Diversity and Abundance of a Pollinator Group: Hawkmoths (Lepidoptera: Sphingidae) in Forests and Surrounding Farmlands, East Usambara Mountains, Tanzania
Adelaide E Sallema1 and Shabani S Selemani1,2*
1Department of Collection, National Museum and House of Culture, National Museum of Tanzania, Tanzania
2Biological and Marine Sciences Unity, Marian University College, Tanzania
Submission: September 22, 2022; Published: October 03, 2022
*Corresponding author: Shabani S Selemani, Biological and Marine Science Unity, Marian University College, P. O. Box 47, Bagamoyo, Tanzania
How to cite this article: Adelaide E S, Shabani S S. Diversity and Abundance of a Pollinator Group: Hawkmoths (Lepidoptera: Sphingidae) in Forests 02 and Surrounding Farmlands, East Usambara Mountains, Tanzania. Int J Environ Sci Nat Res. 2022; 31(1): 556307. DOI 10.19080/IJESNR.2022.31.556307
Abstract
Pollinators are now facing a drastic biodiversity fall in various parts of our world. In Africa data to measure and monitor their biodiversity trend is still inadequate and concentrated more on some insect pollinator groups, especially bees and butterflies. In this study, hawkmoth diversity and abundance in East Usambara Mountains were assessed. Data collection was conducted from three different habitat types that included thick vegetation assemblage with canopy cover, edges of the forest, and farmlands between 2017 and 2019. Hawkmoths were captured using a UV-light trap 250W mercury bulb in 12 sampling sites. A total of 35 hawkmoth species and 991 individuals belonging to the family Sphingidae were identified. The highest hawkmoth abundance was recorded from forest edges (p < 0.05). Canopy cover and farmlands showed lower hawkmoth abundance. Accordingly, higher Shannon Weiner diversity indices were recorded from canopy cover and forest edges (p < 0.05). Farmlands had the lowest Shannon Weiner diversity index. Differences in diversity and abundance of hawkmoths among three habitat types suggest that accelerating anthropogenic factors has led to the destruction of natural habitats. Meanwhile, natural habitat destruction has often been found to exert adverse effects on biodiversity. Moreover, the decline in biodiversity of pollinator insects like hawkmoths hinders the vital pollination service to both crops and wild flora. East Usambara Mountains are still an important refuge for many species that become restricted to different environments due to climate change and anthropogenic pressure, hence calling for regular pollinator conservation and monitoring. This study will provide the baseline data on hawkmoth species richness, abundances and diversity for potential conservation and monitoring programs.
Keywords: Hawkmoths; Abundance; Diversity; East Usambara mountains; Habitat types; Pollinator group; Pollinator conservation
Introduction
Insect pollinators contribute greatly to plant pollination compared to other pollinator groups but sadly they are now facing drastic declines in various parts of our world [1-3]. In Africa, data to measure and monitor their trend is still inadequate and concentrated more on some insect pollinator groups especially bees neglecting other groups such as hawkmoths. Hawkmoths provide vital ecosystem services as pollinators of both crops and wild flora [4]. They are insects from order Lepidoptera family Sphingidae. They pollinate many plants like pawpaw (Carica papaya and Asimina triloba), coffee (Coffee spp.), kapok (Ceiba pentandra) and Sphingophilous flowers which include Lantana spp., Lonicera spp., and Silene spp. [4-6]. There are about 4% of all the plants in Kenya, including pawpaw, and many different African orchids such as aerangoid orchids which are pollinated by hawkmoths [7,8]. A study conducted in South Africa by Johnson & Raguso [9], together with the findings of Martins & Johnson [10] in East Africa, demonstrates the existence of a guild of African plants that appears to rely almost exclusively on the long-tongued convolvulus hawkmoth Agrius convolvuli for pollination.
Hawkmoths comprise about 1,602 species and 205 genera worldwide [11]. About 70% of these species occur in the Africa and the New World [12,13]. Most species are crepuscular and nocturnal, except for some strictly diurnal hawkmoths, such as species of Aellopos, Hemaris and Macroglossum [14]. Hawkmoths are generally composed of nectarivorous species with little exclusion which makes them a very important pollinator group [15]. Apart from hawkmoths, insect pollinator groups include bees, flies, butterflies, moths, wasps, beetles, and thrips, among others [5,16]. It has been estimated that animal pollinators are considered necessary for the reproduction of 90% of flowering plants and one-third of human food crops [17]. Abundant and healthy populations of pollinators can enhance fruit set, quality, and size, then ultimately in farming increase production per acre, and in the wild, biodiversity and wildlife food sources increase [18]. In the United States year 2005 the value of honey bee and non-honey bee pollination services was worth an estimated US $ 15 billion and $ 5-6 billion respectively per year [19].
Currently, there is a persistent decline in the abundance and diversity of Lepidopterans in some countries such as Tanzania and the United States [20,21]. Furthermore, pollinating hawkmoth declines of eight species had been reported in the Northeastern United States which may have ecological effects on both the plants pollinated by these species and vertebrate predators of the moths [22]. In Tanzania, quantification of Lepidopterans to check their diversity trend is at an insufficient level even in the areas where the biodiversity hotspots of the world such as the East Usambara Mountains. Unfortunately, East Usambara Mountains have been under considerable pressure as a result of expanding human population and demands for arable land [23-25]. These activities have led to the clearance of a large part of the forests as well as fragmentation and hence a decrease in biodiversity [26,27]. Moreover, the fragmentation of forests leads to the decline of pollinator diversity of endangered plant species found in the East Usambara Mountains such as Mesogyne insignis [28]. The study by Notø [29] showed anthropogenic disturbance in the East Usambara Mountains affecting the abundance and diversity of fruit-feeding butterflies (Lepidoptera; Nymphalidae). It is recently reported that the abundance of some Lepidoptera pollinating families at farmlands in East Usambara Mountain is significantly dwindling compared to woodland [30].
Data is required to understand how these threats affect the pollinator groups and how to manage agricultural and wild landscapes to secure pollination in such circumstances. Data is needed to provide information for the management and decisionmaking agencies about various pollinator groups. These key knowledge gaps are targeted focusing on hawkmoths. Sampling took place in a key ecosystem area, the East Usambara Mountains. Forests and the agroecosystems surrounding the forests were sampled for the hawkmoth species as little information exists on the diversity, abundance, and status of this pollinator group in forests and the surrounding farmlands.
Materials and Methods
Study site
A sampling of hawkmoths was conducted in the East Usambara Mountains situated in northeastern Tanzania about 40km from the town of Tanga region between 4º45′-5º20′S and 38º26’-38º48′E (Figure 1), with an altitudinal range of 130m to 1506m [24,31]. The rainfall distribution of East Usambara Mountains is bi-modal, peaking between March and May and between September and December [32]. The dry seasons are from June to August and January to March, however, precipitation occurs in all months [33]. Rainfall is greatest at higher altitudes and in the southeast of the mountains, increasing from 1,200 mm annually in the foothills to over 2,200mm at higher altitudes [34].
A total of 12 sampling sites located in 3 different habitat types including thick vegetation assemblage with canopy cover, forest edges, and farmlands were sampled between 2017 and 2019 (Figure 1). The study sites were georeferenced using Garmin eTrex 30x GPS and validated with google Maps (Table 1). The dominant crops found in the farmlands were Camellia sinensis, Elettaria spp., Amomum spp., Cinnamomum spp., Saccharum spp., Musa spp., Piper nigrum, Artocarpus heterophyllus, Syzygium aromaticum, and Dioscorea spp. The dominant vegetation compositions in canopy cover were tree species such as Cephalosphaera usambarensis, Leptonychia Usambarensis, Allanblackia stuhlmannii, and Annickia kummeriae. Meanwhile, at the edges of the forest, the dominant vegetations were grassy verges such as Cynodon spp., Pennisetum purpureum, Lantana camara, Clidemia hirta, and tree species such as Eucalyptus spp., and Maesopsis eminii.
Hawkmoth sampling
Sampling took place for the period of 60 days during long rain (April-June) and short rain (November-December). Hawkmoths were sampled with the help of a UV-light trap consisting of 250W mercury bulbs. Lamps were connected to a 250V generator to ensure stable voltage output. Lamps were placed 1.5m above ground in front of a 3 x 2m white sheet which was attached vertically to two wooden poles. This installation was placed at each site with a white sheet facing only the particular sampling site. The techniques used to sample hawkmoths almost resemble those described in Kingstone & Nummelin [35], Ferro & Romanowski [36], and Lourido et al. [37]. All three habitat types were surveyed between 18:00 and 24:00 hours, 5 days each. Hawkmoths attracted to the light trap were captured by direct handling or with an insect net. Sampled specimens were killed and then placed in envelopes for later preparation and species identification in the Biology laboratory at the National Museum of Tanzania. Identifications were made to species level from keys, illustrations, and guides [4,38,39].
Statistical analysis
Species diversity was calculated using Shannon-Wiener diversity index (H’) [40,41]. The Shannon-Wiener diversity index is given by the formula:
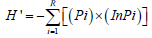
While 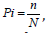 Where Pi is proportional to species ith relative to the total number of species R. n is the number of individuals in a
species, and N is the total number of individuals in a sample.
Where Pi is proportional to species ith relative to the total number of species R. n is the number of individuals in a
species, and N is the total number of individuals in a sample.


Pielou’s evenness index was used to measure species evenness of the hawkmoth species in a habitats type [42] Pielou’s evenness was computed using the following formula:
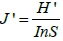
Where J 'is Pielou’s evenness index, H ' is a Shannon-Wiener index and S is species richness.
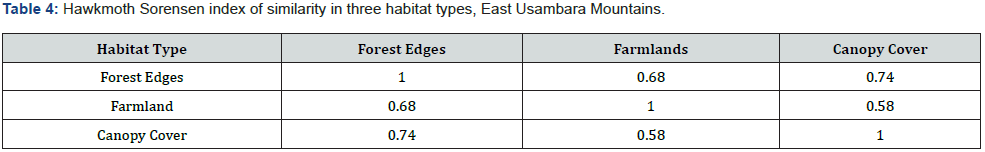
Sørensen index of similarity was used to measure the similarity of species communities among pairs of habitats [43]. Sørensen index of similarity was calculated using the following equation:
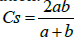
Where CS explains the coefficient of similarity, a is the number of species found in habitat A; b is the number of species present in habitat B and ab is the number of species shared by the two habitats.
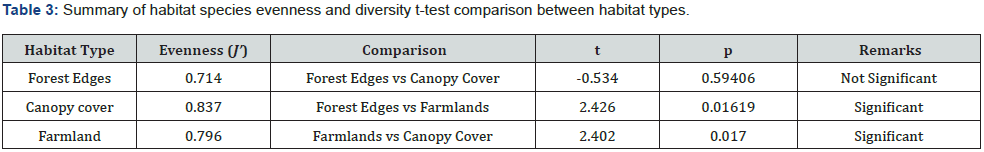
Shapiro-Wilk normality test was used to test the normality of the data by which the normal distribution of the data was verified (p > 0.05). The abundance of hawkmoths among habitats was analyzed by One-way ANOVA test. Comparisons of abundance between pairs of habitats were conducted by Dunn’s post hoc test. A special t-test (diversity t-test) was used to compare the species diversity between pairs of habitats. Paleontological Statistics (PAST) version 3.20 was used for all mentioned above analyses [44]. The statistical significance level for accepting all null hypotheses was 5% (α = 0.05). Results were summarized using tables and graphic presentations.
Results
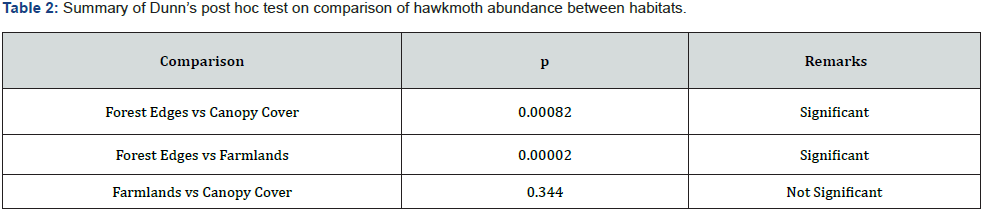
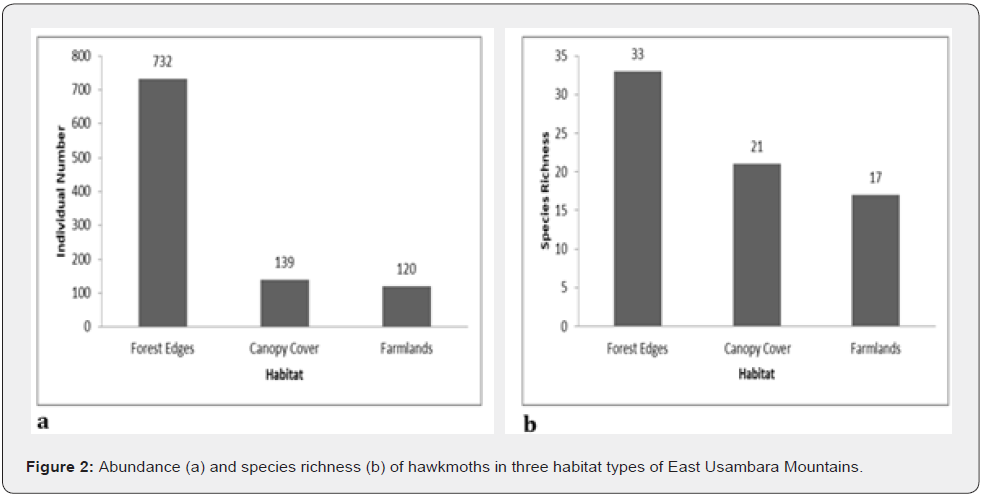
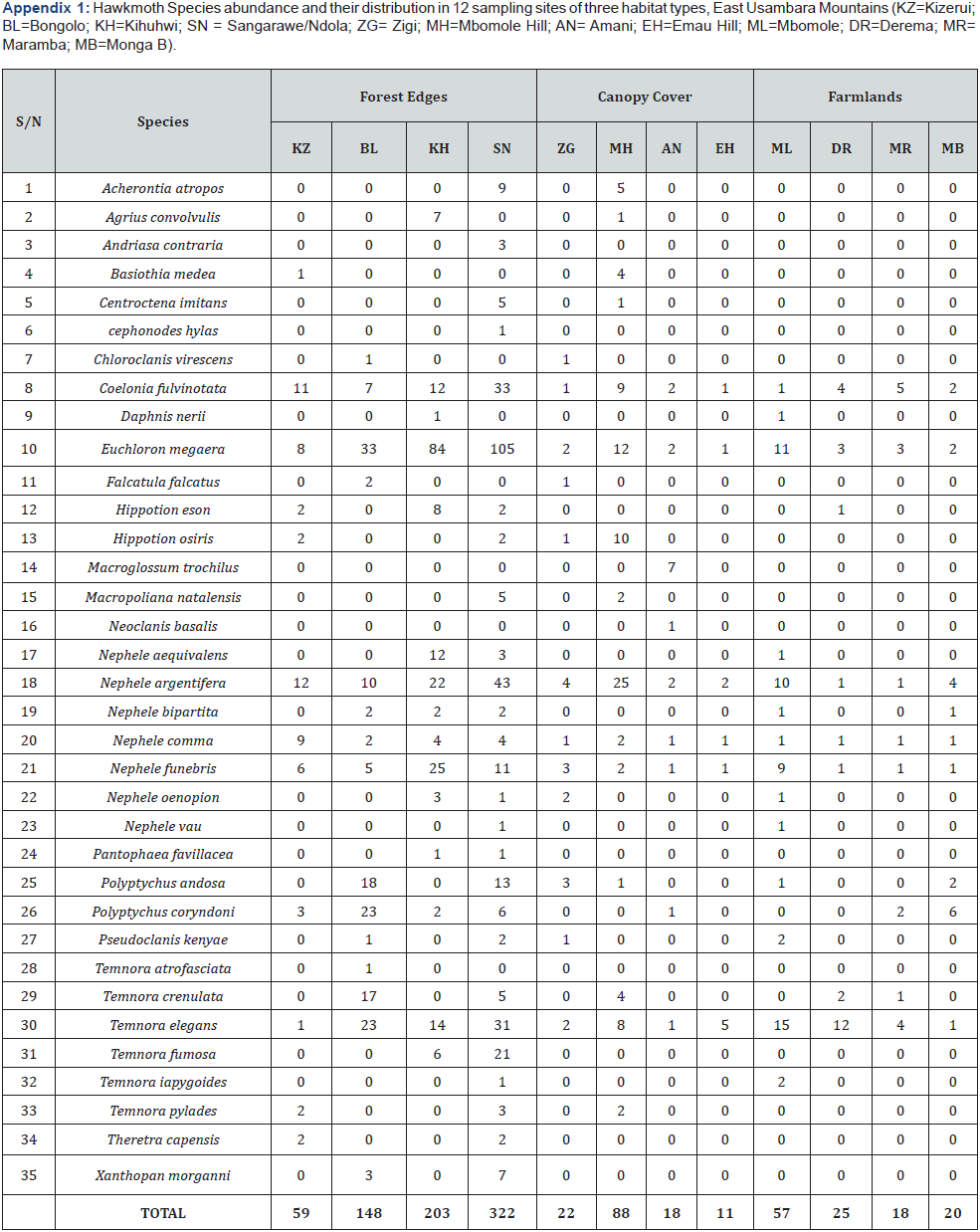
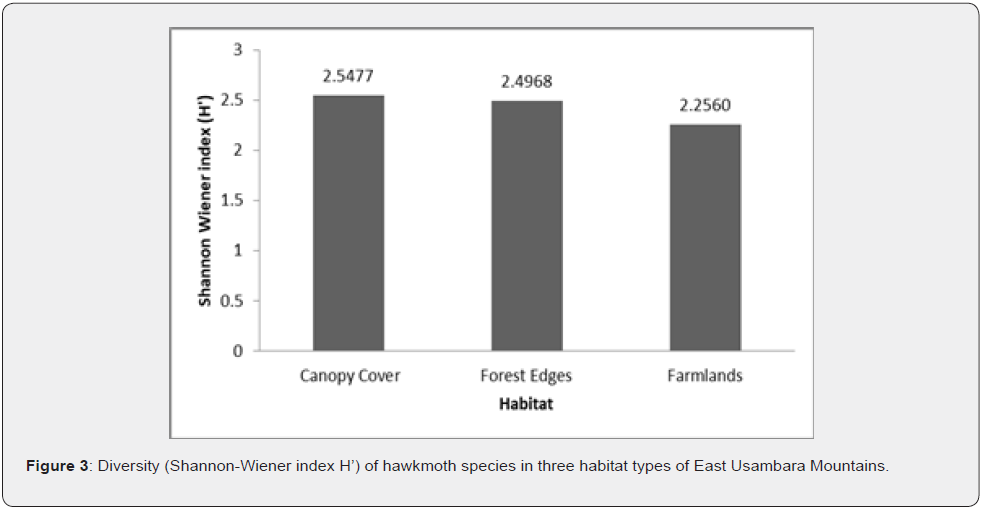
In total, 991 hawkmoth individuals were collected belonging to 35 species from all sites in three habitat types (Appendix 1). The abundance and species richness of Sphingids was highest in forest edges, followed by closed canopy cover, and lowest in farmlands (Figure 2). The differences in abundance among habitats were statistically significant (One-way ANOVA test, F = 5.531, p= 0.0052). Abundance comparisons between pairs of habitats to check where statistically significant differences exist by using Dunn’s post hoc test shown in Table 2 below:
Discussion
Hawkmoth species sampled in the East Usambara Mountains by this study represent about 63% of all Sphingid species sampled on the lower slopes of the Uluguru Mountains located in the eastern part of Tanzania [35]. Moreover, this study includes about 21% of all Sphingid species known from Tanzania [38]. The most common species were Euchloron Megaera, Coelonia fulvinotata, Nephele argentifera, Nephele comma, and Temnora elegans. 4 species were the least abundant and very uncommon among sphingid species detected in all sites. These species were Centroctena imitans, Cephonodes hylas, Neoclanis basalis, and Temnora atrofasciata (Appendix 1). On the other hand, Euchloron Megaera and Nephele comma were recorded as among the most common and abundant species by Kingstone & Nummelin [35] and Kioko et al. [45] concordant with this present study. None of the Sphingid species caught were endemic to Tanzania in this study, although species such as Chaerocina livingstonensis and Macropoliana scheveni are known to be endemic to Tanzania [35,46].
The abundance and species richness of hawkmoths showed a declining trend as one moves from the forest edges through canopy cover to farmlands (Figure 2). Additionally, farmlands had the lowest Shannon Wiener index of diversity compared to those indices of the canopy cover and the forest edges (Figure 3). A study by Selemani [30] showed that farmlands in the East Usambara Mountains had lower hawkmoth abundance compared to woodland. These findings of the lowest species abundance, species richness, and species diversity regarding farmlands, indicate this habitat type experienced severe disturbance and probably from anthropogenic pressure. Furthermore, farmland disturbances have adverse impacts on both pollinators and crop pollination, and this negatively affects crop production and commodity markets [47]. On the other hand, Forest edges had a significantly higher abundance than canopy cover (Table 2). It might be due to a nectar source plant; Lantana camara, an invasive plant species which was predominant at the forest edges. This implies either declines of native nectar plants in the forest canopies or higher competition shown by invasive nectar plants against native nectar plants in pollination ecology. Hawkmoths had been observed extending their forages on Lantana camara and other introduced invasive species such as Tithonia diversifolia in various African countries such as Madagascar and Kenya [10,48]. Higher Sorensen indices of similarity between habitat types propose a wide and almost even distribution of the majority of hawkmoth species in the East Usambara Mountains recorded by this study.
Conclusion and Recommendations
The findings of this present study showed that forest edges and canopy cover were providing potential support to the diversity and abundance of hawkmoths in the East Usambara Mountains. Hence, attention should not only be given to these two forest habitat types but also to the farmlands and other habitat types to boost the diversity of various pollinator groups. Due to the higher species similarities of the hawkmoths on farmlands and forest habitat types, agroforestry should be encouraged. This is because, if the farmlands are managed well, they could be a valuable conservation asset in the difficult trade-off situation between the conservation of biodiversity and the sustainability of rural livelihood. The diversity of hawkmoths and all Lepidoptera species, in general, is directly connected to the availability of specific host plants and not every plant in a particular habitat type. Therefore, for effective and efficient conservation of this pollinator group, a study of the knowledge of hawkmoth host plants should be conducted in the East Usambara Mountains.
Acknowledgment
The authors would like to thank Huruma Mtemela, Agnes Robert and Rashid Njechele for their help with the field and laboratory works. We thank the National Museum of Tanzania (NMT) management for the support of this study. We acknowledge the Botanical Garden of Amani Nature Reserve and the Tanzania Forestry Service Agency branch of Amani Nature Reserve for allowing fieldwork to be conducted.
Funding
This study was conducted with financial support from JRS Biodiversity Foundation.
References
- Potts SG, Biesmeijer JC, Kremen C, Neumann P, Schweiger O, et al. (2010) Global pollinator declines: Trends, impacts and drivers. Trends Ecol Evol 25: 345-353.
- Ollerton J, Erenler H, Edwards M, Crockett R (2014) Extinctions of aculeate pollinators in Britain and the role of large-scale agricultural changes. Science 346(6215): 1360‑
- Macgregor CJ, Evans DM, Fox R, Pocock MJO (2016) The dark side of street lighting: impacts on moths and evidence for the disruption of nocturnal pollen transport. Global Change Biology 23(2): 697‑
- Kitching IJ, Cadiou JM (2000) Hawkmoths of the world: an annotated and illustrated revisionary checklist (Lepidoptera: Sphingidae). Cornell University Press, Ithaca.
- Eardley C, Gimmill B, Gikungu M, Kagoiya R, kinuthia W, et al. (2004) Crops browse and pollinators in Africa; An initial stock-taking African pollinators initiative. Food and Agricultural Organization, Rome.
- Martins DJ, Johnson SD (2009) Distance and quality of natural habitat influence hawkmoth pollination of cultivated papaya. International Journal of Tropical Insect Science 29(3): 114-123.
- Martins DJ, Johnson SD (2007) Hawkmoth pollination of aerangoid orchids in Kenya, with special reference to nectar sugar concentration gradients in the floral spurs. American Journal of Botany 94(4): 650‑65
- Martins DJ (2014) Our Friends the Pollinators; A hand Book of Pollinator Diversity and Conservation in East Africa. Nature Kenya, the East Africa Natural History Society, National Museums of Kenya and Museum Hill, Nairobi, p. 18.
- Johnson SD and Raguso RA (2016) The long-tongued hawkmoth pollinator niche for native and invasive plants in Africa. Annals of Botany 117(1): 25-36.
- Martin DJ and Johnson SD (2013) Interactions between hawkmoths and flowering plants in East Africa: polyphagy and evolutionary specialization in an ecological context. Biological Journal of the Linnean Society 110(1): 199-213.
- Kitching I, Rougerie R, Zwick A, Hamilton C, St Laurent R, et al. (2018) A global checklist of the Bombycoidea (Insecta: Lepidoptera). Biodiversity Data Journal 6: e22236.
- Schreiber H (1978) Dispersal centres of Sphingidae (Lepidoptera) in the neotropical region (Biogeographica, 10). Springer, Dordrecht, pp. 35-36.
- Kawahara AV, Mignault AA, Regier IC, Kitching IJ, Mitter C (2009) Phylogeny and biogeography of hawkmoths (Lepidoptera: Sphingidae) evidence from five nuclear genes. PLOS ONE 4(5): e5719.
- Johnson SD, Moré M, Amorim FW, Haber WA, Frankie GW, et al. (2016) Plant-pollinator interactions from flower to landscape; The long and the short of it; A global analysis of hawkmoth pollination niches and interaction networks. Functional ecology 31(1): 101-115.
- Miller WE (1997) Diversity and evolution of tongue length in hawkmoths (Sphingidae). Journal of the Lepidopterists’ Society 51(1): 9‑
- Marks R (2005) Native pollinators. Natural Resources Conservation Services, Wildlife Habitat Management Institute, Maryland.
- Buchmann SL, Nabhan GP (1996) The forgotten pollinators. Island Press, Washghton D.C, pp. 131-144.
- Ley EL, Buchmann S, Kauffman K, Jacobsen O, Rager M, et al. (2013) Selecting plants for pollinators; A regional guide for farmers, land managers and gardeners in the Hawaiianislands province. The Pollinator Partnership/North American Pollinator Protection Campaign, San Francisco, p. 3.
- Gullan PJ, Cranston PS (2010) The insects: An outline of Entomology. Fourth edition, Wiley-Blackwell, West Sussex, p. 3.
- Sallema AE (2015) Impact of climate change and land use on local butterfly pollinators: A case of Dar es Salaam, Tanzania. Tanzania Wildlife Research Institute (TAWIRI) Scientific Conference, Tanzania.
- Haddad N, Wagner D (2017) Persistent decline in the abundance and diversity of Lepidoptera. News of the Lepidopterists’ Society 59: 94-101.
- Young B, Auer S, Ormes M, Rapacciuolo G, Schweitzer D, et al. (2017) Are pollinating hawk moths declining in the Northeastern United States? An analysis of collection records. PLOS One 12(10).
- Hamilton AC (1989) The place and the problem. In: Hamilton AC, Bensted-Smith R (Eds.), Forest Conservation in the East Usambara Mountains, Tanzania, International Union for Conservation of Nature, Gland and Cambridge, United Kingdom, pp. 29-33.
- Beharrell NC, Fanning E, Howell KM, Staddon SC, Stanwell-Smith D (eds.) (2002) East Usambara forest monitoring: A test of procedures at Semdoe and Kwamgumi forest Reserves: East Usambara Conservation Area Management Programme Technical Paper 54. Frontier Tanzania, London, Dar es Salaam.
- Newmark WD, Senzota RBM, (2003) Power to detect trends in ecological indicators in the East Usambara Mountains Tanzania. African Journal of Ecology 41(4): 294-298.
- Yanda PZ, Shishira EK (2009) Peoples’ response to diminishing natural resource base in the Pare-Usambara area, North Eastern Tanzania. Scientific Research and Essays 4 (12): 1536-1542.
- Sylvander N (2010) Implementation of REDD+ in community forestry In the East Usambara Mountains, Tanzania - possibilities and challenges. World Wildlife Fund, Finland.
- Olotu MI, Ndangalasi HJ, Nyundo BA (2011) Effects of forest fragmation on pollination of Mesogyne insignis (Moraceae) in Amani Nature Reserve forests Tanzania. African Journal of Ecology 50: 106-116.
- Notø C (2014) Effects of anthropogenic disturbance of Afromontane Forest on fruit-feeding butterflies (Lepidoptera: Nymphalidae) in Amani Nature Reserve Tanzania. (Master Thesis), Norwegian University of Life Sciences (NMBU), Norway.
- Selemani SS (2018) Assessment of abudance and diversity of Lepidoptera pollinators of selected families in Amani Nature Researve East Usambara Mountains Tanzania. (MSc Dissertation), University of Dar es salaam (UDSM), Tanzania.
- Burgess ND, Butynski, TM, Cordeiro NJ, Doggart NH, Fjeldså J, et al. (2007) The biological importance of the Eastern Arc Mountains of Tanzania and Kenya. Biological Conservation, 134(2): 209-231.
- Griffiths CJ (1993) The geological evolution of East Africa. In: Lovett JC, Wasser SK (Eds.), Biogeography and ecology of the rain forests of eastern Africa. Cambridge University Press, Cambridge, pp. 9-22.
- Hamilton AC (1989) The climate of the East Usambaras. In: Hamilton AC, Bensted-Smith R (Eds.), Forest Conservation in the East Usambara Mountains, Tanzania, International Union for Conservation of Nature, Gland and Cambridge, pp. 97-107.
- Doody KZ, Howell KM, Fanning E (eds.) (2001) Amani Nature Reserve: A biodiversity survey: East Usambara Conservation Area Management Programme Technical Paper No. 52. Frontier Tanzania; Forestry and Beekeeping Division and Metsähallitus Consulting, Dar es Salaam, Tanzania and Vantaa, Finland, pp. 1-2
- Kingston AJ, Nummelin M (1998) Seasonality and Abundance of Sphingids in a Garden on the Lower Slopes of the Uluguru Mountains in Morogoro Township in Tanzania. Journal of East African Natural History 87(1): 213-220.
- Ferro VG, Romanowski HP (2012) Diversity and composition of tiger moths (Lepidoptera: Arctiidae) in an area of Atlantic Forest in southern Brazil: is the fauna more diverse in the grassland or in the forest? ZOOLOGIA 29(1): 7-18.
- Lourido GM, Motta C da S, Graça MB, Rafael JA (2018) Diversity patterns of hawkmoths (Lepidoptera: Sphingidae) in the canopy of an ombrophilous forest in Central Amazon, Brazil. Acta Amazonica 48(2): 117-125.
- Darge Ph (2012) Tanzania; An Exceptional Biodiversity in Africa: An Illustrated catalogue of the hawkmoths of Tanzania (Lepidoptera, Sphingidae) Volume 1.
- Carcasson RH (1976) Revised' catalogue of the African Sphingidae (Lepidoptera) with Descriptions of the East African species. National Museum, Nairobi.
- Shannon CE, Wiener W (1949) The mathematical theory of communication. University of Illinois Press, Urbana, p. 177.
- Shannon CE, Weaver W (1963) The mathematical theory of communication. University of Illinois Press, Urbana.
- Pielou EC (1966) The measurement of diversity in different types of biological collections. Journal of Theoretical Biology 13: 131-144.
- Sorensen T (1948) A method of establishing groups of equal amplitude in plant sociology based on similarity of species content and its application to analyses of the vegetation on Danish commons. Biologiske Skrifter/Kongelige Danske Videnskabernes Selskab 5: 1-34.
- Hammer Ø, Harper DAT, Rian PD (2001) PAST: paleontological statistic software package for education and data. Palaeontologia Electronica 4: 1-9.
- Kioko EN, Musyoki AM, Luanga AE, Kioko MD, Mwangi EW, Monda L (2021) Geographical and temporal distribution of hawkmoth (Lepidoptera: Sphingidae) species in Africa. Biodiversity Data Journal 9: e70912.
- Darge Ph (2008) Contribution to the biodiversity of the Albertine Rift in Tanzania: some data on the Lepidoptera Saturniidae and Sphingidae. Saturnafrica 1: 4-15.
- Kevan PG, Phillips TP (2001) The economic impacts of pollinator declines: an approach to assessing the consequences. Conservation Ecology 5(1): 8.
- Wasserthal LT (1997) The pollinators of the Malagasy star orchids Angraecum sesquipedale, A. sororium and compactum and the evolution of extremely long spurs by pollinator shift. Botanica Acta 110(5): 343-359.






























