Hyperkalemia Management in the Oncology Patient: A Case of Sodium Polystyrene Sulfate Induced Bowel Perforation
Kathleen Sullivan*, Elena Mead, Meaghen Finan and Jinru Shia
Memorial Sloan Kettering Cancer Center, USA
Submission: February 05, 2017; Published:June 19, 2017
*Corresponding author: Kathleen Sullivan, Memorial Sloan Kettering Cancer Center, 1330 First Avenue, #1405, New York, NY 10021, USA, Tel: (908^ 370-8886; Email: kate.sullivan3511@gmail.com
How to cite this article: Kathleen S, Elena M, Meaghen F, Jinru S. Hyperkalemia Management in the Oncology Patient: A Case of Sodium Polystyrene Sulfate Induced Bowel Perforation. J Anest & Inten Care Med. 2017; 3(2) : 555610. DOI: 10.19080/JAICM.2017.03.555610
Abstract
Kayexalate (sodium or calcium polystyrene sulfate) is a cation-exchange resin commonly used to treat hyperkalemia in patients with renal dysfunction. It works by exchanging its bound sodium with potassium in the colon to promote potassium excretion in the stool. This occurs over hours to days, and is known to cause adverse digestive effects including anorexia, nausea, vomiting, and constipation. Bowel necrosis and perforation is an uncommonly recognized, though devastating complication of kayexalate administration. We present a case of a postoperative surgical oncology patient who developed a bowel perforation associated with oral kayexalate administration. We also review the literature to further delineate the relationship between the use of kayexalate and bowel perforation and necrosis, particularly in a surgical oncology patient where this complication has been less frequently documented.
Keywords: Kayexalate; Hyperkalemia; Bowel perforation; Bowel necrosis; Sodium polystyrene sulfate; Calcium polystyrene sulfate
Introduction
Hyperkalemia is a condition commonly encountered in medical and surgical patients and can induce life-threatening cardiac arrhythmias if left untreated. Kayexalate is a cation-exchange resin frequently used to treat this condition. Although rare, there is a known relationship between kayexalate administration and bowel necrosis and it is therefore important to consider this in a patient with abdominal pain who has been treated with oral kayexalate [1,2]. We present a case of a surgical oncology patient who developed spontaneous bowel perforation in the setting of kayexalate administration.
Case Report
The patient is a 60 year old male with past medical history significant for hypertension, hyperlipidemia, diabetes mellitus type II requiring insulin for glucose control and complicated by diabetic nephropathy (baseline creatinine 1.0), congestive heart failure with ejection fraction 30-40%, non-obstructive coronary lesions, moderate pulmonary hypertension and locally advanced left renal cell carcinoma (14cm mass causing renal vein thrombosis and retroperitoneal lymphadenopathy). The patient was not a candidate for neoadjuvant chemotherapy given his multiple medical comorbidities.
He underwent a left radical nephrectomy with regional lymphadenectomy with significant intra-op findings of left colon ischemia requiring left segmental colectomy with primary anastomosis. His postoperative course was significant for severe sepsis requiring initiation of vasopressor support and piperacillin- tazobactam, new onset atrial fibrillation requiring amiodarone, and acute kidney injury resulting in hyperkalemia. He was initially treated in the post-anesthesia care unit for hypotension with crystalloid, albumin and low dose phenylephrine which was weaned off over several hours. The patient was subsequently transferred to the ward. Hyperkalemia was treated with oral kayexalate 15 grams, of which the patient received six doses over the course of postoperative days #3-5 on a six hourly dosing regimen. On postoperative day #7, the patient developed acute abdominal pain and was found to have feculent output from the surgical drains. He was taken back to the operating room for exploratory laparotomy, left colectomy, transverse colostomy and mucous fistula. Postoperatively, he had continued vasopressor requirement with norepinephrine and vasopressin, he remained intubated and required intensive care unit admission. His antibiotic coverage was broadened to vancomycin, meropenem, and micafungin. Pathology of the left colon specimen revealed transmural necro-inflammation, exudative serositis and serosal fibrosis, as well as basophilic crystalloid particles consistent with kayexalate at the site of perforation, which was proximal to the prior viable-appearing anastomosis (Figure 1). Thus, a diagnosis of kayexalate-induced colon ischemia and necrosis was made.
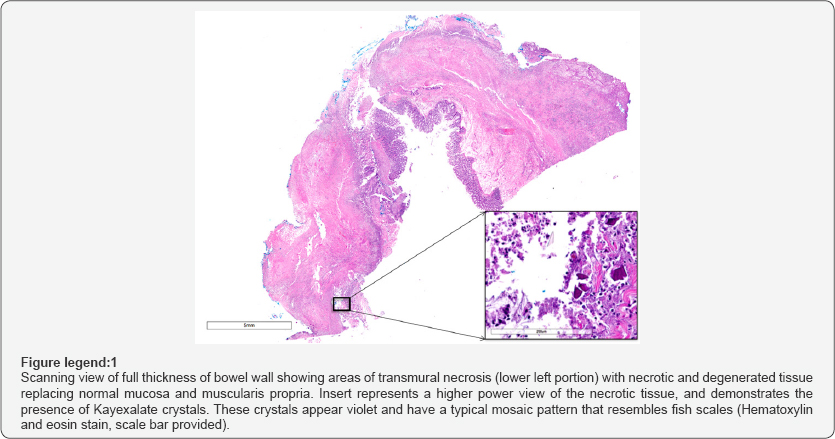
The patient had improvement in his clinical status, was weaned off vasopressor and ventilatory support, demonstrated improvement in his renal function and cardiac function with ejection fraction to 52% and he was discharged from the intensive care unit on post-operative day #6 to the ward, then discharged home with physical and occupational therapy services on post-operative day #19 . The remainder of his course has been unremarkable.
Discussion
Kayexalate is a cation-exchange resin which was first approved by the Food & Drug Administration in 1958 and has since been widely used to treat hyperkalemia [3]. Sorbitol is an osmotic laxative which historically has been added to kayexalate formulations to reduce the incidence of constipation, however there is a well-documented association with kayexalate- sorbitol and bowel necrosis [4,5]. In 2006, the Food and Drug Administration issued a black box warning on kayexalate-sorbitol products, and this formulation has subsequently been removed from the market over the last 10 years [4,5].
Despite the removal of sorbitol from most kayexalate formulations however, there have been numerous case reports of kayexalate administration and bowel necrosis. The incidence is thought to be 0.27% overall, and up to 1.8% during the postoperative period [2,3,6]. There have also been multiple case reports of upper gastrointestinal ulceration due to oral kayexalate administration, although none of these cases have required surgical intervention [7]. The symptoms of intestinal injury have been documented to occur between 3 hours and 11 days following the administration of kayexalate, and mortality from this complication is >30% in cases with gastrointestinal injury [3,5].
Kayexalate can be administered orally or rectally, and works by exchanging its bound sodium with potassium in the colon to promote potassium excretion in stool. Its effect is seen within hours to days, and thus it is not indicated for use as sole therapy in severe hyperkalemia. Although the mechanism of kayexalate induced bowel necrosis is unknown, one suspected mechanism involves the elevated renin levels seen in patients with renal failure who develop hyperkalemia. Renin activates angiotensin II which causes splanchnic vasoconstriction and can predispose the colon to non-occlusive ischemia, especially following dramatic electrolyte and fluid shifts [8]. Norepinephrine, the initial vasopressor indicated in septic shock, is also known to reduce the splanchnic blood flow and this can worsen intestinal vasoconstriction. However, the bowel necrosis and perforation described in cases of kayexalate administration is distinguished from ischemic necrosis by the pathological presence of kayexalate crystals in the bowel wall. Basophilic crystals with a mosaic pattern on Hematoxylin & Eosin stain is pathognomic for the presence of kayexalate.
We present this case to raise clinical suspicion of bowel necrosis and perforation in a patient with abdominal pain, particularly in a surgical oncology patient where this complication has been less frequently documented, after the administration of oral or rectal kayexalate [9,10]. Early diagnosis and prompt surgical intervention is critically important in this rare yet devastating complication of kayexalate administration due to the significant morbidity and mortality associated with this condition. In addition, we recommend the use of alternative treatment strategies for hyperkalemia, including insulin-glucose, diuretics, calcium, bicarbonate, inhaled beta-adrenergic agonists and emergent dialysis in severe, life-threatening hyperkalemia as these are safer and more efficacious therapies when implemented in a timely manner.
Conflict of Interest
We have no financial interest or any conflict of interest.
References
- Dardik A, Moesinger RC, Efron G, Barbul A, Harrison MG (2000) Acute abdomen with colonic necrosis induced by kayexylate-sorbitol. South Med J 93(5): 511-513.
- Gerstman BB, Kirkman R, Platt R (1992) Intestinal necrosis associated with postoperative orally administered sodium polystyrene sulfonate in sorbitol. Am J Kidney Dis 20(2): 159-161.
- Harel Z, Harel S, Shah PS, Wald R, Perl J, et al. (2013) Gastrointestinal adverse events with sodium polystyrene sulfonate (kayexalate) use: a systematic review. Am J Med 126(3): 264.
- Chatelain D, Brevet M, Manaouil D, Yzet T, Regimbeau JM, et al. (2007) Rectal stenosis caused by foreign body reaction to sodium polystyrene sulfonate crystals (kayexalate). Ann Diagn Path 11(3): 217-219.
- McGowan CE, Saha S, Chu G, Resnick MB, Moss SF (2009) Intestinal necrosis due to sodium polystyrene sulfonate (kayexalate) in sorbitol. South Med J 102(5): 493-497.
- Watson M, Baker TP, Nguyen A, Sebastianelli ME, Stewart HL, e.t al (2012) Association of prescription of oral sodium polystyrene sulfonate with sorbitol in an inpatient setting with colonic necrosis: a etrospective cohort study. Am J Kidney Dis 60(3): 409-416.
- Sterns RH, Rojas M, Bernstein P, Chennupati S (2010) Ion exchange resins for the treatment of hyperkalemia: are they safe and effective? J Am Soc Nephrol 21(5): 733-735.
- Chou YH, Wang YH, Hseih MS (2016) Colonic necrosis in a young patient receiving oral kayexalate in sorbitol: case report and literature review. Kaohsiung J Med Sci 27: 155-158.
- Abraham SC, Bhagavan BS, Lee LA, Rashid A, Wu TT (2001) Upper gastrointestinal tract injury in patients receiving kayexalate (sodium polystyrene sulfonate) in sorbitol: clinical, endoscopic, and histopathological findings. Am J Surg Pathol 25(5): 637-644.
- Rogers FB, Li SC (2001) Acute colonic necrosis associated with sodium polystyrene sulfonate (kayexalate) enemas in a critically ill patient: a case report and review of the literature. J Trauma 51(2): 395-397.






























