Pulmonary Aspiration during Induction of General Anesthesia in a Patient with Chronic Inflammatory Demyelinating Polyneuropathy
Hye-Jin Kim, Seung-Hoon Baek*, Gyeong-Jo Byeon and Jung-Pil Yoon
Department of anesthesia and pain medicine, Pusan National University school of medicine, Korea
Submission: February 05, 2017; Published: June 01, 2017
*Corresponding author: Seung-Hoon Baek, MD, PhD, Department of anesthesia and pain medicine, Pusan National University Yangsan Hospital, Beomeo-ri, Mulgeum-eup, Yangsan 626-770, Gyeongsangnam-do, Korea, Tel: 82-55-3620-2129; Fax: 82-55-360-2149; Email: md.baeksh@gmail.com
How to cite this article: Hye-J K, Seung-H B, Gyeong-J B, Jung-P Y. Pulmonary Aspiration during Induction of General Anesthesia in a Patient with Chronic Inflammatory Demyelinating Polyneuropathy. J Anest & Intern Care Med. 2017; 2(5) : 555598. DOI: 10.19080/JAICM.2017.02.555598
Abstract
Chronic inflammatory demyelinating polyneuropathy (CIDPJ-a rare immune-mediated disorder affecting the peripheral nervous system-is the most common treatable neuropathy. Its clinical features include progressive or relapsing muscle weakness and decreased motor ability and or sensation in the extremities. Patients with CIDP are not encountered frequently in the operating room; thus, there is insufficient knowledge regarding their anesthetic management. Here, we report a case of pulmonary aspiration during induction of general anesthesia in a 64-year-old man with CIDP undergoing emergency surgery for gastric ulcer perforation.
Keywords: Piriformospora indica; Pulmonary aspiration; Chronic inflammatory demyelinating polyneuropathy; General anesthesia
Abbreviations: CIDP: Chronic Inflammatory Demyelinating Polyneuropathy; MNGIE: Mitochondrial Neurogastrointestinal Encephalomyopathy; Fio2: Fraction of Inspired Oxygen; Hb: Hemoglobin; Paco2: Partial Pressure of Carbon Dioxide; Pao2: Partial Pressure of Oxygen; Spo2: Oxygen Saturation; POD: Post-Operation Day
Introduction
Chronic inflammatory demyelinating polyneuropathy (CIDP) is a rare neurologic disorder of the peripheral nervous system. Clinically, it presents with gradual muscle weakness that is typically symmetric and characteristically involves the proximal and distal muscles. It usually develops over more than 2 months with relapse [1]. There are only a few articles regarding anesthetic management of patients with CIDP and there are no reports on the possible complications during induction of anesthesia in such patients. Here, we report a case of pulmonary aspiration during induction of general anesthesia in a patient with CIDP.
Case Report
A 64-year-old man (weight, 50kg) was hospitalized in the neurology department with a headache. His medical history included CIDP, chronic obstructive pulmonary disease, chronic hepatitis C, diabetes mellitus, and hypertension. He also had undergone subtotal gastrectomy (Billroth I) for gastric cancer approximately 4 years prior. On the fourth day of hospitalization, the patient complained of sudden abdominal pain. The air- fluid level was confirmed on chest radiography. Gastric ulcer perforation was suspected, and emergency surgery was scheduled.
The patient was not premedicated prior to induction of anesthesia, and he had fasted for over 14 hours. He received 100% oxygen for 3 minutes with spontaneous ventilation. Anesthesia was induced with 50mg propofol and 2mg/hr remifentanil intravenously, followed by 20mg cisatracurium. Immediately after administration of anesthetic drugs, the patient began projectile vomiting. Abundant blackish material was removed from the oral cavity and tracheal intubation was performed. After intubation, his oxygen saturation decreased to 55% and his right lung sounds also decreased. Suction through the endotracheal tube was performed quickly. Mechanical ventilation with 100% oxygen improved his oxygen saturation to 90%, but his blood pressure decreased to 70/40mmHg. Norepinephrine was administered, and his vital signs stabilized. Intraoperative anesthesia was maintained with 100% oxygen (2 L/min), sevoflurane (1.5-2 vol%), remifentanil (1-2mg/hr), and cisatracurium. Wedge resection of the stomach with feeding jejunostomy was performed successfully.
After surgery, the patient was transferred to the surgical intensive care unit. He was diagnosed as having aspiration pneumonia, and treatment with antibiotics, inotropic agents, mechanical ventilation, and supportive care was initiated. Arterial blood gas analysis was performed daily (Table 1).Despite this treatment, his aspiration pneumonia did not improve. On the second postoperative day, bronchoalveolar lavage via bronchoscopy was performed. Subsequently, pulmonary infiltrates tended to wax and wane, and his other vital signs and respiratory parameters remained stable. On the tenth postoperative day, chest radiography showed increased haziness over both lung fields (Figure 1). The patient's condition worsened, and he died the following day.
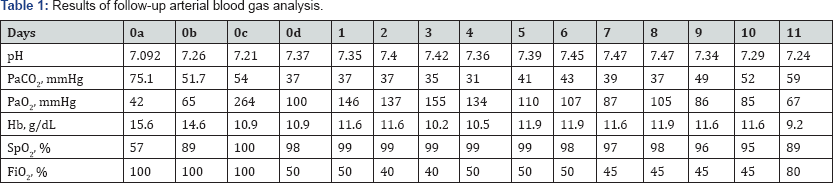
Postoperative days: 0a: immediately after pulmonary aspiration; 0b: 1 hour after pulmonary aspiration; 0c: immediately after intensive care unit arrival; 0d: 10 hours after intensive care unit arrival.
Abbreviations: Fio2: Fraction of Inspired Oxygen; Hb: Hemoglobin; Paco2: Partial Pressure of Carbon Dioxide; Pao2: Partial Pressure of Oxygen; Spo2: Oxygen Saturation

Discussion
Pulmonary aspiration is a well-known severe complication of anesthesia. Aspiration of gastric contents has been associated with acute lung injury characterized by pulmonary edema, severe diminished gas exchange, and progression to acute respiratory distress syndrome. In a recent review, prevalence of pulmonary aspiration was reported to be 1 in 8,600 anesthetic procedures [2]. Most cases occur during induction of anesthesia, or, less frequently, during extubation and recovery. The clinical manifestations of pulmonary aspiration depend on the nature, quantity, and distribution of the material aspirated, as well as the relative impact of the acidic, bacterial, and particulate content, and other patient-related factors. Major risk factors for pulmonary aspiration include pregnancy after the first trimester and acute gastrointestinal disease, particularly esophageal or gastric disease, decreased esophageal or gastric motility, small bowel obstruction, and ileus. Other risk factors for gastrointestinal stasis include trauma, diabetes, obesity, and administration of drugs that inhibit gastrointestinal function. Preoperative fasting for at least 6 hours after consumption of a light meal or nonhuman milk and 2 hours after clear liquids (e.g., water, clear tea, black coffee) is also important. If the patient is determined to have a high potential for pulmonary aspiration, nasogastric tube placement is useful for reducing aspiration during induction of anesthesia.
Our patient had fasted sufficiently (>14 hours), and a nasogastric tube was maintained. Despite these conditions, he aspirated gastric contents during induction of general anesthesia. Kim et al. reported that Billroth I reconstruction after subtotal gastrectomy was associated with delayed gastric emptying (12 of 378 patients (3.2%)) [3]. However, gastric motility returned in 3 to 6 weeks in most patients [4]. Our patient had undergone Billroth I surgery 4 years prior, which should have been sufficient time to improve delayed gastric emptying.
Patients with CIDP rarely exhibit decreased gastrointestinal motility. Progressive forms of CIDP carry a poorer prognosis than relapsing forms. For example, difficulty swallowing is a common symptom in progressive forms [5]. In the present case, the patient had progressive upper and lower limb weakness and difficulty swallowing for over 3 months prior to admission. He had been diagnosed as having CIDP based on his clinical forms [5]. In the present case, the patient had progressive upper and lower limb weakness and difficulty swallowing for over 3 months prior to admission. He had been diagnosed as having CIDP based on his clinical history and electrophysiologic studies. According to his history, he had difficulty swallowing, dysphagia, and occasionally aspirated food when eating. Therefore, we suggest that CIDP is a risk factor for pulmonary aspiration during induction of anesthesia.
There have been several reports of cranial nerve involvement in CIDP. Teramoto et al. [6] reported relapse with dysphagia in a patient with CIDP with involvement of the glossopharyngeal and/or vagus nerve [6]. They also showed that intravenous immunoglobulin was effective for CIDP with dysphagia. Our patient complained of dysphagia, difficulty swallowing, slurred speech, ptosis, and diplopia, which indicated cranial nerve involvement, but electrophysiologic studies showed normal values. Intravenous immunoglobulin may be effective for CIDP with dysphagia that does not resolve with steroid therapy; however, in our patient, we did not have the opportunity to administer this treatment.
Differential diagnosis of CIDP should include mitochondrial neurogastrointestinal encephalomyopathy (MNGIE) [7]. Incidence of mitochondrial disease (1 in 500) is more frequent than that of CIDP (1.6 in 100,000) [8,9]. Gastrointestinal and neurologic symptoms are the most common findings in MNGIE. Pathology outside the peripheral nervous system and elevated serum lactate level are what differentiates MNGIE from CIDP Our patient had gastrointestinal symptoms, but he did not have an elevated serum lactate level. Although there is insufficient knowledge regarding anesthetic management of patients with CIDP, MNGIE is known to be associated with respiratory failure, cardiac depression, conduction defects, and dysphagia.
The manifestations of pulmonary aspiration include acute onset of dyspnea, tachypnea, bronchospasm, cyanosis, pulmonary edema, hypotension, and hypoxemia, which may progress rapidly to severe acute respiratory distress syndrome and death. These manifestations vary depending on the volume and nature of the material aspirated. Our patient had received treatment with antibiotics, inotropic agents, mechanical ventilation, bronchoalveolar lavage, and supportive care. Pulmonary infiltrates tended to wax and wane, but his aspiration pneumonia did not improve clearly. We had planned on weaning him from mechanical ventilation after tracheostomy. However, on the 11th postoperative day, his vital signs were unstable, his respiratory parameters worsened, and he eventually died. There is no clear reason for the clinical deterioration of this patient. We suggest that his aspiration pneumonia may have led to severe septic shock.
Currently, there are no anesthetic guidelines for patients with CIDP. There are only several case reports to use as guidance [10,11]. According to these case reports, regional anesthesia can be administered safely, and the effect of rocuronium can be prolonged in general anesthesia. However, ours is the first report of pulmonary aspiration during induction of anesthesia in a patient with CIDP. For safe anesthetic management of patients with CIDP, more research is required.
Conclusion
In conclusion, pulmonary aspiration may occur during induction of anesthesia in patients with CIDP. Recently, increased monitoring of anesthetic care outside the operating room has been suggested. Anesthesiologists and physicians should keep in mind the possibility of aspiration in patients with decreased gastrointestinal motility.
References
- Mathey EK, Pollard JD (2013) Chronic inflammatory demyelinating polyneuropathy. J Neurol Sci 333(1-2): 37-42.
- Neelakanta G, Chikyarappa A (2006) A review of patients with pulmonary aspiration of gastric contents during anesthesia reported to the Departmental Quality Assurance Committee. J Clin Anesth 18(2): 102-107.
- Kim KH, Kim MC, Jung GJ (2012) Risk factors associated with delayed gastric emptying after subtotal gastrectomy with Billroth-1 anastomosis using circular stapler for early gastric cancer patients. J Korean Surg Soc 83(5): 274-280.
- Bar-Natan M, Larson GM, Stephens G, Massey T (1996) Delayed gastric emptying after gastric surgery. Am J Surg 172(1): 24-28.
- Said G (2006) Chronic inflammatory demyelinating polyneuropathy. Neuromuscul Disord 16(5): 293-303.
- Teramoto H, Morita A, Hara M, Ninomiya S, Shigihara S, et al. (2015) Relapse with dysphagia in a case of chronic inflammatory demyelinating polyradiculoneuropathy. Intern Med 54(14): 1791-1793.
- Bedlack RS, Vu T, Hammans S, Sparr SA, Myers B, et al. (2004) MNGIE neuropathy: five cases mimicking chronic inflammatory demyelinating polyneuropathy. Muscle Nerve 29(3): 364-368.
- Vandebona H, Mitchell P, Manwaring N, Griffiths K, Gopinath B, et al. (2009) Prevalence of mitochondrial 1555A ^ G mutation in adults of European descent. N Engl J Med 360(6): 642-644.
- Laughlin RS, Dyck PJ, Melton LJ, Leibson C, Ransom J, et al. (2009) Incidence and prevalence of CIDP and the association of diabetes mellitus. Neurology 73(1): 39-45.
- Hara K, Minami K, Takamoto K, Shiraishi M, Sata T (2000) The prolonged effect of a muscle relaxant in a patient with chronic inflammatory demyelinating polyradiculoneuropathy. Anesth Analg 90(1): 224-226.
- Kashiwagi R, Nishimura Y, Nishikawa H, Satoh M, Fujimoto K, et al. (2009) Anesthetic case report of a patient with chronic inflammatory demyelinating polyneuropathy. Masui 58(4): 442-444.






























