Quantitative Estimation of Aloin in Aloe Vera and its Commercial Formulation by using UV Spectroscopy
Prachi Jitendra Patel* and Madhuri Hinge
Department of Quality Assurance, Rofel Shri G M Bilakhia College of Pharmacy, India
Submission: September 03, 2019; Published: September 19, 2019
*Corresponding author: Prachi Jitendra patel, Rofel Shri G M Bilakhia College of Pharmacy, Vapi, Gujarat
How to cite this article: Prachi Jitendra Patel, Madhuri Hinge. Quantitative Estimation of Aloin in Aloe Vera and its Commercial Formulation by using UV Spectroscopy. Int J Environ Sci Nat Res. 2019; 21(3): 556064. DOI: 10.19080/IJESNR.2019.21.556064
Abstract
In Indian Ayurvedic system of medicine the leaf of Ghritkumari has been extensively used as a cathartic agent to treat severe stomach constipation. The aloe vera plant is used as food supplement and also used in cosmetic products to treat different skin problems. In order to develop reliable quality control marker to check its potential toxicity and carcinogenic activity and to check the commercial samples of Aloe vera for authenticity and purity, UV Spectroscopy analysis has been done. It has been found that aloin is present in a few commercial samples. HPLC, Infra-Red and thin layer chromatography methods have been reported for aloin determination in commercial samples. Therefore, UV Spectrophotometric method was developed for determination of aloin in commercial formulations. This method gives reproducible results and found linear in the range 80-180μg/ml. The method was found to be simple, precise and accurate for estimation of aloin from aloe vera and commercial formulations containing aloin
Keywords: Aloin; UV-Spectroscopy; Commercial formulation
Introduction
It is an important medicinal leaf of plant of Aloe barbadensis miller, known as curacao aloes, belonging to family liliaceae. Aloe vera contains varieties of chemical constituents from which anthraquinone glycoside is major constituent. Aloin is the principle active constituent of aloe vera, which contain mixture of glycosides. Aloin is official drug in Indian Pharmacopoeia and British Pharmacopoeia. HPLC, UV Spectroscopy, Infra-Red and thin layer chromatography methods have been reported for aloin determination.
Aloin is also known as Barbaloin. Aloin is Bitter, yellow brown colour compound. Aloin an active compound has laxative, anti-inflammatory and anti-cancer effect. Aloe vera is used both internally and externally for curing various ailments. Aloin is used in various cosmetic products, used to treat acne, sunburns, itchy rash, psoriasis and oral submucous fibrosis. Therefore, in order to develop reliable quality control marker to check its potential toxicity and carcinogenic activity and to check the commercial samples of Aloe vera for authenticity and purity is essential.
International aloe science council (IASC) has developed a set of identify, purity and quality standards for aloin containing products certified by IASC are specifically prepared in manner to assure aloin is present in above a minimum level and to ensure the absence of adulteration (Figure 1).
Mechanism of action of aloin
Effect on inflammation: Aloin, dose dependently inhibit inducible nitric oxide synthase mRNA expression and nitric oxide production. With inhibition of NO production, it did not suppress PEG2 production.
Effect on antioxidant system: Antioxidant effect of aloin on free radical induced DNA breaks. The greater reducing activity of aloin at lower concentrations, however, free radical scavenging activity of aloin gradually predominated over its reducing power, resulting in the protection of DNA.
Nutrients in aloe vera
a) Vitamin: A, B, C & E, M,
b) Choline: Helps metabolism,
c) Minerals: Calcium, Phosphorus, Potassium, Iron, Sodium, Chlorine, Zinc,
d) Mono and Polysaccharides: Aldopentose, Cellulose, Glucose, L-rhamnose, Mannose,
e) Fatty acids: cures Burns and wounds,
f) Salicylic Acid: Anti-Bacterial,
g) Amino Acid: Body needs 22 amino acids, out of 20 amino acids are provided by Aloe Vera,
h) Lignin: A strong ability to penetrate human skin,
i) Saponins: Work saponifying agent and antiseptic,
j) Enzymes: Phosphatase-Amylase,
k) Bradykinase: Immune building, Catalase prevents accumulating of water in system,
l) Cellulase: Cellulose digestion, Creatine,
m) Phosphokinase: muscular enzyme
n) Lipase: digestion, Nucleotidase, Alkaline phosphate,
o) Protease: hydrolyses proteins into their constitute elements.
Materials and Methods
Apparatus and instrument
Spectrophotometric was carried out by using UV-Visible spectrophotometer having two matched quartz cells with 1cm light path.
Model: - shimadzu-1800, Software: -UV probe, version 2.42. Glass wares (Borosilicate) Electronic analytical balance (REPTECH), Ultrasonicator (LOBA Life), Magnetic stirrer.
Materials and reagents
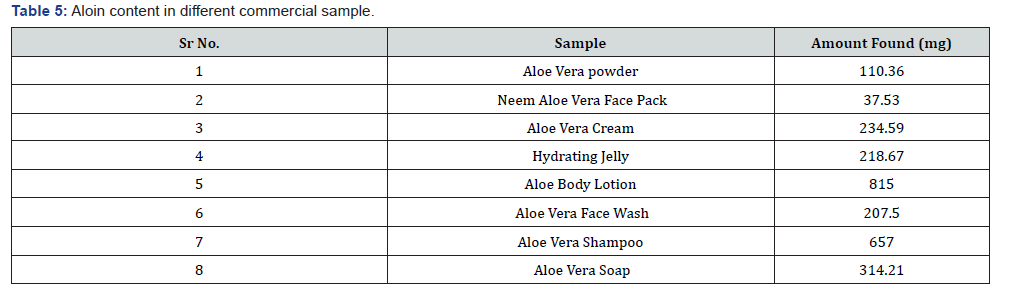
Methanol, N-hexane, Dis. Water, Butanol all chemicals were used as an analytical grade. The aloe vera dry powder was collected from local market. The marketed sample viz., Neem Aloe vera Face Pack, Aloe Vera Cream, Hydrating Jelly, Aloe Vera Gel, Aloe Fresh Body Lotion, Aloe Vera Face Wash, Aloe Vera Shampoo, Aloe Vera Soap were produced from local market.
Extraction methods for commercial formulations
For commercial samples Neem Aloe vera Face Pack, Aloe Vera Cream, Hydrating Jelly, Aloe Vera Gel, Aloe Fresh Body Lotion, Aloe Vera Face Wash, Aloe Vera Shampoo, Aloe Vera Soap were dissolved in 100ml methanol by using magnetic stirrer for 1h and transferred into separating funnel respectively.
For the removal of oily and fatty material, to that added 100ml of n-hexane and shacked well and set aside for separation of two distinguish layers and collected methanol layer into a conical flask respectively. These concentrated methanol extracts were dissolved in a minimum amount of water and transferred into separating funnel respectively.
To that added 100ml of butanol and shacked well and set aside for separation of two distinct layers.
Collect the butanol layer into round bottom flask. Again, added 100ml of butanol to that water layer and collected the butanol layer into the same round bottom flask respectively and concentrated under sun light
These butanol extracts Neem Aloe vera Face Pack, Aloe Vera Cream, Hydrating Jelly, Aloe Vera Gel, Aloe Fresh Body Lotion, Aloe Vera Face Wash, Aloe Vera Shampoo, Aloe Vera Soap were dissolved in the minimum amount of methanol and transferred into 10ml of volumetric flask and made up the volume with methanol.
Method
Preparation of sample solution: The sample solution of extracted aloin from different commercial formulation (Oil, Face pack, Cream, Jelly, Lotion, Gel, Face wash, shampoo, soap) were prepared by dissolving 1mg in 10ml of methanol in 10ml volumetric flask. The solution was scanned in UV range of 200- 400nm
Preparation of standard solution: The standard solution of aloin were prepared by dissolving 1mg in 10ml methanol in 10ml volumetric flask. The solution was scanned in UV range of 200-400nm
Method validation
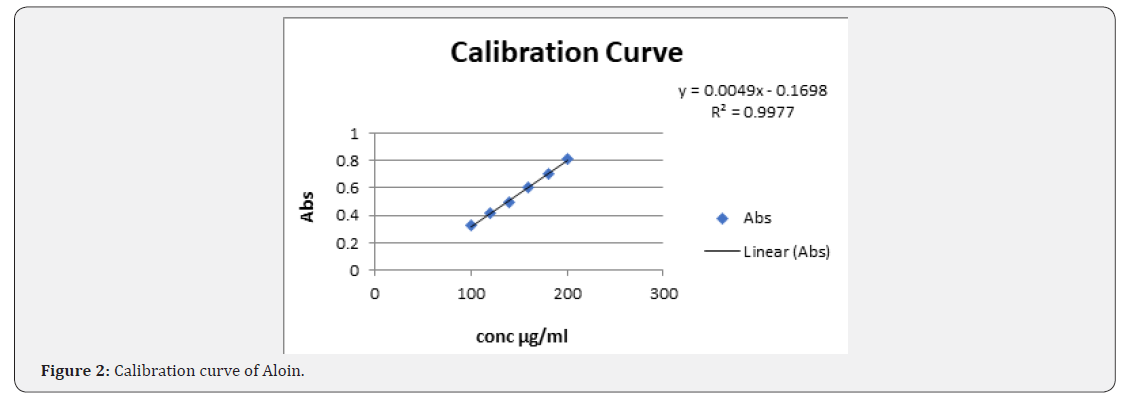
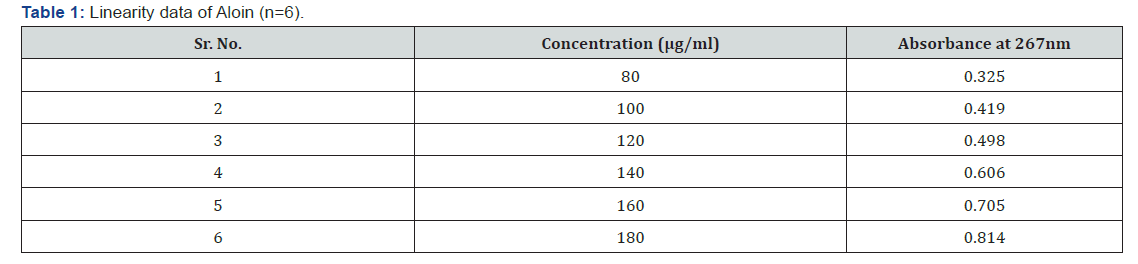
Linearity: The solution was prepared by pipetting 0.8, 1.0, 1.2, 1.4, 1.6 and 1.8ml from working standard stock solution in to 10ml volumetric flask and the volume was adjusted to mark with methanol to produce 80-180μg/ml respectively. The absorbance of solution was measured at 267nm. Calibration curve were generated by taking the response vs. concentration.
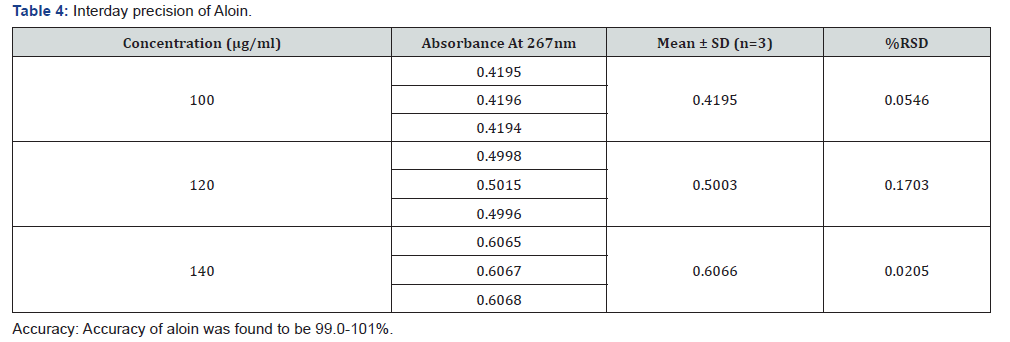
Precision: The precision of an analytical procedure expresses the closeness of agreement (results) between a series of measurements obtained from multiple sampling of the same homogenous sample under the prescribed conditions. Precision of the methods was studied at three level as at repeatability, intermediate precision (Day to Day and analyst to analyst) and reproducibility.
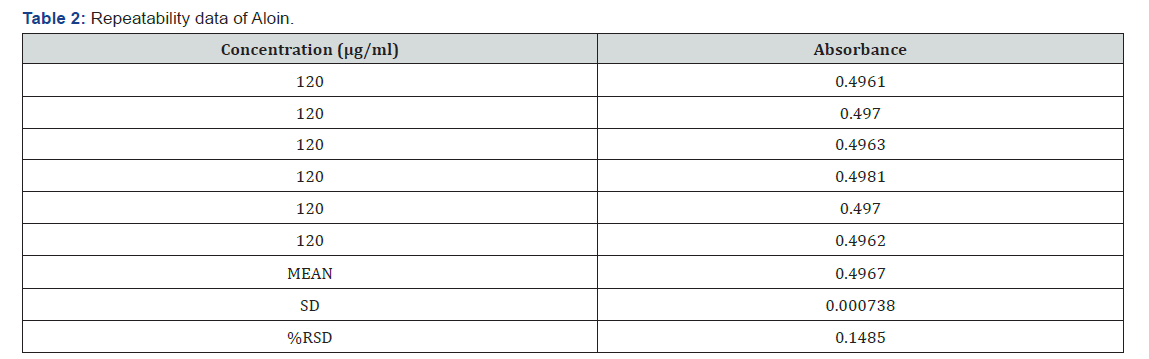
Repeatability: Aliquots of 1.2ml of working standard solution of aloin (1000μg/ml) was transferred to 10ml volumetric flask and volume was adjusted to distilled water to get concentration of 120μg/ml. the absorbance of solution was measured spectrophotometry six times and %RSD was calculated.
Intraday and interday: Aliquots of 0.8, 1.0 and 1.2ml of working standard solution of aloin (1000μg/ml) was transferred to 10ml volumetric flask and volume was adjusted to distilled water to get concentration of 10, 20 and 30μg/ml. The absorbance of solution was measured spectrophotometry three times and %RSD was calculated. For intraday, the analysis was carried out at different intervals on the same day and for interday, the analysis was carried on different days.
Result and Discussion
Calibration curve of standarnd aloin are shown in Figure 2. In all the sample maximum absorbance occur at 267nm (Table 1-5) [1-12].

Conclusion
The spectrophotometric methods were developed and validated as per ICH guidelines. These validated methods were new, rapid, accurate, precise and reproducible. The standard deviation and % RSD calculated for the proposed methods are within limits, indicating high degree of precision of the methods. Hence, it can be concluded that the developed spectrophotometric methods are accurate, precise and can be employed successfully for the estimation of Aloin in different commercial formulation.
Conclusion
Due to poor management, poultry waste especially feathers have become one of the major pollutants. Traditional feather degradation reduces the overall quality of proteins and destroys essential amino acids. Biodegradation of feathers is found to be an efficient, cost-effective, and environmental friendly method for bioconversion of feather waste into useful products. The use of keratinolytic microorganisms to degrade poultry feather has emerged as a sustainable and alternative tool to meet this challenge.
References
- Kokate CK, Purohit AP, Gokhle SB. Pharmacognosy. (13th edn), Nirali Prakashan, p. 9.9.
- Pandey DK, Tabarak Malik, Banik RM (2012) Quantitative estimation of barbaloin in aloe vera and its commercial formulation by using HPTLC. Int J Med Aroma Plants, pp. 420-427.
- Gaurav D, Dhonde, Gaurao D Wandhare, Manashri N Jirapure, Nilesh S Dumore. Extraction of Aloin from Aloe Vera by using soxhlet apparatus. International Journal of Engineering Applications and Technology.
- Sharma V, Shukla VJ, Prajapati PK (2012) Quantitative estimation of aloin from pharmaceutical dosage by HPTLC. International Journal of Pharmaceutical Sciences 3(1): 104-108.
- Mulay S, Ogale S (2018) Quality control Study in various nutraceutical Aloe vera Formulations, ACTA Scientific pharmaceutical Sciences 2(8): 4-9.
- https://pubchem.ncbi.nlm.nih.gov/compound/Aloin-A#section=Chemical-and-Physical-Properties.
- Patel K, Patel DK (2013) Medicinal agent, Pharmacological activities and analytical aspects of aloin: a concise report. Journal of Acute Disease 2(4): 262-269.
- Surjushe A, Vasani R, Saple DG (2008) Aloe Vera: A short review. Indian J Dermatol 53(4): 163-166.
- ICH, editor Q2Atext on validation of analytical procedure. Fed Regist: 1995.
- ICH, editor validation of analytical procedure: text and methodology Q2 (R1). ICH Harmonized tripartite Guideline; 2005.
- (2014) Indian Pharmacopoeia, Government of India, Ministry of Health and Family Welfare, (7th edn), The Indian Pharmacopoeia commission, Ghaziabad, Volume II, p. 1014.
- (2003) British Pharmacopoeia, The department of Health, Social Services and Public Safety, (4th edn) The British Pharmacopoeia commission, Volume I, p. 64.






























