Montmorillonite versus Kaolinite Clay Minerals in a Sustainable Metal Recovery: Contaminated Soil Washing Technology
L Mgbeahuruike*, MU Dike, RF Njoku Tony, EI Emereibeole, HC Uzoma and EF Anegbode
Environmental Sciences, Federal University of Technology Owerri, Nigeria
Submission: March 15, 2021; Published: May 21, 2021
*Corresponding author: L Mgbeahuruike, Environmental Sciences, Federal University of Technology Owerri, PMB 1526, Nigeria
How to cite this article: L Mgbeahuruike, MU Dike, RF Njoku T, EI Emereibeole, HC Uzoma. Montmorillonite versus Kaolinite Clay Minerals in a Sustainable Metal Recovery: Contaminated Soil Washing Technology. Int J Environ Sci Nat Res. 2021; 28(1): 556229. DOI: 10.19080/IJESNR.2021.28.556229
Abstract
Increased remediation of heavy metal contaminated soil is becoming attractive due to unprecedented degradation of soil systems. Therefore, sustainable remedial approach and the study of contaminant geochemistry are topical areas of intense research. In this study, clay minerals, a constituent part of the soil system has been evaluated as an underlying influence on soil cleaning. Two contaminated soils namely, Copsa mica (SCM) and Campina (SCP) sourced from Romania were subjected to leaching behaviour using two different chelating agents; ethylene diamine tetraacetic acid, (EDTA) and citric acid (CA) on a dynamic system configuration for the purpose of soil cleaning. Geochemical characteristics and metal quantification were conducted on the samples using x-Ray powder diffraction (XRD) and inductive coupled plasma-optical emission spectroscopy (ICP-OES) respectively. Results showed dissimilarity in metal leaching behaviour with higher percentage (four-fold) recovery observed for SCM more than SCP, regardless of extractants used. The presence of montmorillonite clay and other observed mineral oxides influenced the extraction of the targeted metals in SCP other than SCM – induced kaolinite clay.
Keywords: : Heavy metals; Montmorillonite; Kaolinite; Efficiency; Extractants; Contamination; Leaching
Introduction
Soil washing is a remediation technique that has been developed as one of the ex situ traditional methods for heavy metal contaminated soils. The effectiveness of this technique largely depends on the contaminant chemistry and micro-scale heterogeneity (such as soils with reactive minerals as well as fine grained soils) of the subsurface. Indeed, studies have shown that soil washing is efficient on soils with higher permeability [1]. However, the processing of these contaminated soils for washing can be technically challenging because of the presence of clay minerals found in the soils. In most cases, clayey soils are difficult to remediate as a result of low permeability and complex composition the clay is made of. A comprehensive review on the effect of clays associated with soil remediation has been documented [2]. It has been observed that the presence of clays can adversely affect both the operational process and overall economic benefit of remediation performance following solution absorption (water) which changes the rheology, froth stability, and increases reagent consumption, due to high surface area resulting to poor metal recovery [2,3].
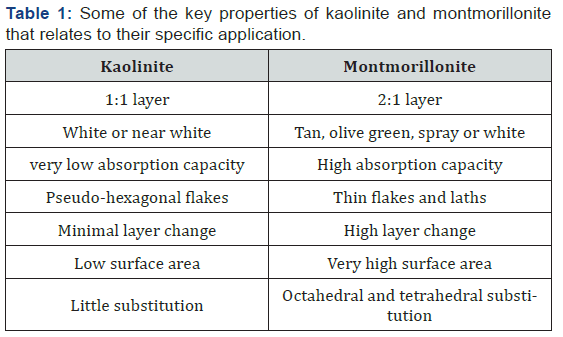
Clay minerals are phyllosilicates which are made up of various combinations of polymeric sheets stacked on each other and bound together. It consists of both a tetrahedral and octahedral sheets. The tetrahedral sheet is made up of silicon-oxygen tetrahedrons with shared basal oxygen molecules. While an octahedral sheet is made up of aluminium-oxygen octahedrons with shared apical and basal oxygen. These tetrahedral or octahedral sheets bond together through hydrogen bonding resulting to 1:1 or 2:1 layered structure. The 1:1 layer structure is referred to as kaolinite clay. It consists of a unit made up of one octahedral and one tetrahedral sheet. The kaolinite layers are strongly held together such that, they are electrically neutral, and therefore, will not attract positively or negatively charged species (ions) and they rarely swell in water.
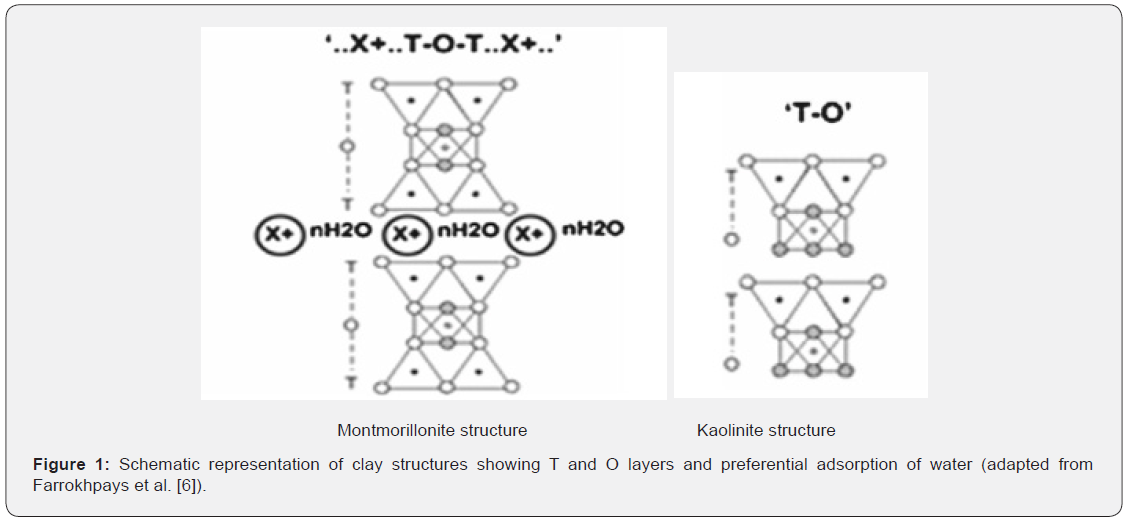
On the other hand, the 2:1 layered structure, which is referred to as the montmorillonite (smectite group) clay, consists of two tetrahedral sheets with one bound to each side of an octahedral sheet. The montmorillonite layers are held loosely, creating voids for water penetration, a phenomenon that gives rise to inter-crystalline swelling (a process in which the interlayer distance can increase to 1nm). Due to ion substitution in the layers, some of these clay minerals have a negative charge on the surface and will attract water molecules or hydrated cations readily. The negatively charged surfaces of 2:1 clay attracts positively charged water molecules, allowing the water molecules to enter between the layers as shown in Fig.1. However, the fundamental building blocks of kaolinite and montmorillonite are very similar for both the tetrahedral and octahedral sheets, except for the arrangement and compositions of the octahedral and tetrahedral sheets which, accounts for the major and minor differences in the physical, chemical and mechanical properties that controls the application of a particular clay mineral [4]. Some of these properties are highlighted in Table 1 below.
Studies [5] have also shown that most clay, especially the montmorillonites, exhibits rheological behaviour that is occasioned by inter-crystalline swelling in the presence of water and the resultant formation of thixotropic gel. This phenomenon is caused when charges occurring in the inner structure of the fluid form flocs and aggregates under the influence of Vander Waals and electrostatic forces.
Clay minerals also exhibit absorptive tendencies for metals over a higher pH range due to their crystallo-chemical properties. Several studies on soil chemistry have demonstrated the ability of clays and clay minerals to selectively absorb metal cations (Zhu and Alva 1993; Carey et al. 1996). In addition, extensive research has assessed the metal absorption characteristics of natural and/or modified clays and mineral clays (Fu et al. 1991; Smith et al. 1996) for their different mechanisms of mineral sorption, including outer-sphere complexes, inner-sphere complexes and multi-nuclear surface complexes (O’Day et al. 1994; Scheidegger et al. 1996).
The aim of this study is to assess the desorption behaviour and geochemical intricacy associated with the remediation of heavy metal contaminated sites of a montmorillonite and/or kaolinite clay soil. Leaching trends will be explored to determine desorption mechanisms associated with chemical reactions and diffusion processes in a dynamic system. Results from this study can provide technical and theoretical insight on contaminant dynamics that affects remediation efficiency for soil washing technique (Figure 1).
Materials and Experimental Methods
Chemicals and materials
The reagents used for this study were of analytical grade. Three base metals (Cd, Cu and Pb) were selected as target metals because of their toxicity and ubiquitous nature in contaminated sites. Leaching agents: ethylene diamine tetraacetic acid (EDTA) and citric acid (CA, were purchased from Sigma Aldrich and Alfa Aeser respectively. All mineral acids used in the analysis were of analytical or spectral grade. Soil samples used in this study, were sourced from Romania and were designated as Copşa-mića (SCM) and Campina (SCP) soils of metallurgical smelter and industrial waste sites respectively. Brief background of SCM and SCP sources is described below:
a) Copşa-mića (SCM)
SCM is a study site located within an industrial town in Sibiu County, Transylvania, Romania. The physiographic characteristic is influenced by the hauling of sand/aggregates activities carried out on daily basis. SCM is considered one of the most polluted towns in the province considering its industrial activity status. The Carbosin and Sometra metallurgical smelter are the two major factories of prominence in the province in the early 1960s, although their operation activities are now moribund. They are known as major pollutant sources responsible for the emission of black carbon and non-ferrous substances.
b) Campina (SCP)
SCP is a study site located within an industrial town housing both the Romanian star oil and the central Orion foundry in Prahova County, Romania. The physiographic characteristics are attributed to the historic dumping of industrial waste from sulphur dioxide and steel manufacturing activities which apparently defines the soil as technosol.
Experimental methods
The soils (SCM and SCP) were analysed for their physical and chemical properties (Table 2) using standard techniques and approaches. For brevity, pH, specific surface area (SSA), particle size distribution, cation exchange capacity (CEC), and baseline metal profile were determined as described in Mgbeahuruike et al.[3]. All metal concentrations were determined by ICP-OES and sample concentrations were corrected for matrix effects as reported in Mgbeahuruike et al. [3]. The percentage metal leached from the soil was calculated using equation 1:
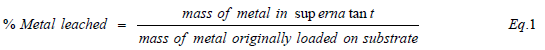
To ensure repeatability, this procedure was carried out in triplicate for all substrates.

Experimental methodology
Measured samples (20g ± 0.0005g) of SMC and SCP were subjected to column leaching experiment with the leaching agents (EDTA and CA) used to extract the metals of interest. The leaching behaviour of the three metals of interest were investigated by subjecting the SCM and SCP samples to an in situ leaching process (not shown here), in a glass chromatographic column (dimension: internal diameter 2cm and 30cm in height) with a sintered disc frit to control flow rate as discussed in Mgbeahuruike[3]. The leaching trend for these metals were monitored over a leaching time interval of 30, 60, 90, 120, and 180min using EDTA and CA with molar concentrations of 0.034 and 0.033 (moldm-3) respectively. After mixing of the soil and solution at the specified intervals, the suspensions were centrifuged at 3000rpm, to enable separation of the liquid from the solid. Following centrifugation, 1mL aliquot (washing solution) from the supernatant in each of the tubes were filtrated with a syringe filter (0.45µm) into a plastic vial of 15mL capacity and later made up to 10mL with deionized water. Subsequently, the filtrates were analysed by ICP-OES and the selected metal concentrations retained in the adsorbent phase (mgkg-1) were calculated.
The results are expressed in mass of metal per kg soil extracted after 180min.
EDX measurement
The clay minerals (montmorillonite and kaolinite) were analysed using EDX spectrometer PW 4025-miniPal-panalytical. This instrument is a non-destructive device and it is simple, inexpensive and rapid. The miniPal 2s detector combines high efficiency with a high resolution. The measurement was successfully performed with the following experimental parameters: emission current max: 1mA; High voltage tube: max 30kv; Power: max 9w (Figure 2).
Results and Discussion
Soil description
Table 2 shows the physicochemical characteristics of the soils (SCM and SCP) under consideration. The CEC and SSA values of SCM (21.6meq/100g & 14m2/g) and SCP (16.7meq/100g & 23m2/g) are relatively similar and falls within the range of a kaolinite clay values in agreement with existing literature. The CEC and SSA are common factors that determine the physical, mechanical and flow properties of soils and which generally influence their adsorption capacity. Therefore, it is expected that both soils may exhibit similar leaching tendencies for targeted metals.
In comparison of mineral profiles determined using X-ray diffraction for the SCM and SCP, quartz and kaolinite were confirmed as dominant crystalline phases present in both soils (diagram not shown). However, significant differences in mineral phases such as goethite, graphite, and calcite were identified in both soils too. The presence of kaolinite in SCP and SCM is an indication that the ability of these soils to retain cations will be solely influenced by CEC. However, the presence of goethite (Fe2O3) and graphite found in the SCM and SCP are available as varieties of natural mixture that can play important roles in chemical and physical attributes of the soil (Pinheiro-Dick & Schwertmann 1996). The prevalence of Fe3+ can enhance the aggregate stability of kaolinite clay by impacting on its leaching potentials (Bronick & La 2005).

The pH of SCM and SCP are observed to be alkaline inclined, 7.2 and 8.3 respectively. This phenomenon may affect the leaching dynamics of these soils because pH is found to influence the metal retention ability of a soil with metals typically being less likely to be leached from an alkaline phase than their acidic counterparts, though changes in geochemical environment could play a domineering role in the mobilization of metals [7].
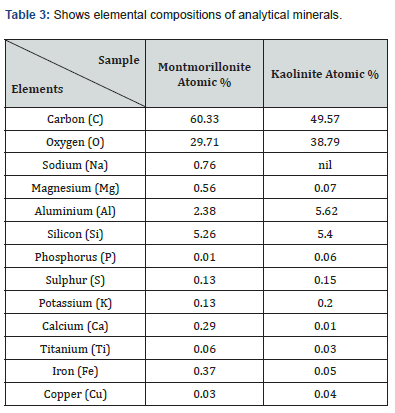
Analysis of soluble metal concentrations of SCM and SCP show significant variations in the values of the targeted metals. Ni and Cu in SCP were 2 and 3 folds higher than same metals in SCM respectively. However, Pb concentration was substantially higher in SCM than its counterparts in SCP. The variation in metal concentrations could reflect the differences observed in anthropogenic activities and inputs between the individual sites. Also, the compositions of soil samples contained in Table 3, have montmorillonite (bentonite group) and kaolinite clay as main dominant minerals of the soils under investigation. It could be observed that the composition ranges ancillary elements (Fe, Cu, Ca, S, P and K) to dominant elements (Si, Mg, C and O). The presence of these trace elements may significantly affect the leaching potentials of the targeted heavy metals.
Column leaching process
In situ extraction leaching was conducted for SCM, and SCP, using the column approach and a solid liquid ratio of 1:10 to investigate the extraction of the metal species of interest. The leaching trend was determined by leaching concentrations (mgkg-1) of targeted metals from the two soils after leaching intervals of 30, 60, 90, 120, and 150min contact time with selected extractants (EDTA and CA). The results (indicating the mass of metal per kg soil extracted after 150min, as well as the % extraction efficiency for each metal) are calculated using equation 1 and are summarized in Table 4 and displayed graphically in Figure 3.

Leaching in natural soils (SCM and SCP)
The metal leaching as summarized in Table 4 and graphically illustrated in Figure 3a & 3b for SCM, showed different extraction efficiencies across the two extractants (EDTA and CA) investigated. Cd, Cu, and Pb extracted from SCM were prominently leached by EDTA as displayed in Figure 3a. The leached metals are reported as 22.9mgkg-1 (44%), 66.9mgkg-1 (65%), and 881.8mgkg-1 (55%) for Cd, Cu, and Pb respectively. The box plot shows differential extraction behaviour across the metals under consideration. Significant variation was observed for in the concentrations of metal leached which was expected, considering the metal burden (mgkg-1) by the individual metals under consideration. However, the average leaching efficiency for these metals were relatively similar and further illustrated by the median distribution prominently skewed to the right for the Cd, Cu and Pb (Figure 3a).
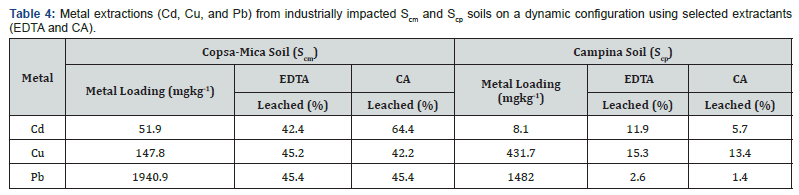
For SCP, EDTA leaching over the three metals were observed to be prominently poor as summarized in Table 3 and displayed in Figure 3b. Cd, Cu, and Pb leached 0.96mgkg-1 (11.5%), 66.1mgkg-1 (15%) and 39.1mgkg-1 (2.6%) respectively. The average leaching efficiency was significantly poor for Pb compared to Cd and Cu. This is further illustrated in the boxplot (Figure 3b), showing the distribution for the three metals being skewed to the right with Cd and Cu displaying similar extraction behaviour different from that observed for Pb. The leaching capacity of SCP was about four times lower than the recovery efficiency of SCM regardless of the extractants investigated. This poor extraction efficiency exhibited by SCP could be attributed to the physicochemical properties described in Table 2. The CEC and surface area of SCP are significantly lower than those of SCM, and as such, should be expected to leach poorly. Also, considering the results shown in Table 3 and graphically illustrated in Figure 3a & 3b, it could be deduced that the presence of montmorillonite in SCP attracted much of the cations (Cd, Cu, and Pb) to the region close to a negatively charged mineral surface by ion exchange reaction. Moreover, the presence of flocs and aggregates of mineral oxides (goethite, calcite) formed in the interlayer of the montmorrillonite could intercalate these cations by causing a swelling tendency due to its crytallo-chemical properties which eventually could prominently interfere with the leaching dynamics as have been observed in SCP [8]. The enhancement efficiency on the desorption of Cu and Pb demonstrated by EDTA over Cd could be attributed to its complexation affinity for both Cu (Log K = 18.8) and Pb (Log K = 17.9) compared to Cd (Log K = 10.7) (Gupta et al. 2000 ) [9]. The effect of stability constants observed for metal complexes appears to be effective considering the fact that kaolinite dominates the SCM soil, having its interlayer structure overlapped by hydrogen bonds (1:1 layer), thereby enabling leaching due to its low viscosity and absorptivity (Table 1). This phenomenon provides the extractants access to reach most of the metals at the outer layer surfaces (labile phases), where the attractive forces are more sites specific with reduced hydration layers around the cations, exposing significant fraction of the targeted metals for rapid dissociation [10,11].
In the case of CA, the extraction efficiencies of Cd, Cu and Pb from SCM and SCP are summarized in Table 4 and also displayed in Figure 3c & 3d. For SCM, it was observed that CA also prominently exhibited extraction capability and efficiency as observed with EDTA-SCM. CA efficiently leached 32.9mgkg-1 (64%), 66.9mgkg-1 (45%), and 881.8mgkg-1 (45%) of Cd, Cu and Pb respectively. The box plot in Figure 3c shows the differential variation in extraction behaviour of the individual metals. The extraction and median distribution are observed to be skewed to the right for Cu and Pb while Cd remained fairly symmetrical. However, for SCP, leaching potential for CA was observed to be significantly poor for all metals as summarized in Table 4 and Figure 3d. The average leaching efficiency for the metals were below 15% and reported as 0.46mgkg-1 (5.7%), 58.4mgkg-1 (13.4%) and 0.5mgkg-1 (1%) for Cd, Cu and Pb respectively. The box plot in Figure 3d shows the differential extraction behaviour across the metals having Cd and Pb prominently skewed to the right while Cu remained fairly symmetrical. Generally, the poor leaching efficiency by EDTA and CA observed for SCP was evidently attributed to physicochemical characteristics mentioned earlier. Although, it was observed that better recovery efficiency was achieved for CA-Cd when compared to the other metals leached with EDTA. This reason was adduced to higher stability constant (Log K = 11.3) and acid effect reaction of citric acid (CA). Also, the reason for the acid effect of CA is better substantiated with the mobilization of CdL2- instead of precipitating Cd(OH)2. However, during the metal leaching of Scp with the extractants, it was observed that effervescence of CO2 was eminent, an indication that suggests the dissolution of CaCO3 minerals according to the reaction of Eq.2. The presence of Ca2+ in the reaction can interfere in the leaching process which incidentally could undermine recovery efficiency of the process. It has been confirmed that CA is more likely to be found complexed more rapidly with calcium, followed by iron and magnesium being the lowest to metabolise (Kiom et al. 2000; Lensbouer et al. 2008).

The presence of the different forms of minerals Fe in SCP could also influence the physical and chemical processes that occurred in the soil (Carrillo-Gonzales et al. 2006), and subsequently influenced the extraction efficiency of the leaching process. For instance, the formation of magnetite (Fe3O4) in soils as described in Eq.3 has the capacity to adsorb heavy metals in an aqueous medium through physical and chemical interactions [12]. Recent studies [13,14] have demonstrated high affinity of EDTA (Log Kedta-Fe =25.1)) for iron oxides which affects colloidal dispersability, affect the decrease in high surface energy, and consequently decrease iron particles agglomeration (Jackson & Milter 2000) [15-21].
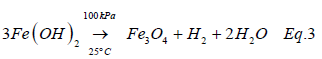
Conclusion
In this study, a soil washing technique was applied to remediate selected metal contaminated soils from historic dumping sites of metallurgical smelter and industrial waste sites. The EDX spectra revealed the various trace elements inherent in both kaolinite and montmorillonite clays which played significant roles in influencing the leaching potentials of SCM and SCP respectively. It was noted that higher percentage recovery efficiency was observed for SCM more than for SCP, a phenomenon attributed to the crytallo-chemical properties of montmorillonite over the kaolinite in SCP. Though, the presence of ancillary minerals (calcite and goethite (Fe2O3) in SCP also played a retarding role in the extraction of these metals by forming stable complexes with the extracting substances.
Conflicts of Interest
The authors declare that they have no conflict of interest regarding the proposed publication of this manuscript.
Acknowledgement
This study was funded by Tertiary Education Trust Fund (TET Fund) Nigeria as part of postgraduate study at Manchester Metropolitan University, UK.
References
- Mao X, Yang X, Shao X, Han F, Chang T, et al. (2018) Enhanced techniques of soil washing for the remediation of heavy metal-contaminated soils. Agricultural Resource 7: 99-104.
- Ndlovu B, Farrokhpay S, Bradshaw D (2013) The effect of phyllosilicate mineral on mineral processing industry. International Journal Mineral Process 125: 149-156.
- Mgbeahuruike LU (2018) An investigation into soil pollution and remediation of selected polluted sites around the globe. PhD thesis, Manchester Metropolitan university, UK.
- Tanchuling MA, Khan MRA, Kusakabe O (2005) Sorption of zinc in bentonite, illite and kaolin clay using column leaching test. Contaminated Soils, Sediments and Water, pp. 251-263.
- Murray HH (1991) Overview clay minerals. In: Chemica 2013, Brasbane.
- Farrokhpay S, Ndlovu B, Bradshaw D (2016) Behaviour of swelling clays versus non-swelling clays in flotation. Minerals Engineering 96-97: 50-66.
- Gadd JM (2008) Transformation and mobilization of metals, metalloids, and Radionuclides by microorganisms. In: Violinte A, Huang PM, Gadd GM (Eds.), Biophysico-chemical processes of metals and metalloids in soil Environments. Willey-Jupac series, Vol. 1 John Willey and Sons, Hoboken, NY, USA, pp. 53-96.
- Ndlovu B, Farrokhpay S, Forbes E, Bradshaw D (2015) Characterization of kaolinite colloidal and flow behaviour via crystallinity measurements. Powder Technology 269: 505-512.
- Lombi E, Zhao FI, Dunham SJ, McGrath SP (2001) Phytoremediation of heavy metal contaminated soils: natural hyperaccumulation versus chemically enhanced phytoextraction. Journal of Environmental Quality 30: 1919-1926.
- Laired DA (1996) Model for the crystalline swelling of 2:1 phyllosilicates. Clays and Clay minerals 44: 553-559.
- Laired DA (1999) Large charge influence on the hydration of expandable 2:1 phyllosilicates. Clays and Clay Minerals 47(5): 630-636.
- Tang SCN, Lo IMC (2013) Magnetic nanoparticle: essential factors for Sustainable environmental applications. Water Resources 47(8): 2613-2432.
- Warner CI, Addleman RS, Cinson AD, Droubay TC, Engelhard MH, et al. (2010) High-Performance, Super paramagnetic nanoparticle-based metals sorbents for removal of Contaminants from natural waters. ChemSuSChem 3(6): 749-757.
- Yang J, Zeng Q, Peng L, Lei M, Song H, et al. (2013) La-EDTA coated Fe3O4 Nano-material: Preparation and application in removal of fisfate from water. Journal of Environmental Sciences 25(2): 413-418.
- Balan E, Allard T, Boizot B, Morin G, Muller JP (1999) Structural Fe3+ in natural kaolinites: new insights from electron paramagnetic resonance spectra fitting at X and Q-band frequencies. Clays and Clay Minerals 47(%): 605-616.
- Evans LJ (1989) Chemistry of metal retention by soils. Environmental Science and Technology 23(9): 1046-1056.
- Labanowski J, Monna F, Bermond A, Cambier P, Fernandez C, et al. (2008) Kinetic extraction to assess the mobilization of Zn, Pb, Cu, and Cd in a metal contaminated soil: EDTA versus Citrate. Environmental Pollution 152(3): 693-701.
- Muller JP, Manceau A, Allard T, lldefonse PH, Hazemann JL (1995) Crystal chemistry of kaolinite and Fe-Mn oxides: relationship with formation conditions in low-temperature systems. American Journal of Science 295(9): 1115-1155.
- Murray HH (2000) Traditional and new applications for kaolin, smectites and palygoskite: general overview. Applied Clay Science 17(5-6): 207-221.
- Mūller vonmoos M, Loken T (1989) The shearing behaviour of clay. Applied Clay Science 4(2): 125-141.
- Petruzzelli D, Hellferich FG (1993) Migration and fate of pollutants in soils and subsoils. NATO ASI Series, vol. G 32 Springer-Verlag Berlin Heidelberge, Germany.






























