Impact of a Non-Invasive Respiratory Support Strategy and Switches in Therapy During Acute Respiratory Failure Due to SARS-COV-2
J Marin-Corral1,2*, FJ Parrilla-Gomez1*, A Castellvi-Font1, P Perez-Teran1, L Picazo1, S Pascual-Guardia3,4, A Rodriguez5, JC Ballesteros6, S Sancho7, L Socias8, E Diaz9,10, A Albaya-Moreno11, C de Haro4,9,10, O Roca4,9,10, MI Restrepo2,12**, JR Masclans1,13** and SEMICYUC COVID19 Group
1Critical Care Department, Hospital del Mar, Critical illness research group (GREPAC), Institut Hospital del Mar d’Investigacions Mediques (IMIM), Spain
2Division of pulmonary diseases & Critical Care Medicine, South Texas Veterans Health Care System and University of Texas Health San Antonio, USA
3Respiratory Medicine Department-Muscle Wasting and Cachexia in Chronic Respiratory Diseases and Lung Cancer Research Group, Hospital del Mar, Spain
4Centro de Investigacion en Red de Enfermedades Respiratorias (CIBERES), Instituto de Salud Carlos III (ISCIII), Spain
5Critical Care Department, Hospital Universitari Joan XXIII, Spain
6Critical Care Department, Hospital de Salamanca, Spain
7Critical Care Department, Hospital La Fe, Spain
8Critical Care Department, Hospital Son Llatzer, Spain
9Servei de Medicina Intensiva, Parc Tauli Hospital Universitari, Spain
10Departament de Medicina, Universitat Autònoma de Barcelona, Spain
11Critical Care Department, Hospital de Guadalajara, Spain
12Division of Pulmonary & Critical Care Medicine, University of Texas Health San Antonio, USA
13Medicine and life science department (MELIS), Universitat Pompeu Fabra, Spain
* and ** contributed equally to the work
Submission: August 01, 2023; Published: August 11, 2023
*Corresponding author: Francisco Jose Parrilla Gomez, MD, Critical Care Department, Hospital del Mar, Passeig Maritim 25-29, E-08003 Barcelona, Spain
How to cite this article: Marin-Corral J, Parrilla F, Castellvi A, et al. Impact of a Non-Invasive Respiratory Support Strategy and Switches in Therapy During Acute Respiratory Failure Due to SARS-COV-2. Int J Pul & Res Sci. 2023; 6(4): 555694. DOI: 10.19080/IJOPRS.2023.06.555694
Abstract
Introduction: Non-invasive respiratory support (NIRS), high flow nasal cannula (HFNC) and non-invasive ventilation (NIV), have been used prior to mechanical ventilation (MV). We aimed to describe the use of initial NIRS and their subsequent changes and assess its impact on clinical outcomes among critically ill patients with acute respiratory failure (ARF) secondary to SARS-CoV-2 admitted to ICU.
Methods: Prospective, multicenter, observational cohort study comprising a secondary analysis of patients included in the SEMICYUC COVID-19 registry. All patients with ARF due to SARS-CoV-2 admitted to ICU and receiving respiratory support were enrolled. NIRS failure, ICU and hospital mortality, and length of stay (LOS) were evaluated.
Results: Among 3889 included patients, 43% initially underwent NIRS (75% HFNC and 25% NIV). NIV as first choice was associated with higher failure (74.4% vs 61.2%, p<0.001) and higher ICU and hospital mortality rates than HFNC (27% vs 20%, p=0.003 and 29% vs 21%, p=0.002). NIV to HFNC switch showed the lowest failure (43%), lowest ICU and hospital mortality rates (18% and 19%) and the shortest LOS (9 days [6-15] vs 21 days [14-32]). HFNC to NIV switch did not significantly worsen outcomes compared to HFNC failure, but patients who failed after the switch showed the highest mortality (42% both ICU and hospital) and the longest LOS (24 [13-34] and 30 [23-48]).
Conclusion: NIRS is common in the ICU and switching NIRS strategies is a frequent practice within different scenarios. Physicians should bear in mind the possibility of outcomes worsening when failure occurs after switching.
Keywords: Non-invasive positive pressure ventilation; Nasal cannula; Acute respiratory failure; Covid-19; SARS-COV-2
Abbreviations: NIRS: Non-Invasive Respiratory Support; HFNC: High Flow Nasal Cannula; NIV: Non-Invasive Ventilation; MV: Mechanical Ventilation; ARF: Acute Respiratory Failure; LOS: Length of Stay; COPD: Chronic Obstructive Pulmonary Disease; SD: Standard Deviations; OR: Odds Ratios; CI: Confidence Intervals
Introduction
During the SARS-CoV-2 pandemic, severe acute respiratory failure (ARF) generated an unprecedented demand for intensive care resources in order to deliver respiratory support, and in many cases during the first wave of the pandemic led to ICU saturation. Although invasive mechanical ventilation (MV) remains the main support treatment in severe hypoxemic ARF with increased work of breathing, non-invasive respiratory support (NIRS) is commonly used as first strategy and can be applied by high flow nasal cannula (HFNC) and non-invasive ventilation (NIV). Several studies have suggested that HFNC is effective in patients with ARF, showing an improvement in ventilatory parameters and reducing the need for intubation, with a potential enhancement in patient survival compared to conventional oxygen therapy and NIV [1-6]. NIV has been extensively shown to improve hypercapnic ARF in patients with chronic obstructive pulmonary disease (COPD) and acute pulmonary oedema and has also shown benefits in hypoxemic ARF in so far as intubation can be avoided, but without clear benefits in mortality rates [7-15].
Despite its benefits, NIRS use carries the risk of patient self-inflicted lung injury and delayed intubation that detrimentally affects clinical outcomes [16-21]. In this regard, NIRS failure in patients with hypoxemic ARF is associated with increased mortality, especially when NIV is used to treat patients with pneumonia [12,19,20,22]. However, there are no formal recommendations in international guidelines regarding the procedure to follow when NIRS is started, and most importantly regarding when the treatment should be escalated [7]. This may lead to considerable differences between clinicians in terms of deciding which NIRS strategy to select as a first choice, when to consider it to have failed, and the timing of intubation and NIRS weaning, which may all impact clinical outcomes [23].
Moreover, in the SARS-CoV-2 scenario, pandemic overload meant that decisions had to be made based on resource availability, including switches of NIRS strategy in order to reserve MV for refractory cases. Studies have provided conflicting results about the efficacy of different NIRS in COVID-19 patients, and the currently available data are unable to identify the best NIRS first choice approach in these patients [24-29]. Moreover, little is known about the influence on outcomes of the use of more than one type of NIRS. In the present study, we sought to describe the use of initial NIRS (HFNC or NIV) and their subsequent changes and assess its impact on clinical outcomes in critically ill patients admitted to the ICU with hypoxemic ARF due to SARS-CoV-2 pneumonia.
Methods
Study design
Prospective, multicenter, cohort and observational study based on a secondary analysis of patients included in the SEMICYUC COVID-19 registry (the Spanish Society of Critical Care Medicine and Coronary Units). Between 1 February 2020 and 1 June 2021, data for all patients admitted to 73 Spanish ICUs due to ARF with COVID-19 pneumonia were recorded. COVID-19 was diagnosed by the presence of a SARS-CoV-2 positive rt-PCR test. The identities of patients were anonymized, and the review board waived the need for informed consent due to the non-interventional nature of the study. The registry was designed and conducted in accordance with the amended Declaration of Helsinki and the study was approved by the Institutional Review Boards of the participating centers.
Participants and study groups
All patients admitted to the ICU due to ARF with confirmed SARS-CoV-2 infection who received respiratory support at ICU admission were eligible for enrollment. Patients in whom the sequence of respiratory support at ICU admission could not be determined and those aged <18 years were excluded from the analysis. Patients who were initially treated with NIRS were classified according to the first NIRS choice used, HFNC or NIV. In each group, patients were subsequently classified as: 1) NIRS success; 2) NIRS failure, when MV was required or when the patient died having a do not intubation order (DNI); or 3) change of NIS, either HFNC-to-NIV or NIV-to-HFNC. The NIRS patients switched to a different NIRS method and were stratified according to success or failure. Patients who were directly intubated and mechanically ventilated were analyzed independently.
The primary outcome of the study was NIRS failure, described as the need for invasive MV or when patients were DNI. Secondary outcomes were all-cause mortality and ICU and hospital length of stay (LOS). These outcomes were evaluated in all the population and separately in each of the previously described patient subgroups. Comparisons of mortality and LOS between NIRS and MV groups excluded patients who were DNI and are shown in the Online Supplement.
Data collection
Demographics (age and gender), presence of comorbidities and usual treatment were registered, as well as severity of illness (APACHE and SOFA scores), biomarkers, respiratory status (PaO2/FIO2 ratio and presence of ≥ 3 quadrants infiltrated on chest radiography) and presence of shock at ICU admission, which was defined as the need for vasopressors [30], and COVID-related treatment received. Data collection also included ventilatory data and outcomes such as respiratory support and type of NIRS used as first choice, subsequent NIRS changes, NIRS failure, ICU and hospital mortality and ICU and hospital LOS.
Statistical analysis
Categorical variables were expressed as frequencies and percentages, and continuous variables as means and standard deviations (SD) when data were normally distributed, or otherwise as median and interquartile range (25th-75th percentile). Differences between groups were analyzed using the chi square test or Fisher’s exact test for categorical variables, and ANOVA, Student’s t-test or the Mann-Whitney U-test for continuous data. Statistical significance was set at a p-value ≤ 0.05. Logistic regression was used to assess independent factors associated with NIRS failure and mortality. Variables that were clinically relevant and presented significant differences in the bivariate analysis were included in the logistic regression. Odds ratios (OR) and 95% confidence intervals (CI) were calculated. The data were analyzed using the Statistical Package for the Social Sciences (SPSS, Chicago, IL, USA) 18 version.
Results
Distribution of the population
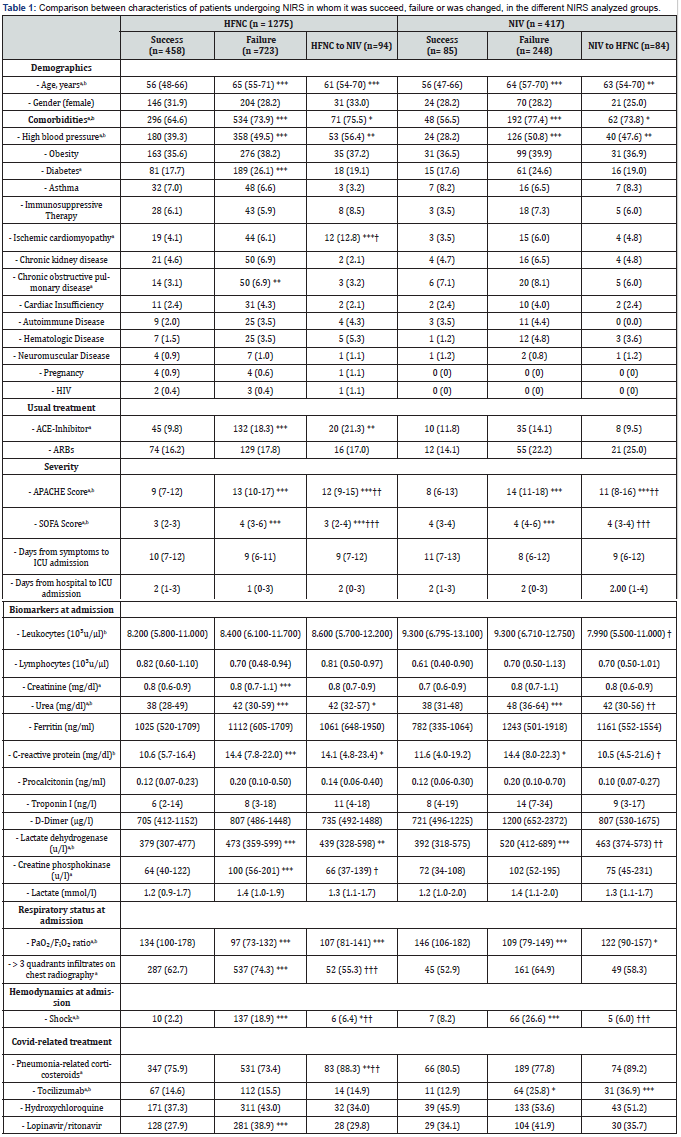
From a total of 3889 patients admitted to the ICU for ARF and confirmed SARS-CoV-2 infection needing respiratory support, NIRS was used as the first respiratory support strategy in 1692 (43.5%) and MV in 2197 (56.5%) (Figure 1). Among NIRS patients, HFNC was used in 1275 (75.3%) patients and NIV in 417 (24.7%). Switch of NIRS was present in 11% of total NIRS and HFNC-to-NIV shifts were less frequent than NIV-to-HFNC shifts (7.4% vs 20.1%, p<0.001) (Figure 1). Thirty-six patients (2.1% of all NIRS patients) were DNI and were distributed equally in the two groups (HFNC 2.0% and NIV 2.4%). Main characteristics of patients included in the different groups are described in the Online Supplement (OS Table 1).
NIRS failure
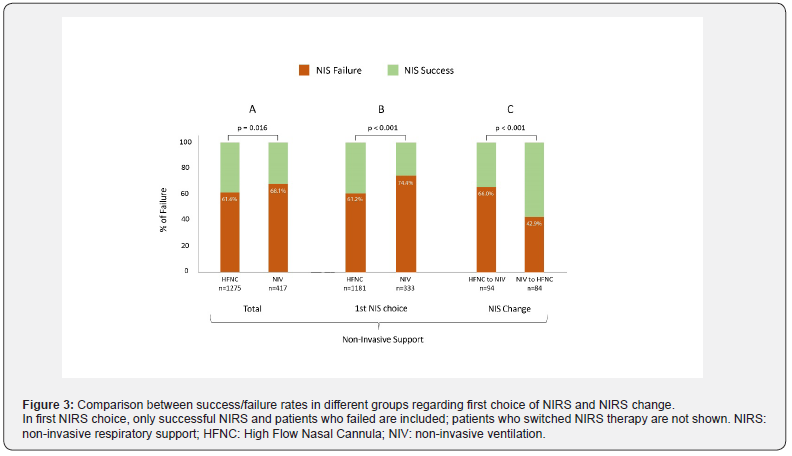
Among all patients who received NIRS at ICU admission (n=1692), 63.2% failed (n=1069). The characteristics of NIRS success and NIRS failure groups are described in detail in the OS Table 2. Patients with successful NIRS were more likely to have received HFNC as the first NIRS choice, and they also presented higher rates of NIRS switch. However, the most frequently used switch was NIV-to-HFNC. Figure 2 shows a detailed flow chart of NIRS failure, indicating the initial NIRS therapy used and the subsequent changes. NIRS failure rates were higher in the NIV group compared with HFNC group (68.1% vs 61.6%, p=0.016). Taking into account only the first choice of NIRS therapy, NIV group had higher failure rate than HFNC group (74.4% vs 61.2%, p<0.001) (Figure 3A and 3B). Among patients in whom NIRS was changed, the HFNC-to-NIV switch presented higher failure rates than NIV-to-HFNC (66% vs 42.9%, p<0.001) (Figure 3C). Table 1 shows the characteristics of patients regarding NIRS failure, success, or switch in both initial NIRS groups, which are described in the Online Supplement.
Data expressed as frequencies and percentages [n (%)] or medians and interquartile ranges (IQR or 25th-75th percentile).
a: ANOVA test with p≤0.05 for HFNC group. b ANOVA test with p≤0.05 for NIV group. * p≤0.05 vs Success; ** p≤0.01 vs Success; *** p≤0.001 vs Success; † p≤0.05 vs Failure; †† p≤0.01 vs Failure; ††† p≤0.001 vs Failure.
HFNC: High Flow Nasal Cannula; NIV: Non-Invasive Ventilation; HIV: Human Immunodeficiency Virus; ACE: Angiotensin Converting Enzyme; ARBs: Angiotensin II Receptor Antagonists; APACHE: Acute Physiology and Chronic Health Evaluation; SOFA: Sequential Organ Failure Assessment.
NIRS mortality
Overall mortality in patients who received NIRS at ICU admission was 21.9% (n=371). Patients who received NIV as the first NIRS therapy were more likely not to survive (30.5% vs 23%, p<0.01) (Table 2). Characteristics of survivors and non-survivors in the different subgroups are shown in Table 2 and described in the Online Supplement.
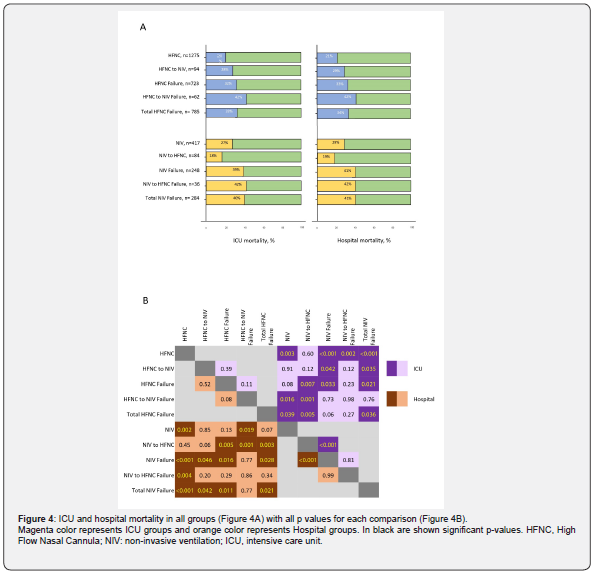
. ICU and hospital mortality rates of each group and subgroups with their comparisons and significances are shown in Figure 4. The NIV group presented higher ICU and hospital mortality rates than the HFNC group (27% vs 20%, p=0.003 and 29% vs 21%, p=0.002). As shown in Figure 4A, ICU and hospital mortality rates in HFNC group were 20% and 21% respectively. When HFNC failed, these figures rose to 32% and 33% respectively. When HFNC was switched to NIV, ICU and hospital mortality rates rose to 28% and 29%, but when this switch failed both these rates rose to 42%. Mortality rates tended to be higher in the HFNC-to-NIV failure group than in the HFNC failure group (ICU mortality 42% vs 32%, p=0.11, all-hospital mortality 42% vs 33%, p=0.08).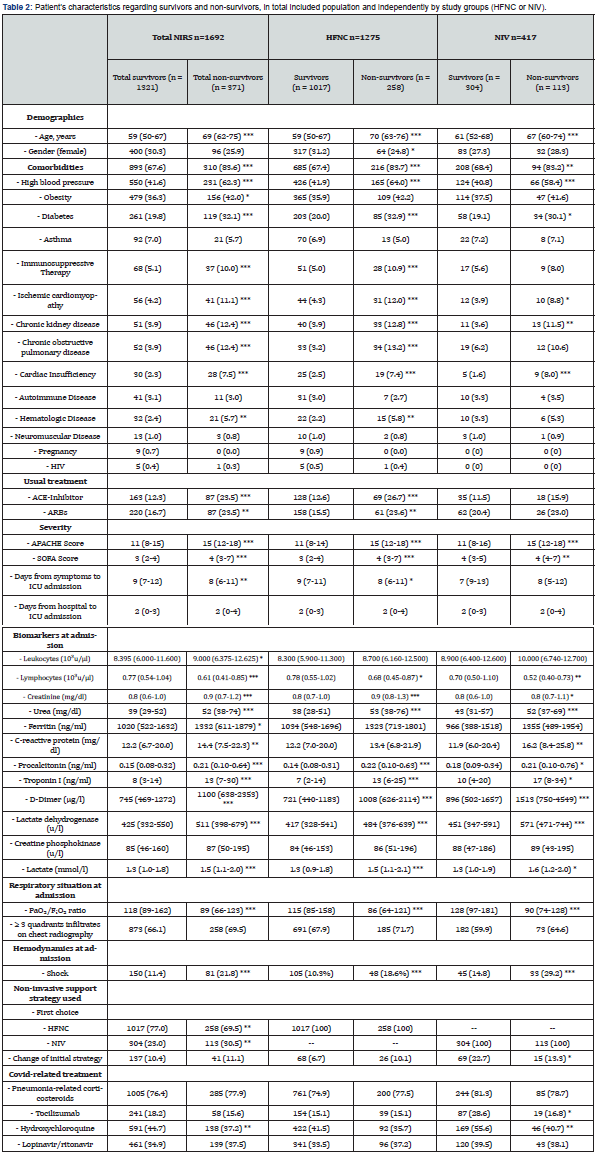
Data expressed as frequencies and percentages [n (%)] or medians and interquartile ranges (IQR or 25th-75th percentile). * p≤0.05; ** p≤0.01; *** p≤0.001.
NIS: Non-Invasive Respiratory Support; HFNC: High Flow Nasal Cannula; NIV: Non-Invasive Ventilation; HIV: Human Immunodeficiency Virus; ACE: Angiotensin Converting Enzyme; ARBs: Angiotensin II Receptor Antagonists; APACHE: Acute Physiology and Chronic Health Evaluation; SOFA: Sequential Organ Failure Assessment.
ICU and hospital mortality rates in the NIV group were 27% and 29% respectively. When NIV failed, these rates rose to 39% and 41% respectively. In contrast, when NIV was switched to HFNC, ICU and hospital mortality rates fell to 18% and 19% respectively, but when this switch failed these rates rose to 42% in both cases. No differences were found in mortality rates between NIV failure and NIV-to-HFNC failure groups (39% vs 42%, p=0.81 and 41% vs 42%, p=0.99). In patients who underwent NIRS switch, ICU and hospital mortality rates were higher in HFNC-to-NIV than in NIV-to-HFNC [28% vs 18%, p=0.12 and 29% vs 19%, p=0.06]. Other significant differences between groups are shown in Figure 4B.
NIRS length of stay
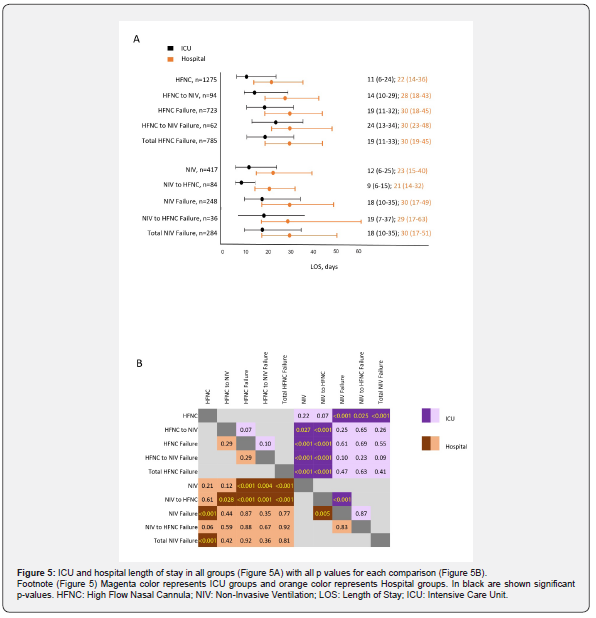
Overall NIRS LOS was 12 days (6 - 24) for ICU and 22 days (14 - 37) for hospital. There were no differences in ICU and hospital LOS among the patients that received any form of NIS. However, NIRS failure groups (i.e., failure of HFNC, HFNC-to-NIV, NIV, or NIV-to-HFNC) were associated with longer ICU and hospital LOS. In contrast, the NIV-to-HFNC switch group had the shortest ICU and hospital LOS (Figures 5A and 5B). Additionally, non-survivors had longer ICU and hospital LOS than survivors in the different NIRS groups (OS Figure 2).
Discussion
The key findings of this study in patients with SARS-CoV-2 admitted to the ICU due to ARF are that: 1) Use of NIRS is common in the ICU and switching NIRS strategies is a frequent practice within different scenarios. 2) HFNC was the most frequently used NIRS at admission and was less likely to be changed as initial NIRS support than NIV; 3) NIV at admission was associated with higher failure and mortality rates than HFNC; 4) The switch from HFNC to NIV did not present worse outcomes than HFNC failure, but when it failed, patients showed the highest mortality and the longest LOS rates of all subgroups.
In our cohort of patients, those who were initially treated with NIRS had lower disease severity, lower mortality, and shorter LOS than those who were initially mechanically ventilated. According to previous data, our results showed that NIRS failure were associated with worse outcomes [12,19,20,31]. Moreover, the outcomes worsening was more important after NIRS strategy switches, with similar mortality rates to patients who were first intubated and ventilated.
Our data show that switching NIRS in patients with ARF is a common practice in the ICU and that NIV-to-HFNC is more frequent than HFNC-to-NIV. NIRS switches have been described during the post-extubation period and in non-SARS-CoV-2 patients with ARF, but pandemic situation allowed us to evaluate the impact of NIRS switches when the use of NIRS had to be maintained to reserve MV for the most severe cases [32,33]. In the light of our results, we can speculate that even though ARF was the reason for starting NIRS, the decision to switch from one NIRS therapy to another was based on the particular situation of the patient. In this regard, patients switched from NIV to HFNC presented different baseline and evolution characteristics to those who were switched from HFNC to NIV. These characteristics may have been the reason for the switch. In this regard, the subgroup of patients who were switched from NIV to HFNC presented milder clinical characteristics at admission, similar to those in the HFNC group; they also presented the lowest failure and mortality rates and the shortest LOS. These findings may suggest that successful switches from NIV to HFNC are mostly performed as a de-escalation process during NIV weaning.
In contrast, our results suggest that failure after the switch from NIV to HFNC is the worst scenario, due to the major increase in mortality rates and LOS almost to the level of patients who were ventilated with MV at ICU admission and were more severely ill. In this case, the switch to HFNC might be used as a rest tool to increase NIV tolerability, as previously described by Frat et al. [32] in non-SARS-CoV-2 patients [32]. Nevertheless, in the subgroup of patients who were switched from HFNC to NIV, the move could be understood as an escalation of NIRS therapy. Although our results suggest that this switch did not worsen outcomes compared with patients who failed HFNC without switching, it should be considered that when failure occurs after the switch (in 66% of cases) mortality and LOS drastically increase, reaching the rates recorded in initially mechanically ventilated patients (who tended to be elderly, had more severe illness and poorer clinical parameters at admission).
This study has several limitations that should be mentioned. First, it is a secondary analysis of a multicenter database which was not directly designed to evaluate the prognostic implications of NIRS switches. Although the results of this study suggest that NIRS therapy switching was common in clinical practice during the pandemics, it should be evaluated in the post-pandemic routinary ICU practice. Moreover, a lack of relevant information is present as the clinical parameters before the decision to intubate, the NIRS parameters at the time of failure, and the dynamic NIRS changes, which are key issues that future studies should consider. For instance, information regarding the reasons for NIRS switches and timing would help to elucidate whether NIRS therapies were used as de-escalation or escalation strategies. Second, we did not have information on the level of ventilatory support (CPAP or Pressure Support mode), the interface used or the use of heat humidifiers during NIV which might have influenced the outcomes [34]. However, diverse groups of subjects from several hospitals in Spain were enrolled and classified using a pragmatic approach, and the study results suggest differences in clinical practices in real-life settings. Moreover, these results show the need to further investigate switched NIRS groups in order to be able to implement new recommendations in future clinical guidelines.
In conclusion, this multicenter study shows that HFNC is the first NIRS used in most patients with ARF due to SARS-CoV-2 admitted to the ICU, and that it is associated with better outcomes regarding NIRS failure and mortality than NIV. We also report that the switch of NIRS strategies in these patients was a common practice in Spanish ICUs during pandemics, above all from NIV to HFNC. Moreover, the transition from HFNC to NIV or NIV to HFNC seems to respond to different needs or reasons that should be explored in greater depth in future studies. Although switching strategies do not seem to worsen outcomes, when making this decision, physicians should bear in mind the significant association with worse outcomes when failure occurs after switching. Further studies are also needed to better understand decisions regarding NIRS initiation and switching, as well as to identify predictive factors related to NIRS responders or non-responders before generalizing these findings in clinical practice.
Acknowledgments
We thank Marta Gas, Anna Salazar and all the health personnel (doctors, nurses, auxiliaries and the entire logistical team involved) for their dedication and commitment in helping to collect the data that made this study possible, and Victor Boutonnet and Llesmil Ahuirand for help with the data processing.
Conflict of Interest Statements
JRM: grants, travels and non-financial support from Fisher & Paykel, personal fees from Dextro; OR: research grant from Hamilton Medical AG and Fisher & Paykel Healthcare Ltd, speaker fees from Hamilton Medical AG, Fisher & Paykel Healthcare Ltd, Aerogen Ltd and Ambu, and non-financial research support from Timpel; all unrelated to this submission. None of the other authors have any conflicts of interest to declare.
References
- Frat J-P, Thille AW, Mercat A, Girault C, Ragot S, et al. (2015) High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med 372(23): 2185-2196.
- Agarwal A, Basmaji J, Muttalib F, Granton D, Chaudhuri D, et al. (2020) High-flow nasal cannula for acute hypoxemic respiratory failure in patients with COVID-19: systematic reviews of effectiveness and its risks of aerosolization, dispersion, and infection transmission. Can J Anesth 67(9): 1217-1248.
- Ni Y-N, Luo J, Yu H, Liu D, Ni Z, et al. (2017) Can High-flow Nasal Cannula Reduce the Rate of Endotracheal Intubation in Adult Patients With Acute Respiratory Failure Compared With Conventional Oxygen Therapy and Noninvasive Positive Pressure Ventilation?: A Systematic Review and Meta-analysis. Chest 151(4): 764-775.
- Rochwerg B, Granton D, Wang DX, Helviz Y, Einav S, (2019) High flow nasal cannula compared with conventional oxygen therapy for acute hypoxemic respiratory failure: a systematic review and meta-analysis. Intensive Care Med 45(5): 563-572.
- Oczkowski S, Ergan B, Bos L, Chatwin M, Ferrer M, et al. (2022) ERS clinical practice guidelines: high-flow nasal cannula in acute respiratory failure. Eur Respir J 59(4): 2101574.
- Lewis SR, Baker PE, Parker R, Smith AF (2021) High-flow nasal cannulae for respiratory support in adult intensive care patient. Cochrane Database Syst Rev 3(3): CD010172.
- Rochwerg B, Brochard L, Elliott MW, Hess D, Hill NS, et al. (2017) Official ERS/ATS clinical practice guidelines: noninvasive ventilation for acute respiratory failure. Eur Respir J 50(2): 1602426.
- Davidson AC, Banham S, Elliott M, Kennedy D, Gelder C, et al. (2016) BTS/ICS guideline for the ventilatory management of acute hypercapnic respiratory failure in adults. Thorax 71(Suppl 2): ii1-35.
- Cao Z, Luo Z, Hou A, Nie Q, Xie B, et al. (2016) Volume-Targeted Versus Pressure-Limited Noninvasive Ventilation in Subjects with Acute Hypercapnic Respiratory Failure: A Multicenter Randomized Controlled Trial. Respir Care 61(11): 1440-1450.
- Crimi C, Noto A, Princi P, Esquinas A, Nava S (2010) A European survey of noninvasive ventilation practices. Eur Respir J 36(2): 362-369.
- Ozsancak UA, Sidhom SS, Khodabandeh A, Ieong M, Mohr C, et al. (2014) Use and outcomes of noninvasive positive pressure ventilation in acute care hospitals in Massachusetts. Chest 145(5): 964-971.
- Demoule A, Girou E, Richard J-C, Taille S, Brochard L (2006) Benefits and risks of success or failure of noninvasive ventilation. Intensive Care Med 32(11): 1756-1765.
- Bourke SC, Piraino T, Pisani L, Brochard L, Elliott MW (2018) Beyond the guidelines for non-invasive ventilation in acute respiratory failure: implications for practice. Lancet Respir Med 6(12): 935-947.
- Keenan SP, Sinuff T, Cook DJ, Hill NS (2004) Does noninvasive positive pressure ventilation improve outcome in acute hypoxemic respiratory failure? A systematic review. Crit Care Med 32(12): 2516-2523.
- Munshi L, Mancebo J, Brochard LJ (2022) Noninvasive Respiratory Support for Adults with Acute Respiratory Failure. N Engl J Med 387(18): 1688-1698.
- Grieco DL, Menga LS, Eleuteri D, Antonelli M (2019) Patient self-inflicted lung injury: implications for acute hypoxemic respiratory failure and ARDS patients on non-invasive support. Minerva Anestesiol 85(9): 1014-1023.
- Brochard L, Slutsky A, Pesenti A (2017) Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. Am J Respir Crit Care Med 195(4): 438-442.
- Spinelli E, Mauri T, Beitler JR, Pesenti A, Brodie D (2020) Respiratory drive in the acute respiratory distress syndrome: pathophysiology, monitoring, and therapeutic interventions. Intensive Care Med 46(4): 606-618.
- Carrillo A, Gonzalez-Diaz G, Ferrer M, Martinez-Quintana ME, Lopez-Martinez A, et al. (2012) Non-invasive ventilation in community-acquired pneumonia and severe acute respiratory failure. Intensive Care Med 38(3): 458-466.
- Kang BJ, Koh Y, Lim CM, Huh JW, Baek S, et al. (2015) Failure of high-flow nasal cannula therapy may delay intubation and increase mortality. Intensive Care Med 41(4): 623-632.
- Roca O, Caralt B, Messika J, Samper M, Sztrymf B, et al. (2019) An Index Combining Respiratory Rate and Oxygenation to Predict Outcome of Nasal High-Flow Therapy. Am J Respir Crit Care Med 199(11): 1368-1376.
- Bellani G, Laffey JG, Pham T, Madotto F, Fan E, et al. (2017) Noninvasive Ventilation of Patients with Acute Respiratory Distress Syndrome. Insights from the LUNG SAFE Study. Am J Respir Crit Care Med 195(1): 67-77.
- Delbove A, Darreau C, Hamel JF, Asfar P, Lerolle N (2015) Impact of endotracheal intubation on septic shock outcome: A post hoc analysis of the SEPSISPAM trial. J Crit Care 30(6): 1174-1178.
- Wendel-Garcia PD, Mas A, Gonzalez-Isern C, Ferrer R, Manez R, et al. (2022) Non-invasive oxygenation support in acutely hypoxemic COVID-19 patients admitted to the ICU: a multicenter observational retrospective study. Crit Care 26(1): 37.
- Ranieri VM, Tonetti T, Navalesi P, Nava S, Antonelli M, et al. (2022) High-Flow Nasal Oxygen for Severe Hypoxemia: Oxygenation Response and Outcome in Patients with COVID-19. Am J Respir Crit Care Med 205(4): 431-439.
- Perkins GD, Ji C, Connolly BA, Couper K, Lall R, et al. (2022) Effect of Noninvasive Respiratory Strategies on Intubation or Mortality Among Patients with Acute Hypoxemic Respiratory Failure and COVID-19: The RECOVERY-RS Randomized Clinical Trial. JAMA 327(6): 546-558.
- Frat J-P, Quenot J-P, Badie J, Coudroy R, Guitton C, et al. (2022) Effect of High-Flow Nasal Cannula Oxygen vs Standard Oxygen Therapy on Mortality in Patients with Respiratory Failure Due to COVID-19: The SOHO-COVID Randomized Clinical Trial. JAMA 328(12): 1212-1222.
- Grieco DL, Menga LS, Cesarano M, Rosa T, Spadaro S, et al. (2021) Effect of Helmet Noninvasive Ventilation vs High-Flow Nasal Oxygen on Days Free of Respiratory Support in Patients With COVID-19 and Moderate to Severe Hypoxemic Respiratory Failure. JAMA 325(17): 1731-1743.
- Ospina-Tascon GA, Calderon-Tapia LE, Garcia AF, Zarama V, Gomez-Alvarez F, et al. (2021) Effect of High-Flow Oxygen Therapy vs Conventional Oxygen Therapy on Invasive Mechanical Ventilation and Clinical Recovery in Patients With Severe COVID-19. JAMA 326(21): 2161-2171.
- Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, et al. (2016) The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315(8): 801-810.
- Bellani G, Laffey JG, Pham T, Fan E, Brochard L, et al. (2016) Epidemiology, Patterns of Care, and Mortality for Patients With Acute Respiratory Distress Syndrome in Intensive Care Units in 50 Countries. JAMA 315(8): 788-800.
- Frat J-P, Brugiere B, Ragot S, Chatellier D, Veinstein A, et al. (2015) Sequential application of oxygen therapy via high-flow nasal cannula and noninvasive ventilation in acute respiratory failure: an observational pilot study. Respir Care 60(2): 170-178.
- Wang S-Y, Liang H-W, Lu G-S, Jiang Z-J, Zhang B-Z, et al. (2021) Effect of sequential high-flow nasal cannula oxygen therapy and non-invasive positive-pressure ventilation in patients with difficult weaning from mechanical ventilation after extubation on respiratory mechanics. Ann Transl Med 9(15): 1251.
- Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP (2016) Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome a randomized clinical trial. JAMA - J Am Med Assoc 315(22): 2435-2441.






























