Assessing Exposure of Small-Bodied Fish to Polycyclic Aromatic Compounds from Crude Oil Spills using Biliary Metabolites
Jamie Dearnley1, Lisa Peters1, Gregg Tomy1, Lauren Timlick2 and Vince Palace1,2*
1 University of Manitoba, Canada
2 International Institute for Sustainable Development – Experimental Lakes Area (IISD-ELA), Canada
Submission: May 02, 2022; Published: June 23, 2022
*Corresponding author: Vince Palace, University of Manitoba & International Institute for Sustainable Development – Experimental Lakes Area (IISD-ELA), Winnipeg, Canada
How to cite this article: Jamie D, Lisa P, Gregg T, Lauren T, Vince P. Assessing Exposure of Small-Bodied Fish to Polycyclic Aromatic Compounds from 02 Crude Oil Spills using Biliary Metabolites. Int J Environ Sci Nat Res. 2022; 30(4): 556293. DOI: 10.19080/IJESNR.2022.30.556293
Abstract
Knowledge gaps pertaining to the remediation of freshwater lakes impacted by oil spills have persisted despite recent record highs for oil production and transportation across vulnerable regions in North America. The Freshwater Oil Spill Remediation Study (FOReSt), conducted at the IISD-Experimental Lakes Area in Canada, is focusing on the efficacy of minimally invasive methods for remediating oil spills in freshwater boreal lakes. In this study, the impacts and remediation of diluted bitumen (dilbit) and conventional heavy crude oil (CHV) spills were investigated. Two common species of small-bodied fish, fathead minnows (Pimephales promelas) and finescale dace (Chrosomus neogaeus), were used to assess exposure to oil constituents in enclosed shoreline ecosystems impacted by model oil spills. Exposure to polycyclic aromatic compounds (PACs), the most toxicologically relevant compounds in oil, were assessed in fish by measuring metabolite concentrations in bile. Finescale dace and fathead minnows from oil treated enclosures each had biliary pyrene metabolite concentrations that were positively correlated with pyrene concentrations in the water of the enclosures. Three months after the initial spills, fish in the enclosure receiving dilbit had significantly more biliary PAC metabolites than fish in reference enclosures that did not receive oil. PAC exposure in experimental fish was not always related to aqueous PAC concentrations, which may have implications for future remediation efforts and assessments of future freshwater oil spills using bile metabolites.
Keywords: Biliary metabolites; Finescale dace; Ecological recovery; Heavy crude oil spill; Shoreline ecosystems; Polycyclic aromatic compounds; Baited minnow traps
Introduction
Additional information is required regarding the efficacy of methods for remediation, reclamation, and restoration of freshwater habitats affected by oil spills [1]. In response, the IISD-Experimental Lakes Area, in Northwest Ontario, Canada, initiated the Freshwater Oil Spill Remediation Study (FOReSt) to examine the utility of using minimally invasive remediation methods to degrade oil that is not removed by initial oil spill cleanup methods (i.e., residual oil). In year 1 of the study shoreline enclosures were treated with model spills of crude oil and, following a primary cleanup, were remediated using a monitored natural recovery (MNR). Unlike laboratory-based studies, lake enclosures encompass entire model ecosystems while still allowing for a controlled experimental environment. This study will help inform remediation efforts of shorelines affected by future unintentional oil releases.
Determining exposure and recovery of oil spill impacted sites is challenging. While chemical recovery can be readily determined, biological and ecological recovery is more difficult to assess. Measuring biliary polycyclic aromatic compound (PAC) metabolites in fish is a powerful tool for assessing short-term exposure and metabolism of oil constituents [2,3]. PACs are the compounds of greatest concern with respect to toxicity of oil to fish. In fish, PACs are generally metabolized by enzymes of the cytochrome P4501A family [4]. These enzymes are found in several tissues of fish but are at their highest concentration in the liver. As a result, many PAC metabolites are excreted and accumulated in the gallbladder and can be quantified in bile. Numerous studies have linked biliary PAC concentrations to short-term PAC exposure and they are a commonly used biomarker in environmental monitoring and laboratory studies [3-5]. In the present study biliary metabolite concentrations in fish were used to assess the efficacy of a remediation method on different oil products and to delineate and to delineate the routes of PAC exposure to fish using parent PAC concentrations in enclosure water and sediment.
Small-bodied fish are especially useful for monitoring studies because they tend to have small home ranges, exhibit high site fidelity and feed within localized foodwebs. They are often abundant and many have widespread geographical distributions, making them even more useful for comparative studies. Two small-bodied fish species, fathead minnows (Pimephales promelas) and finescale dace (Chrosomus neogaeus) were used as exposure indicators for the current study. The species are widely distributed across North America and are likely to be found at a high proportion of potential spill sites in the continent, especially those in the northern latitudes [6,7]. Dose-dependent responses to PACs have previously been demonstrated in fathead minnows [8].
In this study, four shoreline enclosures were constructed and stocked with fathead minnows. Two enclosures were unoiled and used as references, one received a model diluted bitumen (dilbit) spill, and one received a model conventional heavy crude oil spill. Primary clean-up of surface oil was initiated 72 hours after oil introduction to reflect realistic response times and the sole secondary remediation activity was MNR. MNR relies upon microbial break down of petrochemicals following initial clean-up of surface oil, is utilized in situations where human intervention would negatively impact the spill location more than the oil itself or in ecosystems where natural remediation is expected to proceed quickly, and is best accompanied by increased monitoring and recovery trajectory modeling [1,9]. Fish were retrieved at two different time points to assess ongoing PAC exposure using bile metabolites, the concentrations of which were compared between the enclosures and to the parent PAC concentrations in sediment and water. Bile metabolites from both time collection periods indicated ongoing exposure to PACs. This study’s approach mirrors the historic and ongoing use of biliary PAC metabolites in assessing exposure from freshwater and marine oil spills globally.
Methods and Materials
Location and enclosures
The study setting and shoreline enclosures have previously been described [10]. Briefly, the study was conducted at the International Institute for Sustainable Development-Experimental Lakes Area (IISD-ELA) in the boreal forest of Northwest Ontario, Canada. IISD-ELA encompasses 58 small lakes and their watersheds, which are set aside for scientific research. The region is sparsely populated so lakes are largely unaffected by anthropogenic impacts and are representative of pristine boreal lakes at-risk for oil spills across the country. Experimental work took place on Lake 260, a hydrologically isolated bedrock lake with a maximum depth of 14.4m, a volume of 1.764 × 106m3, and a surface area of 34 hectares [11].

This study commenced in spring 2018 with two enclosures (Curry Industries, Winnipeg, Canada) deployed on sand/organic shorelines in Lake 260. Each enclosure extended 10m from the shoreline into the lake with a width of 5m (Figure 1). The enclosures were comprised of floating collars from which impermeable polypropylene curtains extended down to the lake bottom where they were sealed to the underlying sediments using sandbags to prevent the exchange of water between the cells or the general lake environment. Each enclosure was divided down the middle by another curtain to create two side by side enclosure cells, each 2.5m wide × 10m long (note: from this point forward, any use of the word “enclosure” refers to these half-enclosure cells). A skirt protruding from the end of the floating collar extended 5m onto the shoreline and was also pinned into place with sandbags. Each of the enclosures was protected with fine grade mesh to prevent unwanted interactions with native fauna beyond the scope of the experiment.
Oil weathering and application
Two oil products were used in the study: Western Canadian Select (CHV) and Cold Lake Blend (dilbit). To simulate spill scenarios and weathering of oil prior to deposition on a shoreline, both oil products were weathered over 25cm of lake water (220L) in 1m diameter stainless steel evaporation pans. Seven (7kg) of CHV and dilbit were weathered for 36h in separate evaporation pans after which the oils were retrieved with stainless steel spoons and placed into glass bottles for transport to the enclosure site. Details regarding the chemical characterization of the pre-weathered and weathered oils are provided in Palace et al. (2021). The oil was weathered from June 14-16 and application to enclosures occurred on June 16, 2018. With a weathering period of this duration, the partial-to-total evaporation from the oil of volatile compounds such as benzene, alkyl-benzenes, toluene, xylenes, 2-3 ring PAHs and <C17 alkanes can be expected, leading to an increased viscosity of the product [1,12,13].
Each oil product was to the surface of the water in its designated enclosure, approximately 1 meter approximately 1 meter from the shoreline and evenly distributed over the full width of the enclosure. In total, 1460g of CHV and 1563g of dilbit were applied to their respective enclosures. In accordance with realistic response times to oil spill scenarios, no remediation action was taken for 72 hours. After this time had elapsed, free floating oil was recovered from the water surface using oleophilic sorbent pads (Spill Ninja Oil Only Pads, MEP Brothers, Winnipeg, Canada). No attempt was made to remove oil from vegetation, the enclosure walls or from shoreline substrates. Following the sorbent pad cleaning, monitored natural recovery (MNR) was applied as the sole remediation method to address the residual oil [9]. The enclosures were monitored for 3 months, and the study concluded when oiled shoreline substrates were removed and the mesocosms were dismantled fifteen weeks later, on October 4.
Water and sediment sampling and PAC testing
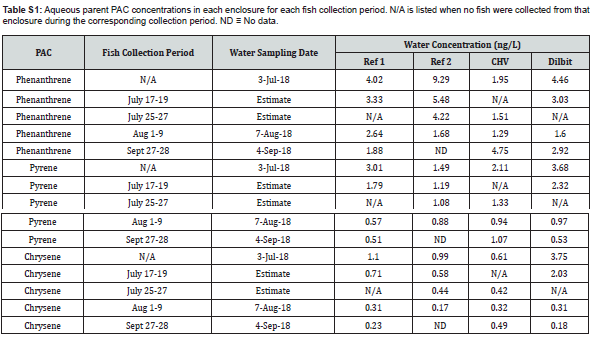
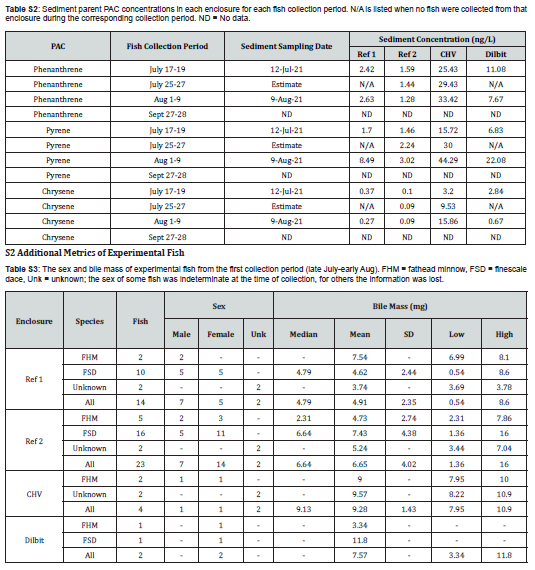
Water was collected for Water was collected for quantitation of alkanes, PACs, and alkylated-PACs 3 days prior to the introduction of oil to determine baselines, and then on days 1, 2, 3, 5, 9, 13, 18, 53, and 80 after oil application. Water samples were pumped from a depth of approximately 10cm, three meters from the shoreline. Samples were collected in glass amber bottles with head space minimized and stored at 4°C prior to transport to Winnipeg where extraction occurred within seven days of collection. Surface grab samples of sediment and soil were collected monthly, stored in amber glass bottles at -80°C until analysis, and analyzed for the same oil constituents as the water. PAC concentrations were measured in water and sediment extracts using GC-MS/MS at the Centre for Oil and Gas Research and Development in Winnipeg as described by Idowu et al. (2017).
Fish stocking, recovery, and gall bladder collection
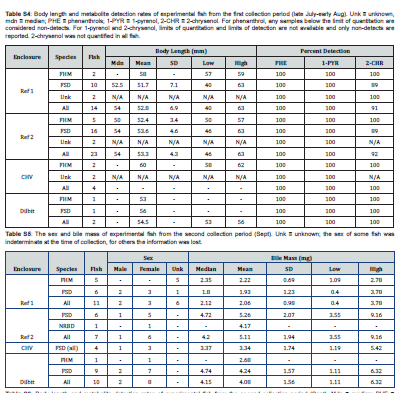
Baited minnow traps were used to remove any native fish trapped inside the enclosures during their installation. Prior to oil application, each enclosure was stocked with 12 fathead minnows (6 male, 6 female) captured from Lake 260. Fish were anesthetized with pH buffered MS-222 (pH=7.0) (0.1g/L), weighed and measured, had their sex determined, and their caudal fin was clipped as an identifying mark. They were allowed to recover in fresh lake water before being introduced into their randomly assigned enclosure. The fish were released into their enclosures in Lake 260 on the day of oil introduction. A first round of fish harvesting occurred July 17–Aug 9, beginning one month after oil introduction. Fish were collected using baited minnow traps set overnight inside the enclosures. Recovered fish were transported to the on-site laboratory (~6km drive) in oxygen enriched lake water, where they were sacrificed using an overdose of pH buffered MS-222 (0.4g/L), measured, weighed, and had their gall bladders excised. Gall bladders were stored on dry ice in 0.5ml Eppendorf tubes and transported back to Winnipeg where they were stored in the dark at -80°C with exposure to light minimized during transportation. Following this first collection period, the enclosures were restocked with fish in the same method as they were in June. A final round of fish harvesting occurred September 11-28 prior to the decommissioning of the mesocosms.
Preparation of bile samples
Preparation and HPLC-F analysis of bile followed methods of Jonsson et al. [14] and Grung et al. [15], each acknowledging Krahn et al. [16] as providing an initial analytical framework. Due to the small size of each gallbladder, bile was determined by weight. For analysis, gallbladders were allowed to thaw and were weighed on an analytical balance (0.0001g) while still in the microcentrifuge tube used for storage. Once weighed, each microcentrifuge tube had 50μL of 0.5% ascorbic acid buffer (pH: 5.0) added, the contents were vortexed, and then centrifuged for 5min at 9,600g. Gallbladders were then minced within their tubes to ensure full extraction of bile and the resulting bile/buffer mixture was transferred to a 1.5mL microcentrifuge tube containing 5μL of β-glucuronidase solution to liberate conjugated forms of PAC metabolites. The samples were vortexed and incubated at 37°C in the dark for 2-5 hours. Following incubation, 50μL of methanol containing 0.5% ascorbic acid was added, the tubes were briefly vortexed and then centrifuged for 5min at 9,600 g with the supernatant analyzed. Gallbladders that appeared to have larger volumes of bile (>10μL) were further diluted with additional buffer and 0.5% ascorbic acid methanol (in a 1:1 ratio). During incubation, tubes used for gall bladder storage were dried and re-weighed to determine the mass of the empty gallbladders and to allow calculation of bile mass.
HPLC-F analysis
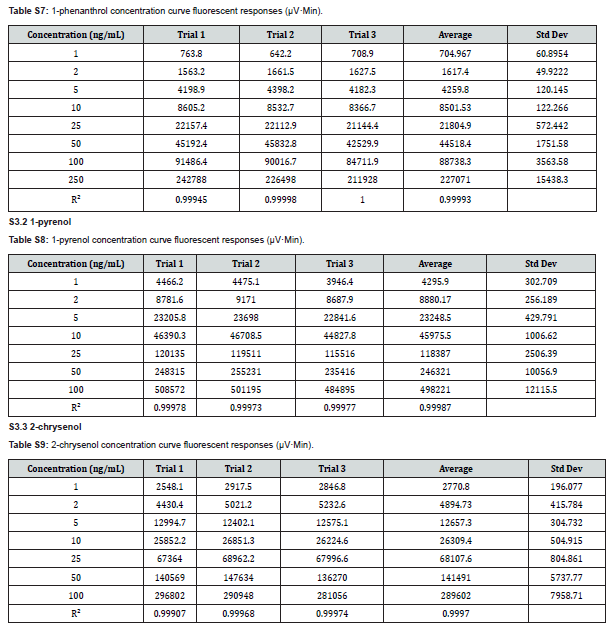
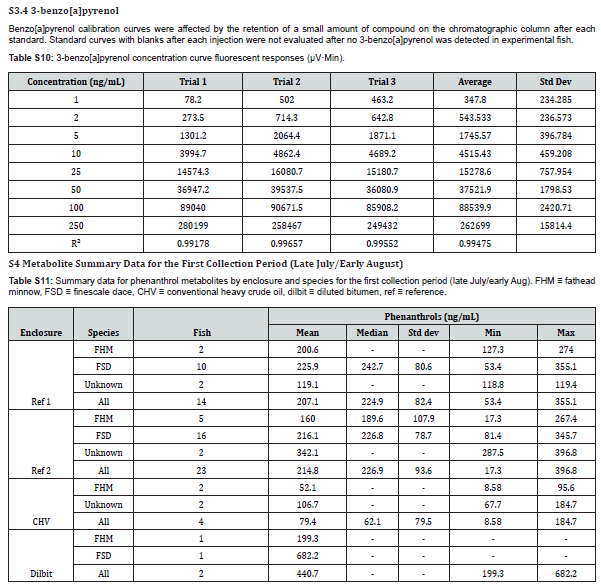
The HPLC system consisted of a Varian ProStar 410 Autosampler, Varian ProStar 210 pumps, ZORBAX RR Eclipse XDB-C18, 80Å, 2.1 x 50mm, 3.5µm column, and a Varian ProStar 363 fluorescence detector. Optimum excitation/emission (ex/em) wavelength pairs for each analyte were determined by direct injection into the fluorescence detector with a syringe pump while scanning through a range of emission or excitation wavelengths. The optimal ex/em pairs used were 275/361nm for 2-naphthol, 300/383nm for 1-phenanthrol, 340/384nm for 1-pyrenol, 265/380nm for 2-chrysenol, and 381/435nm for 3-benzo[a]pyrenol. Ultrapure water (A) and methanol (B) comprised the mobile phase for the elution. The elution profile was as follows: 0-1 minute, isocratic 38% B; 1-35 min, 38-95% B linear gradient; 35-37min, isocratic 95% B; 37-45min, 95-38% B linear gradient; 45-50min, isocratic 38% B. The flow rate was 0.30mL/min. The chromatography column was held at 40°C during analysis and the autosampler delivered 10μL for each injection.
Calculations
The concentrations of 1-pyrenol were calculated from an average response factor obtained from a 10ng/mL mixed standard containing all analytes run at the beginning, middle, and end of each sample run. The concentration of 2-chrysenol was calculated from the recovery of the metabolite in a reference sample spiked to 10ng/mL with each analyte. In some samples, run on a newer chromatographic column, 2-chrysenol could not be quantified in experimental samples due to a changed elution pattern and interference. In these samples, a consistent peak was observed 30 seconds after the 2-chrysenol peak. This was presumed to be a different isomer of chrysenol, was dubbed x-chrysenol, and was quantified in the same manner as 2-chrysenol. The concentration of phenanthrols was estimated based on the average response factor of 1-phenanthrol in the 10ng/mL mixed standard. When other isomers were detected, the response factor of 1-phenanthrol was corrected with the relative response factor of the isomer in question. When isomers coeluted, their concentrations were estimated based on the relative amount of each co-eluting isomer in fully resolved samples. For calculations, the density of bile was assumed to be 1.00g/mL, which is slightly higher than the 0.975g/mL reported for sheepshead minnow (Cyprinodon Variegatus) [14].
Statistical analysis
Statistical tests were performed using RStudio software with statistical significance accepted at p < 0.05. The parametric least squares regression and Welch’s 2-sided t-test were run as appropriate, with the Shapiro-Wilk test performed as necessary to assess the normality of data for t-tests. The assumptions of normality and homogeneity of variance were confirmed graphically using histograms and quantile-quantile plots of the residuals and a heteroscedasticity plot of the residuals, respectively. Metabolite data was natural log-transformed as necessary to meet the assumptions of the models and achieve the best possible fit. When group comparisons were required, the assumptions of the parametric ANOVA were not met by the data of this study, thus the non-parametric Kruskal-Wallis test was run instead in conjunction with a post-hoc Holm corrected Mann-Whitney-Wilcoxon test.
Results
A total of only 10 of 96 fin-clipped fish were recovered from the experimental enclosures for the combined retrieval periods. However, many unexpected finescale dace were also captured. These fish were either not captured during the original removals, infiltrated the enclosures after construction or hatched from eggs laid prior to enclosure construction. Regardless of their origin, all fish with a body length > 37cm were assumed to be resident in the enclosures and were subject to biliary metabolite analysis, regardless of species (see Supplemental Information for body sizes). In total, bile from 75 individual fish was analyzed, though statistical analysis was hampered by uneven recoveries among the different enclosures and time periods. Nevertheless, significant differences in PAC metabolite concentrations were observed among different enclosures and aqueous PAC concentrations were found to predict fish exposure.
Method validation
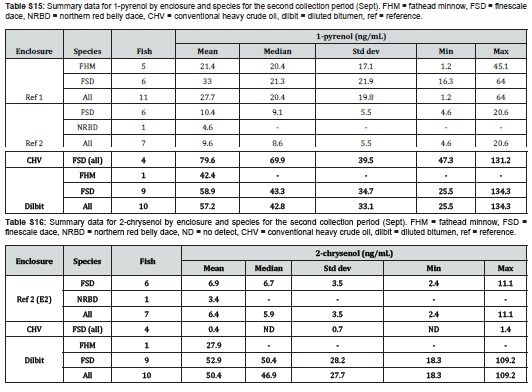
The analytic method for PAC metabolites in bile was validated according to Eurachem guidelines [17]. The determined limits of detection (LOD) and limits of quantitation (LOQ) are shown in Table 1. Both were obtained from replicate measurements of each analyte in reference material (archived fish bile from Lake Winnipeg) after spiked reagent blanks yielded poor, unrepresentative recoveries in comparison. A high concentration of 1-pyrenol and non-detect of 2-chrysenol in the reference material prevented accurate determination of their LOD and LOQ. For each analyte, linearity was demonstrated over the range 1-250ng/mL (see Supplemental Information). The method was shown to be robust to changes in incubation times (1-8 hours) and bile volume (3-10μL; 5μL was the default volume for method development). The method was also robust to chromatography column changes (Waters XSelect HSS T3 XP Column, 100Å, 2.5µm, 2.1mm X 50mm, specifically), though slight changes in elution time of other compounds in bile samples sometimes led to interference with 2-chrysenol. While 2-naphthol standards demonstrated excellent potential for analysis, interference in bile samples precluded their quantitation in this study. Triphenylamine, often used as an internal standard in HPLC-F biliary metabolite analysis [18,19], was tested for use in this method, but demonstrated unsuitably low recoveries. In its absence, each sample run contained a sample of reference bile and a sample of reference bile spiked to achieve an additional 10ng/mL of each metabolite, which assessed metabolite recoveries and the efficiency of the enzymatic decoupling of parent metabolites.
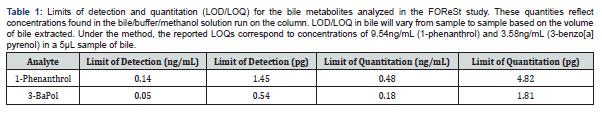
Metabolite concentrations
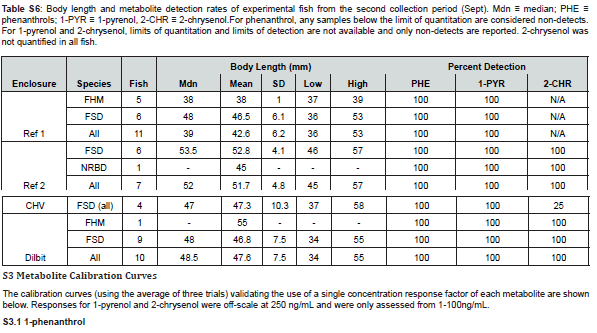
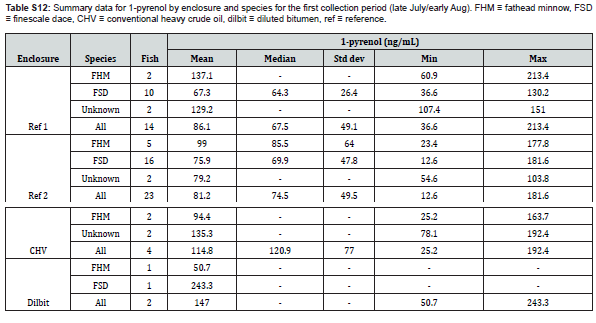
with the exception of 3-benzo[a]pyrenol, which was not reliably detected in any sample, the targeted metabolites were detected in samples, and at concentrations above the determined limits of quantitation when applicable. The fish from the two different collection periods are shown separately and the disparity in fish collections across the different enclosures are readily apparent (Figure 2 & 3). No documentation was available for several fish collected in late July/early August and these are labelled as unknown, though they are likely finescale dace or fathead minnows. Additional information regarding fish size, sex, and bile mass recoveries of experimental fish are compiled in Supplemental Information.
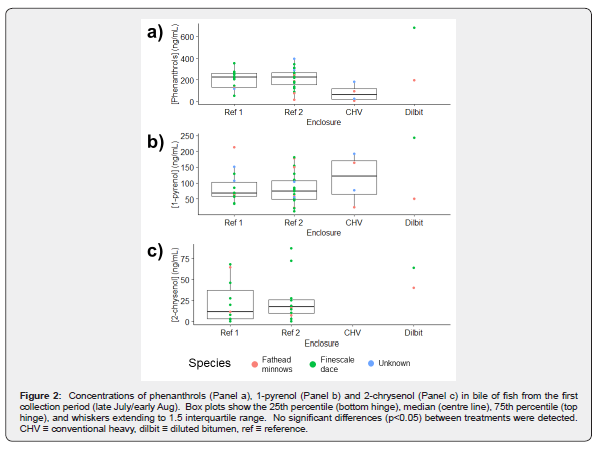
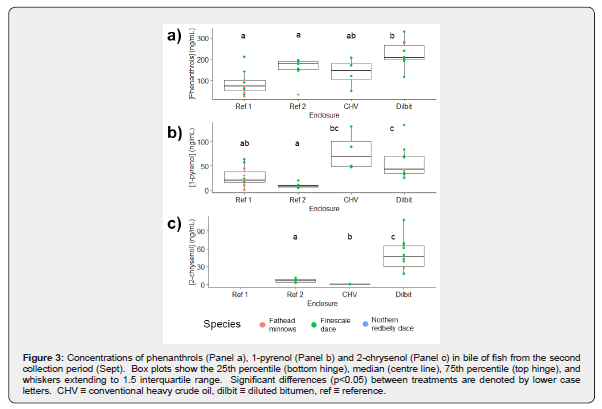
Comparisons between species
Uneven fish recoveries, with respect to both quantity and species, across the enclosures meant determining the equivalency of biliary metabolite concentrations between fathead minnows and finescale dace was of critical importance. Only Reference Enclosure #2 in late July/early Aug (5 FHM, 16 FSD) and Reference Enclosure #1 in September (5 FHM, 6 FSD) had sufficient quantities of both fish for meaningful comparisons. While no significant differences in metabolite concentrations between the two species was found, the large confidence intervals indicate the differences between the two species could be significant relative to the measured concentrations (Table 2). Thus, the two species are treated separately whenever possible. Statistical analyses using all fish are also presented, though conclusions from these models cannot be considered as robust as species-specific ones. Similarly, due to differences detected at the second sampling period, we did not pool results from fish in the two reference enclosures.
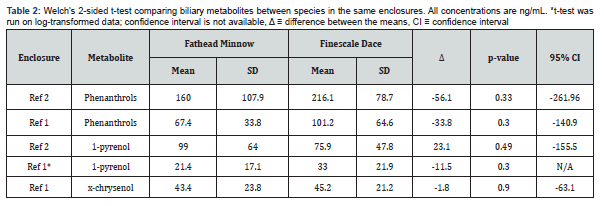
Comparisons between enclosures for late July/early August (1st Sample Period)
No significant effect of enclosure on metabolite concentrations was found for any of the analytes (phenanthrols: H(3)=6.88, p=0.08; 1-pyrenol: H(3)=1.45, p=0.69; 2-chrysenol: H(2)=2.22, p=0.33) for the first collection period. The CHV enclosure was omitted from the test for 2-chrysenol due to a lack of data.
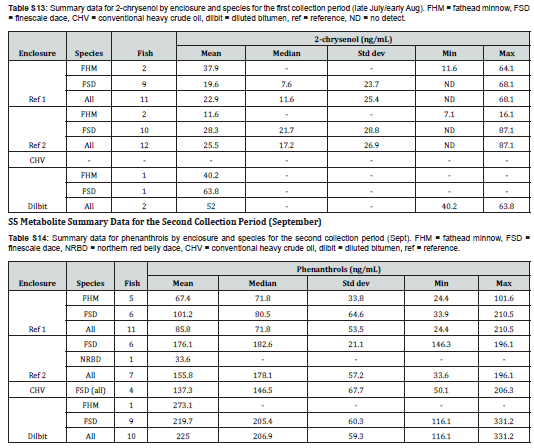
Comparisons between enclosures for September (2nd Sample Period)
The quantities of fish in the second collection period allowed for comparison among finescale dace, in addition to all fish. For finescale dace, significant effects of enclosure on metabolite concentration were found for each analyte except phenanthrols (H(3)=9.6, p=0.022). For 1-pyrenol (H(3)=15.6, p=0.001), the Dilbit and CHV enclosures were found to have significantly higher concentrations than Reference Enclosure #2 (p=0.002 and p=0.048) but not Reference Enclosure #1. For 2-chrysenol (H(2)=15.4, p<0.001) each of the three enclosures for which data was available were found to be significantly different from each other.
Significant effects of enclosure on metabolite concentration were also found for each metabolite using all species in each enclosure. For phenanthrols (H(3)=15.6, p = 0.001), pairwise comparison indicated the Dilbit enclosure fish had higher metabolite concentrations than both reference enclosures (p=0.002, p=0.034,). 1-pyrenol (H(3)=19.1, p<0.001) concentrations were found to be significantly different between the Dilbit enclosure and the two reference enclosures (p=0.040, p<0.001) as well as between the CHV enclosure and Reference Enclosure #2 (p=0.030). Differences between the two references (p=0.053) and the CHV enclosure and Reference Enclosure #1 (p=0.053) were marginally significant. For 2-chrysenol, Reference Enclosure #1 was not included in the analysis due to a lack of data however metabolite concentrations were affected by enclosure (H(2)=17.0, p<0.001) in the other three. Metabolite concentrations of 2-chrysenol were found to be significantly different in each of the three enclosures.
Effects of sample time, sex and fish size on bile metabolites
There were few significant differences in concentrations of metabolites in finescale dace between the first and second sample times. While differences were observed for phenanthrol in Reference Enclosure #1 and for 1-pyrenol in both reference enclosures, no differences were observed for the CHV enclosure, likely as a consequence of small sample size (n=4 for both collection periods). The Dilbit enclosure was omitted from the analysis due to lack of fish in July/Aug, and 2-chrysenol was only available for both time periods for Reference Enclosure #2.
Only two enclosures, both references from the first sampling period, had a sufficient quantity and sex distribution of one species of fish to assess for differences in metabolite concentrations between sex. In Reference Enclosure #1, no differences between sex were found for any of the metabolites (p=0.265-0.918). In Reference Enclosure #2, no differences between sex were found for 2-chrysenol (p=0.738), however male finescale dace (M=282.1ng/mL) were found to have higher phenanthrol concentrations than females (M=186.1ng/mL, p=0.002). Significant differences were also found between males (M=121.7ng/mL) and females (M=55.1 ng/mL) for 1-pyrenol (p=0.026).
Finescale dace in Reference Enclosure #1 and Reference Enclosure #2 from the first collection period and in the Dilbit enclosure from the second collection period were investigated for a relationship between metabolite concentration and fish size (body length). For Reference Enclosure #1 in July/August, the regression was not significant for any of the metabolites (phenanthrol: R² = 0.044, F(1,8)=0.61, p=0.456; 1-pyrenol: R² <0.001, F(1,8)<0.01, p=0.965; 2-chrysenol: R² = 0.029, F(1,7)=0.78, p=0.408). For Reference Enclosure #2, however, the regression was significant (R² = 0.294, F(1,14)=7.24, p=0.018), as body size did predict phenanthrol metabolite concentration (β=-10.1±8.0 (95% CI), p=0.018), with smaller fish having larger metabolite concentrations. This significance was not observed for 1-pyrenol (R² = 0.096, F(1,14)=2.60, p=0.129) nor 2-chrysenol (R² = 0.058, F(1,8)=0.49, p=0.502). In the Dilbit enclosure in September a significant relationship (R² = 0.510, F(1,7)=9.3, p=0.019) was found with fish size predicting phenanthrol concentration (β=6.1±4.7, p=0.019). Contrary to Reference Enclosure #2 in July/August, here phenanthrol concentration increased with fish size. No such relationship was found for 1-pyrenol (R² = 0.056, F(1,7)=0.58, p=0.472) nor 2-chrysenol (R² = 0.075, F(1,7)=1.65, p=0.240).
Metabolite concentrations and sediment and water PAC concentrations
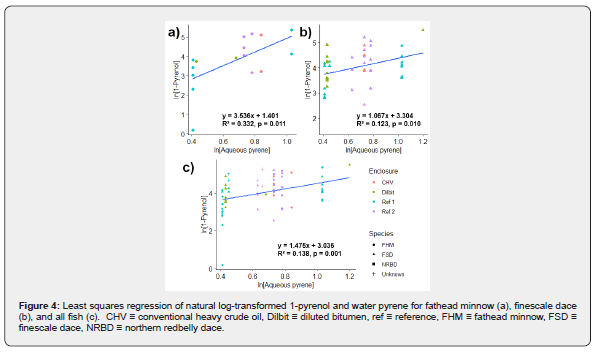
Least square regressions were performed to probe the relationship between water PAC concentrations and biliary metabolite concentrations across each of the enclosures. Water was collected monthly, though sample collection times did not always overlap with fish collections (see Supplemental Information). For fish collected within a week of a water sampling date, concentrations from that date were used in the analysis. Water concentrations were estimated for fish collected outside of this time range by interpolating between the two most recent sampling dates. Water PAC concentrations in September were an exception, as fish were caught 3 weeks after water sampling, but no later data was available for interpolation. No water data was available for Reference Enclosure #2 fish in September and these fish were omitted from regressions. Separate regressions were run for each of finescale dace, fathead minnows, and all fish. Sediment PAC concentration data (see Supplemental Information) was only available for the first collection period, but models generated with it did not sufficiently meet the assumptions of a regression and have been omitted.
Concentrations of 1-pyrenol in finescale dace, fathead minnows, and all fish combined were significantly and positively predicted by aqueous pyrene concentrations (Figure 4). Aqueous concentrations of phenanthrene and chrysene did not significantly predict biliary concentrations for their respective metabolites.
Discussion
Two and a half months after model oil spills and subsequent remediation, biliary PAC metabolite concentrations in small-bodied fish exposed to model oil spills in shoreline enclosures demonstrated a significant increase in PAC exposure compared to fish in reference enclosures. This was especially noticeable for fish exposed to dilbit, which had significantly higher concentrations of all three metabolites compared to reference fish. Aqueous pyrene concentrations predicted exposure in experimental fish, regardless of species. Statistical analysis in the study, especially for the first collection period, was hampered by low fish recoveries from oiled enclosures.
The correlation between aqueous pyrene and biliary pyrene concentrations in this study is particularly notable in assessing spill remediation efforts and is also significant within the wider literature. Studies of biliary metabolites in wild fish, when using targeted analyses, often focus on 1-pyrenol as this compound accounted for 76% of biliary PAC metabolites in a study of European eels (Anguilla) [20,21]. Our results validate 1-pyrenol’s use as a biomarker in wild fathead minnows and finescale dace, thereby permitting comparison of the concentrations of metabolites in the experimental fish to those measured in other studies of PAC pollution or future oil spills. If aqueous concentrations of pyrene can be lowered in an environment following remediation, whether in response to an oil spill or commercial pollution, small-bodied fish should experience less uptake and less exposure. This has important implications for fish health. Pyrene itself has been linked to toxic outcomes in fish and threshold biliary 1-pyrenol concentrations have been identified for several species of predatory marine fish above which chronic health effects may occur [20,22]. All fish in this study were below the lowest of these threshold concentrations, 483 ng/g bile, which was identified for Atlantic Cod (Gadus morhua). The relationship between aqueous pyrene and its biliary metabolite concentrations may also be characteristic of other PACs in crude oil, particularly alkylated PACs, though metabolites of these compounds were not quantified in this study. Nevertheless, alkylated PACs are characteristic of petrogenic PAC sources and are often more toxic than unalkylated analogues [23,24]. In the case of fathead minnows, the pyrene model explained much of the variance in the population (R²=0.332). The models were less predictive in finescale dace and in all species combined. The difference may be an artifact of the sample size; a smaller population of fathead minnows (n=15) potentially allows for less variation compared to the larger population of finescale dace (n=45) across all enclosures. Alternatively, these differences may stem from species-species differences in behaviour, metabolism, and/or habitat preferences. Sex, size, and seasonal differences may also contribute non-PAC related variation for each species, none of which parent PAC modelling attempts to explain.
Unlike pyrene, aqueous concentrations of phenanthrene and chrysene did not predict biliary metabolite concentrations. This lack of relationship may be attributed to several factors. For one, aqueous exposure may simply be a more prominent route of uptake for pyrene compared to the other PACs studied. In a seven-day exposure of juvenile fathead minnows to different concentrations of water accommodated fractions of dilbit (0%, 3%, 10%, 30%), a nominal concentration range of 1.72-157.4ng/L for phenanthrene was required to observe a significant difference in phenanthrol between exposure groups [25]. This is much narrower than the range of aqueous phenanthrene concentrations in this study, though literature has also shown aqueous exposure to a mixture of PACs with a pyrene range of 0.6-6.0 ng/L was sufficient to observe significant differences in exposure for Atlantic cod (Gadus morhua) [26]. Alternatively, aqueous uptake through the gills, compared to other routes of exposure, may have been less prominent for other PACs than for pyrene in this study. Dermal uptake of PACs through sediment and ingestion through diet are alternate routes of exposure for fish [27] and parent PACs in these media were not fully quantified in this study. Lab studies have observed uptake of benzo[a]pyrene from sediment in fathead minnows and noted fish disturbance of the sediment led to higher aqueous concentrations of benzo[a]pyrene in aquaria [28]. The PACs in the dynamic ecosystem of the enclosures may also differ in their bioavailability, driven by differences in their KOW [29]. Lastly, lack of correlations between metabolite concentrations and parent PACs may simply be a product of fluctuating experimental conditions. The variance in aqueous concentrations, even within a collection period, is readily apparent (see Supplemental Information) and limited the study’s ability to assess exposure concentrations for the fish. A study with a large exposure range would be more resilient to this margin of error, but this study’s relatively narrow concentration gradient reduces its predictive usefulness. The lack of correlation between some metabolites in this study, however, is not a failure but rather an important finding. Fluctuations are part of natural systems and illustrate the difficulties presented in modelling actual spill scenario exposures. While this study has demonstrated that modelling can be an effective tool for predicting fish exposure to PACs after an oil spill, it has also shown that direct measurement can be the most reliable tool in assessing fish exposure.
Benzo[a]pyrene is a PAC of notable concern for fish health and one of its metabolites, benzo[a]pyrene-7,8-diol-9,10-epoxide, is particularly carcinogenic [30]. In experimental fish one benzo[a]pyrene metabolite, 3-benzo[a]pyrenol, was targeted but was not detected in any of the bile. This is supported by laboratory exposure of fathead minnows to benzo[a]pyrene, where Grimard et al. [8] found no significant difference between biliary concentrations of 3-benzo[a]pyrenol in fish exposed to 30ng/L benzo[a]pyrene compared with an unexposed control group. In our study, the maximum aqueous concentration of benzo[a]pyrene between the four enclosures at either collection point was <0.4ng/L.
Direct comparison of metabolite concentrations between fish in enclosures aids interpretation of the results of modelling. In September, all species of Dilbit enclosure fish were significantly more exposed to all three parent PACs than the reference enclosure fish. When only finescale dace were considered, these differences still held for pyrene and chrysene, despite aqueous PAC concentrations being nearly identical in Reference Enclosure #1 and the Dilbit enclosure during fish collection (Reference Enclosure #2 concentrations were not available). For chrysene, the Dilbit enclosure fish (all species or finescale dace) only had significantly higher metabolite concentrations than the CHV enclosure, despite aqueous parent PAC concentrations being, at minimum, double in the CHV enclosure than in the Dilbit or Reference #1 enclosures. Similarly, CHV 1-pyrenol concentrations were only significantly higher than the Reference Enclosure #2 fish, with no differences observed for phenanthrols. For 2-chrysenol, concentrations in the CHV fish were significantly lower than those of Reference Enclosure #2. This data demonstrates that while aqueous PAC concentrations can help predict some exposure to fish, other environmental factors play an important role in governing fish uptake, as evidenced by the significant differences between populations with recent exposure to nearly identical aqueous PAC concentrations in this study. Dietary inputs, which were not measured in this study, are a probable candidate to explain some of these differences. Whereas fish can quickly metabolize PAC compounds, lower trophic level organisms unable to process PACs may serve as continual sources of exposure after water concentrations return to baseline concentrations. In assessing fish exposure to PACs in the wake of an oil spill and subsequent remediation, measuring water concentrations is not enough. Though they predicted some PAC exposure here, this study indicates that fish must be sampled directly to fully assess ongoing exposure to PACs.
Recent literature contains few comparable studies of biliary metabolites in small-bodied freshwater fish to compare and contextualize the results of this study. Nevertheless, median concentrations of 1-pyrenol in oiled enclosure fish in this study were well below median values of common minnow (Phoxinus phoxinus) residing in sedimentation ponds subject to road runoff, which were greater than 1000ng/g [15].
In addition to aforementioned predictors of PAC exposure, other factors relevant to this study can influence biliary concentrations of PAC metabolites. Variables such as species, size, and sex each have the potential to contribute to variability. Previous research on biliary metabolites has identified both size and sex as potentially influencing measured concentrations. We recently reviewed two studies that reported a significant impact of sex on metabolite concentrations and another six studies that did not [3]. While most of these studies were on marine fish, no difference between sexes was found among five species of freshwater fish within the Athabasca/Slave River system [31]. Correlations, or lack thereof, between fish size are less commonly reported but have been identified in several studies [22,32]. Many studies on wild fish selectively sample their fish based on size and sex and these factors may have contributed variation to the data in the current study. While species were treated independently whenever possible, sex and size were not considered in analyses as they did not consistently demonstrate significant regressions or differences between populations in this study.
Seasonal variation in biliary PAC metabolite concentrations are frequently reported in wild fish [3] and may have been observed in this study. Significant differences were noted for several metabolites between July/Aug and September in finescale dace from the two reference enclosures. Finescale dace breed in the spring from April to June while fathead minnows breed in July and August at our study site [6,33]. Reproductive efforts may have affected the uptake and metabolism of PACs in late July/August compared to late September. Alternatively, lower parent PAC concentrations in the enclosures for the later collection period may be responsible. Significant differences in metabolite concentrations between the two collection periods may be attributable to a combination of both factors.
Due to low recaptures of tagged fish, the residence time for many experimental fish is unknown but is not expected to have influenced results. A widely cited study on sheepshead minnow suggests peak PAC metabolite concentrations are reached within 2-6 days of exposure [15,22,34]. Similarly, at PAC concentrations relevant to this study, exposed fathead minnows reached peak metabolite concentrations within 7 days [8]. Thus, it is assumed fish collected have been in the enclosures long enough to yield PAC metabolite concentrations reflective of the conditions, despite the unknown residence times.
Conclusion
Analysis of PAC metabolites in bile of small-bodied fish can provide information regarding their exposure after an oil spill, but additional work is required to fully understand routes of exposure. After remediating model oil spills in shoreline enclosures, fish were collected and analyzed for biliary PAC metabolites to assess their exposure to PACs. Between all four experimental enclosures, be they reference or oiled, aqueous pyrene concentrations significantly predicted biliary 1-pyrenol levels in both species studied. The study also found that additional factors influencing fish exposure to PACs can supersede modelling based on water and sediment concentrations of PACs and create significant differences between populations. Although each species is apt to serve as a monitoring species for PACs, simply measuring the water concentrations of PACs in the wake of an oil spill and its remediation is insufficient to assess exposure to resident minnows.
References
- Lee K, Boufadel M, Chen B, Foght J, Hodson P, et al. (2015) The Behaviour and Environmental Impacts of Crude Oil Released into Aqueous Environments. In Royal Society of Canada. Ottawa, ON.
- Beyer J, Jonsson G, Porte C, Krahn MM, Ariese F (2010) Analytical methods for determining metabolites of polycyclic aromatic hydrocarbon (PAH) pollutants in fish bile: A review. Environmental Toxicology and Pharmacology 30(3): 224-244.
- Dearnley JM, Killeen C, Davis RL, Palace VP, Tomy GT (2020) Monitoring polycyclic aromatic compounds exposure in fish using biliary metabolites. Critical Reviews in Environmental Science and Technology 52(4): 1-45.
- Collier TK, Anulacion BF, Arkoosh MR, Dietrich JP, Incardona JP, et al. (2013) Effects on Fish of Polycyclic Aromatic Hydrocarbons (PAHS) and Naphthenic Acid Exposures. In: KB Tierney, AP Farrell, CJ Brauner (Eds.), Organic Chemical Toxicology of Fishes. (1st edn), Vol. 33, pp. 195-255.
- Sarasquete C, Segner H (2000) Cytochrome P4501A CYP1A in teleostean fishes. A review of immunohistochemical studies. The Science of the Total Environment 247(2-3): 313-332.
- Gidmark NJ, Simons AM (2014) Cyprinidae: Carps and minnows. In: WLJ Melvin, BM Burr (Eds.), Freshwater Fishes of North America. Volume 1: Petromyzontidae to Catostomidae, Vol. 1, pp. 354-450.
- Wakefield N (n.d.). Pimephales promelas (fathead minnow). Centre for Agriculture and Bioscience International Invasive Species Compendium.
- Grimard C, Mangold-Döring A, Schmitz M, Alharbi H, Jones PD, et al. (2020). In vitro-in vivo and cross-life stage extrapolation of uptake and biotransformation of benzo[a]pyrene in the fathead minnow (Pimephales promelas). Aquatic Toxicology 228: 105616.
- Magar VS, Chadwick DB, Bridges TS, Fuchsman PC, Conder JM, et al. (2009) Monitored Natural Recovery at Contaminated Sediment Sites. In Environmental Security Technology Certification Program.
- Palace VP, Peters LE, Berard-Brown N, Smyth P, Shanoff T, et al. (2021) The Freshwater Oil Spill Remediation Study (FOReSt): 2018 Pilot Study at the Experimental Lakes Area, Canada. International Oil Spill Conference Proceedings 2021 (1): 667537.
- Brunskill GJ, Schindler DW (1971) Geography and Bathymetry of Selected Lake Basins, Experimental Lakes Area, Northwestern Ontario. Journal of the Fisheries Research Board of Canada 28(2): 139-155.
- Fingas M (2015) Handbook of Oil Spill Science and Technology. In Handbook of Oil Spill Science and Technology.
- Gros J, Nabi D, Wu B, Wick LY, Brussaard CPD, et al. (2014) First Day of an Oil Spill on the Open Sea: Early Mass Transfers of Hydrocarbons to Air and Water. Environmental Science & Technology 48(16): 9400-9411.
- Jonsson G, Beyer J, Wells D, Ariese F (2003) The application of HPLC-F and GC-MS to the analysis of selected hydroxy polycyclic hydrocarbons in two certified fish bile reference materials. Journal of Environmental Monitoring 5(3): 513-520.
- Grung M, Petersen K, Fjeld E, Allan I, Christensen JH, et al. (2016) PAH related effects on fish in sedimentation ponds for road runoff and potential transfer of PAHs from sediment to biota. Science of the Total Environment 566-567(0349): 1309-1317.
- Krahn MM, Burrows DG, Ylltalo GM, Brown DW, Wlgren CA, et al. (1992) Mass Spectrometric Analysis for Aromatic Compounds in Bile of Fish Sampled after the Exxon Valdez Oil Spill. Environmental Science and Technology 26(1): 116-126.
- Magnusson B, Örnemark U (2014) The Fitness for Purpose of Analytical Methods. In Eurachem Guide.
- Holth TF, Nourizadeh-Lillabadi R, Blaesbjerg M, Grung M, Holbech H, et al. (2008) Differential gene expression and biomarkers in zebrafish (Danio rerio) following exposure to produced water components. Aquatic Toxicology 90(4): 277-291.
- Nahrgang J, Brooks SJ, Evenset A, Camus L, Jonsson M, et al. (2013) Seasonal variation in biomarkers in blue mussel (Mytilus edulis), Icelandic scallop (Chlamys islandica) and Atlantic cod (Gadus morhua)-Implications for environmental monitoring in the Barents Sea. Aquatic Toxicology 127(9037): 21-35.
- Kammann U, Akcha F, Budzinski H, Burgeot T, Gubbins MJ, et al. (2017) PAH metabolites in fish bile: From the Seine estuary to Iceland. Marine Environmental Research 124: 41-45.
- Ruddock PJ, Bird DJ, McEvoy J, Peters LD (2003) Bile metabolites of polycyclic aromatic hydrocarbons (PAHs) in European eels Anguilla anguilla from United Kingdom estuaries. Science of the Total Environment 301(1-3): 105-117.
- Davies IM, Vethaak D (2012) Integrated marine environmental monitoring of chemicals and their effects (Issue November 2012).
- Abrajano TA, Yan B, O’Malley V (2013) High Molecular Weight Petrogenic and Pyrogenic Hydrocarbons in Aquatic Environments. In Treatise on Geochemistry: Second Edition (2nd edn), Vol. 11, pp. 481-509.
- Hodson PV (2017) The Toxicity to Fish Embryos of PAH in Crude and Refined Oils. Archives of Environmental Contamination and Toxicology 73(1): 12-18.
- Dearnley JM (2022) Assessing exposure of small-bodied fish to experimental crude oil spills in contained shoreline environments using biliary polycyclic aromatic compound metabolites. University of Manitoba.
- Grung M, Holth TF, Jacobsen MR, Hylland K (2009) Polycyclic aromatic hydrocarbon (PAH) metabolites in Atlantic cod exposed via water or diet to a synthetic produced water. Journal of Toxicology and Environmental Health - Part A: 72(3-4): 254-265.
- Logan DT (2007) Perspective on ecotoxicology of PAHs to fish. Human and Ecological Risk Assessment 13(2): 302-316.
- McCarthy JF, Burrus LW, Tolbert VR (2003) Bioaccumulation of benzo(a)pyrene from sediment by fathead minnows: Effects of organic content, resuspension and metabolism. Archives of Environmental Contamination and Toxicology 45(3): 364-370.
- Achten C, Andersson JT (2015) Overview of Polycyclic Aromatic Compounds (PAC). Polycyclic Aromatic Compounds 35(2-4): 177-186.
- Zhu S, Li L, Thornton C, Carvalho P, Avery BA, et al. (2008) Simultaneous determination of benzo[a]pyrene and eight of its metabolites in Fundulus heteroclitus bile using ultra-performance liquid chromatography with mass spectrometry. Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences 863(1): 141-149.
- Ohiozebau E, Tendler B, Hill A, Codling G, Kelly E, et al. (2016) Products of biotransformation of polycyclic aromatic hydrocarbons in fishes of the Athabasca/Slave river system, Canada. Environmental Geochemistry and Health 38(2): 577-591.
- Vethaak AD, Baggelaar PK, van Lieverloo JHM, Ariese F (2016) Decadal Trends in Polycyclic Aromatic Hydrocarbon (PAH) Contamination Assessed by 1-Hydroxypyrene in Fish Bile Fluid in the Netherlands: Declining in Marine Waters but Still a Concern in Estuaries. Frontiers in Marine Science 3: 215.
- Stasiak R, Cunningham GR (2006) Finescale Dace (Phoxinus neogaeus): a technical conservation assessment.
- Jonsson G, Bechmann RK, Bamber SD, Baussant T (2004) Bioconcentration, biotransformation, and elimination of polycyclic aromatic hydrocarbons in sheepshead minnows (Cyprinodon variegatus) exposed to contaminated seawater. Environmental Toxicology and Chemistry 23(6): 1538-1548.
Supplementary Information
S1 Aqueous and Sediment Parent PAC Concentrations in the Enclosures