Our Scalp Block Results in Craniotomy Cases
Ebru Canakci1*, DoguŞ Unal2, Timur Yildirim3 and Ali Yilmaz3
1Department of Anesthesiology, Ordu University, Turkey
2Department of Anesthesiology, Ordu State Hospital, Turkey
3Department of Neurological Surgery, Ordu University, Turkey
Submission: April 10, 2017; Published: May 12, 2017
*Corresponding author: Ebru Canakci, Department of Anaesthesia and Reanimation, Ordu University ,School of Medicine, Bucak Town Nefs-i Bucak Street 52200/Ordu/Turkey, Tel: +90-05322651687; Fax: +90-0452-2250190; Email: canakciebru@gmail.com
How to cite this article: Ebru C, Dogus U, Timur Y, Ali Y.Our Scalp Block Results in Craniotomy Cases. J Anest & Inten Care Med. 2017; 2(4) : 555592. DOI: 10.19080/JAICM.2017.02.555592
Abstract
Aim: The aim of this study is to investigate the effect of scalp block performed with 0,5% of bupivacaine in craniotomy cases in preventing hemodynamic response due to the incision and its effect on postoperative analgesia and the need of analgesics.
Materials and method: The study was planned as a randomized, placebo controlled, double-blind study. 40 patients between the ages of 40-85 with ASA II-III classification were included in the study for elective craniotomy after the approval of the ethics committee and informed consents of the patients were received and they were separated into two groups (Group S: 20ml 0,9% normal saline, n=20), (Group B: 20ml0, 5% Bupivakain, n=20). Scalp block was performed 15 minutes before craniotomy. The mean arterial pressure (MAP) and heart rate (HR) of the patients wererecorded. Postoperative complications observed in the recovery room (bradycardia, hypotension, drug allergy, nausea, vomiting) were recorded. Pain was evaluated in postoperative conscious patients in the 2, 4, 8, 12, 16 and 24th hours with a 10cm visual analogue scale; and it was planned to administer 75mg of intramuscular meperidine if the VAS score was 5 and above in the postoperative period.
Result: During the craniotomy the MAP and HR values of the patients in Group S were significantly higher than Group B (p<0.05 respectively p=0.002, p=0.029). The VAS scores were also significantly higher in Group S compared to Group B in the postoperative 1, 2, 4, 6 and 12th hours (p<0.05 respectively p=0.022, p=0.031). Use of additional medication in Group S due to perioperative hypertension and tachycardia was significantly higher in comparison with Group B (p<0.001). Similarly, in terms of postoperative analgesic need, Group B had significantly less need for analgesics. Regarding the complications, however, no significant difference was found between the two groups.
Conclusion: In conclusion, scalp block ensures the stabilization of hemodynamic responses by reducing the sympathetic response in the intraoperative period in craniotomy cases and helps reduce the pain in the early postoperative period. We think that bupivacaine can be effectively used in scalp block procedures.
Keywords: Scalp block; Craniotomy; Bupivacaine
Introduction
The aim of neuroanesthesia is to prevent the increase of intracranial pressure without distorting the cerebral autoregulation and to ensure convenient surgical conditions and a safe anesthesia for the patient by maintaining a sufficient level of cerebral perfusion pressure (CPP). Anesthetic agents have obvious effects on cerebral metabolism, cerebral blood flow, cerebrospinal fluid (CSF) dynamics, intracranial volume and pressure [1]. As the intracranial pressure is directly related to the blood pressure, it is crucial to prevent the elevation of blood pressure due to any reason whatsoever in craniotomy patients. Radical elevations in systemic arterial pressure may temporarily distort cerebral autoregulation and, if not prevented, may cause cerebral edema by increasing the pressure in cerebral capillaries. Particularly, in interventions related to lesions involving intracranial areas, the detrimental effects of acute hypertension are more obvious as intracranial compliance is already decreased. Especially if the autoregulation capacity of cerebral vessels is already compromised, this increase will lead to an increased intracranial pressure [2]. Since increased intracranial pressure may cause a decrease in cerebral perfusion pressure or a shift effect in the brain, it should absolutely be prevented before durotomy [3].
Painful stimulants and sudden increases in blood pressure and heart rate cause herniation, cerebral aneurism and arteriovenous malformation rupture by increasing the intracranial pressure, and lead to ischemia in subarachnoid hemorrhage patients, who develop vasospasms, and an increase in the potential morbidity risk. Furthermore, hemodynamic instability will lead to adverse effects in those with atherosclerotic heart disease in the preoperative period [4,5]. Cranial surgical procedures involve continuous change in the intensity of painful stimulants, therefore they require a very close monitoring of the level of anesthesia [3,6].
The aim of scalp block is to block the nerves innervating the scalp at their exit points from the scalp before they form branches with the use of local anesthetic agents. Minor and major occipital nerves innervating the scalp, supraorbital and supratrochlear nerves, zygomaticotemporal nerve, auriculotemporal and major auricular nerve are blocked. As a result, the transmission in the fibers located in the nerve trunk in the area where the drug is delivered is blocked. Scalp block was first defined by Pinosky and bupivacaine was used as the local anesthetic agent. The most frequently used agent has also been bupivacaine in further studies, however there are also some studies performed with lidocaine, ropivacaine and levobupivacaine [5,7,8].
Mainly bupivacaine, ropivacaine and lidocaine have been used for scalp block in the studies [8-10]. Scalp block is a difficult technique requiring the use of local anesthetics in high volumes, which in turn increases the risk of local anesthetic toxicity in patients [9,11]. In awake craniotomies, an average of 150-175mg of levobupivacaine is used for the scalp block and maximum concentration measured in the plasma is 0.98-2.51µg/ml and the time needed to reach this level of concentration is 5-15 minutes and no central nervous system or cardiovascular system toxicity is observed at this level of concentration. It is reported that post-craniotomy pain is less than the pain experienced after operations such as lumbar laminectomy or fascial reconstruction ]12[. However, in contradiction to the general opinion, moderate or severe pain after craniotomy is reported to be quite common and it is observed that this pain is very intense particularly in the first 2 hours after craniotomy [13]. For the treatment of this pain, either local anesthetics are injected to the scar area or systemic nonsteroidal anti-inflammatory agents, drugs such as ketamine, opioids, or tramadol are given. In the meantime, there is still an ongoing search for an ideal analgesic agent and or approach in craniotomy cases complaining of severe pain. If the patient is conscious and have a perception of pain, postoperative analgesia should absolutely be used [14-16]. Bupivacaine is an amide type local anesthetic and was developed by Ekenstom et al. in 1963, it is available as hydrochloride salt in the market. It provides analgesia without motor block in low densities. Since it is highly fat-soluble, its systemic absorption is slow. It is metabolized in the liver except for a small portion excreted through the kidneys. It becomes effective within 5-10 minutes. This duration may reach up to 20 minutes in caudal and peridural injections. Motor and sensorial blockade may last up to 3 hours. It reaches maximum plasma concentration after 30-45 minutes. Its half-life is 9 hours in adults. It is one of the longest acting local anesthetics (5-16 hours). Scalp block is a method used to relieve pain in the early postoperative period and to help ensure hemodynamic stabilization in the intraoperative period [17]. In this study we aimed to investigate the effect of scalp block performed with bupivacaine in elective craniotomy cases in preventing hemodynamic response due to incision, and its effect on postoperative analgesia and the need for analgesics.
Materials and Method
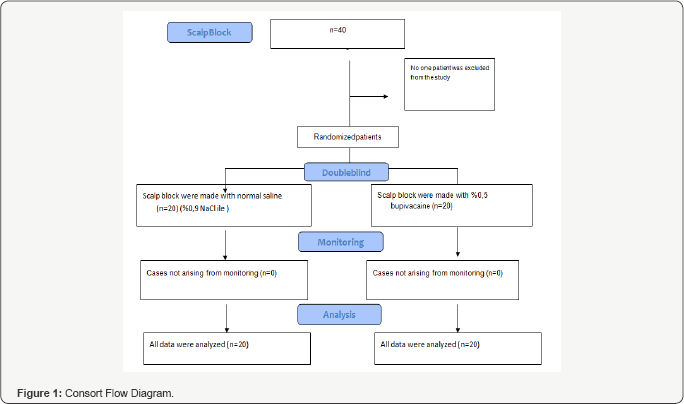
The ethics committee approval was received from the Clinical Studies Ethics Committee of Samsun Ondokuz Mayis University, Faculty of Medicine (Approval number B.30.2.ODM.0.20.08/1192). The study was planned between June/1/2015-December/31/2015. The study was started after receiving the consents of the patients planned to be included in the study. 40 patients, who were accepted at the Neurosurgery clinics of Ordu University Training and Research Hospital and Ordu State Hospital for elective craniotomy due to intracranial mass, were included in the study. Our study was a multicenter, randomized, placebo controlled, double-blind study. 40 patients to undergo elective craniotomy in the study were between the ages. of 40-85, in ASA II-III groups according to the risk classification of the American Society of Anesthesiologists (ASA) defining the physical condition of the patients. All the patients were informed about the study beforehand, and written consents were received from the volunteers who accepted to participate in the study. Those who had a systemic disease under ASA IV risk class, who had allergy against bupivacaine, advanced stage organ failure, alcohol and substance addiction and who were below the age of 40 and over the age of 85 were excluded from the study In our study, patients were not excluded from the study and all data were analyzed. Attached consort diagram drawn for our scientific work (Figure 1).
The patients were randomized with the sealed envelope method before the induction into 2 groups each comprising 20 patients; 20ml of 0.5% bupivacaine (Group B) and 20ml of 0.9% normal saline as the control group (Group S). Preoperative routine monitoring of the patients was done with Datex-Ohmeda Cardiocap™/5 (GE, Finland) device, followed by electrocardiogram (ECG), peripheral oxygen saturation (SpO2) and noninvasive blood pressure monitoring. Before the induction of anesthesia all the patients were premedicated with 0.05mg/kg of intravenous midazolam. After the induction of anesthesia with 2-3mg/kg of intravenous propofol, 2µg/kg of intravenous fentanyl and 0.6mg/kg of intravenous rocuronium, invasive arterial monitoring was performed by inserting a 20G intra-arterial cannula into the radial artery. Anesthesia was maintained with 6mg/kg/h of propofol infusion, 0.15mg/kg of intravenous rocuronium and 0.25µg/kg/min of continuous intravenous infusion of remifentanil. The patients were exposed to mechanical ventilation to reach an EtCO2 level of 30-35 mmHg with an air mixture of 50% O2. Once the baseline hemodynamic values were recorded before and after the induction, scalp block was performed. Skull-pin head holder was placed 5 minutes after the block was done. The medication to be used for the scalp block was prepared in a 20ml syringe by an anesthesiologist, who would not attend the surgery. 20ml of normal saline was put in the syringe for Group S. After being numbered according to the results of randomization, the responsible anesthesiologist made the injections with a 23G needle on the outer layer of the skull by inserting the needle into the skin with a 45° angle. Supraorbital and supratrochlear nerves were blocked by the injection of a 2ml solution on the bilateral supraorbital notch above the eyebrows. Bilateral auriculotemporal nerves were blocked by injecting a 2ml solution at 1.5cm anterior to the ear at tragus level. Bilateral postauricular nerves were blocked by injecting a 3ml solution at 1.5cm posterior to the ear at tragus level. Finally; the major, the minor and the third occipital nerves were blocked by injecting a 3ml solution at the intersection point of the midsection of the line between protuberentia occipitalis and mastoid process, and the upper nuchal line. Skull-pin head holder was placed by the neurosurgeon 5 minutes after the block.
Regarding the systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial blood pressure (MAP), heart rate (HR), peripheral oxygen saturation (SpO2) and end- tidal carbon dioxide (ETCO2) of the patients; the time when the patient was taken into the operating room before the scalp block was accepted as 0 min (= control value). After the scalp block was done, in the 1st, 5th and 10th minutes and then in the 20th, 30th, 40th, 50th, 60th, and 70th minutes, all the parameters were recorded until the end of the operation with 10-minute intervals.
Postoperative pain was evaluated in patients, who were conscious after the operation, in the 2nd, 4th, 8‘h, 12th, 16th and 24th hours with a 10cm visual analogue scale (0 is no pain, 10 is the worst possible pain). It was planned to give 75mg of intravenous diclofenac sodium to patients with a VAS score above 2 and 75mg of intramuscular meperidine to patients with a VAS score above 5. Postoperative analgesic needs and the amount of analgesics used were recorded.
Decrease of SpO2 below 94% for 45 seconds was accepted as hypoxia and elevation of ETCO above 45mmHg was assessed as hypercapnia. Hypertension was accepted as an increase of SBP by 20% above the control value and tachycardia was defined as a heart rate of at least 20% above the control value, and it was planned to administer 2µg/kg of intravenous fentanyl and to increase the propofol infusion dose to 9mg/kg/h. It was planned to administer 0.01mg/kg of bolus intravenous nitroglycerin if SBP and HR were still 20% above the control values.
Hypotension was accepted as an SBP value of 20% of the control value and less and 5-10mg of intravenous ephedrine was planned to be administered. Bradycardia was assessed as 20% below the control value or a value less than 40beats/ minute, and 0.5mg of intravenous atropine was planned. 10mg of intravenous metpamid was planned for the treatment of postoperative nausea and vomiting, and in the case of blurred vision or tinnitus the plan was to keep the patient in the recovery room for a longer period of time and observe.
SPSS for Windows 21.0 package program was used for the statistical analysis of this study. For measurable parameters (age, weight, amount of remifentanil, duration of anesthesia, duration of operation) the Kolmogorov-Smirnov test was used in order to identify whether the distribution was normal or abnormal. For those with normal distribution, Student t test was employed in independent groups to see whether there were differences between the groups. Data, such as gender and ASA, were analyzed with the Chi-square test. Heart rate and MAP data were assessed with repeated measures analysis of variance. In cases of differences, the comparison between the groups was done with the intergroup Posthoc-Scheffe test. Intragroup control values of HR and MAP, for which it was determined that the time factor was crucial according to the repeated measures analysis of variance, were compared by using the Post hoc Bonferroni test. Mann-Whitney U test was employed for the comparison of postoperative VAS scores between the two groups. For statistical analyses p<0.05 was accepted as significant.
Results
None of the 40 patients in the study was excluded from the study. The age, body weight, height, gender, ASA classification of the patients, duration of anesthesia and operation, the total amount of remifentanil used during the operation were found to be similar. All the patients were referred to the surgical intensive care units of Ordu University Training and Research Hospital and Ordu State Hospital postoperatively (Table 1). Demographic characteristics of the groups did not indicate any statistically significant difference.
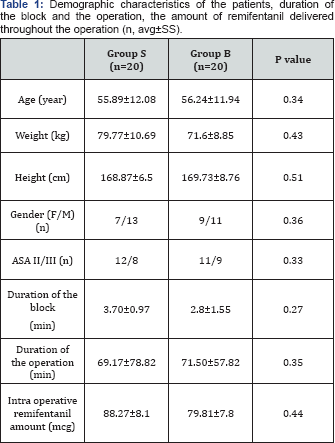
The average values of the heart rate (HR) according to the time of measurement are given in Table 2. While there was no difference between the groups in the control measurements in terms of average heart rates, the HR value after intubation was found to be significantly lower in Group B as compared to Group S (p<0,05). When the intragroup HR values measured at different times were compared according to the control value, it was observed that the average HR values were not statistically different from the control HR average values in both groups. The average values of the mean arterial pressure (MAP) according to the measurement times are given in Table 3. While there was no significant difference between the groups in terms of mean arterial pressure values in the control measurements, the MAP values acquired in the intraoperative 20th and 30th minutes after the scalp block were found to be significantly lower in Group B compared to the control group (normal saline group) (p<0,05). When the intragroup differences were investigated, on the other hand, intraoperative MAP values measured in the 10th, 20th, 30th, 40th, 50th, 60th, and 70th minutes after the scalp block in Group B were observed as significantly lower than the control values (p<0.05).
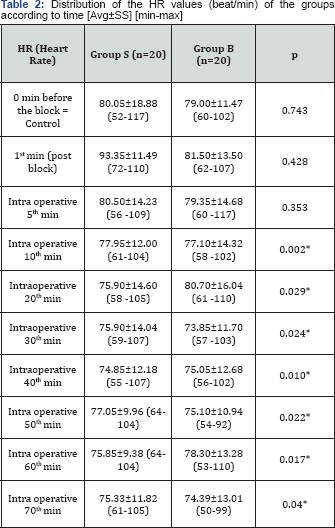
*p<0.05: in comparison with Group S.
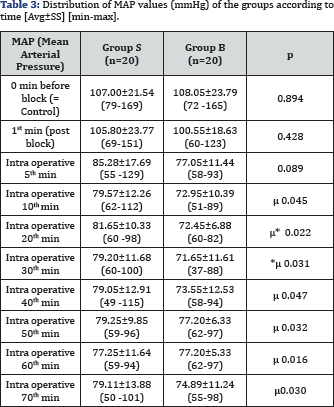
*p<0.05: in comparison with Group S, µ: p<0,05: in comparison with the control measurement values.
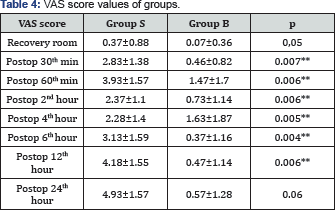
**p<0.01: in comparison with Group S
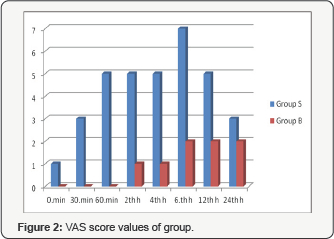
Postoperative pain assessment results of the conscious patients according to the visual analogue scale are given in Table 4 and Figure 2. The VAS scores acquired in the postoperative 30th min, 60th min, 2nd, 4th, 6th and 12th hours were found quite significantly lower in Group B as compared to Group S. The values in the recovery room and postoperative 24th hour were close to the statistical significance level in Group B (p=0.05 and p=0.06 respectively). No serious complications such as nausea, vomiting, bradycardia, and hypotension was observed in any of the patients in the postoperative period. In Group B, except for the patient, who needed 75mg of diclofenac sodium, no patients required meperidine. In Group S, on the other hand, 12 patients (60%) required meperidine in addition to diclofenac sodium particularly in the postoperative 12th and 24th hours.
Discussion
There is a common belief that those undergo neurosurgery suffer from minimum postoperative pain and need analgesics. While it is obvious that this group of patients experience relatively less pain when compared to those underwent orthopedic surgery or thoracic surgery, more than 60% of these patients feel moderate to severe postoperative pain. In a study conducted by Benedittis et al.90% of the patients suffered from post-craniotomy pain in the first 12 hours, which sometimes extended to 48 hours [18]. Our results overlap with those reported in the study of Benedittis et al. Likewise, in our study, there were many patients with a VAS score of 5 and above in the control group (normal saline group, Group S) particularly in the 12th and 24th hours. In a retrospective study conducted by Quient et al. postoperative pain in elective craniotomy patients was assessed in the first 24 hours. In the first 2 postoperative hours, 18% of the patients complained about severely distressing pain; 37% of the patients had severe, 29% had medium and 4% had mild pain. Only 12% of the patients did not describe a post-craniotomy pain in the first 24 hours [19]. Persistent post-craniotomy headache has also been identified and its incidence increases with postoperative unsuccessful analgesia [20,21]. In a study by Kaur et al. [21] 22 out of 126 supratentorial surgery patients developed persistent headache. 7 of these cases (5.6%) had headache for a period of longer than 2 months but shorter than 1 year; 15 cases (11.9%) had headaches for more than a year in the postoperative period.
In neurosurgery cases, laryngoscopy, skull-pin applications, interventions to the periosteum and dura cause painful stimulants. Even in cases with sufficient anesthetic depth, skullpin application and skin incision lead to acute hypertensive response [2,22]. Following the skull-pin placement, efferent pain sensation generated from the periosteum results in severe acute hypertensive response due to sympathetic system activation and eventually, intracranial pressure increases . This technique, which was defined for the first time by Pinosky et al. [10] and had not been implemented in practice before, was compared in a prospective, randomized, double-blind study in terms of the effect of scalp block performed by using 0.5% bupivacaine and normal saline on hemodynamic response to skull-pin placement and on the anesthesia need; and they were able to show that scalp block was successful in controlling the hemodynamic response to skull-pin placement. In our study, we followed the exact description of Pinosky et al. [10] while performing the scalp block. Lee et al. investigated the effect of scalp block performed with 0.25% bupivacaine under general anesthesia on hemodynamics and plasma catecholamine metabolites. 16 elective craniotomy patients were included in this prospective, randomized, double-blind study. One group underwent scalp block procedure with normal saline under general anesthesia induced with isoflurane and 50% N20-02, while the other group underwent the same procedure with a total of 20ml of 0.25% bupivacaine. Looking at the heart rate and mean arterial pressure measurements, it was revealed that scalp block led to more stable hemodynamics and decreased the need of intravenous or volatile anesthetics [23]. We used 0.5% bupivacaine and obtained more stable perioperative hemodynamics just as Lee et al. did in their study.
Gazoni et al. [8] compared perioperative results of the scalp block performed with ropivacaine in patients, who had supratentorial brain tumor, with remifentanil. In the prospective, randomized, double-blind study, while one group received 0.5% ropivacaine during the scalp block procedure, theother group received remifentanil infusion. Although, it was reported that scalp block did not bring along significant advantages in terms of postoperative pain and narcotic analgesics need when compared to remifentanil infusion, it was observed that hemodynamic parameters (MAP, HR) were more stable with the scalp block procedure.
Geze et al. compared the effects of scalp block and local infiltration on hemodynamics and stress response in craniotomy cases with skull-pin placement. In this prospective, randomized, placebo-controlled study, one group had scalp block with 0.5% bupivacaine and another group had local anesthetic infiltration with 0.5% bupivacaine; in the control group, on the hand, in order to prevent excessive hemodynamic responses, after an IV bolus of 0.5µg/kg remifentanil or a loading dose of 500µg/kg/ min esmolol, an IV infusion of esmolol 50µg/kg/min for 4 min was administered. In the study, it is reported that in the scalp block group, increase in blood pressure and heart rate due to skull-pin placement was prevented; there was no need for an additional anesthetic and antihypertensive agent, and blood pressure and heart rate were more stable when compared to the local infiltration and the controlgroups. When the groups are compared in terms of their metabolic and endocrine responses to surgery, stress response was significantly lowered after skull-pin placement in the scalp block group in comparison with the control group. In our study, we also used 0.5% bupivacaine and obtained well-matched results with that of Geze et al. We also observed perioperative hypertension and tachycardia in patients included in the normal saline group. 86% of the patients have pain with somatic features indicating that the source of pain is pericranial muscles and soft tissue. It is also known that local anesthetics administered before the skin incision on scalp have preemptive analgesic effect [24]. Therefore, scalp block is a technique that can be preferred to be used as a stand-alone analgesic method or to decreasethe dose of analgesics [25]. Taking all these remarks into account, we interviewed our conscious patients in the postoperative 30thmin, 1st, 2nd, 4th, 6th, and 24th hours on VAS and the use of additional analgesics. Our VAS scores were significantly lower in the bupivacaine group compared to the normal saline group.
Ayoub et al. [26] investigated the efficacy of scalp block in a group of 50 patients following remifentanil-based anesthesia. In this double-blind study, anesthesia was induced with 1-3mg/ kg of propofol and 1.0µg/kg of IV bolus remifentanil; followed by 0.1µg/kg/min of intravenous remifentanil infusion. The patients were randomized into two groups; one group having scalp block with bupivacaine or lidocaine and the other group having 0.1mg/kg intravenous morphine during dural closure at the end of the surgery. As an additional analgesic agent, codeine was administered subcutaneously in both groups. Both groups had similar pain scores. There was no significant difference between the two groups in terms of the total dose of codeine administered and the first codeine dose. While there was no difference between the groups in terms of confusion, nausea and vomiting was higher in the morphine group. The authors indicated that scalp block offered the same analgesic quality with a postoperative hemodynamic profile similar to morphine. Scalp block is an adjuvant method that can be used in order to avoid nausea and vomiting experienced with opioids [26]. Similarly, Bala et al. performed scalp block in 40 supratentorial craniotomy patients with bupivacaine or placebo following skin closure and they used intramuscular diclofenac or intravenous tramadol as analgesic. Patients without a scalp block had moderate to severe pain and had more frequent needs of additional analgesics. In this study too, it was revealed that the pain scores recorded after 6 hours were equal [27]. In our study, on the other hand, we obtained pain-free postoperative periods of over 12 hours, even extending to 24 hours. Scalp is a highly vascularized area, there are some studies analyzing the rate of transmission of local anesthetics applied to this area to the systemic circulation [7,28]. In our study, bupivacaine was administered very slowly in order to avoid drug toxicity, as it was required in high volumes in scalp block procedures and prior to the administration of bupivacaine, needle aspiration was performed in order to avoid accidental intra-arterial injection. After making sure that there was no blood, local anesthetic agent was injected. Although the patients were not monitored for QT intervals, there was no arrhythmia or asystole observed in routine ECG monitoring. In the postoperative period, no findings such as blurry vision, tinnitus or convulsion indicating systemic toxicity were reported.
Conclusion
In conclusion, in craniotomy cases, scalp block provides stabilization of hemodynamic responses by decreasing sympathetic response intraoperatively and helps reduce the early postoperative pain. Therefore, we believe that scalp block should play a more important role in anesthesiology practices and should be performed in all craniotomy patients.
References
- Kayhan Z, Klinik Anestezi (2004) Baski Istanbul: Logos Yayincilik. 760783.
- Shapiro HM (1972) Acute intraoperative intracranial hypertension in neurosurgicalpatients: mechanical and pharmacologic factors. Anesthesiology 37(4): 399-405.
- Warner DS, Hindman BJ, Todd MM, Sawin PD, Kirncher J, et al. (1996) Intracranial pressure and hemodynamic effects of remifentanil versus alfentanil in patients undergoing supratentorial craniotomy. Anesth Analg 83(2): 348-53.
- Murthy HS, Rao GS (2001) Cardiovascular responses to scalp infiltration with different concentrations of epinephrine with or without lidocaine during craniotomy. Anesth Analg 92(6): 1516-1519.
- Levin R, Hesselvik JF, Kourtopoulos H, Vavruch L (1989) Local anesthesia prevents hypertension following application of the may field skull-pinhead holder. Acta Anaesthesiol Scand 33: 277-279.
- Gesztesi Z, Mootz BL, White PF (1999) The use of remifentanil infusion for hemodynamic control during intracranial surgery. Anesth Analg 89(5): 1282-1287.
- Costello TG, Cormack JR, Mather LE (2005) Plasma levobupivacaine concentrations following scalp block in patients undergoing awake craniotomy. Br J Anaesth 94(6): 848-851.
- Gazoni F, Pouratian N, Nemergut E (2008) Effect of ropivacaine skull block on perioperative outcomes in patientswith supratentorial brain tumors and comparison with remifentanil: a pilot study. J Neurosurg 109(1): 44-49.
- Mathiu D, Beaudry M, Martin R, McLelland H, Robert B, et al. (2003) Effect of local anesthetic agent bupivicaine prior to application of the skull-pin holder for craniotomies. J Neurosurg 98(6): 1194-1197.
- Pinosky ML, Fishman RL, Reeves ST, Harvey SC, Patel S, et al. (1996) The effect of bupivacaine skull block on the hemodynamic response to craniotomy. AnesthAnalg 83(6): 255-261.
- Costello TG, Cormack JR, Mather LE, LaFerlita B, Murphy MA, et al. (2005) Plasma levobupivacaine concentrations following scalp block in patients undergoing awake craniotomy. Br J Anaesth 94(6): 848851.
- Dunbar P, Visco E, Lam AM (1999) Craniotomy procedures are associated with less analgesic requirements than other surgical procedures. Anesth Analg 88(2): 335-340.
- Quiney N, Cooper R, Stoneham M, Walters F (1996) Painaftercraniotomy. A time for reappraisal? Br J Neurosurg 10(3): 295-299.
- Graham AC, Reid MM, Andrews PJD (1999) Perception of pain experienced and adequacy of analgesia following elective craniotomy. Anaesthesia 54(8): 814-815.
- Verchere E, Grenier B, Mesli A, Siao D, Sesay M, et al. (2002) Postoperative pain management after supratentorial craniotomy. Neurosurg Anesthesiol 14(2): 96-101.
- Kayaalp O (2002) Rasyonel Tedavi Yonunden Tibbi Farmakoloji. 10. Baski. Ankara: Hacettepe-Taj Kitapfilik Ltd Sti, Turkey, 792-804.
- Nemergut EC, Durieux ME, Missaghi NB, Himmelseher S (2007) Pain management after craniotomy. Best Pract Res Clin Anesthesiol 21(4): 557-573.
- DeBenedittis G, Lorezetti A, Migliore M, Spagnoli D, Tiberio F, et al. (1996) Postoperatif pain in neurosurgery a plot study in brain surgery. Neurosurgery 38(3): 466-468.
- Quiney N, Cooper R, Stoneham M,Walters F (1996) Pain after craniyotomy. A time for reappraisal? BR J Neurosurg 10(3): 295-299.
- Gee JR, Ishaq Y, Vijayan N (2003) Postcraniyotomy Headache 43: 276278.
- Kaur A, Selwa L, Fromes G, Roos DA (2000) Persistent headache after supratentorial craniyotomy. Neurosurgery 47(3): 633-636.
- Bedford RF, Persing JA, Pobereskin L, Butler A (1980) Lidocaine or thiopentone for rapid control of intracranial hypertension. Anesth Analg 59(6): 435-437.
- Geze S, Yilmaz AA, Tuzuner F (2009) The effect of scalp block and local infiltration on the haemodynamic and stress response to skull- pin placement for craniotomy. Eur J Anaesthesiol 26(4): 298-303.
- Kissin I (2000) Preemptive analgesia. Anesthesiology 93: 1138-1143.
- Nguyen A, Girard F, Boudreault D, Fugere F, Ruel M, et al. (2001) Scalp nerve blocks decrease the severity of pain after craniotomy. Anesth Analg 93(5): 1272-1276.
- Ayoub C, Girard F, Boudreault D, Chouinard P, Ruel M, et al. (2006) Comparison between scalp nerve block and morphine for transitional analgesia after remifentanil-based anesthesia in neurosurgery. Anesth Analg 103(5): 1237-1240.
- Bala I, Gupta B, Bhardwaj N, Ghai B, Khosla VK (2006) Effect of scalp block on postoperative pain relief in craniotomy patients. Anaesth Intensive Care 34(2): 224-227.
- Costello TG, Cormack JR, Hoy C, Wyss A, Braniff V, et al. (2004) Plasma ropivacaine levels following scalp block for awake craniotomy. J Neurosurg Anesthesiol 16(2): 147-150.






























