Response of Zooplankton Population to Environmental Forcing: Case of Southern and Northern Coasts of Sfax, Tunisia
Salma Kmiha-Megdiche, Zaher Drira* and Habib Ayadi
Salma Kmiha-Megdiche, Zaher Drira* and Habib Ayadia
Submission: December 06, 2019; Published: January 06, 2020
*Corresponding author: Zaher Drira, LR18E530 Marine Biodiversity and Environment, Department of Life Sciences, Sfax Faculty of Sciences, University of Sfax, Soukra Road Km 3.5. BP 1171 - PO Box 3000 Sfax, Tunisia
How to cite this article: Salma Kmiha-Megdiche, Zaher Drira, Habib Ayadi. Response of Zooplankton Population to Environmental Forcing: Case of Southern and Northern Coasts of Sfax, Tunisia. Int J Environ Sci Nat Res. 2020; 23(2): 556110. DOI: 10.19080/IJESNR.2020.23.556110
Abstract
A comparative approach of hydrological variables in order to clarify the abundance and diversity of the zooplankton community was investigated on a spring cruise in the Northern and Southern coastal waters of Sfax coupled with water-quality. Environmental and zooplankton data were taken across the tidal front TF, covering well-mixed MZ and frontal interface FI waters. Zooplankton abundance, structure, specific composition and life histories were analyzed in relation to depth, temperature, salinity, orthophosphate, major and trace elements and chlorophyll-a. Zooplankton-specific composition did not vary across the different zones but, due to differences in relative abundance in Southern and Northern part. Three main copepod assemblages were revealed and characterized by the distribution of physico-chemical variables and chl-a. A strong divergence of zooplankton group was deduced through both multivariate analyses (NMDS, PCA and CCA), reflecting changes in zooplankton between the three typical zones. Results suggest that environmental parameters related differential responses of zooplankton should be considered when zooplankton monitoring is performed to assess the effect of climate forcing and pollution in coastal environments. The small number of copepods studies in this area makes our contribution useful for further global effect of climate forcing and pollution in coastal environments in the Mediterranean Sea.
Keywords: Southern coast of Sfax, Northern coast of Sfax, Zooplankton, Copepod, Monitoring, Environmental factors, Pollution, Tidal front, Well-mixed, Frontal interface
Introduction
Due to their rapid response to environmental changes zooplankton organisms are useful as bio-indicators for a variety of natural and anthropogenic stressors [1]. Many characteristics of zooplankton community assemblages make them useful as bio-indicators, especially the diversity of their response to stress due to their high taxonomic diversity. In all aquatic ecosystems, zooplankton have a key position in the trophic food web [1-3] and in biochemical cycles [4,5]. Zooplankton, being the major consumer of primary producers, plays an important role as a carbon and energy carrier to the high trophic levels such as other invertebrates, fish larvae and fish as secondary producers [6-11]. Planktonic crustaceans can be used as a good tracer of pollution and eutrophication phenomena in order to evaluate the water quality in the marine ecosystems [12,13]. Anthropogenic disturbance of near shore ecosystems appears to be the most widespread phenomena threatening coastal ecosystems [14]. In coastal marine environments, sewage discharge can affect several ecosystems in multiple ways by changing structural and functional attributes of biodiversity [14,15].
The Gulf of Gabes (GG), located in eastern Mediterranean Sea at the southeast of Tunisia (Figure 1) [16], extends from Ras Kaboudia in the north to the Tunisian/Libyan border in the south with a coastline of more than 400km. It is one of the most important fishing areas in Tunisia [17] and in the Mediterranean Basin [18,19]. Studies on metal pollution in coastal waters and their ecological impacts on natural ecosystems have increased in the last few decades. The GG has been polluted by numerous contaminant sources [20-22]. The Sfax region is an important industrial pole located in the GG in the Southeast of Tunisia, currently is considered one of the most polluted areas in the Mediterranean sea, according to a recent study carried out by Hamza-Chaffai et al. [23], Illou [24], Louati [25], Mkawar et al. [26], Gargouri et al. [27], Mezghani-Chaari et al. [28], Rekik et al. [29], Zaghden et al. [30] and Ben Salem et al. [11] still undergoes a strong anthropogenic pressure emanating from intensive urbanization, industrial wastes, over-fishing and a huge phosphogypsum deposit which affect the zooplankton communities. The south coast of Sfax as an important ecosystem has received an increased attention due to its obvious role on the fishery. Studies on diversity of zooplankton are scarce in this region and the few works mostly deal with horizontal distribution and for limited taxa, or specific populations.Since the year 2007, the Northern coast of the Sfax City has undergone a mega project of depollution and management (TAPARURA) based on the excavation and backfilling of a coastal line of about 4 km which led to the creation of about 420 ha of artificial beaches. Whether the source of heavy metals is natural or anthropogenic, once in the aquatic environment, these pollutants have a great affinity for water, phosphates, represent one of the major sources of seawater contamination [31,32] and are, therefore, considered among the possible major dangers threatening the vulnerable marine ecosystem in this area. Consequently, studies aiming to assess the impacts of anthropogenic activities on aquatic ecosystems must consider the quality of marine water, which can reflect the quality of marine environment and represent an essential source of information in major marine monitoring programs [33]. The coastal area surrounding the Southern coast of Sfax has experienced negative impacts on biodiversity, such as the local reduction and disappearance of some species of zooplankton. In spite of the seriousness of the ecological situation of the Gulf of Gabes, dumping phosphogypsum into the marine environment still occurs. Within this context, the present study was carried out to assess the environment quality in the South and North coastal area of Sfax, which is the most threatened and significantly impacted by industrial activities. The main objectives of this study are: How did environmental parameters forcing
(i) Zooplankton population
(ii) Affect the spatial trend of discrepancy between the Northern and Southern coasts of Sfax?
Methods and Materials
Study site
Sfax is one of the countries which give attention to environmental problems. It is a heavily industrialized city, located on Southeastern coast of Tunisia. It is a part of the GG (34° 43’ N 10° 46’ E) in the Mediterranean Sea. Its coastline extends along about 140km from the Taparura zone in the North to Thyna in the South. The Northern coast of Sfax, including the recently restored Taparura zone, expands along 12km from the phosphate treatment plant (NPK) to Sidi Mansour encompassing the continental shelf (between 34.66 °N and 34.78 °N).
Sampling sites were selected in the described Northern and Southern coasts of the Sfax City. The selected study area covers a representative zone extending from the South to North. The three recognized zones, mixed zone (MZ: North), frontal interface (FI: was chosen as an interface between North/South) and tidal fronts (TF: South) area of Sfax. The sources of heavy metals are evident in the South coastal area. The major anthropogenic sources of heavy metals in South coast are industrial and urban waste, wastewater discharges, agricultural activity and all other human activities in FI and TF. Upstream of the city of Sfax, agricultural activity is important, and industries are present. This pollution has strong negative impacts on marine life and on the fishery resources. There are the large industrial complexes around the South coast of Sfax city and significantly further to south and the North along the coast (Figure 1A).
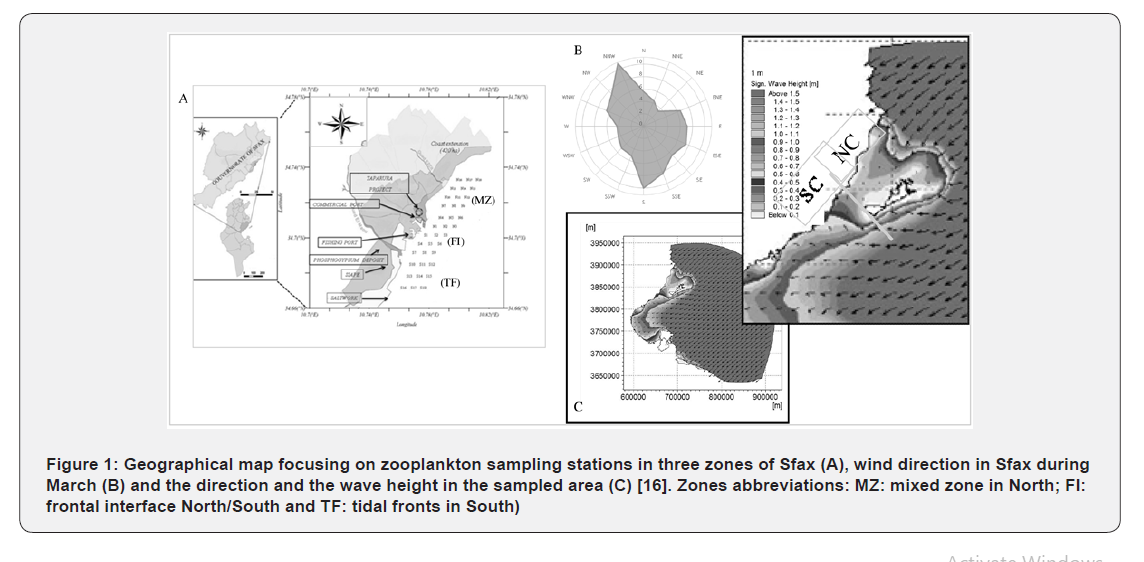
Sampling
Field sampling was carried out during a spring cruise conducted on the 22nd of March 2013 in three different zones: FI (depth = 4 - 4.5m); MZ (depth = 6 - 6.8m) and TF (depth = 3- 4.5m). Seawater samples were collected on the surface layer from 36 coastal stations (North: N1-N18, South: S1-S18) (Figure 1). At each station, water samples for the several physical (temperature, salinity, transparency, suspended particulate matter (SPM), pH and conductivity) chemical and (NO2 -, NO3 -, NH4 +, PO4 3- and Si(OH)4), T-P) and biological (chlorophyll-a) analyses were collected at the surface layer. Samples for nutrient analyses (120ml) were preserved immediately upon collection (-20°C, in the dark) and those for phytoplankton enumeration (1 l) were preserved with the Lugol (4%) iodine solution [34] and stored in the dark at low temperature (4 °C). Before assaying, 2 l of chlorophyll-a samples were filtered through 47mm GF/C filters. Analyses were estimated by spectrometry method, after the extraction of the pigments in acetone (90%). Samples for trace metal analyses were collected using 4-l Nalgene polycarbonate bottles. The bottles were opened below the water surface to avoid sampling of the surface microlayer. They were extensively washed with 1 M hydrochloric acid (HCl) and Milli-Q water before use to prevent the contamination of the bottles, rinsed three times with the respective sample before filling, and placed in cold and dark conditions after collection. Zooplankton was collected at the previously mentioned stations using a cylindro-conical net (30cm aperture, 100cm height, 100μm mesh size) equipped with a Hydro-Bios flowmeter. The net was towed obliquely from a depth near the bottom to the surface at each station in the Northern (6.61 ± 2.39m in depth) and Southern coasts (6.25± 2.44m in depth) at a mean speed of 1m s−1 for 10min. Zooplankton samples were rapidly preserved in a buffered formaldehyde solution (2%). They were stained with Rose Bengal to identify the internal tissues of the different zooplankton species and also to facilitate copepod dissection.
Sample analyses
Temperature, salinity and pH were measured immediately after sampling by means of a multi-parameter kit (Multi 340 i/SET). Water transparency was measured with a Secchi disc. Turbidity and of electrical conductivity were measured with a conductimeter. In laboratory nutrients (NO2 -, NO3 -, NH4 +, PO4 3- and Si(OH)4), total-nitrogen and total phosphate (after transformation into NH4 + and PO4 3−, with potassium persulphate at 120 °C, respectively) were analysed with a BRAN and LUEBBE type 3 autoanalyser and concentrations were determined colorimetrically using a UVvisible (6400/6405) spectrophotometer [35]. The concentrations of SPM were determined by measuring the dry weight of the residue after filtration of 1 l of seawater onto Whatman GF/C membrane filters (nominal porosity 0.45μm). Samples of trace metals , i.e., cadmium (Cd), cobalt (Co), chrome (Cr), copper (Cu),iron (Fe), nickel (Ni), lead (Pb), manganese (Mn) and zinc (Zn) were placed immediately in sealed and clean polyethylene bags and stored at 4 °C. In the laboratory, samples were analyzed by means of flame atomic absorption spectrophotometry (AAS) (Perkin Elmer A-Analyst 200 instrument copyright @ 2007, version 6 model). Some characteristics of the metal trace analysis, e.g., absorption wavelength λ (nm) were detected using UV radiation. Seawater samples were filtered under vacuum filtration with a 0.2 μm porosity filter and underwent acid attack. The blank used to analyze the different heavy metals was composed of 100ml of water and 2 or 3 drops of HCl. Chlorophyll-a concentrations were estimated by spectrometry, after the extraction of the pigments in acetone (90%). The concentrations were then estimated using the equations of Scor-unesco [36]. For phytoplankton analyses, subsamples (50ml) were enumerated under an inverted microscope using the Utermöhl [37] method. Quantitative zooplankton samples were counted according to Rose [38], Bradford-Grieve [39] and Costanzo et al. [40]. The different copepod species were sorted into four demographic classes (nauplii, copepodids, adult males and adult females). Enumeration was performed under a vertically mounted deep-focus dissecting microscope (Olympus TL 2). The mean and standard deviations (mean ± SD) of physicochemical and biological variables were obtained from the water surface column for Northern and Southern coastal stations.
Statistical analysis
The abundance of each zooplankton group (mean ± standard deviation) was analyzed in relation to the different zones (TF, FI and MZ) of The South and North system and only the most frequent zooplankton groups were plotted. Then, the zooplankton assemblage structures at different stations across the study area were analyzed using multivariate analysis on the abundance of those adults and juveniles identified at the species level (PRIMER 5 software package; Clarke & Warwick [41]). Data were transformed [log (x + 1)] to reduce the influence of the most abundant species, and a Bray–Curtis similarity matrix were constructed on the abundance data followed by cluster analysis and non-metric multidimensional scaling (NMDS). The stress coefficient (s) of the NMDS ordination indicates excellent representation (s < 0.05), adequate ordination (s < 0.2) or arbitrary ordination (s > 0.3; Clarke & Warwick [41]).The BIO-ENV procedure was used to relate environmental data with zooplankton assemblage structure. Moreover, to explain the relationship between physical (temperature, salinity, pH, conductivity, suspended particulate matter SPM and turbidity), chemical (PO4 3- and T-P, major element (Fe and Mn), trace metals (Zn, Pb, Cu, Ni and Cd) and biological (chl-a) parameters, we used a canonical correspondence analysis (CCA) [42] assessed over 36 stations.
To investigate the influence of the three zones on zooplankton specific composition, mean species richness (adult species number) was calculated and analyzed at each zone of the South to North system. To evaluate the null hypothesis of no difference in richness between different stations, analysis of variance was used (ANOVA; Zar [43]) and to identify significant differences extending from the South to North with biological and physicochemical parameters. Also, were used values of maximum depth to locate zooplankton species turnover between each station along all transect. Diagrams and contour plots of the physicochemical and biological parameters were performed using the interpolation method with Surfer software version 11. Finally, all zooplankton groups and development stages were analyzed in relation to using’s correlations [43]. Spearman correlation analysis was implemented to determine the relationship between environmental data (depth, temperature, salinity, turbidity, six investigated trace metals in the surface water column and chl-a concentration).
Results
Environmental data
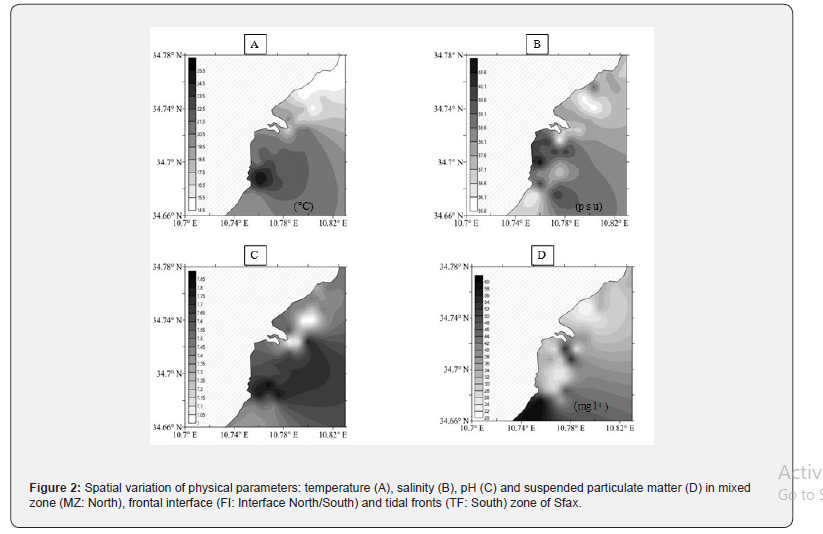
The comparison of the environmental factors (temperature, salinity, nutrients and their ratios) reveals clear differences between the Northern and Southern areas. The water temperature values were dissimilarly distributed on both sampled areas (Figure 2) with mean values of 16.77 ± 2.10 °C in the North and 22.16 ± 1.54 °C in South (Table 1). Salinity was higher in the South (36 - 41 psu) than in the North (35.75 - 39psu). In the Southern part, salinity was inversely correlated with transparency (r = - 0.582; p < 0.05). The pH values were generally more alkaline in the South (7.42 - 7.84) than in the North (7.00 - 7.78) (Table 1; Figure 2). A significant positive correlation was observed between pH and temperature in the Southern part (r = 0.651, p < 0.05). In contrast, negative correlations were found between pH and temperature in the Northern area (r = - 0.561, p < 0.05). SPM variations showed a difference between these two areas. In the North, it varied from 21.55mg l-1 to 38.29mg l-1 (Table 1). However, it varied from 40.60mg l-1 to 89.04mg l-1 in the South (Table 1; Figure 2). Turbidity was relatively higher (62.37 ± 40.79 N.T.U) in the Southern than in the Northern area (18.65 ± 10.86 N.T.U) (Table 1). The conductivity was higher in the South (44.08 10-2 ± 19.79 10-2μS cm-1) than in the North (0.06 10-2 ± 0.07 10-2 μS cm-1; Table 1). In the South, the conductivity was positively correlated with transparency (r = 0.579, p < 0.05) and with SPM (r = 0.545, p < 0.05) but a weak correlation was observed in the Northern area. According to our results, some of the determined physicochemical variables (salinity, SPM, water temperature and pH) affected the distribution of meso-zooplankton directly, whereas others indirectly.

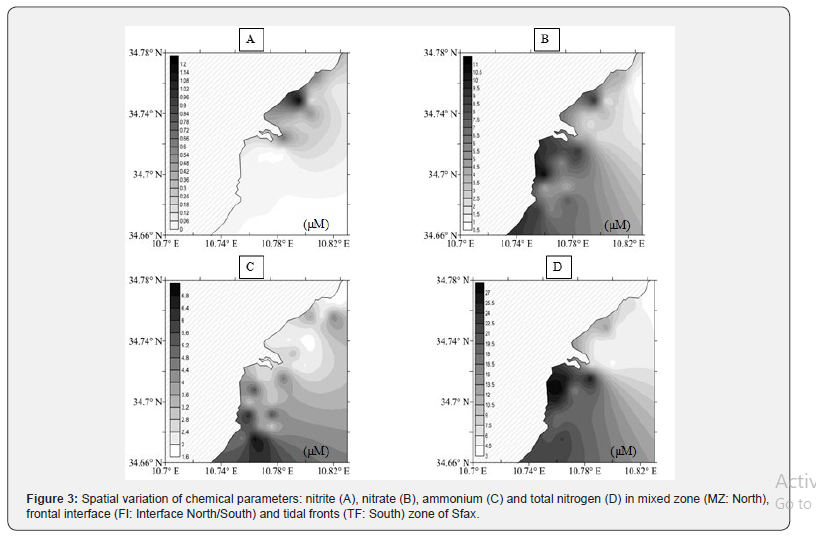
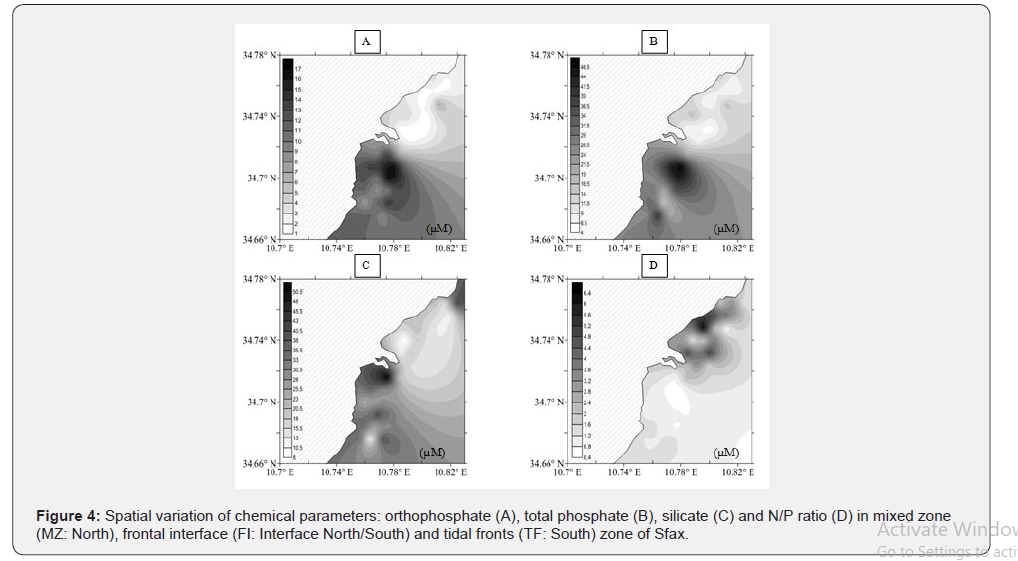
The chemical variables analyzed during this study are summarized in Figure 3. The nutrient compounds were more important in the South than in the North. In fact, the nitrogen concentration (T-N) was higher in the Southern (21.25 ± 3.17μM) than in the Northern (15.08 ± 4.14μM) coast. Nitrate (NO3 -) and ammonium (NH4 +) concentrations, which represented the highest inorganic nitrogen content, showed a converse trend between the sampled areas and were more important in the polluted area (Southern part). Nitrite (NO2 -) concentrations, which were lower than the other inorganic nitrogen forms, were relatively lower 0.005 ± 0.01μM in the non-restored Southern and higher 0.39 ± 0.3μM in the restored Northern coasts. Ammonium (NH4 +) concentrations showed higher values in the South 4.25± 1.45μM and lower values between 2.15 ± 0.33μM in the North (Table 1). As for total phosphate (T-P) concentrations, the mean value was more important in the South (29.96 ± 9.11μM) than in the North (10.05 ± 1.94μM) (Table 1). The orthophosphate (PO4 -) concentration was more abundant in the South (11.52 ± 2.85μM), than in the North (2.41 ± 0.70μM). Furthermore, the ANOVA test showed that PO4 - differed significantly between both sampled areas (F = 151.53, p < 0.001, df = 4.427) (Table 1). The mean Silicate (Si (OH)4) value was more important in the South (31.53 ± 9.13μM) than in the North (17.17 ± 6.73μM) (Table 1). Total phosphate (T-P) and orthophosphate (PO4 -) concentrations illustrated the same distribution pattern, with a high content in the polluted South. The N/P ratio of DIN (DIN = NO2 - + NO3 - + NH4 +) to DIP (DIP = PO2 +) fluctuated between 1.05 ± 0.37μM in the South and 1.60 ± 0.64μM in the North. This value was less than the Redfield ratio (16), suggesting a potential N limitation for phytoplankton and an excess of phosphate compounds.
Chlorophyll-a concentrations were ranged from 3.24 × 10-2 ± 2.60μg l-1 in the Southern to 0.18 × 10-2 ± 0.33μg l-1 in the Northern (Table 1). A slight difference in phytoplankton abundance between the Northern restored area (17.27 102 ± 5.33 102 cells l-1) and the Southern non-restored area (16.98 102 ± 4.01 102 cells l−1) (Table 1). According to the results, the significant chl-a content at the Southern part may indicate an abundance in algal biomass. This was probably due to the decreased transparency and increased phosphorus levels. There were significant negative correlations between chl-a and phosphorus levels (r = 0.590, p<0.01) and chl-a and turbidity in the study (r = - 0.452, p < 0.01).
Zooplankton community
Abundance and specific composition
A summary of the meso-zooplankton taxa observed throughout the study period is given in Table 2. The community structure of meso-zooplankton was diverse in our study area.Zooplankton abundance was very high in the whole study area. The zooplankton community was dominated by copepods in both areas (73% in the Northern and 85% in the Southern parts of total zooplankton abundance). In this study, the quantitative parameters of the zooplankton varied between Southern (TF: tidal fronts) and Northern (MZ: mixed zone; FI: interface Northern and Southern) coast. During the survey period, adult copepods were predominant in both regions (Southern part: 33.7 103 ± 11.98 103 in d m-3; Northern part: 30.9 103 ± 31.21 103 in d m-3). In the South, adult females were dominant (23.61 103 ± 8.62 103 in d m-3) as compared to adult males (10.18 103 ± 3.5 103 in d m-3) and were dominant in three zones. However, the Northern part was characterized by a clear co-predominance of females (12.13 103 ± 13.1 103 in d m-3) and males (11.18 103 ± 11.14 103 in d m-3; Table 1). Juvenile stages of copepods (copepodids and nauplii), occurred in lower numbers in mixed zone MZ situated in the Northern part (11.11 103 ± 14.18 103 in d m-3), but dominated in frontal interface FI situated in the Southern (28.8 103 ± 0.48 103 in d m-3).

A total of 36 copepod species belonging to 17 families were identified throughout the study period belonging to four different orders namely calanoida, cyclopoida, harpacticoida and Poecilostomatoida (Table 2). The maximum values of calanoida abundance were detected in the North (MZ) 72.8% dominated by P. grani Sars 22.53, O. similis Claus 16.27; P. parvus Claus 14.01; P. latisetosa Krichagin 10.40; A. italica Steuer 6.27and A. bifilosa Giesbrecht 6.43%. The rest of the zooplanktonic groups were represented by gelatinous were permanent components of the holoplankton, dominated by chaetognatha (Parasagitta elegans Tokioka 33.08 %) and appendicularia (Oikopleura dioica Fol 22.01 %).
Among the second cyclopoida order was a permanent group in three zones. It was composed of the highest abundance 49.33% in FI contributing to the dominance and persistence of O. similis 20.4; O. nana Giesbrecht 15.92 and O. plumifera Baird 13.01% were the three most abundant copepod species. Meroplanktonic larvae dominated in this zone mainly at FI stations, had high abundance of Fish egg 24.01; decapoda 20.09 and gasteropod larvae 16.15%.
Harpacticoida displayed high density 47.57% compared to other groups in TF and were mostly represented by abundant of H. littoralis Sars 16.17; E. acutifrons Dana 12.6 and T. battagalii Volkmann-Rocco 11.22 % in coastal water and by polychaeta larvae 20.01; foraminifera 15.20 and euphausiida (Euphausia lucens Hansen 14.02 %).
Influence of environmental factors on zooplankton distribution
The depth, Salinity, SPM and some elements trace of the water column was the environmental factor that best correlated with most zooplankton groups and its development stages (Table 3). Moreover, this variable was the one that best explained the three zooplankton assemblages defined (Spearman’s rank correlation, r = 0.65), which coincided with this zone of the South and North parts (Figure 4). Although the spatial pattern of some zooplankton groups was observed in relation to depth (Figure 4), which could be related to different chl-a and/or temperature values, only cyclopoid and harpacticoid adults were correlated with these variables (Table 3). Therefore, differences in nutriments concentrations would be the main factor responsible for the zooplankton spatial pattern across the study area.

Species richness did not differ between zones (Figure 4 & 5) did not vary between stations because the same zooplankton species were present all along the transect. Thus, specific composition did not change along stations.

Discussion
How did environmental parameters forcing the spatial trend of discrepancy between the Northern and Southern coasts of Sfax?
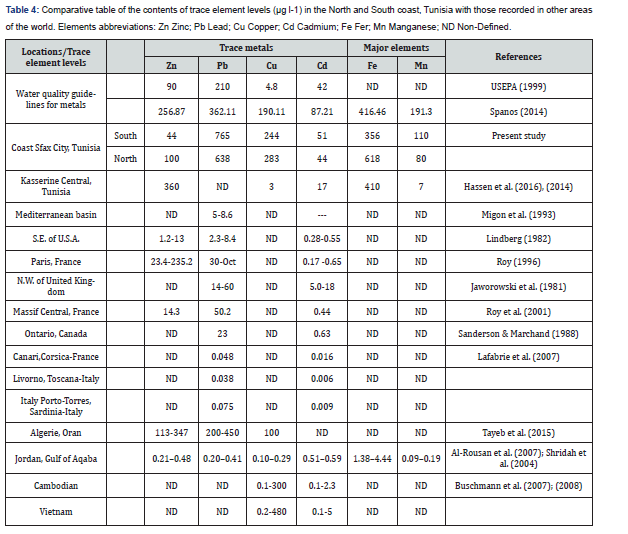
Our study provides the first detailed taxonomic of the zooplankton communities’ discrepancy between the Northern to the Southern coasts and to study the role of certain environmental variables in structuring zooplankton communities. Given that the study area occurs within the same water mass, species composition did not across the different part of the Southern (TF) and the Northern (MZ and FI) but relative abundance of zooplankton groups led to differences in assemblages’ structure among zones. We showed that water quality parameters (T, Salinity, orthophosphate, turbidity major and trace metals) had an important relationship with the zooplankton distribution pattern. Our results showed higher values for temperature as usually founded in arid to semi-arid Mediterranean areas [44]. In fact, a significant difference was observed in water temperature from Northern to Southern part (ANOVA; F = 76.86, p < 0.001, df = 3.027). In addition, water temperature is one of the most important ecological factors affecting the abundance, composition and diversity of zooplankton communities [45-49]. The low pH values can reasonably be attributed to the industrial activity still settled along the Southern in TF and IF coast whereas it was reduced in the North part (MZ). Zooplankton composition follows changes in salinity [50]. In fact, neritic species are favoured by higher salinity in TF and FI. Conductivity variation was the main physical feature driving the spatial trend of chemical compounds such as the orthophosphate content (ANOVA; F =86.55, p < 0.001, df = 7.171). Moreover, a major difference with respect to the pre-restoration study concerns the high orthophosphate concentration (mean value = 10.28μM; Rekik et al. [51]) prior to phosphogypsum containment and which was 20 times lower (mean value = 0.57μM) in MZ after restoration. As expected, we found lower orthophosphate concentrations in the North part (3.81 ± 2.34μM l-1) than prior to restoration (10.28 ± 9.94μM; Rekik et al. [29]) in TF, a result of the phosphogypsum containment and algal consumption. Inorganic nitrogen concentration values such as (NO2 - , NO3 - and NH4 +) were higher in the Southern part (TF and IF) due to high anthropogenic influences of the polluting activities in the zone. Several authors have noted that it is difficult to designate a single abiotic parameter which controls the biological processes in the marine eosystem [52-54]. The total concentrations of Pb, Fe, Cu, Mn, Cd and Zn in surface water of North to the South are presented in Table 4. The abundance order of trace metals concentrations was found to be Pb>Fe>Cu>Mn>Cd>Zn with mean concentrations of 765- 638; 356-618; 244-284; 110-80; 51-44 and 44-100μg l-1 respectively, those derived from anthropogenic source. High level of heavy metals has leaded to serious health effects in the region of Sfax City. Correlation analysis indicated higher concentrations of heavy metals (Fe and Pb) were found in TF stations, located in the South near the marine discharge zone of Chemical Group phosphogypsum. The other pollutants may originate from other industrial wastes influenced by coastal currents. The comparison of heavy metals contents recorded herein with those reported in water from different regions of the world showed that the Cd and Zn concentrations in surface water of are remarkably high in the region of Sfax City. The total metal contents in sea water found in the present work are comparable with those found in the literature (Table 4). Our study showed that, the concentrations of the other heavy metals analyzed (i.e., Cr, Cu and Pb) were found to be low compared to those reported in other regions of the world [55-57]. Our study showed that urban waste in the region of South is under the standard limit, causing a significant marine pollution. Increasing levels of pollution in coastal region has harshly harm the biodiversity of the marine system. Most of pollutants found its way to the food chain and may effect in bioaccumulation of toxic substances and pathogenic microorganisms in addible seafood sorts [31]. Metals are a group of the most important pollutants which cause environmental degradation in coastal areas. Heavy metals are deemed serious pollutants because of toxicity, persistence, and non-degradability in the environment. The importance of heavy metals in coastal environments derives from both their potential toxic effects and excessive anthropogenic sources which can equal or exceed natural input. Our results support the hypothesis that the coastal area of the Sfax city has significant local geochemical or hydrodynamic conditions of the body of water under examination [32]. Our work shows the importance of using several possible biomonitors when studying the quality of ecosystems, such as copepods, bivalve molluscs (filtering organisms), and herbivores (gastropod molluscs).
How did zooplankton population affect the spatial trend of discrepancy between the Northern and Southern coasts of Sfax?
In the present study, copepods formed the dominant group in meso-zooplankton, both in species composition and richness. They constituted 73 to 85% of total meso-zooplankton. Similar results were found by other works studies from the Gulf of Gabes (2005-2007; 69-83%; Drira et al. [9]), the Mediterranean lagoon of Bizerte (2006; 70 %; Sakka-Hlaili et al. [58]), the Northern coast of Sfax before restoration activities (2007; 61%; Rekik et al. [29]); the Southern coast of Sfax (2008; 66%; Ben Salem et al. [1]), the Algerian coast [59], the Southeastern Mediterranean region in Alexandria, Egypt (2011-2012; 63%; Abou-Zaid et al. [60]) and in the Eastern Indian Ocean of Australia (2015, 85%; McKinnon et al. [61]). The physico-chemical features affected the spatial distribution of meso-zooplankton mainly copepods considered as good biological indicators of water masses [62,63]. In many studies on pelagic copepods, temperature and/or food supply appeared to be the main variables which control fecundity [4,64,65]. Our study aimed at identifying spatial changes linked to environmental environmental forcing over the spring period. However, our data refer to the three typical zones (mixed MZ, frontal interface FI and Tidal front TF) were identified in the study area. As occur in TF located in the Southern part, the higher harpacticoida, polychaeta larvae, euphausiida and foraminifera abundance and high values of PO4 3- (r = 0.583; p < 0.05), Fe, Pb and salinity values were found, this zone is characterized the presence of suspension detritus SPM (r = 0.548; p < 0.05) and turbidity (r = 0.602; p < 0.05) high nutrient concentrations of PO4 3-, which are resuspended from the bottom to the surface by the effect of the strong tidal mixing of trace metals of Pb (r = 0.580; p < 0.05) ; Ni (r = 0.511; p < 0.05) and Fe (r = 0.524; p < 0.05) and existence of micro-streams cause the surface layer to flow in the spring, the success of many planktonic organisms would be difficult because of the mechanical stress due to high turbidity, conductivity and nutrient concentrations in this polluted zone (TF). Despite the low phytoplankton abundance registered in this area, it is known that the tidal mixing favors diatom development while dinoflagellates develop better in less turbulent waters. Although diatoms are considered a high-quality food for many zooplankton groups [66], in TF zone, they are diluted along most of the water column with the consequence that their concentration is not enough to support large zooplankton abundances. Moreover, in highly turbulent environments, zooplankton would not only be limited by food availability but also by its swimming ability, affected by the strong mixing effect that in turn diminish predator–prey encounters [67-72]. Despite turbulent conditions, high conductivity and temperature, our results show that adults and juveniles of harpacticoid copepods seem to do better shoreward, in the TF zone than the frontal interface FI. Harpacticoid copepods reached their highest abundances at the TF zone, being the dominant zooplanktonic group. An overall dominance of harpacticoids such as H. littoralis, E. acutifrons and T. battagalii found in the present study area of coastal habits, showing its affinity to orthophosphate concentration; their success in the TF zone could be favored for some characteristics of their life strategies; females of those species carry the eggs in sacks instead of releasing them freely in the water [73-81] diminishing their dispersion in this polluted and turbulent in coastal waters.
Furthermore, these copepods do not depend on phytoplankton for survival because they feed mainly on small particles associated with the microplankton community, which usually grows on suspended detritus [1] that characterizes this coastal zone.
The highest abundances of copepod nauplii were observed at the surface of the frontal interface FI of the South and North system. Although the specific identification of nauplii stages cannot be reached in the present work, we might suggest that this area could be an important breeding zone for all copepod species that inhabit this frontal interface FI system. As this FI is a convergence area, copepod nauplii could be transported from other zones of the system and concentrate at the FI because of physical forcing.
Furthermore, as this zone is characterized by a high and constant nutrient supply, which are provided from the mixed zone MZ, it is an optimal area for phytoplankton production, which is in turn food for copepods. Cyclopoida revealed pronounced fluctuations in their density and recorded their highest density at very low values of depth the coast due to the important proliferation of oithonids, mainly the neritic taxa. Also in FI is also characterized by the dominance of cyclopoida copepods, whose juvenile and adult abundances have a tendency to increase towards this zone. Thus, convergence processes that occur in the FI could benefit the survival of these zooplankton groups, given the high concentration and varied availability of food there. Small copepods connect the classical and microbial foodwebs and are used as prey for other pelagic carnivores. Hence they exhibit various reproductive and feeding strategies which help to maximize population size, in order to counter heavy losses due to predation [73]. The unique contribution of the four euryhaline species of the family Oithonidae (O. similis; O. nana; O. plumifera and O. sp.) was markedly evident in FI suggests their high adaptability to trophic and hydrologic conditions in this interface zone. The highest copepod eggs production rates and egg concentrations of cyclopoida occurred at the FI indicating that the front is also a favorable area for increases of secondary production These coexisting species exhibited certain strategies (e.g., shifting the feeding habit) in their opportunistic behaviors, in order to adapt to a highly variable marine environment of this coastal region [48, 81-83]. Copepod nauplii, unlike copepodites and adults, have limited energy reservoirs and inefficient feeding appendages, and given that its survival depends on high concentrations of phytoplankton, they could achieve high survival rates in the FI where the highest chl-a values are registered. These copepods could succeed better in this zone, where high chl-a values were detected, given that they are preferably herbivores in eutrophic environments [84-86]. Other zooplankton groups with high abundances in the FI were Meroplankton. All the groups that dominated in abundance at FI feed on phytoplankton (although only cyclopoids were correlated with chl-a) and they also take advantage of microplankton as a food source [87-89], which is also more abundant at fronts. Meroplanktonic group, mainly of Fish larvae, bivalvia and decapoda larvaea, were also observed in the FI, are usually concentrated and retained at fronts [79,90], but their patterns could also be affected by the distribution of the benthic populations from where they were released. There is not enough knowledge on the benthos of the region as to compare with our results on benthic larvae. The MZ showed the highest abundances of copepodites, juveniles and adults of calanoid copepods. Two calanoid copepod families, namely Paracalanidae and Acartiidae, formed the MZ abundance and were ubiquitously present such as P. grani; P.parvus; P .latisetosa; A. italica and A. bifilosa and thus seem to be well-adapted to the stresses of the MZ environment in our study. The significant role of this calanoida family to the total copepod community from Mediterranean coastal waters was also established by other workers [91-94]. Calanoida copepods have a complex behavior and their swimming patterns that affect both the way they avoid predation [45] and the way they gather food [69,71,95]. Calanoida copepod shifts their feeding habit by morphological alteration/modifications depending on the food availability. The main food for the copepods are the phytoplankton, variability of which depends on environmental conditions in MZ in contrast to physical accumulation of SPM in coastal waters brought through discharge impinging photosynthetic activity, local sinking in the Northern protect zone, controlled by climatic events (e.g., prevailing wind systems direction North/South), spatial nutrient enrichment in the TF and FI, differences in growth rates of individual plankters and nutrient up take and predation patterns of food chain etc could be held responsible. Our finding showed that Acartiidae are small and omnivorous, but they actively consume phytoplankton, which is considered an important component of their diet [96]. They develop mixed feeding mechanisms to incorporate the available food items characterized this MZ. Acartiidae are neritic species favouring shallow Mediterranean environments [11,77]. Gelatinous groups, considered as main copepods predators, were found in the MZ but in very low abundances at the study time. However, gelatinous zooplankton were represented by two species, an oceanic specie of chaetognaths (Parasagitta elegans) and marine species of appendicularia (Oikopleura dioica) densities in this zone as well as other zooplankton predators such as fish larvae. These organisms could have a negative impact on copepods’ abundance. Moreover, very low meroplankton group’s abundances were found in the MZ, this could be explained by the preference of this group for more oceanic habitats. However, their larvae were highly abundant across the Northern part, which suggests the use of the MZ as a reproduction and/or nursery area for this group. Despite all zooplankton species, mainly copepods, would perform better in different zones of the Northern protect zone, the FI and TF appears to be an important breeding area for all copepod species. Nauplii of all copepod groups could be transported to the FI and there retained by physical processes (e.g., convergence), but it is not clear how juveniles and adults of the different copepods species are then transported from the interface zone to the other zones, where better conditions for their development seem to exist. It has been shown that small hake larvae are retained in the MZ zone of a TF through a vertical migratory behavior, taking advantage of the existence of a twolayer water flow that would allow horizontal transport. In that sense, it could be expected that a similar behavior of copepods would be the main mechanism responsible for the movement of this group to the different zones of the TF (Southern part) and IF (Interface South/North) system, allowing them to explore zones profitable for its development. Our results indicate the extreme flexibility of copepods in adapting to a fluctuating environment and thus acting as causal link between environmental change and alteration in biodiversity in Southern and Northern part of Sfax city. During their larval stages, all fish consume zooplankton and some adult fish continue to be at least planktivorous. Hence abundance of meso-zooplankton might affect fish recruitment, although the details of this linkage are only just being revealed. As a consequence of intensified water quality degradation across the coastal regions of Southern, the interspecific competition among the presently dominant tolerant zooplankton and more sensitive ones might get changed. Anthropogenic activities and climate-induced forces may cause replacement of large copepods with small ones and this shift in size structure of the copepod community in TF and FI may have negative impact for existence of other zooplankton community and zooplanktivorous fishes. Hence a long-term trend analyses related to changes in MZ diversity, abundance and production is recommended to identify the complex biota–environment interactions.
In conclusion, abundance, distributional patterns and life histories of zooplankton appear to be strongly influenced by differences in the environmental water column parameters across the Southern and Northern side. In this work, we review three differences group of the copepod assemblages. The principal issues are listed below:
a) MZ and Gelatinous (ubiquitous community, depth 6-6.8m, high Chl-a) characterized by Chaetognatha (Parasagitta elegans); Appendicularia (Oikopleura dioica) and the coastal copepod species P. grani, P.latisetosa, P.parvus, A. italica and A. bifilosa.
b) FI and Meroplankton (Intermediate or interface community, depth 4-4.5m) characterized by high abundance of Fish, Decapoda, Gasteropod and cosmopolitain and abundant euryhaline species O. similis; O. nana; O. plumifera and O. sp.
c) TF poor water community (Costal community, depth 3-4.5m, high turbidity) characterized by Ostracods, euphausiids (Euphausia lucens), Mysidacea and several deep-water copepod taxa H. littoralis; E. acutifrons and T. battagalii.
The formation of the TF controls the food supply in the different zones, offering different physical and feeding mechanism or behavior for zooplankton groups, which respond differently to the environmental settings varying from a highly turbulent to a MZ and stable condition. The different zooplankton groups prevailing in different parts of the TF system could also be related to physical processes such as transport and retention and also to their life history strategies. Furthermore, the existence of zooplankton vertical migrations coupled to a two-layer water flow could be playing an important role in the distribution of different organisms. To overcome this pollution, we recommend allowing the construction of sewage treatment plant urban sanitary water for the region of South and North part of Sfax (Mediterranean Sea). An installation of a release monitoring station to sea and ports in Sfax is more than necessary. This will contribute to the development of risk mapping for better management of shipping, as well as preservation of the marine ecosystem of the Mediterranean. Although the species we studied in the present work and suggested as biomonitors present numerous advantages, more information and more studies are necessary to clarify accumulation patterns.
Acknowledgements
This work was conducted in the Biodiversity and Aquatic Ecosystems UR11/ES72 Research Unit of the Sfax University and supported by the Taparura and “Sfax Integrated Coastal Area Management” SMAP III Projects.
References
- Ben Ltaief T, Drira Z, Hannachi I, Bel-Hassen M, Hamza A, et al. (2015) What are the factors leading to the success of small planktonic copepods in the Gulf of Gabes, Tunisia? Journal of the Marine Biological Association of the United Kingdom 95: 744-761.
- Damotharan P, Perumal NV, Arumugam M, Perumal P, Vijayalakshmi S, et al. (2010) Studies on zooplankton ecology from Kodiakkarai (Point Calimere) coastal waters (South East coast of India). Research Journal of Biological Sciences 5: 187-198.
- Ladhar C, Tastard E, Casse N, Denis F, Ayadi H (2015) Strong and stable environmental structuring of the zooplankton communities in interconnected salt ponds. Hydrobiologia 743(1) : 1-13.
- Bickel SL, Tang KW (2014) Zooplankton-associated and free-living bacteria in the York River, Chesapeake Bay: comparison of seasonal variations and controlling factors. Hydrobiologia 722: 305-318.
- Leandro SM, Tiselius P, Marques SC, Avelelas F, Correia C, et al. (2014) Copepod production estimated by combining in situ data and specific temperature- dependent somatic growth models. Hydrobiologia 741: 139-152.
- Deksne R, Škute A, Meinerte A (2011) Seasonal changes in zooplankton community of the Daugava River. Acta Biologica University 11(1): 61-75.
- Letessier TB, Pond DW, McGill RAR, Reid WDK, Brierley AS (2012) Trophic Interaction of invertebrate zooplankton on either side of the Charlie Gibbs Fracture Zone/Subpolar Front of the Mid-Atlantic Ridge. Journal of Marine System 94: 174-184.
- Dam HG (2013) Evolutionary adaptation of marine zooplankton to global changes. Ann Rev Mar Sci 5: 349- 370.
- Drira Z, Bel-Hassen M, Ayadi H, Aleya L (2014) What factors drive copepod community distribution in the Gulf of Gabes, Eastern Mediterranean Sea? Environ Sci Pollut Res Int 21(4): 2918-29
- Schizas NV, Dahms HU, Ricaurte M, Hwang JS (2014) Population genetic patterns of the copepod Calanus sinicus in the northwest Pacific. Hydrobiologia 741: 153-165.
- Ben Salem Z, Drira Z, Ayadi H (2015) What factors drive phytoplankton, ciliates and mesozooplankton communities variations in a polluted area: the south coast of Sfax (Tunisia)? Environ Sci Pollut Res Int 22(15): 11764-11780.
- Webber MK, Roff JC (1995) Annual structure of the copepod community and its associated pelagic environment off Discovery Bay, Jamaica. Marine Biology 123(3): 467- 479.
- Pagano M, Sagarra PB, Champalbert G, Bouvy M, Dupuy C, et al. (2012) Metazooplankton communities in the Aheatoll lagoon (Tuamotu Archipelago, French Polynesia): Spatiotemporal variations and trophic relationships. Marine Bulletin Pollution 65(10-12): 538-548.
- Short FT, Wyllie-Echeverria S (1996) Natural and human-induced disturbance of seagrasses. Environmental Conservation 23(1): 17–27.
- Pearson TH, Rosenberg R (1978) Macrobenthic succession in relation to organic enrichment and pollution of the marine environment. Oceanography and Marine Biology 16: 229-311.
- Koutitonsky VG, Smeti H, Atoui A, Queiroz T, Toulany E, et al. (2013) Nearshore wave-climate modeling at eroding coastal sites in Angola, Nigeria, Ivory coast and Tunisia. NANO - Nearshore Hydrodynamic Group Report, p. 20.
- CGP (1996) Annuaires des Statistiques des Pêches en Tunisie. Ministère de l'agriculture, Tunisie.
- Zaouali J (1993) Les peuplements benthiques de la Petite Syrte (golfe de Gabès, Tunisie). Résultats de la campagne de prospection du mois de juillet 1990. Etude préliminaire: biocénoses et thanatocénoses ré Mar Life 3 : 47-60.
- Hattab T, Ben Rais Lasram F, Sammari C (2011) Modélisation de l'habitat des ressources halieutiques dans le golfe de gabès et projections selon un scénario de changement global. Bulletin de l'Institut National des Sciences et Technologie de la Mer de Salammbô 38: 55-64.
- Darmoul B, Hadj Ali Salem M, Vitiello P (1980) Effets des rejets industriels de la région de Gabès (Tunisie) sur le milieu ré Bulletin de l'Institut National des Sciences et Technologie de la Mer de Salammbô 7 : 5-61.
- Darmoul B (1988) Pollution dans le golfe de Gabès (Tunisie): bilan des six années de surveillance (1976–1981). Bulletin de l'Institut National des Sciences et Technologie de la Mer de Salammbô 15 : 61-84.
- Ayadi N, Aloulou F, Bouzid J (2014) Assessment of contaminated sediment by phosphate fertilizer industrial waste using pollution indices and statistical techniques in the Gulf of Gabes (Tunisia). Arabian Journal of Geosciences 8(3): 1755-1767.
- Hamza-Chaffai A, Amiard-Triquet C, El-Abed A (1997) Metallothionine like protein: is it an efficient biomarker of metal contamination? A case study based on fish from the Tunisian coast. Archives of Environmental Contamination and Toxicology 33: 53- 62.
- Illou S (1999) Impacts des rejets telluriques d'origines domestique et industrielle sur les environnements côtiers: Cas du littoral Nord de la ville de Sfax (Tunisie). Thèse de l'Université de Tunis II.
- Louati A (2003) Etude de la contamination par hydrocarbures des sédiments de la région de Sfax (Tunisie). Thèse de l'Université de Sfax et de l'Université Pierre et Marie Curie.
- Mkawar S, Azri C, Kamoun F, Montacer M (2007) Impact sur les biophases marines des rejets anthropiques notamment des métaux lourds rejetés sur le littoral nord de la ville de Sfax (Tunisie). Techniques Sciences Méthodes 10: 71-85.
- Gargouri D, Azri C, Serbaji MM, Jedoui Y, Montacer M (2011) Heavy metal concentrations in the surface marine sediments of Sfax Coast, Tunisia. Environmental Monitoring Assess 175(1-4): 519- 530.
- Mezghani-Chaari S, Hamza A, Hamza-Chaffai A (2011) Mercury contamination in human hair and some marine species from Sfax coasts of Tunisia: levels and risk assessment. Environ Monit Assess. 180(1-4): 477-487.
- Rekik A, Drira Z, Guermazi W, Elloumi J, Maalej S, et al. (2012a) Impacts of an uncontrolled phosphogypsum dumpsite on summer distribution of phytoplankton, copepods and ciliates in relation to abiotic variables along the near-shore of the southwestern Mediterranean coast. Mar Pollut Bull 64(2): 336-346.
- Zaghden H, Kallel M, Elleuch B, Oudot J, Saliot A, et al. (2014) Evaluation of hydrocarbon pollution in marine sediments of Sfax coastal areas from the Gabes Gulf of Tunisia, Mediterranean Sea. Environmental Earth Science 72: 1073-1082.
- Ben Salem Z, Ayadi H (2016) Heavy metal accumulation in Diplodus annularis, Liza aurata, and Solea vulgaris relevant to their concentration in water and sediment from the southwestern Mediterranean. Environ Sci Pollut Res Int 23(14): 13895-13906.
- Drira Z, Sahnoun H, Ayadi H (2017) Spatial distribution and source identification of heavy metals in surface waters of three coastal areas (Gulf of Gabes, Tunisia). Polish Journal of Environmental Studies 26: 1057-1069.
- Gao X, Chen CT (2012) Heavy metal pollution status in surface sediments of the coastal Bohai Bay. Water Research 46(6): 1901-1911.
- Bourrelly P (1985) Les Algues d’Eau Douce. Initiation à la Systè Tome II. Les Algues bleues et rouges. Les Euglénieins, Peridiniens et Cryptomonadines. Société Nouvelle des Editions Boubée.
- APHA (1992) Standard Methods for Examination of Water and Wastewater. American Public Health Association, Washington, DC.
- UNESCO (1966) Determination of photosynthetic pigments in seawater. UNESCO, Paris.
- Utermöhl H (1958) Zur Vervollkommnung der quantitative Phytoplankton-Methodik. International Vereining Theory Angew Limnology 9: 1-38.
- Rose M (1933) Copépodes pé In Faune de France 26. Paris: Office Central de Faunistique, p. 372.
- Bradford-Grieve JM (1999) Copepoda. Sub-Order: Calanoida, Family: Acartiidae, Genera: Acartia, Paracartia. ICES Identification Leaflets for Plankton.
- Costanzo G, Campolmi M, Zagani G (2007) Stephos marsalensis new species (Copepoda, Calanoida, Stephidae) from coastal waters of Sicily, Italy. Journal of Plankton Research 22: 2007-2014.
- Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation. PRIMER-E, Plymouth.
- Ter-Braak CJF (1986) Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67(5): 1167-1179.
- Zar JH (1999) Biostatistical Analysis. (4th edn), Prentice-Hall, New Jersey.
- Kchaou N, Elloumi J, Drira Z, Hamza A, Ayadi H, et al. (2009) Distribution of ciliates in relation to environmental factors along the coastline of the Gulf of Gabes, Tunisia. Estuarine, Coastal and Shelf Science 83(4): 414-424.
- Park GS, Marshall HG (2000) The trophic contributions of rotifers in tidal freshwater and estuarine habitats. Estuarine, Coastal and Shelf Science 51(6): 729-742.
- Castro BB, Antunes SC, Pereira R, Soares AMVM, Gonçalves F (2005) Rotifer community structure in three shallow lakes: seasonal fluctuations and explanatory factors. Hydrobiologia 543: 221-232.
- Buyurgan Ö, Altındağ A, Kaya M (2010) Zooplankton community structure of Asartepe Dam Lake (Ankara, Turkey). Turkish Journal of Fisheries and Aquatic Sicenses 10: 135-138.
- Kehayias G, Ramfos A, Ntzialas P, Ioannous S, Bisouki P, et al. (2013) Zooplankton diversity and distribution in a deep and anoxic Mediterranean coastal lake. Mediterranean Marine Science 14: 179-192.
- Sarma D, Das J, Dutta A (2013) Ecology of two riverine wetlands of goalpara district, assam in relation to plankton productivity. International Journal of Play Therapy 4: 219-225.
- Vuorinen I, Hanninen J, Viitasalo M, Helminen U, Kuosa H (1998) Proportion of copepod biomass declines with decreasing salinity in the Baltic Sea. Journal of Marine Science 55(4): 767-774.
- Rekik A, Maalej S, Ayadi H, Aleya L (2012b) Restoration impact of an uncontrolled phosphogypsum dump site on the seasonal distribution of abiotic variables, phytoplankton and zooplankton along the near shore of the south-western Mediterranean coast. Environ Sci Pollut Res Int 20(6): 3718-3734.
- Kaartvedt S, Svendsen H (1995) Effect of freshwater discharge, intrusions of coastal water, and bathymetry on zooplankton distribution in a Norwegian fjord system. Journal of Plankton Research 17(3): 493-511.
- Buyukates Y, Roelke DL (2005) Influence of pulsed inflows and nutrient loading on zooplankton and phytoplankton community structure and biomass in microcosm experiments using estuarine assemblages. Hydrobiologia 548(1): 233-249.
- Abo-Taleb HA, El Raey M, Abou Zaid MM, Aboul Ezz SM, Abdel Aziz NE (2015) Study of the physico-chemical conditions and evaluation of the changes in eutrophication-related in El-Mex Bay. African Journal of Environmental Science and Technology 9(4): 345-364.
- Al-Rousan S, Al-Shloul R, Al-Horani F, Abu-Hilal H (2007) Heavy metal contents in growth bands of Porites corals: Record of anthropogenic and human developments from the Jordanian Gulf of Aqaba. Mar Pollut Bull 54(12): 1912-
- Shridah MA, Okbah MA, El-Dek MS (2004) Trace metals in the water columns of the Red Sea and Gulf of Aqaba, Egypt. Water, Air, and Soil Pollution 153: 115-124.
- Hassen I, Hamzaoui-Azaza F, Bouhlila R (2016) Application of multivariate statistical analysis and hydrochemical and isotopic investigations for evaluation of groundwater quality and its suitability for drinking and agriculture purposes: case of Oum Ali-Thelepte aquifer, central Tunisia. Environmental Monitoring and Assessment 188(3): 135.
- Hlaili SA, Grami B, Gosselin M, Mabrouk HH, Hamel D (2007) Phytoplankton growth and microzooplankton grazing rates in a restricted Mediterranean lagoon (Bizerte Lagoon, Tunisia). Marine Biology 151(2): 767-783.
- Hafferssas A, Seridji R (2010) Relationships between the hydrodynamics and changes in copepod structure on the Algerian coast. Zoological Studies 49(3): 353-366.
- Abou-Zaid MM, El Raey M, Aboul Ezz SM (2014) Diversity of Copepoda in a Stressed Eutrophic Bay (El-Mex Bay), Alexandria, Egypt. Egypt Journal of Aquatic Research 40(2): 143-162.
- McKinnon AD, Duggan S, Holliday D, Brinkman R (2015) Plankton community structure and connectivity in the Kimberley-Browse region of NW Australia. Estuarine, Coastal and Shelf Science 153: 156-167.
- Hsieh CH, Chiu TS, Shih CT (2004) Copepod diversity and composition as indicators of intrusion of the Kuroshio Branch Current into the northern Taiwan Strait in spring 2000. Zoological Studies 43(2): 393-403.
- Sharma S, Siddique A, Singh K, Chouhan M, Vyas A, et al. (2010) Population dynamics and seasonal abundance of zooplankton community in Narmada River (India). Researcher 2(9): 1-9.
- Gómez-Gutiérrez J, Peterson WT, De Robertis A, Brodeur RD (2003) Mass mortality of krill cased by parasitoid ciliates (Brevia). Science 301(5631): 339.
- Sun XH, Sun S, Li CL, Zhang GT (2008) Seasonal and spatial variation in abundance and egg production of Paracalanus parvus (Copepoda: Calanoida) in/out Jiaozhou Bay, China. Estuarine, Coastal and Shelf Science 79(4): 637-643.
- Drira Z, Hamza A, Bel Hassen M, Ayadi H, Bouaïn A, et al. (2010) Coupling of phytoplankton community structure to nutrients, ciliates and copepods in the Gulf of Gabes (south Ionian Sea, Tunisia). Journal of the Marine Biological Association of the United Kingdom 90: 1203-
- Rodriguez V, Vives F (1984) Copepodos de las aguas portuarias de Màlaga. Investigaciones Pesqueras 48(2): 235-254.
- Lakkis S (1990) Composition, diversité et successions des copépodes planctoniques des eaux libanaises (Méditerranée Orientale). Oceanology Acta 16: 489-501.
- Belmonte G, Pati AC (2001) Hatching rate and diapause duration in eggs of Paracartia latisetosa (Copepoda: Calanoida). Journal of Plankton Research 29: 39-47.
- Daly-Yahia MN, Souissi S, Daly-Yahia-Kefi O (2004) Spatial and temporal structure of planktonic copepods in the Bay of Tunis (Southwestern Mediterranean Sea). Zoology Studies 43(2): 366-375.
- Pane L, Boccardo S, Mariottini GL (2005) Mesozooplankton assemblage and First Record of Paracartia grani Sars GO, 1904 (Copepoda: Calanoida) in the Western harbour of Genova (Ligurian Sea). Riv Biology 98: 323-336.
- Yebra L, Bonnet D, Harris RP, Penelope KL, Peijnenburg KTCA (2011) Barriers in the pelagic: population structuring of Calanus helgolandicus and C. euxinus in European waters. Marine Ecology Progress Series 428: 135-149.
- Turner JT (2004) The importance of small planktonic copepods and their roles in pelagic marine food webs. Zoology Studies 43: 255-266.
- Sarmento VC, Jorge P, Santos P (2012) Species of Harpacticoida (Crustacea, Copepoda) from the phytal of Porto de Galinhas coral reefs, northeastern Brazil. Check List 8(5): 936-939.
- Belmonte G, Mazzocchi MG, Prusova IY, Shadrin NV (1994) Acartia tonsa: a species new for the Black Sea fauna. Hydrobiologia 292: 9-15.
- Siokou-Frangou I, Papathanassiou E, Lepretre A, Frontier S (1998) Zooplankton assemblages and influence of environmental parameters on them in a Mediterranean coastal area. Journal of Plankton Research 20(5): 847-870.
- Siokou-Frangou I, Christaki U, Mazzocchi MG, Montresor M, Ribera d’Alcalà M, et al. (2010) Plankton in the open Mediterranean Sea: A review. Biogeosciences 7: 1543-1586.
- Berline L, Siokou-Frangou I, Marasovic I, Vidjak O, Fernandez De Puelles ML, et al. (2012) Intercomparison of six Mediterranean zooplankton series. Progress in Oceanography 97-100: 76-91.
- Boyer S, Arzul I, Bonnet D (2012) Some like it hot: Paracartia grani (Copepoda: Calanoida) arrival in the Thau lagoon (south of France-Mediterranean Sea). Marine Biodiversity Records 5.
- Pansera M, Granata A, Guglielmo L, Minutoli R, Zagami G, et al. (2014) How does mesh-size selection reshape the description of zooplankton community structure in coastal lakes? Estuarine, Coastal and Shelf Science 151: 221-235.
- Contreras JJ, Sarma SSS, Merino-Ibarra M, Nandini S (2009) Seasonal changes in the rotifer (Rotifera) diversity from a tropical high altitude reservoir (Valle de Bravo, Mexico). Jounal of Environmental Biology 30(2): 191-195.
- Gallienne CP, Robins DB (2001) Is Oithona the most important copepod in the world's oceans? Journal of Plankton Research 23(12): 1421-1432.
- Zervoudaki S, Christou ED, Nielsen TG, Assimakopoulou G, Giannakourou A, et al. (2007) The importance of small-sized copepods in a frontal area of the Aegean Sea. Journal of Plankton Research 29(4): 317-338.
- Marshall SM, Orr AP (1966) Respiration and feeding in some small copepods. Journal of Marine Biology Association of the United Kingdom 46: 513-530
- Lawrence D, Valiela I, Tomasky G (2004) Estuarine calanoid copepod abundance in relation to season, salinity, and land-derived nitrogen loading, Waquoit Bay, MA. Estuarine, Coastal and Shelf Science 61(3): 547-557.
- Ben Ltaief T, Drira Z, Devenon JL, Hamza A, Ayadi H, et al. (2017) How could thermal stratification affect horizontal distribution of depth-integrated metazooplankton communities in the Gulf of Gabes (Tunisia)? Marine Biology Research 13: 269-
- Richard S, Jamet JL (2001) An unusual distribution of Oithona nana Giesberecht (1892) (Crustacea: Cyclopoida) in a Bay: the case of Toulon Bay (France, Mediterranean Sea). Journal of Coasts Research 17(4): 957-963.
- Castellani C, Irigoien X, Harris RP (2007) Regional and temporal variation of Oithona biomass, stage structure and productivity in the Irminger Sea, North Atlantic. Journal of Plankton Research 29(12): 1051-1070.
- Temperoni B, Vinas MD, Diovisalvi N, Negri R (2011) Seasonal production of Oithona nana Giesbrecht, 1893 (Copepoda: Cyclopoida) in temperate coastal waters off Argentina. Journal of Plankton Research 33(5): 729-740.
- Anderson DM, Gilbert PM, Burkholder JM (2002) Harmful algal blooms and eutrophication: nutrient sources, composition and consequences. Estuaries 25(4): 704-726.
- Chaalali A, Chevillo X, Beaugrand G, David V, Luczak C, et al. (2013) Changes in the distribution of copepods in the Gironde estuary: A warming and marinisation consequence? Estuarine, Coastal and Shelf Science 134: 150-161.
- Chen MR, Kâ S, Hwang JS (2010) Diet of the copepod Calanus sinicus Brodsky, 1962 (Copepoda, Calanoida, Calanidae) in northern coastal waters of Taiwan during the northeast monsoon period. Crustaceana 83: 851-864.
- Chew LL, Chong VC (2011) Copepod community structure and abundance in a tropical mangrove estuary, with comparisons to coastal waters. Hydrobiologia 666: 127-143.
- Duggan S, McKinnon AD, Carleton JH (2008) Zooplankton in an Australian tropical estuary. Estuaries Coasts 31: 455-467.
- Belmonte G (1992) Diapause egg production in Acartia (Paracartia) latisetosa (Crustacea, Copepoda, Calanoida). Bolletino dizoologia 59: 363-366.
- Benedetti F, Gasparini S, Ayata SD (2016) Identifying copepod functional groups from species functional traits. Journal of Plankton Research 38(1): 159-166.






























