Research Article
Current State of Pharmacodynamic Target Attainment in Critically Ill Patients with Severe Sepsis in Canadian Icus: Prospective Cohort Study
Clarence Chant1, Jennifer Poh2, Lisa Burry3, Anne Julie Frenette4, Lynne Kelly5, Kristi Parmiter6 and SalmaanKanji7
1St. Michael’s Hospital, Canada
2Hospital for Sick Children, Canada
3Mount Sinai Hospital and University of Toronto, Canada
4Universite de Montreal, Hospital , Canada
5London Health Sciences Center, London, Canada
6Eastern Health, St. John’s, Canada
7The Ottawa Hospital and Ottawa Research Institute, Canada
Submission: January 29, 2016; Published: March 25 2016
*Corresponding author: Clarence Chant, PharmD, BCPS, FCCP, FCSHP, St. Michael’s Hospital, Room B-00730 Bond Street, Toronto, ON, M5B1W8, Tel: 416-864-5103; 416-864-6058;Email; Email: chantc@smh.ca
How to cite this article: Clarence C, Jennifer P, Lisa B, Anne-Julie F, Lynne K, et al. Current State of Pharmacodynamic Target Attainment in Critically Ill 005 Patients with Severe Sepsis in Canadian Icus: Prospective Cohort Study. J Anest & Inten Care Med. 2016; 1(1) : 555554. DOI: 10.19080/JAICM.2015.01.555554
Abstract
Objectives: We sought to determine the rate of pharmacodynamic target attainment of antibiotic therapy for critically ill patients with severe sepsis in Canadian ICUs.
Methods: In this multicenter observational study, adult Canadian ICU patients with severe sepsis were prospectively followed. The minimum inhibitory concentration (MIC) of the organism was obtained from hospital’s microbiology laboratory, and pharmacodynamic targets (PD) of the prescribed empiric antibiotics were calculated using population pharmacokinetic parameters to estimate the rate of PD target attainment. The primary outcome was the proportion of patients who attained PD target. We performed sensitivity analysis varying the elimination rate constant (ke), volume of distribution (Vd), and MIC parameters.
Results: Sixty-nine pairs of antibiotic and MIC were evaluated in 43 patients. In the base case, PDtarget attainment was achieved in 92.7% (64/69) of the antibiotic-MIC pairings, or in 88% (38/43) of patients. In sensitivity analysis, 83.7% (36/43) of the patients achieved the desired PDtarget at all variations of the PK or MIC parameters. Clinical failure occurred in three patients despite target attainment.
Conclusions: Current antibiotic doses for treatment of critically ill patients with severe sepsis achieved 88% PD target attainment. These results need to be validated using measured patient PK parameters and a larger sample size of MICs of causative organisms.
Keywords: Pharmacokinetic; Pharmacodynamic; Critically ill; ICU; Severe sepsis
Abbreviations: AUC: Area Under The Curve; ICU: Intensive Care Unit; Ke: Elimination Rate Constant; MIC: Minimum Inhibitory Concentration; PK: Pharmacokinetic; PD: Pharmacodynamic; Vd: Volume of Distribution
Introduction
Optimal use of antibiotics in patients with severe sepsis is paramount, particularly in this era of increasing antibiotic resistance, lack of new antibiotic agents being developed, and the high incidence and mortality associated with severe sepsis [1-3]. In patients with severe sepsis, optimal antibiotic use involved administering the right antibiotic in the optimal dose/regimen in a timely manner [4-5]. In other words, therapeutic failures can still occur even if standard doses of an effective antibiotic are administered promptly. Possible reasons for this include: alterations in pharmacokinetic (PK)/pharmacodynamic (PD) parameters in patients with severe sepsis, compromised organ function as a sequalae of sepsis (e.g. renal and or hepatic dysfunction), and inadequate local antibiotic concentration (e.g. abscess) despite adequate serum concentrations. Therefore, alternative dosing strategies that incorporate both PK parameters of the patient and PD properties of the antibiotic have been advocated, such as the use of continuous or extended interval infusions, particularly for critically ill patients with severe sepsis [6]. This is consistent with the recommendations set out by the Infectious Diseases Society of America, in which dosage optimization is suggested as a possible strategy for improving the usage of current antibiotics and minimizing the development of resistant organisms [7].
Patients with severe sepsis represent a highly vulnerable population with significant risk of morbidity and mortality who might benefit from dosage optimization efforts. However, the extent to which current doses of antibiotic regimens for patients with severe sepsis achieve optimal PD targets is unknown. Literature surrounding this issue consists primarily of non-clinical studies demonstrating theoretical benefits, nonrandomized or controlled clinical studies with positive results. Furthermore, the use of extended/continuous infusion in an attempt to optimize the probability of PD target attainment also yielded conflicting results as reported in a recent systematic review of randomized controlled trials [6]. The objective of this study is to determine the rate of attainment of pre-defined PD targets for critically ill patients with severe sepsis.
Materials and Methods
Study design, setting and patient population
This multicenter, prospective, observational trial enrolled patients admitted to an ICU between September 2012 and June of 2013. Patients ≥ 18 years old with severe sepsis were eligible for enrollment into the study. Severe sepsis is defined using the standard consensus criteria [8]. Patients were excluded if they were: 1) Immunocompromised, defined as: those with an absolute neutropenia count of less than 500/mm3, patients with active human immunodeficiency virus infection and not on highly active antiretroviral therapy, patients receiving greater than 20mg of prednisone equivalents per day for greater than one month, patients on concurrent chemotherapy, or on immunosuppressive therapy post organ transplantation, 2) on antibiotics for less than 24 hours, and 3) receiving only antifungal or antiviral medication. Immunocompromised patients were excluded as the management of their infections often require different approaches and thus limit generalizability.
Data collection
For each participating ICU, all patients were screened daily and enrolled patients were followed starting from the initiation of empiric antibiotics for the treatment of their severe sepsis for 14 days, or until discharge from the ICU or death/withdrawal of care, whichever occurred first. Data was collected prospectively only for the first episode of infection for each patient regardless of when it occurred during their ICU stay, or if the patient was readmitted to the ICU. A standardized case report form was used to ensure all relevant information was collected for the index episode of infection including microbiology results. The MIC of the causative organism was determined by the participating hospital’s microbiology laboratory using current local techniques. Causative organism was defined as the isolate identified from the presumed infectious source as documented in the medical records. No attempts were made to alter current practices, including antibiotic dosing which were determined by local care team and in accordance with product monographs of each drug. Dosage adjustment for patients with renal dysfunction was determined by local care team as well. Given the multicenter nature of the study, standard definitions were developed for each data point and were made available to all participating sites prior to the beginning of patient enrollment. Clinical cure, reported by each site investigator without independent adjudication, was defined as complete resolution of the signs/ symptoms associated with the infection and discontinuation of the prescribed antibiotics. For patients receiving combination antibiotics, either as initial empiric or definitive therapy, each antibiotic - MIC pairing was evaluated separately.
Pharmacokinetic data used for calculations
A comprehensive review of all published literature was performed for each antibiotic reported by the sites using MEDLINE (1946-2012) using the keywords “drug monitoring”, “pharmacokinetics”, “sepsis”, “critical care”, and “critical illness”. Articles were restricted to those written in English and included patients aged 18 years or older. All abstracts recovered were reviewed by one investigator (J.P.) using the following criteria: [1] describing the use of the antibiotic in question in patients admitted to the ICU or in patients with severe sepsis, and [2] providing pharmacokinetic data of the antibiotic in the population of interest. The hierarchy of choice of PK parameters from the literature, in descending order, is: ICU patients with severe sepsis, general ICU patients, hospitalized patients, and healthy volunteers. The references of included manuscripts were searched for other potential manuscripts that might qualify for inclusion. For qualifying abstracts, full text manuscripts were reviewed by the same investigator (J.P.) for the following data elements: (1) volume of distribution (Vd), and (2) the estimated elimination rate constant (ke). In situations where the Vd was reported in units of liters per kilogram (L/kg), an assumption of 70 kilograms was made. In some published literature, half life (t1/2) was reported instead of ke, and the ke was calculated using the following equation (ke = ln2/t1/2). The un weighted mean values of the Vd and ke for each antibiotic was calculated if there existed more than one study. If values are available for patients on renal replacement therapies (intermittent or continuous modalities) these were also abstracted. These PK parameters were used for calculations in the current study.
Pharmacodynamic target attainment calculations
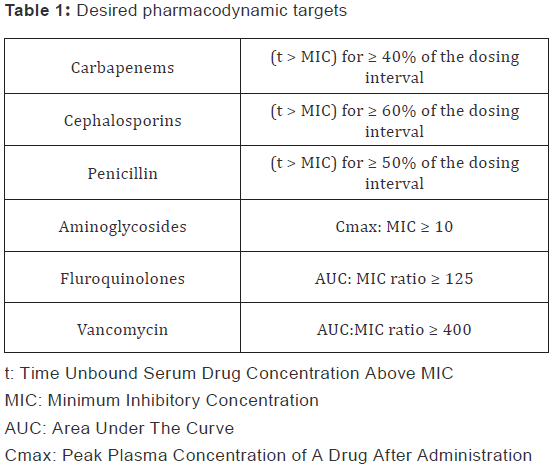
The primary outcome of this study is the proportion of infections treated with an antibiotic regimen that is predicted to achieve the desired PD targets derived from current literature (Table 1) [9,10]. Target attainment was calculated by using PK parameters described above, a one-compartment model, and patient weight. Pharmacodynamic calculations were performed for each individual patient and the antibiotic (empiric or targeted) - MIC pairs. Area-under-the-curve (AUC) was estimated from daily antibiotic dose and patient’s clearance of that antibiotic. For antibiotics that exhibit time dependent killing, time above MIC (t > MIC) was estimated using one compartment kinetic modeling. These calculations yielded the base case scenario used to determine the primary endpoint. Given the inherent variability in PK parameters (see table 1) and in automated methods of MIC determination commonly used at the participating institutions, we conducted sensitivity analysis of the base case scenarios using the following variables and range: 1) one fold increase/decrease in MIC values (common range of error for the automated methods), 2) minimum and maximum literature reported values in PK parameters of clearance and volume of distribution.
The study protocol and waiver of consent was approved by the institutional review boards at each of the participating sites.
Results
Participating sites demographics
Seven Canadian ICUs participated in this study. The ICUs were all in teaching institutions, predominantly closed ICU (6/7), and cared for a mixed population (medical, surgical, and some trauma and neurosurgical). Antimicrobial stewardship programs were present in 4/7 ICUs, and standard sepsis management protocols were present in only 2 ICUs. All but one institution used the VITEK system for automated MIC determination, with Micro scan being used in the remaining ICU. However, only 2/7 ICUs routinely receive the MIC values with the microbiology report. All the ICUs have dedicated clinical pharmacist (s) assigned.
Episodes of inpatient care
Of the patients included, 360 (57%) had at least one episode of inpatient care within the 3 years before the admission to ICU. The ICU patients with pre-existing diseases used significantly more hospital resources 3, 2, and 1 year before admission to the ICU (p= 0.008, <0.001, and <0.001), and up to 3 years after discharge regarding amount of care (number of visits) (p= 0.003), duration of stay (p=0.001), and costs (p=0.002), but there were no differences during the ICU period in that preexisting disease did not increase the cost of the stay in ICU. Most importantly, there was a significant increase in the number of episodes of inpatient care for the 3 years before admission to ICU (p<0.001).
Patient characteristic
Forty-three patients (30 males and 13 females) were enrolled in the study (Table 2). These patients were moderately ill with a mean APACHE II score of 22 ± 6.5. At baseline, the most common site of infection was blood (24 patients), and the most common organ system failure was cardiovascular. A variety of causative organisms were reported, with E. coli being the most common gram-negative organism and methicillin-sensitive S. aureus being the most common gram-positive organism. Seventy eight percent of patients had 3 or more SIRS criteria, and 28% were on renal replacement therapy. Dosages of antibiotics prescribed are outlined in Table 3.
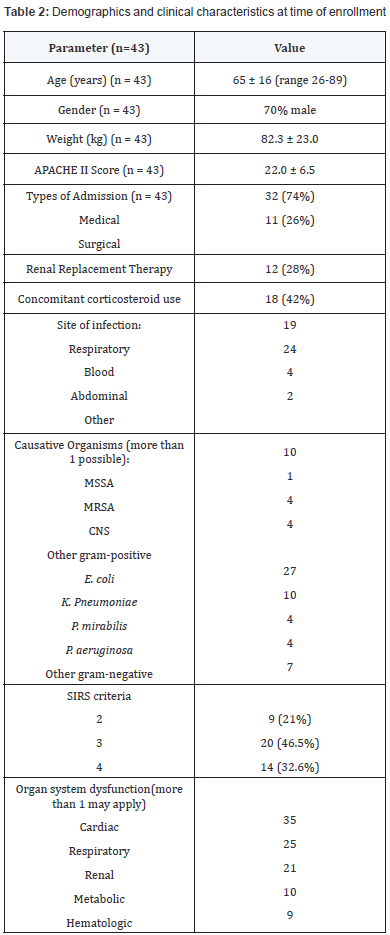
APACHE: Acute physiologic and chronic health adjustment evaluation; SIRS: Systemic Inflammatory response syndrome; MSSA: Methicillin- Sensitive S. Aureus; MRSA: Methicilin-Resistant S. Aureus; CNS: Coagulase-Negative Staphylococci
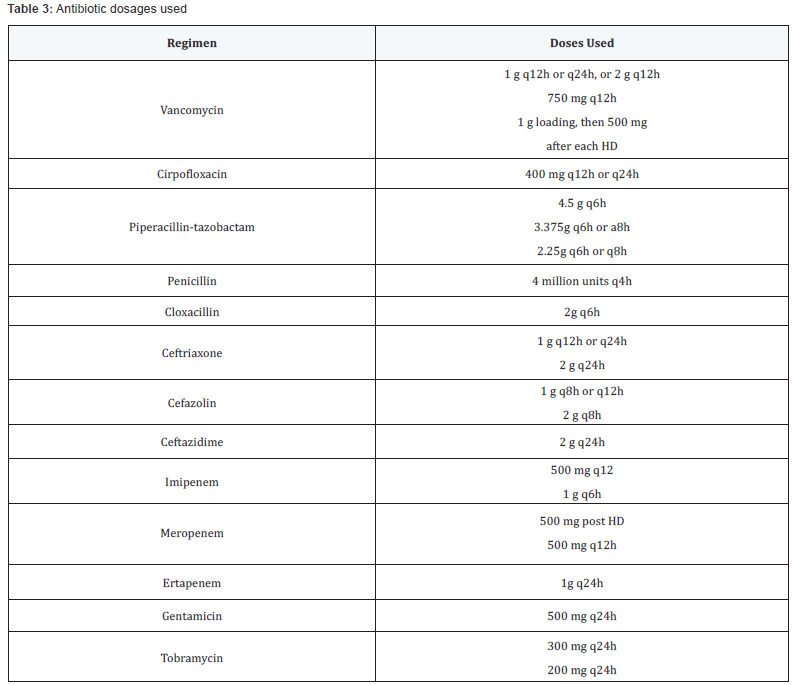
Q24h: Every 24 hours; HD: Hemodialysis
Outcome analysis
Analysis of 69 pairs of antibiotic and MIC were completed in all 43 patients. Pharmacodynamic target attainment was achieved in 91% (63/69) of the antibiotic-MIC pairs, or in 88% (38/43) of the patients in the base case scenario (Table 4). Sensitivity analysis showed that 84% (36/43) of the patients achieved the desired pharmacodynamic targets at all variations of the factors associated with therapeutic failure (one fold increase and decrease in MIC, minimum and maximum ke and Vd). Exploring the 5 patients who did not attain the desired PD targets in the base case scenario, 2 patients were prescribed antibiotics that the organism has intrinsic resistance towards, and 3 of the 5 patients were due to a prescribed conservative tobramycin dosage, but they were also receiving concomitant antibiotics that did attain PD targets. In the sensitivity analysis, 2 more patients failed to attain PD targets when the MIC values were doubled. Three patients were classified as clinical failures, and in all cases PD targets were achieved (Table 5). One patient with clinical failure had concomitant fungemia, one had inadequate source control.
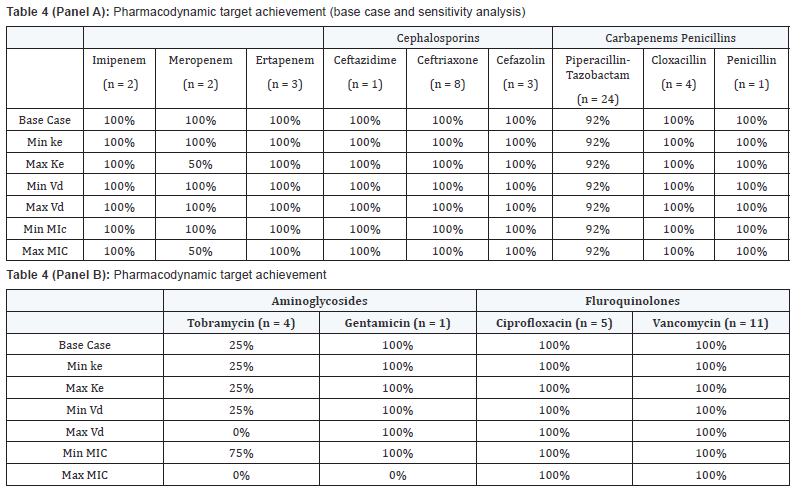
Base case refers to target attainment for average Ke and Vd and laboratory reported MIC. Ke and Vd were varied using min/max values found in literature and MIC were varied with one fold dilution above and below reported MIC
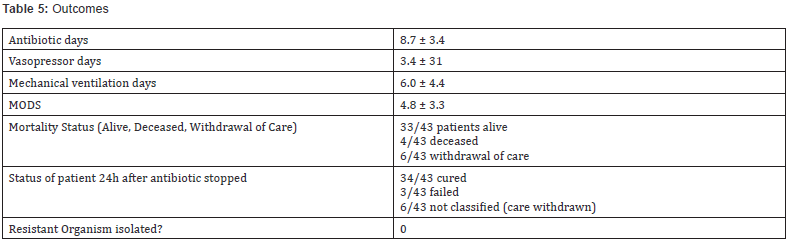
Values are mean ± standard deviation
MODS: Multiple organ dysfunction score
Discussion
In this prospective, observational, multicenter study, using clinical data from 43 critically ill patients with severe sepsis treated with antibiotics, we found that 88% of patients achieved PD targets. In the sensitivity analysis, 84% of patients achieved the desired PD targets at all variations of the Vd, ke and MIC.
To the authors’ knowledge, this is the first multi-center study documenting the proportion of ICU patients with severe sepsis achieving pre-defined PD targets with a variety of antibiotic therapies. The results garnered from this study differs significantly from a single center study that enrolled 19 ICU patients with gram negative sepsis that reported only 16% (3/19) of the patients achieved the desired PD targets [11]. The differences may be explained by the different PD targets used during that study timeframe, smaller sample size, and different microbiologic ecology of the study sites. The recently published DALI study involving 181 culture-positive ICU patients with any infections (out of the total 361 patients included in the study) reported that 16% of patients did not achieve the defined PD target, similar to our findings despite different methodology [12]. The DALI study enrolled a different patient population (severe sepsis vs any infection), evaluated different antibiotics (beta-lactam only), and partially used measured MIC (only 34.2% of the pathogens had an MIC value, and the remaining cases were analyzed using the literature reported MIC90). The DALI study also performed actual PK parameter determination, and used PD targets which were also different (total drug concentration above MIC for 40-60% of dosing interval vs free drug concentration above MIC for 50% of dosing interval). While a 12-16% target non-attainment rate is undesirable from a patient perspective, both the DALI and this study have limitations that preclude precise estimates of the true target non-attainment rates. In our study, target non-attainment did not predict clinical failure, while in the DALI study target non-attainment was found to be a significant risk factor for poor clinical outcomes. Therefore efforts to maximize probability of target attainment may be more appropriately reserved for at risk patient population or organisms with known borderline MIC values, or in patients with PK parameters that would predict failure. This differential importance is consistent with the conflicting literature that demonstrate any clinical benefits of using strategies such as extended/continuous infusions of antibiotics in order to optimize PD target attainment [6,13,14]. While the principles underlying PD based dosing is intuitive and supported by in vitro studies and small case series, it has not been consistently proven in larger randomized controlled trials. Closer examination of these principles and their operationalization in actual clinical practice provides explanation for this discordance. First, the desired PD targets published in the literature are usually derived from single studies with minimal validation by other investigators. Pharmacokinetics data for these antibiotics in critically ill patients is not routinely published, and thus extrapolation from studies in other patient population are often done, which carries potential for significant errors given the known variability in the critically ill population. In addition, MIC values are an important determinant of PD target attainment; given the inaccuracies associated with MIC values determined by automated techniques routinely employed in most hospitals, ascertaining actual PD attainment would only be possible with a significant increase in workload in the microbiology laboratory to perform more accurate MIC determinations. Finally, it is clear even from mathematical calculations that with low MIC values PD target attainment is easily achieved even at conventional doses of antibiotics. Therefore before widespread changes in both microbiology practice and antibiotic dosages are warranted, more rigorous proof-of-concept of PD-based dosing is required.
Strengths of our study include gathering data from multiple ICUs across Canada, providing a broader representation of ICU patients, causative organisms and infectious sources. Standardized data definitions also ensured data accuracy. The Canadian context is important given the different bacterial resistance patterns in different countries especially in comparison to the US. Sensitivity analysis was performed to enhance internal and external validity. Some important limitations of our study are inherent in its design: the naturalistic, observational design precludes proving causality. In addition, the majority of patients received combination antimicrobial therapy. The use of the automated VITEK system to estimate MICs instead of the E-test or traditional Kirby Bauer techniques introduces another layer of error [15]. Use of population pharmacokinetic parameters to estimate PD-attainment may not be representative of every patient enrolled in our study and could not account for any augmented renal clearance, in addition to lack of documented PK values in critically ill patients for some antibiotics. However this is a systemic issue with antibiotic PK in ICU patients in general and not specific to this study. Our study focused solely on conventional administration of antibiotics and did not take into account strategies for optimizing PD targets such as use of continuous infusions, or use of higher than recommended doses to achieve PD targets. Finally this study was designed to test the hypothesis of target attainment, not clinical outcomes, which would require a much larger sample size.
This study demonstrated a reasonable level of PD target attainment with current antibiotic doses in Canadian ICU settings. Studies designed to produce more precise estimates of this target attainment rate are needed to delineate which clinical scenario may warrant the efforts to optimize the probability of PD target attainment.
Acknowledgements
We would like to acknowledge Curtis Harder for assistance with data collection. We would also like to acknowledge all the participating sites: St. Michael’s Hospital, Toronto; Mount Sinai Hospital, Toronto; The Ottawa Hospital, General Campus, Ottawa; London Health Sciences Center, London; Hôpital du Sacré-Coeur de Montreal, Montreal; and Eastern Health Hospital, St. John’s Newfoundland.
Funding
Provided by Medbuy Research and Education funds.
Ethical Approval
All sites received approval from their respective REB.
Authors Contribution
- CC contributed to the design of the study, data collection, data analysis, and wrote the initial draft of the manuscripts and revised subsequent drafts.
- JP and KP contributed to data collection and analysis, and contributed to the draft of manuscripts.
- LB, LK, AJF, SK contributed to study design, data collection and also to the revisions of the manuscripts.
- All authors have approved the final draft of the manuscript.
References
- Hughes JM (2011) Preserving the lifesaving power of antimicrobial agents. JAMA 305(10):1027-1028.
- Martin CM, Priestap F, Fischer H, Fowler RA, Heyland DK, et al. (2009) A prospective, observational registry of patients with severe sepsis: The Canadian Sepsis Treatment And Response Registry. Crit Care Med 37: 81-88.
- Dellinger RP, Levy MM, Carlet JM, Bion J, Parker MM, et al. (2008) Surviving Sepsis Campaign: International guidelines for management of severe sepsis and septic shock: 2008. Intensive Care Med 36(1): 296-327.
- Pea F, Viale P (2009) Bench-to-bedside review: Appropriate antibiotic therapy in severe sepsis and septic shock-does the dose matter? Crit Care 13(3): 14.
- Chant C, Leung A, Friedrich JO (2013) Optimal dosing of antibiotics in critically ill patients by using continuous/extended infusions: a systematic review and meta-analysis. Crit Care 17(6): R279.
- Dellit TH, Owens RC, McGowan JE Jr, Gerding DN, Weinstein RA, et al. (2007) Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America Guidelines for Developing an Institutional Program to Enhance Antimicrobial Stewardship. Clin Infect Dis 44(2): 159-177.
- Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, et al. (2013) Surviving sepsis campaign: International guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 41(2): 580- 637.
- Roberts JA, Lipman J (2009) Pharmacokinetic issues for antibiotics in the critically ill patient. Crit Care Med 37(3): 840-851.
- Lodise TP, Drusano GL (2009) Pharmacokinetics and pharmacodynamics: Optimal antimicrobial therapy in the intensive care unit. Crit Care Clin 27(1): 1-18.
- Mohr JF, Wanger A, Rex JH (2004) Pharmacokinetic/ pharmacodynamic modeling can help guide targeted antimicrobial therapy for nosocomial gram-negative infections in critically ill patients. Diagn Microbiol Infect Dis 48(2): 125-130.
- Roberts JA, Paul SK, Akova M, Bassetti M, De Waele JJ, et al. (2014) DALI : Defining antibiotic levels in intensive care unit patients: Are current beta-lactam antibiotic doses sufficient for critically ill patients? Clin Infect Dis 58(8): 1072-1083.
- Roberts JA, Webb S, Paterson D, Ho KM, Lipman J (2009) A systematic review on clinical benefits of continuous administration of betalactam antibiotics. Crit Care Med 37: 2071-2078.
- Falagas ME, Tansarli GS, Ikawa K, Vardakas KZ (2013) Clinical outcomes with extended or continuous versus short-term intravenous infusion of carbapenems and piperacillin/tazobactam: a systematic review and meta-analysis. Clin Infect Dis 56(2): 272-282.
- Angus DC, Carlet J (2003) Surviving intensive care: a report from the 2002 Brussels Roundtable. Intensive Care Med 29(3): 368-377.
- Jorgensen JH, Crawford SA, Masterson M, Mansell MK, McElmeel ML, et al. (2006) Direct comparison of antimicrobial susceptibility testing by the BD Phoenix, bioMerieux VITEK2, and disk diffusion test methods as compared to results generated by the CLSI broth microdilution test. American Society of Microbiology 2006.






