Nutritional Benefits of Edible Macroalgae from the Central Portuguese Coast: Inclusion of Low-Calorie ‘Sea Vegetables’ in Human Diet
Jelena Milinovic1*, Ana Luisa Fernando2, Bruno Campos1, Bruno Leite1, Paulina Mata1, Mário Diniz3,4, José Sardinha5 and João Paulo Noronha1
1LAQV-REQUIMTE, Department of Chemistry, NOVA School of Science and Technology, Universidade NOVA de Lisboa, Portugal
2MEtRiCS, Departamento de Ciências e Tecnologia da Biomassa, NOVA School of Science and Technology, Universidade NOVA de Lisboa, Portugal
3Associate Laboratory i4HB - Institute for Health and Bioeconomy, NOVA School of Science and Technology, Universidade NOVA de Lisboa, Portugal
4UCIBIO, Applied Molecular Biosciences Unit, Department of Chemistry, NOVA School of Science and Technology, Universidade NOVA de Lisboa, Portugal
5CERENA - Natural Resources and Environment Studies Center, Instituto Superior Técnico, University of Lisbon, Portugal
Submission: July 28, 2021; Published: September 03, 2021
*Corresponding author: Jelena Milinovic, LAQV-REQUIMTE, Department of Chemistry, NOVA School of Science and Technology, Universidade NOVA de Lisboa 2829-516 Caparica, Portugal
How to cite this article: Jelena M, Ana L F, Bruno C, Bruno L, Paulina M, et al. Nutritional Benefits of Edible Macroalgae from the Central Portuguese Coast: Inclusion of Low-Calorie ‘Sea Vegetables’ in Human Diet. Int J Environ Sci Nat Res. 2021; 28(5): 556250. DOI: 10.19080/IJESNR.2021.28.556250
Abstract
In this work, major nutrients were determined in edible green (Codium tomentosum, Ulva rigida), red (Gracilaria gracilis, Osmundea pinnatifida, Porphyra sp), and brown macroalgae (Saccorhiza polyschides, Undaria pinnatifida) collected from the central Portuguese coast. The results showed that selected species had the macroelements (Ca+Mg+Na+K+P) ranged from 85 (Porphyra sp) to 226mg g-1 (Codium tomentosum), and the oligoelements (Cu+Fe+Mn+Se+Zn) ranged from 60 (U. pinnatifida) to 655µg g-1 (O. pinnatifida), thus representing a sustainable source of minerals. The low values of Na/K (0.3-3.7) showed that selected species can be of special nutritional interest for patients with hypertension. The highest contents of Fe (552µg g-1) found in O. pinnatifida and Se (30µg g-1) in S. polyschides, suggest the increased proportion of these oligoelements, by the inclusion of macroalgal species in the diet, rather than some traditionally known food. The total content of proteins (15.5-24%), carbohydrates (52-62%), crude fibres (14-32%), and lipids (0.9-4.8%) indicated that the selected macroalgae were relatively low-calorie food. Analyzed red species were characterized by the higher content of proteins (24% in Porphyra sp), whereas brown species showed increased levels of crude fibres (32% in S. polyschides). The caloric values ranged from 172 (S. polyschides) to 256kcal 100g-1 (Porphyra sp) and they were negatively correlated (R = -0.81) with the ash content in studied species. The correlation between calories and ash indicated the influence of (thallus) morphology of species on their mineral and nutritional composition. Based on their particular nutritional profiles, the studied species can be recommended for selective inclusion in the human diet, ensuring high-quality and health-promoting food.
Keywords: Edible macroalgae; Osmundea pinnatifida; Saccorhiza polyschides; Na/K value; Selenium; Low-calorie food
Introduction
Edible macroalgae (seaweeds) are an important sustainable source of food, nutrients, and food-flavoring products for direct human consumption, accounting for 83-90% of their total production [1]. The species of each taxonomic group of macroalgae, green (Chlorophytae), red (Rhodophytae), and brown (Phaeophyceae), have variable ranges of nutritional components, including macroelements (Ca, Mg, Na, K, P), some oligoelements (e.g., Cu, Fe, I, Mn, Se, Zn), proteins, carbohydrates, and lipids [2-12]. Most polysaccharides are constituents of the cell wall, these are not digested and function as dietary fibres, important for digestion [13,14]. The nutritional composition of macroalgae depends on numerous factors, both biotic (the morphology, the maturity, physical properties of macroalgae, etc.), and abiotic, i.e., environmental conditions [3,11,15].
China is the largest global producer of cultivated macroalgae, of which red and brown species account for more than 93% of those used in nutrition [1]. The main commercial species are red Gracilaria spp., used as a salad vegetable, Porphyra spp. (nori), commonly used in Japanese sushi, and brown Saccharina japonica (kelp), Undaria spp (wakame), contributing to 92% of the global production in 2015 [1]. In addition to these species, the two main commercial species used for direct human consumption are green Ulva spp (sea lettuce) and Codium spp [1,16]. In Asia, these species are considered important food resources (good source of minerals, proteins, and fibres), commonly eaten raw or dried (in salads) or cooked (in soups and stews) [17].
The use of macroalgae in the human diet has strong roots in Asian countries, and their highest consumption was estimated in Japan (at approximately 9.6g a day), slightly more than the size of a typical portion in other Asian cuisines (8.0g per day) [4,18]. Recently, demands for macroalgae as food have been spread to Western countries (Canada, the USA, Nordic countries, Ireland, Iceland, France, Portugal, Spain, etc.), but their consumption is still less than 1% of the fully harvested macroalgae at national levels [19]. The increase in demand for the introduction of macroalgae in gastronomy (phycogastronomy), over the last fifty years has surpassed the capacity to supply the requirements from natural resources. In Canada, the USA, Iceland, and Ireland, different types of macroalgae have traditionally been consumed, thus continuously developing this market. The government and commercial organizations in some Nordic countries and France promoted phycogastronomy by gradually introducing macroalgae in restaurant menus, but still not considering macroalgae as fully-fledged vegetables [20,21]. With its large maritime area, Portugal has a considerable variety of available macroalgal species, and some of them are potential food resources [22,23].
However, so far only a few data on the nutritional composition of several edible species from the Portuguese coasts was reported [8,9,11]. To the best of our knowledge, their complete nutrition profiles, including the caloric values, are not available in the current scientific literature.
In this work, the concentrations of macroelements (Ca, Mg, Na, K, P), oligoelements (Cu, Fe, Mn, Se, Zn), biomolecules (proteins, carbohydrates, crude fibres, lipids) are reported in edible macroalgae collected from the central Portuguese coast (Cape Mondego). In addition to species with wide applications in Asian gastronomy (Codium tomentosum, Ulva rigida, Gracilaria gracilis, Porphyra sp, Undaria pinnatifida), red species Osmundea pinnatifida and brown Saccorhiza polyschides were also selected for the present work because of their abundance along the Portuguese coastline. The nutritional potentials of analyzed species are critically commented on and some recommendations for their inclusion in the regular diet (particularly in Western countries) as low-calorie ‘sea vegetables’ have been also considered.
Materials and Methods
Reagents
All reagents used were of analytical grade from commercial suppliers, as purchased. The standard stock solutions of elements were purchased from Merck Inorganic Ventures, Spectrosol, Alfa Aesar, Plasma Cal, Sigma Aldrich, and VWR chemicals. All solutions were obtained by using ultrapure Milli-Q water (18.2MΩ cm-1), from Millipore Purification System (Watermax W1, Diwer Technologies, Portugal).
Collection and pretreatment of macroalgae
Seven edible macroalgal species were collected from the central Portuguese coast, at the Cape Mondego (40º 11’ N, 08º 54’ W) (Figure 1). The area of Cape Mondego was classified as a Natural Monument in 2007, and it is integrated into the Network of Protected Areas in Portugal [24].
Seven species were sampled, of which two green (Codium tomentosum, Ulva rigida), three red (Gracilaria gracilis, Osmundea pinnatifida, Porphyra sp), and two brown (Saccorhiza polyschides, Undaria pinnatifida). The species were collected in 2019, in two seasons, from spring (G .gracilis, O. pinnatifida), to late summer/early autumn (C. tomentosum, U. rigida, Porphyra sp, S. polyschides, U. pinnatifida), according to their abundance (Figure 2). Two species, red O. pinnatifida and brown S. polyschides lesser-known in global gastronomy, were also selected for the present work due to their abundance along the Portuguese coastline. All species were collected at the mature phase, following the best ecological practices and favourable weather conditions [12].

After harvesting samples were transported in thermal boxes to the laboratory. The samples were washed with distilled water to remove salts, detrital particles, and epiphytes. Then, samples were lyophilized at -50°C for 48h (CoolSafe 9L, LaboGeneTM, Denmark). The lyophilized biomasses were finely ground automatically, and the resulting grains were sifted through the laboratory’s stainless-steel sieve (120 mesh, 0.12mm aperture). The obtained homogeneous fine fractions were kept in labeled recipients and stored at -18°C before the analysis.
Determination of ash content and minerals in macroalgae
The total ash, corresponding to the inorganic material of the sample, was determined gravimetrically after incineration of the lyophilized sample (1.0g) at 550°C for 8h [25]. All results are expressed as a mean mass fraction in % (g 100g-1, referred to sample dry weight, dw), based on three replicate measurements (n = 3). The standard deviation (SD) was low (SD < 0.4%) for determining ash content in all samples.
The macroelements (Ca, K, Mg, Na, P) were analysed using the inductively coupled plasma - optical emission spectroscopy (ICP-OES, Jobin Yvon model Activa M), whereas oligoelements (Cu, Fe, Mn, Se, Zn) and potentially toxic elements (Cd, Hg, Pb) were determined by inductively coupled plasma - mass spectrometry (ICP-MS, Quadrupole Thermo Scientific X Series), as previously described [26,27]. Before ICP-analyses, approximately 0.2g of lyophilized sample was mixed with 2.0mL of HNO3 in a Teflon vessel, and heated in a microwave (CEM MARS 5) according to the following digestion program: within the initial 10min, the temperature increased to 180°C and remained constant for the next 10min. After cooling, 0.5mL of H2O2 was added to the solution, following a new digestion cycle as described. Then, the sample was evaporated to 0.5mL and Milli-Q water was added to the volume of 25mL. The samples were diluted in 2% HNO3 to avoid matrix interferences due to salinity.
Limit of detection (LOD) for Ca, K, Na, and P was 0.006mg g-1, while for Mg was 0.0012mg g-1. LOD for Cu, Se, and Zn was 0.25µg g-1, whereas for Mn and Fe was 0.62 and 1.2µg g-1, respectively. Cadmium, Pb and Hg had LODs equal to 0.012 and 0.031µg g-1, respectively. Quality control of the method was assured by the same analytical procedure performed with blanks, sample duplicates (SD < 10%), and the certified reference materials (CRMs) DORM-4 (n = 24), and TORT-2 (n = 6). The CRMs were used throughout the analytical procedures for the quantification of elements and the control of the quality and traceability of results, as a calibration measurement standard. Obtained recoveries were high, ranging from 91 to 106%. All results are expressed as a mean concentration in mg g-1 (Ca, K, Mg, Na, P) or µg g-1 (Cd, Cu, Fe, Hg, Mn, Pb, Se, Zn), referred to sample dry weight, dw.
The concentrations of potentially toxic elements Cd (0.08-0.63µg g-1 dw), Hg (not detected), and Pb (0.1-0.7µg g-1 dw) in macroalgae were below the threshold limits regulated in the EU (< 3.0µg g-1 dw), thus designating selected macroalgae as edible species [1,28].
Determination of biomolecules in macroalgae
The protein content was determined by the method of Kjeldahl [25,29]. The organic nitrogen conversion factor of 6.25 was used to calculate the total protein content [30].
The lipids in macroalgae were quantified by the modified method based on extraction using a mixture of chloroform, methanol and water [6,31]. Briefly, 0.25 g of the lyophilized sample was mixed with 5.5mL of solution chloroform/methanol/water (1.5:1.2:1, v/v/v), in a glass tube (6mL). The tube was shaken in the vortex (1min) and subsequently the separation of phases was achieved by centrifugation (1700xg for 15min). The chloroform phase was transferred to the vial (previously weighted) and the methanol/water layer was washed with a new portion of chloroform (2.0mL). The combined chloroform phases were evaporated under a nitrogen stream to dryness. The total lipid content was calculated gravimetrically.
The crude (water-insoluble) fibre content was determined by the Weende method [30]. Briefly, a portion of 1.0g of lyophilized sample was boiled in diluted solutions of H2SO4 and KOH, and after, the content of crude fibre was determined gravimetrically.
All results are expressed as a mean mass fraction in % (g 100g-1, dw), based on three replicate measurements (n = 3). For quantified proteins, lipids, and crude fibres SD was lower than 1.0, 0.8, and 3.0%, respectively.
Evaluation of total carbohydrates and caloric values in macroalgae
The total carbohydrate content (expressed in %, dw) was calculated as the weight difference based on the ash, protein, and lipids in the lyophilized sample. Based on results for total protein, lipids, and carbohydrates (less the crude fibre), the energy value of macroalgae was calculated as the sum of these parameters, multiplied by a factor of 4, 9, and 4, respectively [32]. The results are expressed in kcal 100g-1, referred to sample dw.
Data analyses
The relationship between concentrations was assessed by the correlation coefficient (R) at a significant level of 95% (p = 0.05). Comparison of mean values of nutritional composition between macroalgal species was carried out by ANOVA test (p = 0.05). Principal component analysis (PCA) was performed on data to summarize the information of nutritional profiles in studied species. 2-component PCA was applied using the software Unscrambler (v. 10.4, Camo, ASA, Norway). PCA used the nonlinear iterative partial least squares (NIPALS) algorithm and Hotelling’s T2 distribution to calculate the outliers (p = 0.05).
Results and Discussion
Ash and mineral contents in edible macroalgae
As all the macroalgae were harvested from a single site (Cape Mondego), species collected during each season were exposed to the same environmental conditions. G. gracilis and O. pinnatifida were sampled in spring, and the other species in late summer/early autumn of 2019. However, the concentrations of macroelements and oligoelements did not vary significantly between the two sampling seasons. This can be justified for a low variability of the physico-chemical properties in a site (e.g., the temperature of seawater, pH, and salinity). During harvesting seasons, in spring and late summer/early autumn, the seawater temperature was 16°C, pH 7.6 and 8.0, and salinity 31-35ng L-1 and 34-36ng L-1, respectively [12].
The concentrations of ash and minerals (macroelements and oligoelements) in analysed Portuguese macroalgae are presented in Table 1. The ash content ranged from 21% (C. tomentosum, U. pinnatifida) to 29% (S. polyschides). The differences in ash content between the studied species, which were not statistically different, and they corresponded to varying levels of minerals. Similar results for the ash content (20-31%) were obtained for the same edible species (U. rigida, G. gracilis, O. pinnatifida, Porphyra sp, S. polyschides, and U. pinnatifida) collected at different coastline sitess in NW Portugal, Spain, and New Zealand [9,33,34].
Although the sum of macroelements (Ca, K, Mg, Na, and P), accounted for > 99.6% of the total minerals, their individual concentrations varied between the species. In all species, Mg (3.3-19mg g-1) showed higher concentrations than Ca (1.7-14mg g-1), with an exception for S. polyschides in which both elements had similar values. Within three phyla, the contents of Mg were higher in green species, probably due to higher levels of chlorophylls (with Mg ion in the center of the porphyrin ring). Potassium and Na were also in varying concentrations in all species, i.e., 20-106mg g-1 and 27-136mg g-1, respectively. The sum of K and Na represented 70.3-94.3% of the total concentration of macroelements in analyzed samples (Figure 3). The lowest concentrations of K+Na were found in Porhyra sp (7.1mg g-1) whereas the highest in C. tomentosum (195mg g-1) and U. pinnatifida (17.4mg g-1). The Na/K value was less than unity only in G. gracilis (Na/K = 0.3) and S. polyschides (Na/K = 0.9), whereas in the rest of samples this ratio was between 1.3 and 3.7, which is similar to other results previously reported [7-9,35].
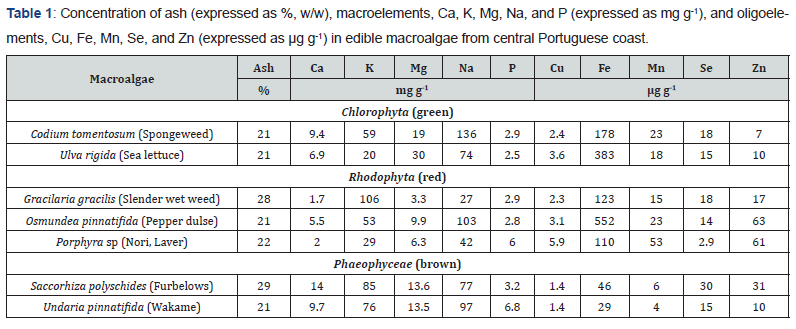

Phosphorous was present at a fairly low level (1.3-7.1% of the sum of macroelements), in all seven species ranging from 2.5 (U. rigida) to 6.8mg g-1 (in U. pinnatifida). The concentrations of P were of the same order of magnitude as Ca, giving the value of Ca/P higher than unity in all species, with an exception in G. gracilis (Ca/P = 0.6) and Porphyra sp (Ca/P = 0.3). The results obtained were similar to those previously reported for the similar species of the same genus, Porphyra columbina, collected from the central pristine beach in Argentina [36].
The highest amounts for the sum of Ca+K+Mg+Na were found in C. tomentosum (223mg g-1) and U. pinnatifida (197mg g-1). In O. pinnatifida (172mg g-1) and S. polyschides (189mg g-1) similar values were observed, while U. rigida and G. gracilis contained less amounts of macroelements (131 and 138mg g-1, respectively). The species Porphyra sp had the smallest sum of the macroelements (79mg g-1). Similar composition of macroelements was observed in the edible macroalgae from the French coastline [16].
Among oligoelements, Cu content was in the lowest range (1.4-5.9µg g-1) whereas Fe was present in a broad interval of concentrations, from 29µg g-1 (U. pinnatifida) to 552µg g-1 (O. pinnatifida). Manganese, Se, and Zn were 3-22 orders of magnitude higher than Cu in edible species studied, i.e., up to 53, 30, and 63µg g-1, respectively. Manganese was found at the highest level in Porphyry sp (53µg g-1), whereas in the same species the lowest content of Se (2.9µg g-1) was registered. In the rhe rest of the species had Se ranged from 14 to 18µg g-1, and its maximum level was found in S. polyschides (30µg g-1). The highest Zn content was obtained in red species O. pinnatifida (63µg g-1) and Poprhyra sp (61µg g-1). Copper was positively correlated with Mn (R = 0.95), and negatively with Se (R = -0.83).
Biomolecules in edible macroalgae
In general, studied macroalgae showed relatively high contents of proteins and carbohydrates, whereas their total lipid level was very low (Table 2). The protein content ranged 15-17%, 18-24%, and 16% in studied green, red, and brown species, respectively. Since proteins are mostly composed of glutamate and aspartate, both amino acids that contribute to the umami taste [4,37,38], the maximum protein levels obtained in red species, correspond to their highest umami amino acid levels, previously found in red species (G. gracilis, O. pinnatifida) [38]. The high levels of proteins found in O. pinnatifida (20%) and Porphyra sp (24%) were similar to other results obtained for these edible species from the Azores archipelago and the Central and NW Portuguese coast [8,9,11]. Similar results were obtained in the edible species of Porphyra collected from a pristine beach within San Jorge Gulf, Argentina [36], NW Iberian coast [2], and in coastlines of Japan, Korea, and China [3]. These results may suggest similar natural characteristics of marine environments, predominantly oligotrophic with good availability of nitrogen to algal populations. Moreover, all results were obtained after the same methodology applied in protein determination (Kjeldahl’s method with the use of N-conversion factor of 6.25).
The total carbohydrates showed the highest values (60-62%) in green species, while the red and brown macroalgae contained lower levels (52-58%). Similar results were obtained in previous studies with several edible macroalgal species from Chile [31]. Red species showed the lowest, whereas brown species were characterized by the highest fibre contents (14-18% and 22-32%, respectively) (Table 2). Due to the lack of fibre content results in previous studies with macroalgae from the Portuguese coastal environments, the comparisons of results are unattainable.
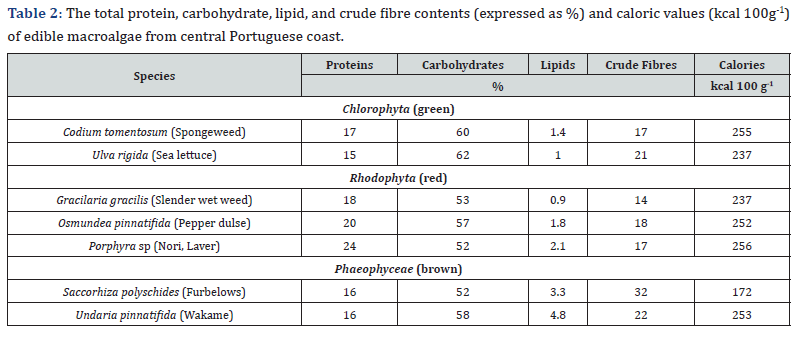
All species showed very low total lipid content (Table 2), and this can be explained by the harvesting season. Namely, the lowest lipid percentage is typical in summer and autumn, whereas the highest values occur in winter [39]. Green and red species showed very low lipid contents, ranging from 0.9% to 2.1%, and slightly higher values were found in brown macroalgae (3.3-4.8%). Low lipid ranges were obtained for the same species of macroalgae by other authors [2,5,7-9,14,33,34].
The caloric values of the studied species varied significantly, from 172kcal 100g-1 (S. polyschides) to 256kcal 100g-1 (Porphyra sp). The calories in macroalgae were statistically negatively correlated to their ash content (R = -0.81). Therefore, a high ash and Ca content in S. polyschides, derived from precipitated calcium carbonate in the thick thallus of this species (Figure 2 & 3) resulted in the low caloric value, whereas Poprhyra sp has a foliose thallus, with lower ash and Ca values and therefore, having a higher caloric content.
The caloric values obtained in this work were lower in comparison to the scarce data reported for the same species, O. pinnatifida and Porphyra sp, collected in North Yorkshire, United Kingdom, which caloric values were respectively 325 and 437kcal 100g-1 [40]. This difference in results can be associated not only to slightly different contents of biomolecules (proteins, lipids, carbohydrates, etc.) in species grown in different environmental conditions but also due to the different methodologies used i.e., the latter values were determined directly by using a bomb calorimeter [40].
Clustering of edible macroalgae based on their nutritional profile
A two-component PCA allowed a good distinction (79%) between the studied species belonging to the three taxonomic phyla (Figure 4). The first principal component (PC-1) accounted for 44% of the variance in data, while the second component (PC-2) explained 35% of the variance. PC-1 represents gradient opposing high concentrations of Se, crude fibre (with positive scores), with that of Fe (with negative score). PC-2 mainly reflects variations in Mg, Na (with positive scores), versus Cu, Mn, Zn (with negative scores). Both green species (C. tomentosum and U. rigida) were positively correlated with Mg, Na, Fe, and carbohydrates, whereas all studied red species were correlated to the P, most of oligoelements (e.g., Cu, Mn, Zn), as well as proteins. The brown species (S. polyschides and U. pinnatifida), also clustered separately, showed a higher affinity for Ca, Se, lipids, and crude fibres. Data for S. polyschides are strongly displaced along the PC-1, and this is likely to be caused by its relatively high Ca, ash, and crude fibre contents, and low caloric values.
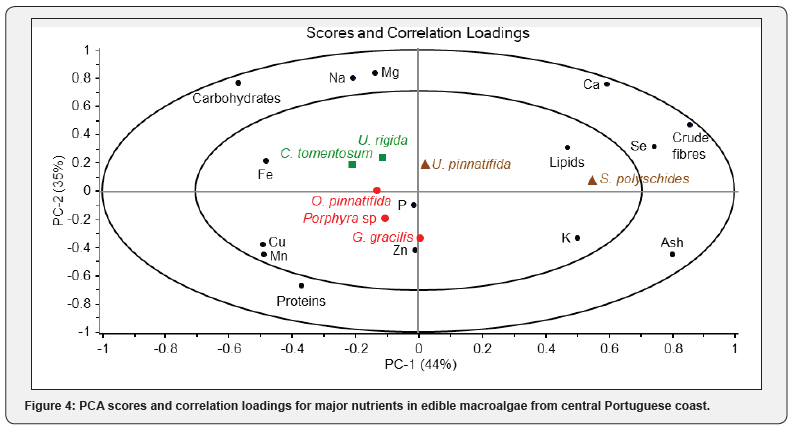
The results presented in PCA plots can serve as a database of a smaller set of nutritional components in selected edible species. According to their specific nutritional composition (e.g., O. pinnatifida had highest Fe, Zn; S. polyschides had highest Ca, Se, ash, and crude fibres), each species can be introduced selectively as unique dietary supplement in the diet.
Inclusion of macroalgae from the Cape Mondego in human diet
Results obtained for both minerals and biomolecules (proteins, carbohydrates) in the studied edible macroalgal species can be considered for their Results and Discussion into the regular human diet with balanced nutritional requirements. The sum of macroelements (Ca+K+Mg+Na+P), ranging from 85mg g-1 (Porphyra sp) to 226mg g-1 (C. tomentosum), and oligoelements (Cu+Fe+Mn+Se+Zn), ranging from 60µg g-1 (U. pinnatifida) to 655µg g-1 (O. pinnatifida), were higher than in some terrestrial vegetables, such as carrots, tomatoes, or potatoes [7]. These results indicate that macroalgae can provide a sustainable source of minerals (macroelements and oligoelements), by their diverse applications in the diet.
Consumption of some macroalgae, can provide significant health effects in terms of increased oligoelements levels. As an example, a portion of 10g (dw) of O. pinnatifida contains 5.5mg of Fe, and thus its addition as a supplement (e.g., to bread, crackers, or biscuits) can increase the proportion of iron in food thus helping in the treatment of iron deficiency anemia [41,42]. Therefore, the addition of O. pinnatifida to food, can be an alternative to the French baguette supplemented with Ulva sp or Palmaria palmata (Fleurence, 2016). The content of Se in all studied species (except in Porphyra sp) was in higher levels (14-30µg g-1), than in some traditionally known food rich in Se (e.g., Brazil nuts contain 0.8-6.9µg g-1) [43]. According to the European Food Safety Authority, for the regulation of immune and reproductive systems, the adequate daily intake of Se was set at 70µg for adults [28], and this amount can be achieved by the inclusion of smaller portions of macroalgae in diet. Nevertheless, it is important to analyse the levels of Se species in macroalgae and for instance, their changes after applying various methods of cooking (e.g., steaming, water blanching, boiling, etc.). The obtained results for Se species can provide information on the optimal use of macroalgal species as food ingredients (e.g., in broths and soups).
The favorable nutrition effects of macroalgae can also be very beneficial for patients with hypertension who require special medical treatments. The Na/K ratio is important to control hypertension for patients suffering from an excessive secretion of K [35]. In the studied samples, the low level of Na/K values (0.3-3.7) can be of special nutritional interest, significantly lower than in common food, such as olives (Na/K~44) [36]. Therefore, the consumption of selected species can provide balanced Na/K diets. As an example, macroalgae can be incorporated in meat products, with less salt than the conventional recipes, provided a promising foodstuff with functional, health and sensory attributes [44].
The recent global estimate for protein sources highlights their higher demand by 2050 [45]. Given the variability of the content of total proteins in macroalgae, they can be an alternative to non-animal protein sources in the future [46]. The total protein content in studied red macroalgae (18-24%) is comparable to plants with a high-protein content, such as carrots, tomatoes, peas, or beans (11-28%) [9,36,47]. Hence, macroalgae can substitute or complement, the role of legumes and cereals to get protein-balanced diets. However, the protein digestibility can differ between the species, and this should be studied in more details. Some results showed that red species had higher protein digestibility than green species, while brown macroalgae had the lowest digestibility [48]. The study of digestibility of proteins in U. pinnatifida showed they were high, i.e., 70% in humans [49].
A portion of 10.0g (dw) of green, red, or brown macroalgae can provide 6.1, 5.4, or 5.5g of total carbohydrates, respectively, which corresponds to a similar portion of peas, tomatoes, and carrots (4.7, 5.8, or 6.3g, respectively) [47]. Compared to terrestrial plants, which contain crude fibres in quantities varying from 8.3% (apples) to 17.5% (cabbage), the studied macroalgae (U. rigida, S. polyschides, U. pinnatifida) contained higher amounts of crude fibres (21-32%), representing a valuable source of fibre, which recommended daily intake (25g for adults) can be achieved by consuming of food supplemented with selected species [14,28].
Macroalgae-derived lipids are known to be highly digestible and this was reported for U. pinnatifida species from Japan, which lipids are 98% digestible in adults [50]. Hence, the inclusion of macroalgae in diet can provide moderate lipid intake and moderate prevention of weight gain, compatible with good health.
Conclusion
The edible green, red, and brown macroalgae examined in this study, have considerable nutritional potentials, with better mineral profiles than some terrestrial plants. Together with high protein, carbohydrate, and low lipid levels, studied species from Cape Mondego, in the central Portuguese coastline, represent healthy, low-calorie ‘sea vegetables’. Given their different nutritional profiles, species belonging to green, red, and brown phylum can be included in diversified macroalgal-based food (e.g., appetizers, snacks, salads, meals, desserts, beverages, etc.) both healthy and attractive to consumers.
The application of edible macroalgae, in particular in Western gastronomy, should be fully exploited, to fit gastronomic purposes. Future studies are important for the nutritional evaluation of various edible species and their digestibility, as well as possible effects as a functional food.
Acknowledgment
This work is financed by national funds from FCT - Fundação para a Ciência e a Tecnologia, I.P., in the scope of the project UIDP/04378/2020 and UIDB/04378/2020 of the Research Unit on Applied Molecular Biosciences - UCIBIO and the project LA/P/0140/2020 of the Associate Laboratory Institute for Health and Bioeconomy - i4HB. This work was also supported by the project “MAR-01.03.01-FEAMP-0016-Alga4Food” which is financed by the European Maritime and Fisheries Fund and co-financed by the Operational program MAR2020 in the field of Sustainable development of Aquaculture in the domains of Innovation, Advice and Productive Investment - Innovation and Knowledge Action.
This research was funded by national funding by Foundation for Science and Technology - FCT, through the individual research grant (2020.04441.BD) of C. Rodrigues. This work was supported by the Mechanical Engineering and resource Sustainability Center - MEtRICs which is financed by national funds form FCT/MCTES (UIDB/04077/2020 and UIDP/04077/2020).
The authors would like to thank to L. Pereira, J. Cotas and staff of the MARE UC - Marine and Environmental Research Center, Faculty of Science and Technology, University of Coimbra, Portugal for the harvest support and macroalgae identification. The authors appreciated collaboration of M.J. Botelho and C. Vale for the results of the ICP-analyses. The authors would like to thank to C. Rodrigues and V.G. Lauriano Souza, for the assistance in sample analysis.
References
- FAO (2018) The global status of seaweed production, trade and utilization. In Globefish Research Program (vol. 124). FAO Rome Italy.
- Sánchez Machado DI, López Cervantes J, López Hernández J, Paseiro Losada P (2004) Fatty acids, total lipid, protein and ash contents of processed edible seaweeds. Food Chem 85(3): 439-444.
- Dawczynski C, Schubert R, Jahreis G (2007) Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem 103(3): 891-899.
- MacArtain P, Gill CIR, Brooks M, Campbell R, Rowland IR (2007) Nutritional value of edible seaweeds. Nutr Rev 65(12 Pt 1): 535-543.
- Kumari P, Kumar M, Gupta V, Reddy CRK, Jha B (2010) Tropical marine macroalgae as potential sources of nutritionally important PUFAs. Food Chem 120(3): 749-757.
- Ginneken VJT, Helsper JPFG, Visser W, Keulen H, Brandenburg WA (2011) Polyunsaturated fatty acids in various macroalgal species from north Atlantic and tropical seas. Lipids Health Dis 10: 104.
- Kumar M, Kumari P, Trivedi N, Shukla MK, Gupta V, Reddy CRK, Jha B (2011) Minerals, PUFAs and antioxidant properties of some tropical seaweeds from Saurashtra coast of India. J Appl Phycol 23(5): 797-810.
- Paiva L, Lima E, Patarra RF, Neto AI, Baptista J (2014) Edible Azorean macroalgae as source of rich nutrients with impact on human health. Food Chem 100: 128-135.
- Rodrigues D, Freitas AC, Pereria L, Rocha Santos AP, Vasconcelos MW, et al. (2015) Chemical composition of red., brown and green macroalgae from Buarcos bay in Central West Coast of Portugal. Food Chem 183: 197-207.
- Miedico O, Pompa C, Tancredi C, Cera A, Pellegrino E, et al. (2017) Characterisation and chemometric evaluation of 21 trace elements in three edible seaweed species imported from south-east Asia. J Food Compos Anal 64(2): 188-197.
- Vieira EF, Soares C, Machado S, Correia M, Ramalhosa MJ, et al. (2018) Seaweeds from the Portuguese coast as a source of proteinaceous material: Total and free amino acid composition profile. Food Chem 269: 264-275.
- Milinovic J, Rodrigues C, Diniz M, Noronha JP (2021) Determination of total iodine content in edible seaweeds: Application of inductively coupled plasma-atomic emission spectroscopy. Algal Res 53: 102149.
- Southgate DAT (1990) Dietary fibre and health. In: Southgate DAT, Waldron K, Johnson IT, Fenwick GR (Eds.) Dietary fibre: Chemical and Biological Apects. The Royal Society of Chemistry, Cambridge, United Kingdom. pp. 282-284.
- Burtin P (2003) Nutritional value of seaweeds. Elec J Env Agricult Food Chem 2: 498-503.
- Pereira L (2009) Guia ilustrado das macroalgas. Conhecer e reconhecer algumas espécies da flora portuguesa. Tipografia Lousanense, Lda, Lousã, Portugal.
- Fleurence J (2016) Chapter 5 - Seaweeds as food. In: Fleurence J, Levine I (Eds.), Seaweed in health and disease prevention. Elsevier Inc, pp. 149-167.
- Senthil A, Mamatha BS, Vishwanath P, Bhat KK, Ravishankar GA (2011) Studies on development and storage stability of instant spice adjunct mix from seaweed (Eucheuma). J Food Sci Tech 48(6): 712-717.
- MHLW (2014) The National Health and Nutrition Survey in Japan (2004–2014) The Ministry of Health, Labour and Welfare.
- McHugh DJ (2003) A guide to the seaweed industry. FAO Fisheries Technical Paper 441. Food and Agriculture Organization of the United Nations, Rome. FAO, Viale delle Terme di Caracalla, Rome, Italy.
- Mouritsen OG, Williams L, Bjerregaard R, Duelund L (2012) Seaweeds for umami flavor in the new Nordic cuisine. Flavour 1: 1-
- CEVA (2014) Algae technology and innovation center.
- Pereira L (2011) A review of the nutrient composition of selected edible seaweeds. Seaweed: Ecology, nutrient composition and medicinal uses. Nova Science Publishers, Inc New York, USA. pp. 15-47.
- Soares C, Machado S, Vieira EF, Morais S, Teles MT, et al. (2017) Seaweeds from the Portuguese coast: A potential food resource? IOP Conference Series: Materials Science and Engineering 231: 012126.
- Rocha J, Brilha J, Henriques MH (2014) Assessment of the geological heritage of Cape Mondego Natural Monument (Central Portugal). Proc Geol Assoc 125: 107-113.
- AOAC (1990) Official Methods of Analysis. Agricultural Chemicals; Contaminants; Drugs (vol I). AOAC, Arlington, USA.
- Henriques B, Lopes CB, Figueira P, Rocha LS, Duarte AC, et al. (2016) Bioaccumulation of Hg, Cd and Pb by Fucus vesiculosus in single and multi-metal contamination scenarios and its effect on growth rate. Chemosphere 171: 208-222.
- Henriques B, Teixeira A, Figueira P, Reis AT, Almeida J, et al. (2019) Simultaneous removal of trace elements from contaminated waters by living Ulva lactuca. Sci Total Environ 652: 880-888.
- EFSA (2017) Dietary Reference Values for nutrients - summary report. EFSA Support Publ 14(12): e15121E.
- Watts S, Halliwell L (1996) Appendix 3 - Detailed field and chemical methods for soil. In: Watts S, Halliwell L (Eds.), Essential Environmental Science, Methods & Techniques. Routledge, London, United Kingdom. pp. 475-505.
- Adrian J, Potus J, Poiffait A, Dauvillier P (2000) Análisis nutricional de los alimentos. Editorial Acribia SA, Zaragoza, Spain.
- Ortiz J, Uquiche E, Robert P, Romero N, Quitral V, et al. (2009) Functional and nutritional value of the Chilean seaweeds Codium fragile, Gracilaria chilensis and Macrocystis pyrifera. Eur J Lipid Sci Tech 111(4): 320-327.
- Nielsen SS (2017) Food analysis. Springer, Cham, Switzerland.
- Smith JL, Summers G, Wong R (2010) Nutrient and heavy metal content of edible seaweeds in New Zealand. New Zeal J Crop Hort 38: 19-28.
- Taboada C, Millán R, Míguez I (2010) Composition, nutritional aspects and effect on serum parameters of marine algae Ulva rigida. J Sci Food Agricult 90(3): 445-449.
- Padam BS, Chye FY (2020) Chapter 2 - Seaweed components, properties, and applications. In: Torres MD, Kraan S, Dominguez H (Eds.), Sustainable seaweed technologies: Cultivation, biorefinery, and applications. Advances in Green Chemistry, Elsevier, pp. 33-87.
- Cian RE, Fajardo MA, Alaiz M, Vioque J, González RJ, et al. (2014) Chemical composition, nutritional and antioxidant properties of the red edible seaweed Porphyra columbina. Int J Food Sci Nutr 65(3): 299-305
- Mouritsen OG (2012) Umami flavour as a means of regulating food intake and improving nutrition and health. Nutr Health 21(1): 56-75.
- Milinovic J, Campos B, Mata P, Diniz M, Noronha JP (2020) Umami free amino acids in edible green, red, and brown seaweeds from the Portuguese seashore. J Appl Phycol 32: 331-339.
- Cherry P, O Hara C, Magee PJ, McSorley EM, Allsopp PJ (2019) Risks and benefits of consuming edible seaweeds. Nutr Rev 77(5): 307-329.
- Marsham S, Scott GW, Tobin ML (2007) Comparison of nutritive chemistry of a range of temperate seaweeds. Food Chem 100: 1331-1336.
- Jimenez K, Kulnigg Dabsch S, Gasche C (2015) Managament of iron deficiency anemia. Gastroenterol Hepatol 11(4): 241-250.
- Marles RJ (2017) Mineral nutrient composition of vegetables, fruits and grains: The context of reports of apparent historical declines. J Food Compos Anal 56: 93-103.
- Kieliszek M, Blazejak S (2016) Current knowledge on the importance of selenium in food for living organisms: a review. Molecules 21(5): 609.
- Gullón P, Astray G, Gullón B, Franco D, Campagnol PCB, et al. (2020) Inclusion of seaweeds as healthy approach to formulate new low-salt meat products. Curr Opin Food Sci 40: 20-25.
- Henchion M, Hayes M, Mullen AM, Fenelon M, Tiwari B (2017) Future protein supply and demand: Strategies and factors infuenicng a sustainable equilibrium. Foods 6(7): 53.
- Geada P, Moreira C, Silva M, Nunes R, Madureira L, et al. (2021) Algal proteins: Production strategies and nutritional and funcitonal properties. Bioresour Technol 332: 125125.
- Bocanegra A, Bastida S, Benedi J, Ródenas S, Sánchez Muniz FJ (2009) Characteristics and nutritional and cardiovasuclar-health properties of seaweeds. J Med Food 12(2): 236-258.
- Mišurcová L, Kráčmar S, Klejdus B, Vacek J (2010) Nitrogen content, dietary fibre, and digestibility in algal food products. Czech J Food Sci 28: 27-35.
- Černá M (2011) Seaweed proteins and amino acids as nutraceuticals. Adv Food Nutr Res 64: 297-312.
- Yamada Y, Miyoshi T, Tanada S, Imaki M (1991) Digestibility and energy availability of Wakame (Undaria pinnatifida) seaweed in Japanese. Jap J Hyg 46(3): 788-794.






























