Antimony Recovery by Electro-Electrodialysis (EED)
Gerardo Cifuentes1*, Gabriel Riveros2, Matías Agurto1, Sebastián Díaz1 and Magdalena Cifuentes-Cabezas3
1Department of Metallurgical Engineering, Universidad de Santiago de Chile, Chile
2Transducto S.A., Chile
3Department of Chemical and Nuclear Engineering, Universitat Politècnica de València, Spain
Submission: September 23, 2022;Published: October 12, 2022
*Corresponding author: Gerardo Cifuentes, Department of Metallurgical Engineering, Universidad de Santiago de Chile, Avda. L.B. O’Higgins 3363, Estación Central, Santiago, Chile
How to cite this article: RGerardo C, Gabriel R, Matías A, Sebastián D, Magdalena C-C. Antimony Recovery by Electro-Electrodialysis (EED).Int J Environ Sci Nat Res. 2022; 31(2): 556310.DOI: 10.19080/IJESNR.2022.31.556310
Abstract
Antimony is present in the electrolyte copper refining as impurity that come from pyrorefinated anodes. To produce metallic copper with high quality it is necessary to have under control all the impurity. In special for the antimony capture from the electrolyte is necessary an ionic exchange (IX) process and later elusion stage of Sb by means of an acid hydrochloric solution, ending the process the antimony eluate liquor is send to precipitation with gypsum and deposited under land. This paper proposes an alternative to the clasical co-precipitation process of antimony, using an electro-electrodialysis (EER) cell with a cation exchange membranes, with this alternative path is possible to recover metallic antimony at the cathode.
This paper presents the laboratory latest results to recover antimony as cathode from hydrochloric industrial eluate electrolytes from IX process at 300A/m2, 600L/min, room temperature and without organic additives. The best result parameters obtained was faradic efficiency of 92.91% at current intensity of 300A/m2, voltage 2.3V and flow rate solution of 300L/min for synthetic 6N HCl solutions with 20g/L of Sb3+. For a real solution the parameters obtained was faradaic efficiency of 96.5% and cathodic composition of Sb 96.04% and 3.72% Bi.
Keywords: Electro-electrodialysis; Antimony; Recovery
Introduction
In the copper electrorefining industry the purification process of copper electrorefining solutions contaminated with antimony (300 to 500mg/L) consists in placing them in contact with an ion exchange (IX) resin to remove Sb from the electrolyte. In this case the last step of IX consist in the use of hydrochloric acid solution as eluate to obtain a rich antimony solution with approximately 20-40g/L of Sb. This eluate is distillated to recovery hydrochloric acid, the rest of the solution with antimony are fixed by co-precipitation with gypsum. The uncontrol of this last operation represents most of the operational problems in the electrolyte treatment plant.
The electro-obtaining process of antimony it is possible in various ways, one of them is by means of an electro-electrodialysis (EED) cell.
The concept of EED using a selective ionic membrane, applying a potential difference between the electrodes, the ions pass through the membrane such as is show in the Figure 1, concentrating and/or purifying allowing to obtain a cathode of high purity. This device has the particularity of working with hydrochloric acid solutions 6N. The concentrations of antimony ions could vary in the range of 2.5 to 80g/L depending on the previous step. An advantage of this process is that during electrolysis, the solution depleted in the metal ion can be reused in the process of the element of interest if necessary.
This alternative process using a cell based on electro-electrodialysis (EED) offer a new form to recovery secondary products in the copper industry.
Methodologyy
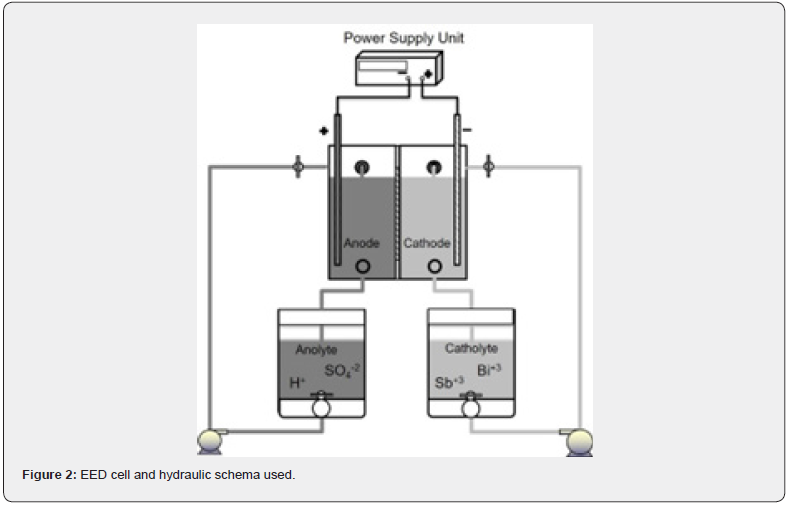
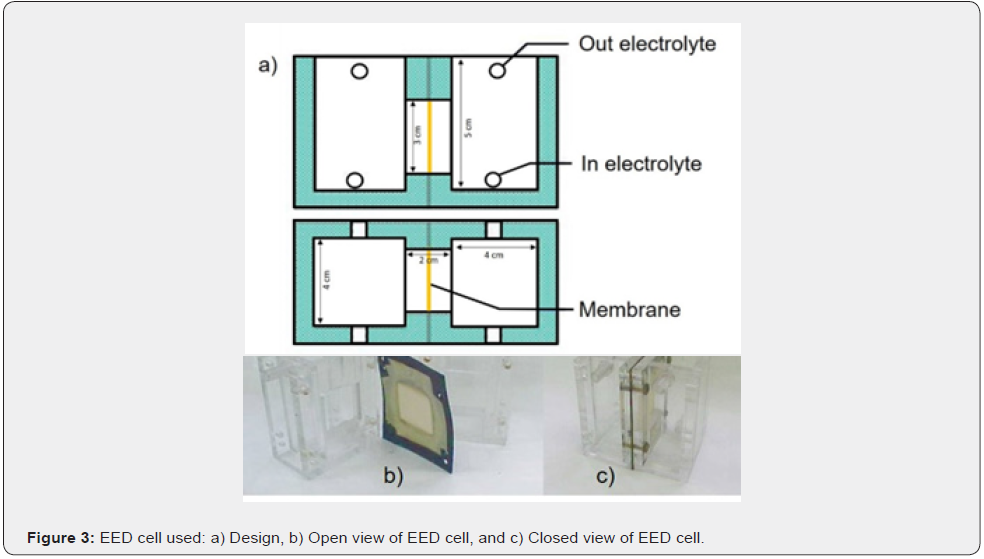
The electro-electrodialysis system used, consists of two compartments separated by a cationic membrane, one of which contains a synthetic 6N of HCl and 20 g/L of Sb3+ and 4g/L of Bi3+ catholyte, where the cathode is located, and the other compartment separated by the 5x5cm Ionac MC3480 cationic membrane, which contains a synthetic 30g/L of H2SO4 anolyte solution. The cathode consists of a 9cm2 316L stainless steel sheet (the back side was insulated with Teflon) and the anode was made of titanium covered with RuO2. All the system is shown in Figure 2. The cathodic current densities used in all the test was 300A/m2. The different time for the tests was 2, 3, 6 and 8 hours at normal pressure and temperature conditions. For validate all the last obtained results, a test was carried out with real solution,an industrial IX eluent from an electrolytic refinery of north Chile´s containing 6N of HCl and 30g/L of Sb3+ and 6g/L of Bi3+, the experimental parameter were the same conditions before mentioned with a test time of 2 hours. Figure 3 shows the EED cell used and their design dimension.
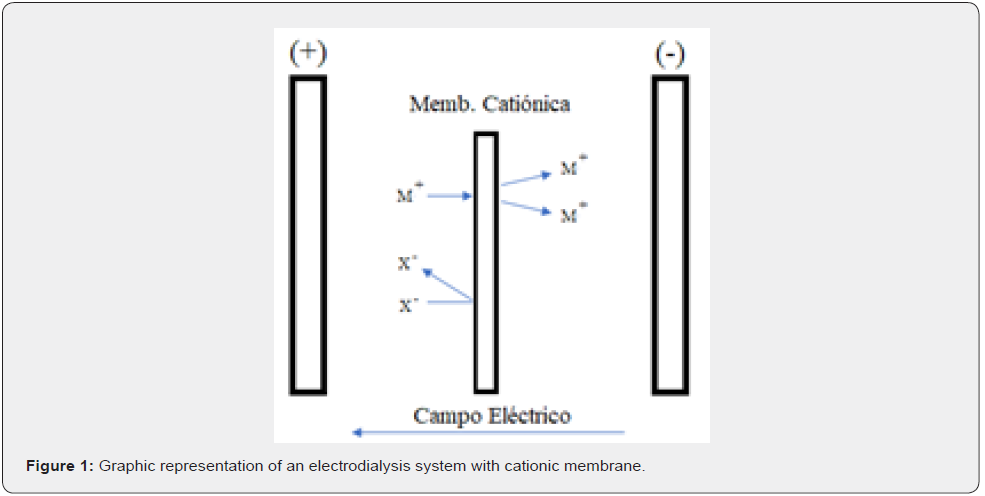
The cell will be connected to a power source, with the positive pole connected to the anode and the negative pole connected to the cathode.
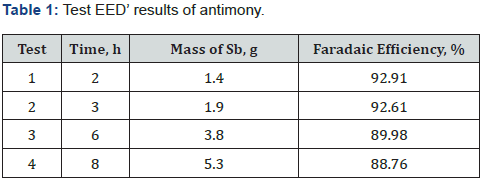
At first, reactive electrodialysis (EED) tests with cationic membrane were performed to determine the best performance of electrode pair against the operating conditions. In these tests direct eluate solution (unconditioned) was used and four different time, see Table 1. The tested electrodes pair (anode-cathode) were stainless steel 304-titanium/RuO2 and stainless steel 316L-titanium/RuO2.
Results and Discussion
Table 1 show the results of the tests using synthetic solution at 300A/m2 current density, it is seen that when increase the time lower is the faradaic efficiency. This is a logical result, because when increase the time of the electrolysis, lower will be the antimony concentration in the solution, increasing the hydrogen evolution reaction like parasite. Ending the test, the final Sb concentration into the solution was 1g/L.
The best result parameters obtained was faradic efficiency of 92.91%, a current intensity of 300A/m2, voltage 2.3V and rate flow solution of 300L/min. The stainless steel 304 was attacked by pitting corrosion during the test so its use was not continued in subsequent tests.
The results of the electrolysis tests with hydrochloric acid 6N solution obtained from the IX elution step from the industry and one electrode pair of stainless steel 316L-titanium/RuO2 are showing in Table 2. In this table It can be seen the Faradaic efficiency and the cathodic composition when metallic Sb and Bi are obtained with EED cell using a real IX eluate solution [1-6].
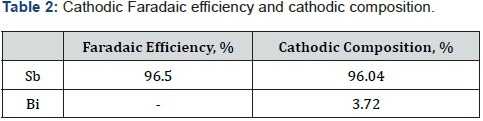
Figure 4 show the cathode 96.04% antimony cathode produced with the best parameters obtained.
Conclusion
From the results obtained, it is inferred that it is possible to electro-obtain metallic Sb + Bi with high quality from hydrochloric acid solutions 6N from unconditioned IX eluent solution. The values seem to be independent of the presence of additives, although its presence plays an important role in terms of the surface quality of the deposit in the majority several cases of electrowinning of metals.

Acknowledgment
The authors express their gratitude to the DICYT of the University of Santiago of Chile and Chemical and Nuclear Engineering Department, University Research Institute for Industrial, Radiophysical and Environmental Safety (ISIRYM) of Polytechnic University of Valencia (UPV), Spain. The authors express their gratitude too to the AQUAMEMTEC-CYTED (Project 318RT0551) and the ERAMIN 2 (Project Sb-RECMEMTEC).
References
- Alafara A, Adekola FA, Folashade AO (2005) Quantitative Leaching of a Nigerian Iron Ore in Hydrochloric Acid. Journal of Applied Sciences and Environmental Management 9(3): 15-20.
- Cifuentes, G., et al. (2018) Copper electrowinning from diluted solutions through EDR cells with high fluidodynamics. Euromembrane 2018, Valencia, 9th -13th July, Spain.
- Cifuentes G, Guajardo N, Hernandez J (2015) Recovery of hydrochloric acid from ion exchange processes by reactive electrodialysis. J Chil Chem Soc 60(4): 2711-2715.
- Cifuentes L, Mondaca C, Casas JM (2004) The effectiveness of membrane systems for the separation of anolyte and catholyte in a lab-scale copper electrowinning cell based on reactive electrodialysis. Minerals Engineering 17(6): 803-809.
- Radulescu, A Ficelco Olteanu, E Panturu, L Grigoras (2008) New Hydrometallurgical Process for Gold Recovery. Sci Environ 53(67): 135-139.
- Riveros F (2014) Eliminación de ion nitrato de soluciones ácidas de cobre mediante electrodiálisis. Trabajo de titulación para obtener el Título de Ingeniero Civil en Metalurgia y Magister en Ciencias de la Ingeniería mención Metalurgia Extractiva. Departamento de Ingeniería en Metalurgia. Universidad de Santiago de Chile, Santiago, Chile.






























