Relevance of the Stereoselective Behavior of Chiral Herbicides in the Environment
A Cervantes Díaz, J L Alonso Prados, C López Goti and P Sandín España*
Unit of Plant Protection Products, INIA, CSIC, Spain
Submission: October 14, 2023; Published: October 25, 2023
*Corresponding author: Pilar Sandín-España, INIA, CSIC. Ctra. La Coruña, Km. 7.5, 28040 Madrid, Spain, Email ID: sandin@inia.csic.es
How to cite this article: A Cervantes D, J L Alonso P, C López G, P Sandín E. Relevance of the Stereoselective Behavior of Chiral Herbicides in the Environment. Int J Environ Sci Nat Res. 2023; 32(5): 556350. DOI 10.19080/IJESNR.2023.32.556350
Abstract
Environmental behavior and toxicity of chiral compounds is a topic of great interest in environmental chemistry. In recent years, the evolution of the stereochemistry of organic compounds has highlighted the importance of considering separately stereoisomers of chiral herbicides when studying their fate and behavior in the environment. As many as 40% of all plant protection products currently used contain chiral active ingredients. Not only the effects on the biological target, but also their biodegradation, environmental fate and toxicity of stereoisomers of the same herbicide may be also different. As such, chiral compounds should be evaluated by using enantiomer-specific techniques and analytical tools.
In this sense, stereoselective separation and analysis with HPLC-MS/MS is one of the most effective and powerful technique for investigate the enantioselective behavior of the isomers. Most of the chiral separation are performed by means of columns based on derivatized amylose or cellulose due to their wide analyte coverage and compatibility with reverse phase and sensitive detection techniques such as mass spectrometry. For sure, research in this field will continue growing due to the necessity of assessing the risks related to exposure to chiral herbicides, which can be considered as emerging environmental contaminants.
Keywords: Chiral herbicide; HPLC; Stereoselectivity; Enantiomers; Diastereoisomers
Introduction
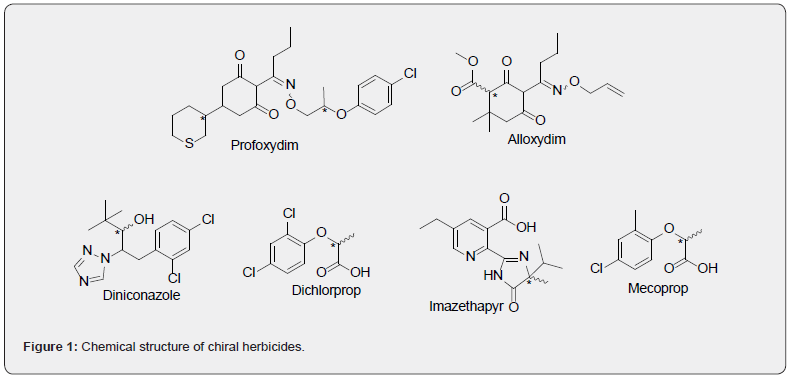
Stereoisomers are isomers that have the same molecular connectivity and differ in the spatial disposition of two or more atoms. These isomers constitute a great concern in the pesticide regulation framework [1]. Though, in the pharmaceutical industry, single enantiomer chiral drug design started in the 1960s with the thalidomide case, chirality in pesticides started in the 1990s and little attention has still been paid so knowledge of their environmental and toxicity remains incomplete [2,3]. As much as 40% of the herbicides currently used contain one or more asymmetric centres in their structure [3,4], and this percentage is expected to increase since the novel developed molecules have more complex structures. When more than one stereogenic centre is present in a molecule, not only enantiomers are generated, but also diastereomers. Diastereomers have different physical- and chemical properties and can behave like different substances in all aspects. Stereoisomers are usually commercialized, applied and released into the environment as mixtures of diastereomers or enantiomers and are usually difficult to separate since the physical and chemical properties are similar and practically identical in the case of enantiomers. Thus, production of a single pure active isomer is much more expensive. In fact, only 7% is sold as pure stereoisomer or as an enriched formulation of the active stereoisomer [4]. The differences in fate and/or toxicity of individual stereoisomers of pesticides is based in the stereoselective interaction with enzymes, receptors or other naturally occurring molecules [2]. Thus, pesticides of the triazole family, showed a completely different biological activity between the two stereoisomers; for example, R isomer of diniconazole (Figure 1) is a fungicide whereas the S isomer is a plant growth regulator [1].
In the case of herbicides, pesticides used to control undesired plants in the crops, a great number of molecules present chiral atoms in their structures.
Imidazolinones were developed in the early 1980s and have been broadly employed on soybeans, cereals, sunflower and lentils. They are chiral herbicides with a stereocenter in the imidazolinone ring. Studies of enantioselectivity in herbicidal activity revealed that the R enantiomer of imazethapyr (Figure 1) exhibited 10-fold more inhibition to acetolactate synthase (ALS) than the S enantiomer [5].
Phenoxypropanoic-acid herbicides are systemic and postemergence herbicides that were introduced in the 1950s. Representative chiral phenoxypropanoic-acid herbicides include mecoprop and dichlorprop (Figure 1). Both herbicides have one stereogenic center. It is known that R enantiomers have greater herbicidal activity than the S enantiomers [6]. Nonetheless, the racemic mixtures are still used, releasing both isomers into the environment with unknown ecotoxicity to non-target plants. Regarding their environmental behavior, the enantiomeric degradation of dichlorprop showed that the R enantiomer was preferentially degraded over the S enantiomer [6].
Diclofop methyl (Figure 1) is a chiral aryloxyphenoxypropionic herbicide first registered in 1982 for control of wild oats and annual grasses in wheat and barley. The R enantiomer exhibits more potent inhibition of acetyl-CoA carboxylase (ACCase) than does the S enantiomer [7].
Cyclohexanedione oxime herbicides (CHD) have been developed during the last 30 years. In general, this family of herbicides is used for post-emergence control of annual and perennial grass weeds in broad-leaved crops, except profoxydim which is used for the control of grass weeds in rice crop [8]. These compounds show a keto-enol tautomerism due to the presence of two ketone groups, as well as two isomers E/Z relating to the alkyl side chain bound to oxime ether group. Alloxydim (Figure 1) was the first herbicide of this family introduced into the market in 1978 and presents one chiral carbon. A bioassay performed by Sandin-España´s group [9] to compare the herbicidal activity of E and Z isomers showed that the Z isomer of alloxydim in the isomeric mixture has little or no phytotoxic effect on the wheat germination whereas E-alloxydim inhibited the seed germination at low doses.
Profoxydim (Figure 1) was the last ciclohexanediones herbicide registered in 1998 and it is applied to rice paddy systems [10]. Profoxydim has two stereogenic center and it is applied as a mixture of four diastereoisomers. Our group has developed a novel chiral chromatographic method for the simultaneous determination of the four profoxydim isomers in rice grain and husk by means of HPLC-MS/MS [11] using a polysaccharide-based chiral stationary phase column. These results will help to provide data of the different toxicities and behavior of each stereoisomer to perform an accurate risk assessment and to gain the final objective of minimizing the risks to human, animal health and the environment.
Regarding environmental behavior, many chiral compounds are released to the environment as racemates and the biodegradation and environmental fate of each isomer may differ [12]. In many cases, only one enantiomer is decomposed, while the other enantiomer is accumulated in the environment. Enantiomeric enrichment in the environment has been reported for many chiral herbicides such as acetamide, triazole, and organophosphorus herbicides [13]. This is due to differences in the biological degradation pathway and rate among isomers. Consequently, environmental fate and behavior of isomers must be assessed separately when main degradation and dissipation routes are of biological origin.
Furthermore, environmental profile is always improved by using only the active isomer since the amount of chemical introduced in the environment is significantly reduced. Ignoring stereoselectivity may cause over- or under-estimation of environmental fate. When chirality is not considered and isomeric mixtures are employed, the use of these products may suppose the emission of a 50-75% of unnecessary products.
Therefore, in order to reduce the burden of herbicides in the media and protect the environment from unintended effects, it is of great significance to separate the stereoisomers of chiral herbicides in order to evaluate the respective properties of each pure isomer, and eventually to market the optical isomer that shows higher biological activity. Standard achiral analysis cannot measure optical isomer compositions, but only the sum total of stereoisomers, giving only partial information. Thus, traditional risk assessments are unreliable if enantioselective behaviors occur. Consequently, it is of utmost importance to develop chiral separation and additional stereoisomer analysis methods for chiral herbicides that can be applied to environmental matrices and agricultural products. However, there are relatively few publications focusing on trace analyses of chiral herbicides in environmental samples and agrochemical products.
At the analytical level, chiral separation of pesticide stereoisomers has been commonly performed by different techniques (gas chromatography, capillary electrophoresis, supercritical fluid chromatography) with the high-performance liquid chromatography (HPLC) technique being the most widely used for chiral separation [13-15].
Furthermore, the development of chiral stationary phases (CSPs) [14] capable of effective chiral recognition of a wide range of stereoisomers is the key point that makes the HPLC technique one of the most effective and the preferred technique for chiral separations in the last two decades [13] to determine herbicides in the environment [4] especially those nonvolatile, polar and thermolabile [6].
The high specificity and sensitivity required for the analysis of stereoisomer residues in very complex agro-environmental matrices is a major analytical challenge. In this regard, LC coupled to MS/MS in pesticide residue analysis has become increasingly popular, as it allows the detection of target compounds in the low ng/L range, and also free of interferences from matrix present in complex samples. Although numerous chiral LC-MS/ MS methods have been applied to the enantioselective analysis of pharmaceutical stereoisomers in recent years [3], applications of these methods on chiral herbicides remain scarce in the literature.
Currently, polysaccharide-based CSPs are the most widely used columns due to their advantages, such as their high versatility, selectivity and reproducibility [16]. More than 95% of racemic compounds have been successfully solved using polysaccharide CSPs in particular, cellulose and amylose-based polysaccharide CSPs, as they are the most versatile and applicable phases [6].
Conclusion
Chiral herbicides can have significant environmental and toxicological effects once these compounds are released in the environment. Biotic processes, such as those driven by microorganism in soil or enzymes in plants play a key role and are responsible for enantio- or diastereoselective transformation modifying the stereomeric proportions. However, most chiral herbicides are mainly manufactured as racemates or mixture of diastereoisomers since the synthesis of the pure optically active isomer is still highly costly. Thus, it is highly important to develop chiral analytical techniques with high efficiency to determine the stereoisomers of herbicides in the environment. Furthermore, not only it is important to develop analytical methods to achieve the required low detection limits but also preparative scale methods to perform further studies with the individual’s isomers. Over the last decades, HPLC technique using the appropriate chiral stationary phases (the polysaccharide ones are the most popular) take the leading role in the chiral separation of herbicides. Thus, it is necessary to continue investigating and developing chiral methodologies to detect these compounds in the environment and to be able to formulate pure optical isomers to be used as agrochemicals.
Acknowledgement
This work was supported by projects RTA2017-00043-00- 00 of the Ministerio de Economía, Industria y Competitividad (MINECO), Spain and Y2020/EMT-6290 (PRIES-CM) of the Comunidad de Madrid, Spain. A. Cervantes-Díaz thanks MCIN for a predoctoral contract (grant PRE2018-085397).
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Oriol Magrans J, Alonso Prados JL, García Baudín JM (2002) Importance of considering pesticide stereoisomerism-proposal of a scheme to apply Directive 91/414/EEC framework to pesticide active substances manufactured as isomeric mixtures. Chemosphere 49(5): 461-469.
- Pérez S, Barceló D (2008) Applications of LC-MS to quantitation and evaluation of the environmental fate of chiral drugs and their metabolites. Trends in Analytical Chemistry 27(10): 836-846.
- Jeschke P (2018) Current status of chirality in agrochemicals. Pestide Management Science 74(11): 2389-2404.
- Lucci E, Bosco C, Antonelli L, Fanali C, Gentili A, et al. (2022) Enantioselective high-performance liquid chromatographic separations to study occurrence and fate of chiral pesticides in soil, water, and agricultural products. Journal of Chromatography A 1685: 463595.
- Chin TE, Wong RB, Pont JL, Karu AE (2002) Haptens and monoclonal antibodies for immunoassay of imidazolinone herbicides. Journal of Agricultural and Food Chemistry 50(12): 3380-3389.
- Ye J, Wu J, Liu W (2009) Enantioselective separation and analysis of chiral pesticides by high-performance liquid chromatography. Trends in Analytical Chemistry 28(10): 1148-1163.
- Nakahira K, Haga M, Uchiyama M, Suzuki K (1990) Comparative effects of Quizalofop and its esters on acetyl-CoA carboxylase and fatty acid biosynthesis in corn leaf chloroplasts. Journal of Pesticide Science 15: 189-197.
- Walter H (2001) Profoxydim: Development of a Herbicide from Laboratory to Field. In: Prado RD, Jorrín JV (Eds.), Uso De Herbicidas En La Agricultura Del Siglo XXI. Córdoba: Servicio Publicaciones Universidad de Córdoba; 2001. pp. 19-30.
- Villaroya Ferruz M, Sevilla Morán B, Cubero Torrado NH, Alonso Prados JL, Sandin España P, et al. (2015) Phytotoxic study of the E/Z isomers of herbicide alloxydim. Congress of Malherbology, Spain.
- (2006) In: Tomlin CDS (Ed.), The Pesticide Manual: A World Compendium. (14th edn), Hampshire: BCPC Publications.
- Sandin España P, Cervantes Diaz A, Mateo Miranda M, Sanchez Argüello P, Cubero Torrado NH, et al. (2022) Development and validation of a chiral HPLC-MS/MS method for simultaneous determination of profoxydim isomers in rice and husk. 11th European Conference on Pesticides and Related Organic Micropollutants in the Environment & 17th Symposium on Chemistry and Fate of Modern Pesticides, Greece.
- Xinglu P, Fengshou D, Zenglong C, Jun X, Xingang L, et al. (2017) The application of chiral ultra-high-performance liquid chromatography tandem mass spectrometry to the separation of the zoxamide enantiomers and the study of enantioselective degradation process in agricultural plants. Journal of Chromatography A 1525: 87-95.
- Ling L, Shanshan Z, Lixia J, Cheng Z, Weiping L (2010) Enantiomeric separation of organophosphorus pesticides by high-performance liquid chromatography, gas chromatography and capillary electrophoresis and their applications to environmental fate and toxicity assays. Journal of Chromatography B 878(17-18): 1264-1276.
- Ward TJ, Ward KD (2010) Chiral separations: fundamental review 2010. Analytical Chemistry 82(12): 4712-4722.
- Maier NM, Franco P, Wolfgang L (2001) Separation of enantiomers: needs, challenges, perspectives. Journal of Chromatography A 906(1-2): 3-33.
- Qian M, Wu L, Zhang H, Wang J, Li R, et al. (2011) Stereoselective determination of famoxadone enantiomers with HPLC-MS/MS and evaluation of their dissipation process in spinach. Journal of Separation Science 34(11): 1236-1243.






























