Soil Remediation by Removal of Cu and Ni with an Unconventional Organic Solution
L Mgbeahuruike1*, LJH Potgieter2, MU Dike1, RF Njoku Tony1, HC Uzoma1 and UC Ndukwe1
1Department of Environmental Sciences, Federal University of Technology Owerri, Nigeria
2School of Chemical and Metallurgical Engineering, University of the Witwatersrand, South Africa
Submission: March 15, 2021;Published: April 07, 2021
*Corresponding author: L Mgbeahuruike, Department of Environmental Sciences, Federal University of Technology Owerri, PMB 1526, Nigeria
How to cite this article: L Mgbeahuruike, LJH Potgieter, MU Dike, RF Njoku T, HC Uzoma, et al. Soil Remediation by Removal of Cu and Ni with an Unconventional Organic Solution. Int J Environ Sci Nat Res. 2021; 27(5): 556221.DOI: 10.19080/IJESNR.2021.27.556221
Abstract
The contamination of soils with toxic metals is a major ecological concern, especially in close vicinity of automobile workshops. This study focuses on the extraction of copper (Cu) and nickel (Ni), using 2, 4-pentanedione (Hacac) dissolved in a non-polar organic solvent (methylated spirit). The objective of the investigation was to investigate the target metals’ mobilization and its secondary Fe mineral dissolution induced by the soil washing treatment. Operating conditions such as soil/solution ratio (1:10), acidity (pH 4.7) and extraction time (150min) were monitored during the process.
Results showed that the optimum extraction efficiency (>50%) for Cu and Ni occurred at low pH. However, the extraction process revealed a simultaneous high Fe dissolution (up to about the 35% of the total Fe content in the soil) in the form of ferric triacetylacetonate (Fe (Hacac)3) complex, suggesting that the predominant fraction of the targeted metals extracted were occluded in the Fe-Mn oxides fraction. The desorption process of the metals extracted included fast and slow phases. The kinetics of the extraction process can be described by an Elovich function (R2 values ≥ 0.93), which is typically associated with a heterogeneous diffusion process. Overall, the test result affirms the effective removal of Cu and Ni with Hacac. The high degree of iron dissolution which occurs simultaneously suggests that a bulk micronutrient chemical evaluation is essential within the soil when an extractive decontamination activity is proposed for site remediation.
Keywords: Heavy metals; Extraction; Leaching; Efficiency
Introduction
Soil contaminated with heavy metals constitutes a serious environmental problem throughout the world [1]. This is most worrisome in developing countries, where intervention values are much higher than in the developed world. Various anthropogenic activities, especially automobile repairs, generate and accumulate substantial metal-bearing wastes around the vicinities where they operate [2] and this creates environmental contamination at unacceptable levels. The impact of these wastes (metals) could be potentially deleterious and could have far reaching consequences due to its mobility, toxicity and persistence in the environment and living organisms exposed to them [3,4]. Although many of these metals occur naturally in the soil, and are intrinsic elements of the earth’s crust, inordinate amounts introduced into the soil can present a potential environmental and health risk, depending on the chemical forms of the metals. The latter aspect controls both its mobility and reactivity in the environment [5].
The forms and pathways presented by some of these metals have greatly increased their chances of human exposure via a source-pathway-receptor mechanism. These contaminants can persist and accumulate in plants and animals after their release into the ecological system due to their stability, biological and non-degradability potentials thereby endangering human and animal health. This phenomenon can be attributed to some of the respiratory-related diseases common in our contemporary world which invariably is implicated to the high burden of heavy metals via the food chain [6]. This exposure could result to acute or chronic diseases with concomitant effects. For instance, Ni exposure during work activity can cause bronchial asthma or other associated occupational lung diseases [7]. Animal studies in the past confirms [8] that nickels are primarily deposited in the lungs particularly when associated with insoluble NiO, followed by moderately soluble Ni3S2 and insoluble NiSO4. Thus, their (Ni, Cu) removal from contaminated soils becomes critically important in the close vicinities of automobile repair shops.
Technologies used for soil remediation include, but not limited to, solidification/stabilization, thermal treatment, physical separation and froth flotation [9]. These processing techniques are associated with high costs, high energy consumptions and high greenhouse gas emissions. However, soil washing has
been developed as one of the ex situ conventional remediation
methods for remediation of heavy metal polluted soils. It works
by dissolving or suspending the undesirable soil contaminants
in a washing solution, which is typically an aqueous solution of
a chelating agent which allows heavy metal desorption from soil
solid phase by forming strong and water-soluble metal ligand
complexes. This approach does not cause deterioration of soil
physico-chemical properties and it is economically efficient [10].
The most commonly used chelating agents are typically weak
organic acids and the aminopolycarboxylic acids, e.g., ethylene
diamine tetraacetic acid [EDTA], because of their high chelating
ability and capacity to enhance the metal extraction from the soil
solid phase [11,12]. However, EDTA is less efficient in extracting
metals bound to the reducible/Fe-Mn oxide fraction in soils
[13,14] and is practically inefficient and unavailable for metals
occluded in the residual fraction of the soil [15]. This observation
was corroborated by the sequential chemical extraction
conducted by Lee & Kao [16]. These authors observed a higher
removal efficiency of heavy metals associated with exchangeable
fractions, than those strongly adsorbed onto the Fe-Mn oxides
fraction. Hence, there is still a need for sustainable and effective
chelating agent that has the extractive capacity to enhance metal
solubility from these recalcitrant soil solid phases. Previous
studies have demonstrated the enormous potential of Hacac for
iron extraction from iron ore fines (11). However, the specific
combined extraction of metals (Cu and Ni) from contaminated
soils has not been previously reported prior to our study making
our work eminently novel.
Acetylacetone (C5H8O2), also referred to as 2,4-Pentanedione
(Hacac), is a chelating agent of ẞ-diketones. It is a volatile ligand
at room temperature with a density of 0.975g/ml, and it boils
at 140oC under atmospheric pressure. Hacac has a bidentate
structure because it can bond to the central iron via both oxygen
atoms.
Currently, a significant increase in environmental pollutants
have occurred around soils surrounding automobile services and
repair activities, as observed and reported by Nwachukwu et al.
[17]. Some of the wastes occluded in the soil include wastes from
solvents used for cleaning metallic parts, asbestos from brake
pads, and corrosion of scrap auto panes discarded indiscriminately
within the working environs. The chemical components of these
wastes can be tremendously toxic when absorbed by the skin,
inhale to the lungs or adsorbed in soils in the vicinity of automobile
workshops. While the automobile activity is of great economic
value, the ecological impact thereof has become a serious problem
affecting land-use, water and public health management.
Several studies [17,18] to assess the heavy metal
concentrations and the ecological and health risk associated with
automobile waste in the Owerri metropolis Nigeria have been
conducted in the past with no sufficient and sustainable data to
date has been presented to potentially remediate or reduce the
concentrations of these heavy metals in the soil to a safe threshold
level. Therefore, the aim of this present work was to evaluate the
soil contamination of Nekede mechanic/automotive workshop
for remediation purposes by leaching of targeted metals (Cu and
Ni) with an unconventional soil washing agent that are more
commonly applied in mineral processing, i.e., Hacac dissolved in
non-polar solvent (methylated spirits). For this purpose, a-column
based configuration was adopted. The efficiency of the washing
treatment, and its secondary effect on Fe dissolution in the soil,
was assessed.
Materials and Method
Study area
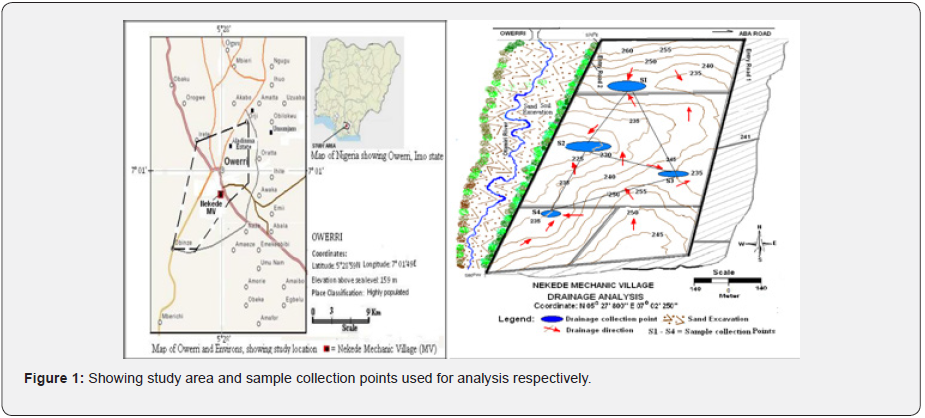
The study area, indicated in Fig.1a, is located in Nekede
mechanic village, which is considered to be the largest functional
mechanic village in South-Eastern Nigeria. Nekede is a town within
the Otamiri watershed and ranges longitudinally between 7o03’
and 7o05’ and 5o26’ and 5o26’ in latitude. The climatic conditions
of the area have the following characteristics: temperature range
of 21oC to 27oC, relative humidity ranges between 60-80%. The
area lies within the sub-equatorial, sub-humid region with March
to October experiencing a rainy season and November through
to February experiencing a dry season. Annual average rainfall
is approximately 1500mm with a monthly average rainfall of
30mm. The communities are mainly agrarians and farm within
the vicinity of the mechanic village. This creates the possibility for
increased levels of heavy metals across the food chain and results
in bioaccumulation of the metals by living organisms.
The soil used in the study was collected from a polluted
automobile site in Owerri, Imo state, Nigeria. Soil samples were
randomly collected at drainage points along the natural flow
direction from the site, within the auto-mechanic surrounding
at each location, as shown in Figure 1b. The sample collection
was done in the order of different depths, i.e. (0-15)cm, (15-30)
cm, and (30-45)cm, within a 10m radius of each other, using
a soil auger. The collected samples were later homogenized to
make a composite sample for the area under investigation. The
homogenized sampled soil was dried at 40oC and sieved through a
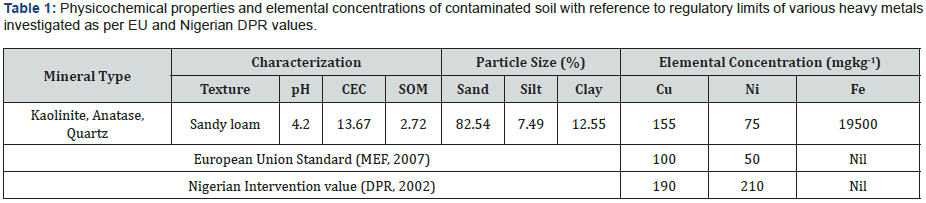
2mm mesh sieve. Soil properties are shown in Table 1.
2,4-Pentanedione (Acetylacetone-Hacac) and industrial
methylated spirit (IMS) used for this study were purchased
from Alfa Aeser. Throughout the investigation, high purity
deionized water (18.2mΩcm) was used. All glassware and
Polytetrafluorethylene (PTFE) containers used were previously
soaked in 10% (v/v) nitric acid for 12h and then rinsed with high
purity deionized water prior to use.
Standard techniques and approaches were employed to
determine the physical and chemical properties of the soil samples.
Total organic content was estimated by Loss on Ignition [19]. The pH was measured at solid: liquid ratio 1: 10 after mechanical
agitation at 120 revolution min-1 using a reciprocating shaker
for a minimum of 60min (Mettler Toledo, [20]. Specific surface
area (SSA) and Cation Exchange Capacity (CEC) were measured
using standard methods by Cokca & Birand [21] and Santamarina
et al. [22], respectively. The particle size analysis was done with
a Malvern mastersizer 2000, and the acid soluble metal content
of the soil was determined after dissolution in aqua regia. The
dissolved Cu, Ni, and Fe concentrations were measured with ICPOES
and sample concentrations were corrected for matrix effects
by procedural blanks, normalized to sample mass and reported as
mgkg-1 dry weight.
The column experiment was performed using an experimental setup (in triplicate) that consisted of a separating funnel as a reservoir, a glass chromatographic column (internal diameter 0.2cm and 30cm in height) equipped with a sintered disc frit to regulate flow rate. The leaching solution (80% Hacac in 20% IMS) was introduced into the column using via the reservoir and a constant head of 10cm3 was used to deliver influent solution at a flow rate of 0.8cm3. min-1. The soil sample was uniformly packed (8cm) using a slurry technique and sandwiched between two thin layers of sand (0.5cm) each. The leachate was collected at selected time intervals over a total leaching time of 150min. Flow and pH (4.2± 0.2) were measured immediately after sample collection. The metal concentrations (Cu, Ni, and Fe) were measured using inductively coupled plasma optical emission spectroscopy (ICP-OES) at the Manchester Metropolitan University. The concentration of Hacac ensured an excess of extractant for each of the target metals concerned. The percentage metal leached from the soil was calculated using equation [1] as follows;
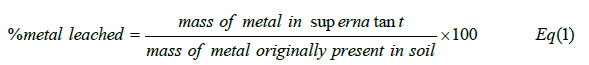
Results and Discussion
Soil characterisation
The soil parameters were characterized based on colour
(Munsell), CEC, mineral profile, and pH as summarized in Table 1.
The soil exhibited a reddish brown colour (Munsell 5YR.5/1),
which indicates the presence of red earths, characterized as
ferric oxide. The CEC gave a low value (13.7), suggesting a low
metal exchange capacity. This could likely influence the leaching
dynamics of the soil. X-ray diffraction reveals quartz, kaolinite,
and anatase as major minerals in the soil. Due to the presence of these highly weathered clay minerals, the ability for the soil to
retain cations maybe solely influenced by surface area. The pH
(4.2) is acidic, which can assists metals in the two leach readily.
The soil bulk geochemistry suggests that the metals of interest
are present at levels higher than the EU regulatory standards, but
lower than that set for DPR (Department of Petroleum Resources)
values in Nigeria.
Column extraction
In situ column leaching experiments with a solid: liquid ratio of 1: 10 were used to investigate the extraction behaviour of Cu, Ni, and Fe bound in and to the soil components. The leaching concentrations of metals (mgkg-1) extracted from the contaminated soil after 30, 60, 90, 120 and 150 minutes contact time with Hacac are summarised in Table 2.
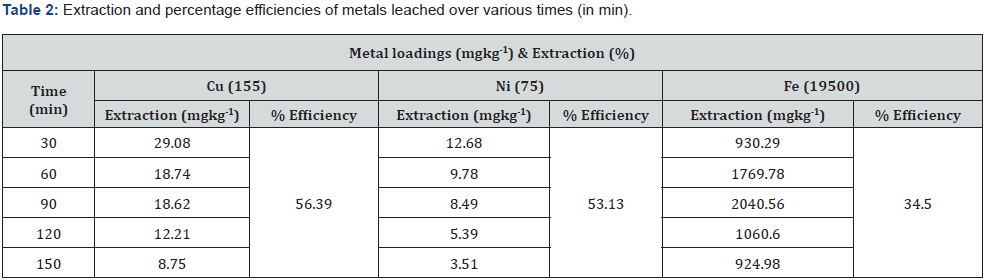
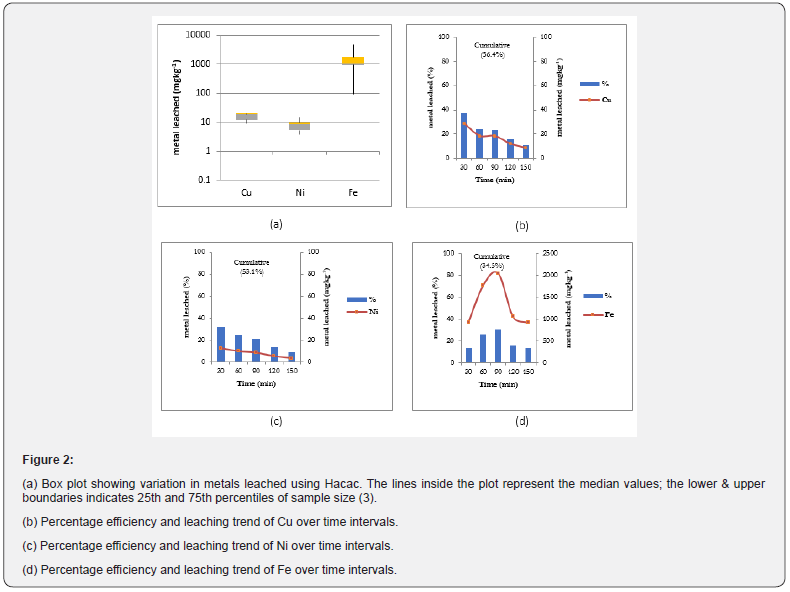
The extraction performance of Hacac at chelant-to-metal molar ratios of 2:1 under acidic conditions is illustrated in Figures 2 (a, b, c, and d). It is clear that there are considerable differences in the metals leached (mgkg-1) and cumulative extraction efficiencies (%) that were observed for the three metals (Cu, Ni, and Fe). Figure 2 (b, c, and d) are graphical representations of the percentage leaching efficiency for the Cu, Ni and Fe over time. For Cu, the amount of leaching indicates a downward trend, with the highest extraction occurring within 30min before eventually flattening off (Figure 2b). The total cumulative leaching is 56.4%. The leaching of Ni was observed to maintain a fairly uniform downward trend with highest extraction occurring at 30min after the start time and then it gradually flattens off (Figure 2c). The total cumulative leaching efficiency was 53.1%. However, Fe leaching exhibited a leaching trend that progressively attained a maximum peak at 90min before displaying a steep drop. The total cumulative extraction was 34.5%, which is quite a bit lower than in the cases of Cu and Ni (Figure 2d). Sequential extraction work reported by Tessier et al. [23], indicates that the extraction behaviour and amounts of each metal leached can be explained by the dominating phases commonly present in the soil, e.g., Fe-Mn oxide, exchangeable and carbonate phases. The effectiveness of Hacac to mobilize Cu and Ni within the short times observed may be attributed to its reaction kinetics, active chemical affinity and specificity of the metal-ligand interaction in the reactions [24]. The simultaneous extraction of Fe is due to the significant affinity between Hacac and Fe (Log Kfe-hacac = 9.30). This is higher than the stability constants for Cu (Log KCu-hacac = 8.24) and Ni (Log KNi-hacac = 4.96) with acac [25], respectively.
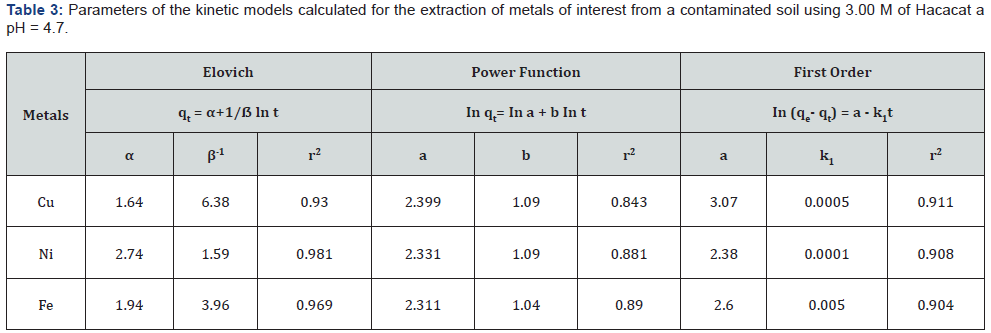
N/B: α and ẞ are Elovich constants, r2 is coefficient of constant determination, a and b are constants (rate coefficient value).
Kinetic studies
From the results presented in Table 2, one can deduce the kinetics of the Cu, Ni, and Fe extraction processes from the contaminated soil. Three models (Elovich, power function and 1st order function) were fitted to the data to describe the extraction dynamics. These models are commonly used in literature [26,27]. The parameters of the three types of models are presented in Table 3. Based on the values of the regression coefficients (R2) it can be concluded that the Elovich model best describes the extraction kinetic behaviour when compared to the other two models [28]. The values of α and β-1 variables (Table 3) were typical of fast and slow desorption processes, an indication that diffused extraction may have occurred. This phenomenon often occurs during coordination interactions in the extraction process [29].
Conclusion
The use of Hacac dissolved in methylated spirits as an unconventional soil remediation solution to extract Cu-Ni from soil at a contaminated site demonstrated an efficient process with average efficiency of >50% Cu and Ni leached. The kinetic extraction was found to involve the initial metal dissolution via pH-dependent, followed by the development of exchange reactions between the metal-Hacac complexes. The kinetics of the extraction process for the heavy metals Cu and Ni obey the Elovich equation. The kinetic model derived in this study could be utilized in the design of a reactor which can be employed for a batch remediation process. While metal recovery has not specifically been addressed in this investigation, metal recovery from the extracted M (acac)x complexes are theoretically possible via one of two routes, which will both simultaneously regenerate the Hacac extractant as part of the process. In the one instance hydrogen gas can be used as metal reactant, which would yield nano-metal particles. In the second case one can use hydrogen sulphide to first precipitate the metals as metal sulphides, and then roast the metal sulphides in a reducing hydrogen atmosphere. This will simultaneously reduce the metal and regenerate hydrogen sulphide for reuse in the precipitation step. A useful follow up of this investigation will therefore be a techno-economic feasibility study of the extraction process and subsequent metal production to ascertain its financial viability.
references
- Mclaughlin MJ, Parker DR, Clarke JM (1999) Metals and micronutrients food safety issues. Field Crop Res 60(1-2): 143-163.
- Abanuz GY (2011) Heavy metal contamination of surface soil around Gebze industial area, Turkey. Microchem Jour 99(1): 82-92.
- Klavins M, Briede A, Rodinov V, Kokorite I, Parele E, et al. (2000) Heavy metals in Rivers of Latvia. Sci Total Environ 262(1-2): 175-183.
- Yuan BZ, Jefferson AM, Baldwin KT, Thorgeirsson SS, Popescu NC, et al. (2004) DLC-1 operates as a tumor suppressor gene in human non-small cell lung carcinomas. Oncogene 23(7): 1405-1411.
- McBride MB, Martinez CE, Topp E, Evans L (2000) Trace metal solubility and speciation in calcareous soil 18 years after no-till sludge application. Soil Sci 165(8): 646-656.
- Tchounwou PB, Centeno JA (2008) Toxicologic pathology. In: Gad SC (Ed.),Handbook of Pre-Clinical Development. New York. NY: John Wiley & Sons, USA, pp. 551-580.
- Estlander T, Kanerva L, Tupasela O, Keskinen H, Jolanki R (1993) Immediate and delayed allergy to nickel with contact urticaria, rhinitis, asthma and contact dermatitis. Clin Exp Allergy 23(4): 306-310.
- Dunnick J, Elwell M, Benson J, Hobbs C, Hahn F, et al. (1989) Lung toxicity after 13-week inhalation exposure to nickel oxide, nickel subsulfide or nickel sulfate hexahydrate in F344/N rats and B6C3F1 mice. Fund Appl Toxicol 12(3): 584-594
- USEPA (2004) Cleaning Up the Nations Waste Sites: Markets and Technology Trends. EPA 542-R-04-015. Technology Innovation and Field Services Division.
- Xu H, Huang X, Zhong T, Chen Z, Yu J (2014) Chinese land policies and farmers’ adoption of organic fertilizer for saline soils. Land Use Policy 38: 541-549.
- Potgieter JH, Kabemba MA, Teodorovic A, Vermaak SSP, Augustyn WG (2006) An investigation into the feasibility of recovering valuable metals from solid oxide compounds by gas phase extraction in a fluidised. Minerals Engineering 19(2): 140-146.
- Zhang H, Zheng LC, Yi XY (2009) Remediation of soil co-contaminated with pyrene and cadmium by growing maize (Zea mays L). Int J Environ Sci Tech 6(2): 249-258.
- Wasay SA, Parker WJ, Van GP (2001) Contamination of a calcareous soil by battery industry wastes II. Treatment. Can J Civ Eng 28: 349-354.
- Papassiopi N, Tambouris S, Kontopoulos A (1999) Removal of Heavy Metals from Calcareous Contaminated Soils by EDTA Leaching. Water Air Soil Pollut 109: 1-15.
- Dermont G, Bergeron M, Mercier G, Lafleche MR (2008) Soil washing for metal removal: a review of physical/chemical technologies and field applications. Journal of Hazardous Material 152(1): 1-31.
- Filgueiras A, Lavilla I, Bendicho C (2003) Partitioning in Environmental Solid Samples. Journal of Environmental Monitoring 4(6): 823-857.
- Nwachukwu MA, Huan F, Alinnor J, Nwachukwu M (2012) A comparative analysis of trace metal pollution parity between sandy and shaly soils: evidence from two mechanic villages in the Imo river basin. J Environ Earth Sci 65: 765-774.
- Amadi PU, Agomuo EN (2018) Oral ingestion risks of heavy metal accumulation at top soils of automobile workshops in Owerri capital city of Imo State, Nigeria Acta Chem. /AS/ 26-1, pp. 21-44
- Goldin A (1987) Reassessing the use of loss-on-ignition for estimating organic matter in non-calcareous soils. Comm Soil Sci Plant Anal 18(10): 1111-1116.
- Gawlik BM, Lamberty A, Pauwels J, Muntau H (2001) Certification of soil-pHsuspension of water and CaCl2) and adsorption coefficient for atrazine, 2,4-D and lindane in six different reference soils (Euro Soil). IRMM, p. 442
- Cokca E, Birand AA (1993) Determination of cation exchange capacity of clayey soils by the methylene blue test. Geotech Test Jour 16(4): 518-524.
- Santamarina JC, Klein KA, Wang YH, Prencke E (2002) Specific surface determination and relevance. Can Geotech Jour 39(1): 233-241.
- Tessier A, Campbell PGC, Bisson M (1979) Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem 51(7): 844-851.
- Podyachev SN, Shagidullin RR, Sudakova SN, Mustafina AR, Syakaev VV, et al. (2006) Synthesis of Tris (β-Diketones) and Study of their Complexation with some Transition Metals. Russ Chem Bull 55(11): 2000-2020.
- Witt K, Lenarcik ER, Urbaniak W (2016) Selective transport of zinc ions through novel polymer inclusion membranes (PIMS) containing β-diketone derivatives as carrier reagents. Sep Sci Technol 51(15-16): 2620-2627.
- Sparks DL, Zelanzy LW, Martens DC (1980) Kinetics of potassium desorption in soil using miscible displacement. Soil Sci Soc USA Jour 44(6): 1205-1208.
- Havlin JL, Westfall DG, Olsen SR (1985) Mathematical models for potassium release kinetics in calcareous soils. Soil Sci Soc USA Jour 49(2): 371-376.
- Li Q, Zhai J, Zhang W, Wan M, Zhou J (2007) Kinetic studies of adsorption of Pb (ll), Cr (lll) and Cu (ll) from aqueous solution by sawdust and modified peanut husk. J Hazard Mater 141(1): 163-167.
- Nowack B (2002) Critical review: environmental chemistry of aminopolycarboxylate chelating agents. Environ Sci Technol 36(19): 4009-4016.






























