Study of Heavy Metals on Humans, Specially Lead (PB) in Commercially Available Sambar Powder in Bengaluru, Karnataka, India
Sripathy L1*, Mohammed Rafi2, Vandana N3 and Dhananjay KP3
1Associate Professor, Department of Chemistry, Vivekananda Degree College, India
2Assistant Professor, Department of Chemistry, Vivekananda Degree College, India
3B. Sc 4th Sem, Vivekananda degree college Rajajinagar, India
Submission: March 11, 2022; Published: April 04, 2022
*Corresponding author: Sripathy L, Associate Professor, Department of Chemistry, Vivekananda Degree College, II Stage, Rajajinagar, Bengaluru, India
How to cite this article: Sripathy L, Mohammed R, Vandana N, Dhananjay K. Study of Heavy Metals on Humans, Specially Lead (PB) in Commercially 02 Available Sambar Powder in Bengaluru, Karnataka, India. Int J Environ Sci Nat Res. 2022; 30(1): 556279. DOI: 10.19080/IJESNR.2022.30.556279
Abstract
There are substantial metals in the sambar powder and based on studies, it is not just for India but elsewhere as well. The content nowadays differs from what was available a few years ago due to fast developments in the fields of industrialization, globalization. Heavy metals are put into food to increase its flavour and taste but can also make people ill. It has been found by a recent report that the concentration of powerful metals in locally available sambar powder ranges from 118-271 micrograms which is hazardous to health. Tests of kabab powder from chosen stores were gathered and tried utilizing the X-beam fluorescence (XRF) strategy. Tests were found to have Iron (Fe), Zinc (Zn), Rubidium (Rb), Strontium (Sr), Zirconium (Zr), Molybdenum (Mo), Lead (Pb) in each example. The degrees of the overwhelming metals in every example were as follows: Iron[120-525], Zinc [25-26], Rubidium[11-33], Strontium[11-41], Zirconium[13-26], Molybdenum[17-32], Lead[18]. There were major differences in these toxic concentrations as they could be up to 9-10 times than what is defined as permissible by WHO. Although other research has examined the presence of metals in sambar powder and found it to be a potentially dangerous substance, few studies conducted in Bengaluru examine the heavy metal concentration present and the dangers it holds. Tests have found some dangerous heavy metals in marked stuffed sambar powder primarily used around Bangalore. The substances of these heavy metals have been surveyed by the x-Ray fluorescent spectrometer method which lead researchers to conclude that constant exposure to these heavy metals leads to danger to health. Since these kabab powders have widespread and is normally utilized across Bangalore it might cause numerous infections like Musculoskeletal, Immunological, Neurological, Reproductive maladies [1]. The amassing of Lead in our body is generally dangerous for human development and advancement so this must be considered as a significant issue moves must be made to control these pollution at the underlying stage.
Keywords: Heavy metals; Sambar powder; Toxicity; Bengaluru
Introduction
Financially motivated adulteration of spices is a longstanding and important public health problem worldwide [1]. There is a growing concern that in some ways food fraud may be riskier than traditional threats to the food supply [2]. Today, adulteration of turmeric with lead chromate, which is vibrant yellow, is a concern in India and Bangladesh. Lead has been the main substantial metal with wide applications for a long time, because of its wide scope of modern utilization in the production of batteries, fuel added substances, pipes, shades, patches, safeguarding and so forth it is a generally expected word related and modern danger all through the world [3]. The defilement of substantial metals in individual food shifts and relies on the weighty metals presented in the development, transport handling and fortress of food. Lately, there has been a developing interest in checking weighty metal defilement in flavours. 'Flavours' can be characterized as the dry piece of plant-like roots, leaves and seeds, which give a food a specific character and pungency. The other innovative handle used to carry the food to the shopper can essentially expand the absolute weighty metal substance of the food. The flavours are being utilized as diet parts to further develop the shading, smell, palatability, and agreeableness of food. Aside from these flavours additionally contain regular parts like uncommon alkaloids, tannins or polyphenols [4]. Flavours assume a significant part in the readiness of many marked and unbranded sambar powders. the principal objective of this review was to decide the tainting of weighty metals exceptionally Lead (Pb) in monetarily utilized marked sambar powders found in our nearby business sectors. Due to the huge measure of flavours burned-through, realize the harmful metals substance in these sambar powders. Utilizing X-ray-refraction fluorescence (XRF) not really settled some poisonous substantial metals like Iron, Zinc, Rubidium, Molybdenum, Zirconium, Strontium and Lead and so forth weighty metal fixation present in these sambar powders as been contrasted and global guidelines WHO/FDA. Qualities were assessed. The investigation of weighty metals is critical as it affects human wellbeing. The fundamental consequences for humans because of these substantial metals defilement is as per the following: stroke, viral disease, iron deficiency, cancer, hyper melancholy, weakness, IQ level is decreased. So, this review should be given an essential significance to decrease the use of substantial metals in the economically utilized sambar powder.
Materials and Methodology
The samples of commercial and normal Sambar powders were collected from local markets (Shops) in Bengaluru. We collected 21 samples widely used in Bangalore. These samples were assayed by the x-ray fluorescence method. The main principle of this x-ray fluorescence spectroscopy (XRF) is based on the fact, that atoms, when irradiated with x-rays, radiate secondary x-rays – the fluorescence radiation. x-ray fluorescence spectrometry is used for elemental analysis based on the detection of emitted x-ray radiation from excited atoms. X-rays are short wavelength electromagnetic radiation. X-ray spectrometer generally utilizes the region of about 0.1-11nm. This technique is a two-step process that begins with the removal of an inner shell electron of an atom. The resulting vacancy is filled by an outer shell electron. The second step is the transition from the outer shell electron orbital to an inner shell electron orbital. This transition is accompanied by the emission of an x-ray photon (Figure 1).
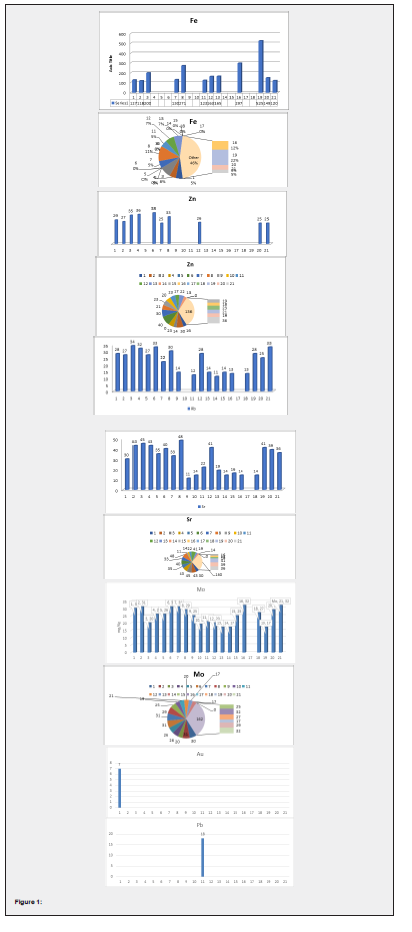
Discussion
The presence of metals has been reported from our study. Contamination may occur accidentally through contaminated irrigation water, fertilizer, air, soil, industrial processing and packaging etc (Table 1).
Zinc
Zinc is a trace metal that is necessary for a healthy immune system. Zinc plays an important role in growth metabolism and regulates metabolic activities. The concentration of zinc was found to be range from 11 to 26, which is less than the permissible level. The deficiency of zinc causes growth retardation, hypogonadism and also affects bone and growth metabolism just as an inordinately high amount of zinc could be more deleterious than nutritious, however, our samples do not exceed the WHO limit [5].

Iron
Iron is also an essential metal and a common metal in humans and plants. They occur predominantly in plants in the form of phytate complexes. They may also be in the form of inorganic iron salts such as ferric hydroxide. Thus, iron has a relatively high than WHO levels in foods. However, the iron level in the collected samples ranged from 120 to 600µg/g. The deficiency of iron causes anaemia, stomach ulcers, fatigue, malabsorption etc. Iron is an essential nutrient for plants. It functions to accept and donate electrons and plays important roles in the electron-transport chains of photosynthesis and respiration. But iron is toxic when it accumulates to high levels. It can act catalytically via the Fenton reaction to generate hydroxyl radicals, which can damage lipids, proteins, and DNA. Plants must therefore respond to iron stress in terms of both iron deficiency and iron overload. Recent advances in our understanding of plant responses to iron stress have resulted from the use of traditional approaches including genetics, biochemistry and cell biology. Iron toxicity from intentional or accidental ingestion is a common poisoning. The acute ingestion of iron is especially hazardous to children. Life-threatening toxicity is associated with pediatric ingestion of potent adult preparations, such as prenatal vitamins. Serious iron ingestion in adults is usually associated with suicide attempts [1,2]. The toxicity of iron affects the heart, liver and also causes diabetes [6].
Copper
Copper plays an important role in the oxidative defence system, on the other hand, chronic copper toxicity can result in severe poisoning [7]. Although copper is an essential element, in trace amounts but can be toxic at higher levels the copper content in the collected samples ranged between 20 to 35 µg/g. their permissible level was reported to be 50µg/g [8]. You could get severe toxicity from ingesting large amounts of copper salts through your skin. Copper can work its way through your internal organs and build up in your brain, liver, and lungs. People who have copper toxicity can become very unwell. Nausea and vomiting are two symptoms of it.
Cadmium
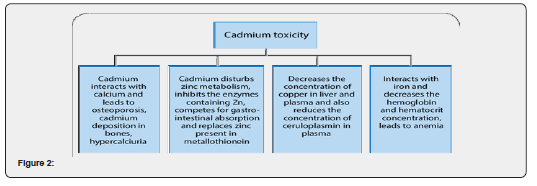
Cadmium is a toxic heavy metal that is produced during the smelting of other metals. Cadmium appears to mimic zinc and to a lesser extent, calcium and cadmium compounds are classified as human carcinogenic [7]. The permissible level of cadmium according to WHO is 0.3µg/g, but the collected samples ranged between 0.5-0.8µg/g, which is higher than the permissible level (Figure 2).
Nickel
Nickel is also a micronutrient essential for the proper functioning of the human body, as it increases hormonal activity, increases bone strength helps in red blood cell production and is involved in lipid metabolism. Nickel is usually present in the RNA and DNA of our body where it functions in association with nucleic acids. Nickel is emitted to the atmosphere from natural sources including windblown dust, volcanoes, vegetation, forest fires and meteoric dust [2]. The principal anthropogenic sources of nickel emissions to the atmosphere include the combustion of coal and oil, municipal incineration, steel and other nickel alloy production and electroplating [1,2]. In urban areas, nickel levels in the ambient air range from 1-10ng m-3. In industrialised areas and large cities levels in the range of 110-180ng m-3 have been recorded [3]. In polluted air, the main nickel compounds appear to be nickel sulphate, nickel oxides, nickel sulphides, and to a smaller extent, elemental nickel [1]. Food and cigarette smoke are the main sources of nickel exposure in the general public [3,5]. Nickel deficiency in the body is rare due to its small needed quantity. The results collected for nickel in the samples ranged from 0.7-0.8µg/g. The deficiency of nickel affects calcium and vitamin B12 metabolism, however, toxicity results in poor bone development, decreased resistance to infection, headache, nausea, vomiting [9,10].
Manganese
Manganese is an essential mineral it is needed by the body in a very small amount. It is important in growth development, bone metabolism, healing wounds, etc. It mainly acts as an anti-oxidant. The manganese content in the collected samples ranged from 70-120µg/g. Manganese toxicity (manganism) is rarely encountered, yet it is important to recognize the consequence of occupational, accidental, or iatrogenic exposures to Mn. The main toxic effects attributable to this metal are extra-pyramidal side-effects that closely resemble symptoms of Parkinson’s syndrome [3]. These adverse effects are secondary to its deposition in specific components of the basal ganglia and alteration of dopaminergic neuronal enzyme activity. Other prominent effects include cardiotoxicity, hepatotoxicity, and increased mortality in infants [4].
Cobalt
Cobalt is a trace element, present in the body in small amounts. Cobalt helps in processing vitamin B12. The collected sample ranged from 40-50µg/g. deficiency of cobalt is strongly related to the disturbance of synthesis of vitamin b12 synthesis, so it might cause anaemia and hypofunction of the thyroid and increase the risk of developmental abnormalities and failure in infants [11]. Toxicity caused by cobalt is extremely rare. Cobalt toxicity can cause effects in haematological, cardiovascular, immunological, gastro internal, dermatological, neurological systems.
Rubidium
Rubidium has no known biological role but has a slight stimulatory effect on metabolism, probably because it is like potassium. The amount of rubidium in the collected samples was found to be 10-40µg/g. Rubidium toxicity causes ingestion, hyper irritation, skin ulcers etc. [12].
Lead
Lead is the most toxic heavy metal owing, to the fact that it accumulates in biological tissues. The lead level in the collected samples ranged from 0-18µg/g which is more than the permissible level. Lead toxicity is due to atmospheric contamination, contaminated irrigation water and the addition of fertilizers and pesticides in an uncontrolled manner. Lead damages health in two ways: - disruption of normal cellular processes, leading to toxicity and bioaccumulation, particularly in the liver and kidney in which the rate of excretion is much slower than the rate of uptake. However, on a severe note, even though lead does not have biological functions, its toxic effects are numerous, including musculoskeletal, renal, ocular, immunological, neurological, reproductive, and developmental effects. Chronic exposure of children to even small amounts affect intelligence [1]. Lead exposure can be particularly harmful to children under five, who absorb
Molybdenum
Molybdenum is a cofactor synthesised by the body and required for the function of four enzymes: - sulphite oxidase, xanthine oxidase, aldehyde oxidase and mitochondrial amidoxime reducing component. Molybdenum content in the collected samples ranged from 15-35µg/g. Molybdenum deficiency and toxicity are rare, possibly because of the body’s ability to adapt to a wide range of molybdenum intake levels [13].
In addition to all these metals, we also found the presence of gold and silver in some particular samples. Silver levels in the collected samples were found to be 50-60µg/g. Gold levels in the collected samples ranged from 4-6 µg/g. Maybe this concentration of these two metals would have been occurred accidentally during manufacturing or while synthesis. Detailed study of these two metals is unknown (Figure 3-5).


Conclusion
The results in our investigation showed that there is no risk from the daily use of the above-mentioned spices if taken in limited quantity. Human needs for spices are very few grams per day and there is no risk of using these spices understudy in the foodstuff. But some results obtained indicate that spices and herbs used in food may contain some toxic metals at relatively high level. Therefore, they should be under continuous observation. The concentrations of trace elements were found to be largely dependent upon the type of sample analyzed and different factors that play an important role in ecosystem functioning. The samples analyzed were rich sources of at least one or more nutritionally essential trace elements (Cu, Zn, Fe, Mn, Cr, Ni, Co). This leads to the conclusion that spices not only add flavour to food, but also enhance its nutritive value by providing several essential elements in bioavailable form. On the other hand, the contribution of the toxic elements Pb, Rb, Sr, Cd to the overall intake from the analyzed spices was very low and could not pose any threat to the consuming population. However, if other Pb, Rb, Sr, and Cd sources are included and if spices consumed are unwashed and contaminated during processing, the daily intake may exceed the recommended levels. Keeping a close watch on these contaminants is recommended to guide consumers against the imminent health risks associated with these heavy metals. we found even Au in one of the samples but did not find the source of this.
Our study concluded that if the spices are taken in a limited dosage, there is no risk from the daily use of these spices. The human need for spices is very few grams per day and they are safe to include in food. However, some results obtained suggest that spices and herbs used in food could contain some toxic metals at relatively elevated levels. Therefore, they should be monitored continuously. Trace element concentrations were found to be largely dependent on sample type.
Acknowledgement
We thank our Principal, Management of Vivekananda Degree College for allowing us to perform and participate in research projects along with academic excellence. We sincerely express our gratitude to Dr. VENKATESH TUPPIL, Adviser for QCI, and FQI, NRCLPI and Mr. Devesh Takur, Scientific Assistance, NRCLPI. Prof. B.M. Nagabhushana, HOD of Chemistry Department, MSRIT for assisting with valuable guidelines.
References
- Moore JC, Spink J, Lipp M (2012) Development and application of a database of food ingredient fraud and economically motivated adulteration from 1980 to 2010. J Food Sci 77(4): R118-R126.
- Layton L (2010) At U.S. dinner tables, the food may be a fraud; deception, mislabelling increase, but FDA lacks enforcement resources. The Washington Post, p. 1.
- Madiwale T, Liebelt E (2006) Iron: not a benign therapeutic drug. Curr Opin Pediatr 18(2): 174-179.
- Sane MR, Malukani K, Kulkarni R, Varun A (2018) Fatal Iron Toxicity in an Adult: Clinical Profile and Review. Indian J Crit Care Med 22(11): 801-803.
- International Programme on Chemical Safety (IPCS) (1991) Chemical. Environmental Health Criteria 108: Nickel. WHO, Geneva.
- Agency for Toxic Substances and Disease Registry (ATSDR) (2005) Toxicological Profile for Nickel. US Department of Health and Human Services. Atlanta, US.
- World Health Organization (WHO) (2000) Air Quality Guidelines for Europe. WHO Regional Publications, European Series, No. 91. (2nd edn), WHO Regional Office for Europe. Copenhagen.
- Shi Z (1994) Nickel carbonyl: toxicity and human health. Sci Total Environ 148(2-3): 293-298.
- Department for Environment Food and Rural Affairs (DEFRA) and Environment Agency (EA) (2002) Contaminants in soil: Collation of toxicological data and intake values for humans. Nickel. Environment Agency. Bristol.
- Pfalzer AC, Bowman AB (2017) Relationships Between Essential Manganese Biology and Manganese Toxicity in Neurological Disease. Curr Environ Health Rep 4(2): 223-228.
- O'Neal SL, Zheng W (2015) Manganese Toxicity Upon Overexposure: a Decade in Review. Curr Environ Health Rep 2(3): 315-328.
- International Zinc Nutrition Consultative Group (IZiNCG), Kenneth H Brown, Juan A Rivera, Zulfiqar Bhutta, Rosalind S Gibson, et al. (2004) Assessment of the Risk of Zinc Deficiency in Populations and Options for Its Control. Food and Nutrition Bulletin 25(1 Supp 2): S99-S203.
- Janet A Novotny (2011) Molybdenum Nutriture in Humans. Journal of Evidence-Based Complementary & Alternative Medicine 16(3): 164-168.






























