Disease: MELD.sOperative data were presented by mean ± SD using Wilcoxon test showing
ANT: An hepatic Time; CIT: Cold Ischemia Time; WIT: Warm Ischemia Time; AGBWR: Actual Graft Body /Weight Ratio; OT: Operative Time.
Data was considered statistically significant, p< 0.05.
NS: Non significant, p> 0.05.
There were statistically significant increase in mean values of intraoperative lactate at the dissection & an hepatic phases, p =0.00, then followed by statistically significant decrease in mean lactate values at the reperfusion & 2hours post reperfusion phases, p=0.04. There were decreases in mean values of post -operative lactate. Regarding IPGF group, mean preoperative lactate was (T0 17.25±3.09) mg/dl. The mean values of serum lactate continue to increase all over the intra and post -operative period. Lactate clearance was positive in non-IPGF group(30.64±15.57%) in contrast to IPGF group in which lactate clearance was in a negative value(-182.88±136.53%) and it was statistically significant, p&#x-003C;0.001, Table 2. In non-IPGF group, there were decreases in SCVO2 values at their perfusion phase without reaching significant value, p= 0.26, then followed by significant decrease at 2hours post reperfusion phase, p=0.00.
Data were presented by mean ± SD using Wilcoxon test, and p<0.05 is considered statistically significant,
NS: non-significant, p> 0.05.
T0: preoperative lactate and Scvo2,
T1 lactate and Scvo2 in dissection phase,
T2: lactate and Scvo2 in anhepatic phase,
T3: lactateand Scvo2 in reperfusion phase,
T4: lactate and Scvo2 in 2 hrs post reperfusion phase,
T5: lactate in POD1,
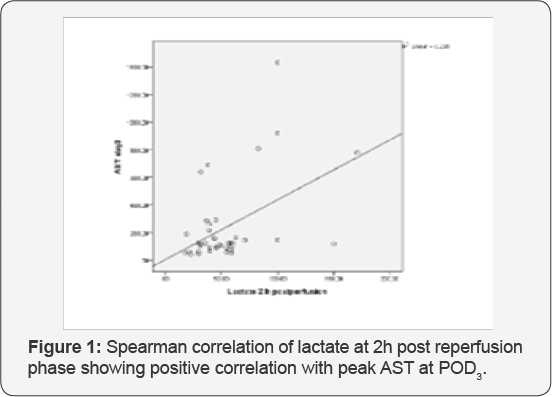
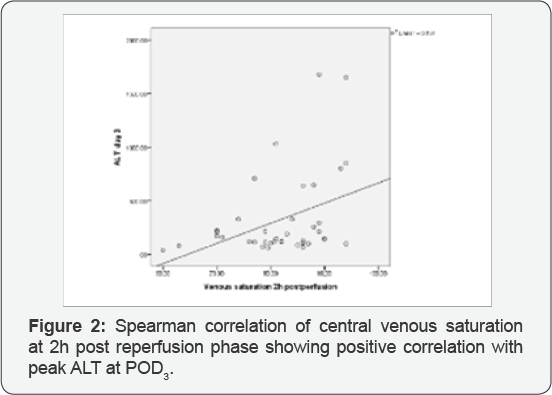
Receiver operating characteristics curve (ROC) analysis resulted in an area under the curve of 0.65 for serum lactate and 0.46 for ScvO2, with a sensitivity and specificity of 83% and 50% for serum lactate, and 94% and 25% for ScvO2. The optimum cutoff values for lactate and ScvO2 predicting IPGF were 31.55% and 66.5 respectively, as presented in Figures 3 & 4. (ROC) analysis for lactate clearance resulted in an area under the curve 92.4%, with a sensitivity and specificity 100% and 78% and cut-off value was 19.59%, presented in Figure 4. Regarding liver enzymes and coagulation, in non-IPGF group, there were increase in liver enzymes and coagulation postoperatively reaching peak level at post- operative day3 then tends to decrease. In contrast with IPGF group, liver enzymes continue to rise with persistant coagulopathy as presented in Table 3.

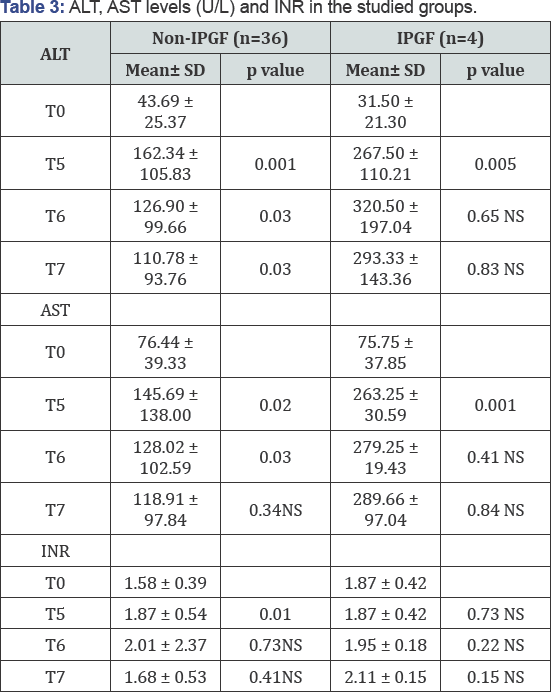
Data were presented by mean ± SD using Wilcoxon test, and p< 0.05 is considered statistically significant,
NS: non-significant, p> 0.05.
ALT: Alanine amino transferase ,
AST: Aspartate amino transferase.
INR: International normalized ratio.
T0: preoperative ALT, AST and INR,
T5: ALT, AST and INR in POD1,
T6: ALT, AST and INR in POD3
T7: ALT, AST and INR in POD5.
Discussion
The main finding in our study was Lactate clearance was positive in non-IPGF group in contrast to IPGF group in which lactate clearance was in a negative value. Early graft function after liver transplantation (LT) is an important prognostic parameter for the individual outcome [4]. Initial poor graft function (IPGF) has been described as a borderline dysfunction with the potential to recover [5], which appears as a form of temporary/ reversible liver insufficiency. Among the parameters most frequently utilized for prediction of early mortality in OLT patients, the perioperative blood lactate levels, INR and the MELD post-OLT seem to be more precise. The serial blood lactate monitoring has been assessed in the critically ill patient in many situations [6] demonstrated that persistent elevation of blood lactate level in septic patients can be a reliable predictor of multiple organ failure syndrome and poor prognosis. Thus, in the ICU setting the study of the behavior of this metabolite in severe sepsis or septic shock is gaining terrain even more, since the reduction of elevated blood lactate level is considered to be the primary target of the treatment of the critically ill patient [7].
Hyperlactatemia has been shown to be associated with increased mortality and morbidity in a critical care setting in patients following liver resection [8]. Temporary absence of the liver during the anhepatic phase results in lactic acidosis until the graft liver is reperfused. Svensson et al. [9] reported similar blood lactate profiles in LT. They demonstrated that the inability of the hepatic graft to reduce lactate levels was associated with primary graft non function, as well as an increased incidence of mortality or morbidity during early postoperative periods. De Gasperi et al. [10] recorded that blood lactate appears to be a useful indicator of hepatic metabolic recovery of its functional capabilities in the immediate postoperative period of OLT. Nishimura et al. [11] & Orii et al. [12] conducted that the lactate clearance is associated to the size of the graft. Bernal et al. [13] reported that arterial blood lactate measurement rapidly and accurately identifies patients who will die from paracetamol-induced acute liver failure and its use can improve the speed and accuracy of selection of appropriate candidates for transplantation. Wu et al. [14] concluded that early postoperative lactate clearance can serve as a prompt and accurate bedside predictor of IPGF. Lactate clearance, especially early clearance as an important index in severe sepsis and septic shock, is reported to perform well in the prediction of prognosis with a high sensitivity and specificity.
We used ROC analysis to determine the best cut off of early lactate clearance rate and its sensitivity and specificity in liver transplant patients as a predictor of IPGF. Our study revealed that change in intraoperative blood ScvO2 after hepatic allograft reperfusion served as good predictor of initial graft function during LT. The oxygen available to the tissues at every stage of the liver transplantation is dependent on the cardiac output and the arterial oxygen content (CaO2). Bleeding, vascular clamping, and hypotension are some of the factors that may adversely affect the oxygen availability with undesirable consequences on graft viability in orthotopic liver transplantation [15]. Abnormal changes in total oxygen consumption are considered as an early indicator for the occurrence of primary nonfunction of the newly transplanted liver. Attempts to correlate changes in oxygen uptake with the function of the newly implanted liver have also had contradictory results [16]. Monitoring hemodynamic parameters during liver transplantation allows control of tissue oxygenation and allograft viability. The significance of oxygen transport and the role of oxygen consumption as a predictor of outcome in a variety of diseases have been discussed but ideal values for oxygen transport are difficult to maintain during liver transplantation.
In patients with chronic liver failure, peripheral vasodilation, mainly in the splanchnic circulation, and a compensatory increase in cardiac output and hence oxygen delivery exist. Severe intrapulmonary and extrapulmonary arterial-venous admixturing characterizes this group of patients, [17] and a rightward shift in the ODC is usually observed in patients with cirrhosis. One quarter of whole-body O2 consumption (VO2) in awake humans is due to splanchnic (liver) metabolism, and this fraction increases during general anesthesia, under which extra splanchnic O2 metabolism is selectively inhibited with relative preservation in the splanchnic bed. The liver remains a major contributor to VO2 even in the'presence of end stage disease, as evidenced by the marked decrease in VO2 after hepatectomy during liver transplantation (OLT). Our study showed that there was a decrease in central venous saturation in the immediate reperfusion phase as the neohepatic graft start to consume oxygen but if a graft fails to function, there was failure to decrease central venous saturation as there was no oxygen consumption by the neohepatic graft.
This agreed with Takaya et al. [18] & Scalea TM et al. [16], showed that in addition to increased lactate or metabolic acidosis, ScvO2 may indicate inadequate oxygen delivery earlier. The occurrence of IPGF is associated with many factors, but confirmation has not yet been made [20]. IPGF is a serious clinical complication after OLT, with elevation of serum aminotransferase. Some patients went to primary graft non function PGNF, [21] which was manifested by hepatocellular necrosis, rapidly rising transaminases, absence of bile production, severe liver-related coagulation deficit, high lactate levels, systemic hemodynamic instability and acute renal failure [22]. Varotti et al. [23] vascular complications represent a second group of early complications after LT associated with high morbidity and mortality .The clinical pictures of these complications may vary considerably, but if not promptly treated they can also lead to graft failure. How to avoid IPGF is the crucial problem that the transplant surgeon must deal with. Gruttadauria S et al. [24] described a condition that we defined as early graft dysfunction (EGD) which could be identified preoperatively and concluded that EGD could be identified preoperatively and was associated with increased morbidity after LRLT.
A prompt recognition of EGD can trigger a timely treatment. Salvalaggio et al. [25] created a grading system for EGD. Lee et al. [26] concluded that donation after cardiac death (DCD) liver allografts have been associated with increased morbidity from primary non-function, biliary complications, early allograft failure, cost, and mortality. Early allograft dysfunction (EAD) after liver transplantation has been found to be associated with worse patient and graft survival. Siniscalch et al. [27] demonstrated that patients with and without post transplantation complications had slightly different mean MELD scores. FilhoI et al. [28] concluded that the MELD post-OLT performed better in predicting early mortality of patients after orthotopic liver transplantation. Wagener et al. [29] demonstrated that the MELD score could be a useful tool not only for pretransplant graft allocation but also for postoperative risk stratification. We noticed in our current study that the duration of anhepatic phase, cold and warm ischemia times were prolonged in patients with IPGF more than patients with non-IPGF. During the anhepatic phase, cytokines, metabolites, and other toxins accumulated in the splanchnic system. Ijtsma et al. [30] concluded that the anhepatic phase duration over 100 minutes to be an independent predictor of graft dysfunction, which was associated with significantly lower patient survival.
Microcirculation of grafted liver could be gradually resumed normal after reperfusion when WIT was less than 30 min, which indicated that hepatic cells held the recovery potency and could regain normal microcirculatory structure after reperfusion if the WIT was less than 30 min. After 45 min of warm ischemia, most hepatic sinus was unobstructed, but there were still some sinus filled with cytoplasm blebs, reticular fibrosis and hemocytes. So 45 min may be the deadline of hepatic warm ischemia. When WIT was more than 60 min, microcirculatory structure of liver graft presented irreversible injuries. [31] During the phase of cold ischemia, loss of mitochondrial respiration and ATP depletion occur consequently though hypothermia reduce the metabolic rate and prolongs the time that anoxic cells can retain essential metabolic function [32]. In agreement with, Totsuka et al. [33] revealed that cold-ischemia time appears to be a good predictor of not only PNF but also of patient and graft survival. There are several limitations in our study. First, continuous monitoring of Scvo2 intraoperatively by a new fibro-optic technology was not available in our study. Second, the unavailability of Swan Gaunaz catheter for collection of mixed venous saturation for good estimation of oxygen consumption by the neograft.
References
- Chen XB and Xu MQ (2014) Primary graft dysfunction after liver transplantation. Hepatobiliary Pancreat Dis Int 13(2): 125-137.
- Burton JR, Rosen HR (2006) Diagnosis and management of allograft failure. Clin Liver Dis 10(2): 407-435.
- Silberhumer GR, Gerd R, Pokorny H, Hetz H, Herkner H, et al. (2007) Combination of extended donor criteria and changes in the Model for End-Stage Liver Disease Score predict patient survival and primary dysfunction in liver transplantation: A retrospective analysis. Transplantation 83(5): 588-592.
- Pokorny H, Gruenberger T, Soliman T, Rockenschaub S, Langle F, et al. (2000) Organ survival after primary dysfunction of liver grafts in clinical orthotopic liver transplantation. TransplInt 13(Suppl 1): S154-S157.
- Maring JK (2005) Studies on predictability of early graft function after liver transplantation. University of Groningen p. 1-13.
- Bakker J, Gris P, Coffernils M, Kahn RJ, Vincent JL (1996) Serial blood lactate levels can predict the development of multiple organ failure following septic shock. Am J Surg 171(2): 221-226.
- Nichol AD, Egi M, Pettila V, Bellomo R, French C, et al. (2010) Relative hyperlactatemia and hospital mortality in critically ill patients: aretrospective multi-centre study. Crit Care 14(1): R25.
- Wiggans MG, Starkie T, Shahtahmassebi G, Woolley T, Birt D, et al. (2013) Serum arterial lactate concentration predicts mortality and organ dysfunction following liver resection. Perioper Med (Lond) 2(1):-21.
- Svensson KL, Persson H, Henrikson A (1989) Whole body gas exchange, amino acid and lactate clearance as indicators of initial and early allograft viability in liver transplantation. Surgery 105(4): 472.
- De Gasperi A, Mazza E, Corti A, Zoppi F, Prosperi M, et al. (1197) Lactate blood levels in the perioperative period of orthotopic liver transplantation. Int J Clin Lab Res 27(2): 123-128.
- Nishimura A, Hakamada K, Narumi S, Totsuka E, Toyoki Y, et al. (2004) Intraoperative blood lactate level as an early predictor of initial graft function in human living donor liver transplantation. Transplantation Proceedings 36(8): 2246-2248.
- Orii R, Sugawara Y, Hayashida M, Yamada Y, Kubota K, et al. (2000) Peri-operative blood lactate levels in recipients of living-related liver transplantation. Transplantation 69(10): 2124-2127.
- Bernal W, Donaldson N, Wyncoll D, Wendon J (2002) Blood lactate as an early predictor of outcome in paracetamol-induced acute liver failure: a cohort study. Lancet 359(9306): 558-563.
- Wu JF, Wu RY, Chen J, Ou Yang B, Chen MY, et al. (2011) Early lactate clearance as a reliable predictor of initial poor graft function after orthotopic liver transplantation. Hepatobiliary Pancreat Dis Int 10(6): 587-592.
- Walsh T, Garden O (2002) Metabolic, cardiovascular and acid-base status after hepatic artery or portal vein reperfusion during orthotopic liver transplantation. Liver Transpl 8(6): 537-544.
- Walsh T, Hopton P, Garden O, Lee A (1998) Effects of graft reperfusion on haemodynamics and gas exchange during liver transplantation. Br J Anaesth 81(3): 311-316.
- Mohamed R, Freeman JW, Guest PJ, Davies MK, Neuberger JM (2002) Pulmonary gas exchange abnormalities in liver transplant candidates. Liver Transpl 8(9): 802-808.
- Takaya S, Nonami1T, Selbyl R, Doyle H, Murray G, et al. (1993) The relationship of systemic hemodynamics and oxygen consumption early allograft failure after liver transplantation.Transpl Int 6(2): 73-76.
- Scalea TM, Hartnett RW, Duncan AO, Atweh NA, Phillips TF, et al. (1990) Central venous oxygen saturation: a useful clinical tool in trauma patients. J Trauma 30(12):1539-43.
- Maring JK, Klompmaker IJ, Zwaveling JH, Kranenburg K, Ten Vergert EM, et al. (1997) Poor initial graft function after orthotopic liver transplantation: can it be predicted anddoes it affect outcome? An analysis of 125 adult primary transplantations. Clin Transplant 11(5): 373-379.
- Mor E1, Klintmalm GB, Gonwa TA, Solomon H, Holman MJ, et al. (1992) The use of marginal donors for liver transplantation.A retrospective study of 365 liver donors. Transplantation 53(2): 383-386.
- Uemura T, Randall HB, Sanchez EQ, Ikegami T, Narasimhan G, et al. (2007) Liver Retransplantation for Primary Nonfunction: Analysis of a 20-Year Single-Center Experience. liver transplantation 13(2): 227233.
- Varotti G, Grazi GL, Vetrone G, Ercolani G, Cescon M, Del et al. (2005) Causes of early acute graft failure after liver transplantation: analysis of a 17-year single-centre experience. Clin Transplant 19(4): 492-500.
- Gruttadauria S, Marsh JW, Vizzini GB, di Francesco F, Luca A, et al. (2009) Early graft dysfunction following adult-to-adult living related liver transplantation: Predictive factors and outcomes. World J Gastroenterol 15(36): 4556-4560.
- Salvalaggio P, Afonso RC, Felga G, Einstein BH (2013) A proposal to grade the severity of early allograft dysfunction after liver transplantation. Einstein 11(1): 23-31.
- Lee DD, Singh A, Burns JM, Perry DK, Nguyen JH, et al. (2014) Early Allograft Dysfunction in Liver Transplantation With Donation After Cardiac Death Donors Results in Inferior Survival. Liver transplantation 20(12): 1447-14453.
- Siniscalchi A, Cuachetti A, Toccaceli L, Spiritoso R, TommasoniE, et al. (2009) Pretransplant model for end-stage liver disease score as a predictorof post-operatve complications after liver transplantation. Transplant Proc 41(4): 1240-1242.
- Filho AB, Nicolini EA, Martins MA, Silva Jr OC (2011) The use of perioperative serial blood lactate levels, the APACHE II and the postoperative MELD as predictors of early mortality after liver transplantation. Acta Cirûrgica Brasileira 26(6): 535.
- Wagener G, Raffel B, Young AT, Minhaz M, Emond J (2013) Predicting early allograft failure and mortality after liver transplantation: The role of the postoperative Model for End-Stage Liver Disease Score. Liver transplantation 19(5): 534-542.
- Ijtsma AJ, Hilst CSVD, Boer MTD, Jong KPD, Peeters PM, et al. (2009) The clinical relevance of the anhepatic phase during liver transplantation. Liver Transpl 15: 1050-1055.
- He XH, Ma Y , Wu LW, Ju WQ, Wu JL, et al. (2004) Safe time to warm ischemia and posttransplant survival of liver graft from non-heart- beating donors. World J Gastroenterol 10(21): 3157-3160.
- Selzner N, Rudiger H, Graf R, Clavien PA et al. (2003) Protective strategies against ischemic injury of the liver. Gastroenterology 125(3): 917-936.
- Totsuka E, Fung J, Lee M, Ishii T, Umehara M (2002) Influence of cold ischemia time and graft transport distance on postoperative outcome in human liver transplantation. Surgery Today 32(9): 792-799.






























