Populations Structure of Mosquitofish Gambusia Affinis (Baird and Girard; 1853) in Four Different Lakes in Benghazi, Libya
Enas Eltaeeb
Houssein Elbaraasi*
Department of Zoology, University of Benghazi, Libya
Submission: July 03, 2019; Published: July 22, 2019
*Corresponding author: Houssein Elbaraasi, Department of Zoology, Faculty of Science, University of Benghazi, Benghazi, Libya
How to cite this article: Enas E, Houssein E. Populations Structure of Mosquitofish Gambusia Affinis (Baird and Girard; 1853) in Four Different Lakes in Benghazi, Libya. Int J Environ Sci Nat Res. 2019; 20(4): 556041. DOI:10.19080/IJESNR.2019.20.556041
Abstract
The aim of this study was to investigate some biological aspects within the populations of mosquitofish Gambusia affinis in four different brackish water lakes which are, Althama (A), Aian Elmajdob (B), Bodizera (C) and Ard Alahlam (D) in Benghazi, Libya. Result of the sex ratio of males to females was 1.1:1 in site A, 2.5:1 in site B, 0.6:1 in site C, and 2:1 in site D. The highest mean total length and weight of female was in Aian Elmajdob lake 4.52 ± 0.34cm, 1.28 ± 0.33g, the lowest mean total length and weight of female was in Bodizera lake 3.44 ± 0.35cm, 0.49±0.01g. However, 3.19 ± 0.29cm, 0.35 ± 0.12g were the highest mean total length and weight of males in Ard Alahlam lake. The lowest mean total length and weight was in Althama lake 2.73 ± 0.28cm, 0.21 ± 0.09g.
Keywords: Population; Mosquitofish; Gambusia affinis; lakes; Benghazi; Libya
Introduction
The mosquitofish Gambusia affinis (Baird and Girard; 1853) belong to poecilidae (live bearers) family. It is a small greenish olive to brown above, grey-blue on sides, and silvery- white below in colors. It has short body with flattened head. Its mouth pointed upward for surface feeding [1,2].
The origin of Gambusia affinis is in fresh and brackish waters of the eastern and southeastern United State of America and Gulf of Mexico [1]. It has been progressively introduced to many countries such as Spain, Eastern European countries, Italy and North Africa as malaria control [3]. It lives in wide range of waters from flowing rivers, to vegetated ponds and lakes, also in backwaters and quiet pools of streams.
The mosquitofish is omnivorous feeds on small aquatic invertebrates including insects, their eggs and larvae, aquatic crustaceans such as cladocerans, ostracods, copepods as well as eggs and fry of small fish, small gastropods and amphibian tadpoles. Its diet may also opportunistically include terrestrial insects which fall into the water, as well as filamentous, algae and detritus [4-9].
G.affinis is short lived animal with a maximum lifespan of just two or three years [10]. This species exhibits sexual dimorphism with females typically being larger than males [11,12].
Mosquitofish are typically occurring at temperatures between 12-29ºC [10]. It is generally found at lower salinity [1]. It can tolerate salinities between 0 and 25ppt [13]. Moreover, Salinity mainly affected mosquitofish females which, despite presenting earlier maturation and higher reproductive investment hence supporting the hypothesis that salinity limits mosquitofish invasive success [14]. The overall aim of this study was to assess the biology and population structure of G. affinis in four different brackish water lakes (Althama, Aian Elmajdob, Bodizera and Ard Alahlam) in Benghazi, Libya this assess lies in studies some biological aspects within the populations.
Materials and Methods
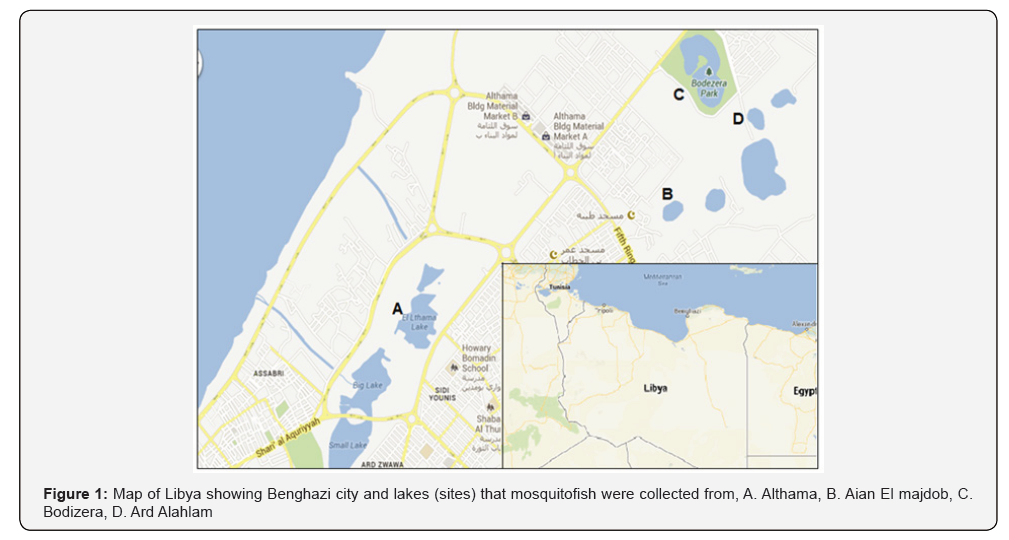
Populations of the mosquitofish Gambusia affinis in four lakes (sites), located in the eastern part of Benghazi city, were studied. The lakes are: Althama (site A), Aian El majdob (site B), Bodizera (site C), and Ard Alahlam (site D) (Figure 1).
A total of 658 specimens were collected by dip net from all sites, about 212 specimens from Althama, 161 specimens from Aian El majdob, 127 specimens from Bodizera and 158 specimens from Ard Alahlam. Samples were kept in 10% formalin and transferred to the Aquaculture and Fisheries lab at the Zoology Department, Faculty of Science, University of Benghazi.
Upon arriving to the lab, fish sex was determined, body weight was taken using a digital balance and the morphological measurements were taken by a rule.
Total length and weight and Length frequency distributions between male and females in the sites were analysis using ANOVA. The sex ratio is given as males: females (M: F), The chi-square (X²) was used to verify the significant differences between the sex ratio of the species within the populations that commonly expected 1:1 sex ratio [15].
The condition factor (K) was calculated by the formula [16]:
K= 100w/ L³
Where W= Weight (g), L= Total length (cm).
The condition factor (K) were analysed using ANOVA followed by Tukey’s Multiple Comparison test. All the statistical analysis and calculations were done by Graph software and Microsoft Excel programs.
Results
Sex ratio
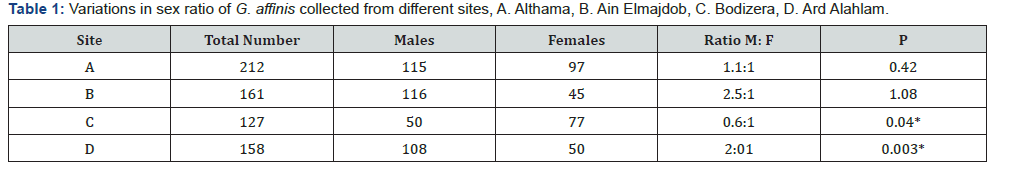
A total of 658 specimens were collected from four different brackish-water lakes located in Benghazi, Libya. Site A. Althama (n=212), Site B. Ain Elmajdob (n=161), Site C. Bodizera (n=127), and Site D. Ard Alahlam (n=158). It was determined (Table 1) that 46% of the samples were females (n=97), 54% males (n=115) in site A; 28% females (n= 45), 72% males (n=116) in site B; 60% females (n=77), 40% males (n=50) in site C; 32% females (n=50), 68% males (n=108) in site D. The sex ratio of males to females was 1.1:1 in site A, 2.5:1 in site B, 0.6:1 in site C, and 2:1 in site D (Table 1) and the analysis showed that in sites A and B the ratio of males and females differ significantly (P=0.42 and P=1.08, respectively), While in site C and D the differences was statistically insignificant (P<0.05).
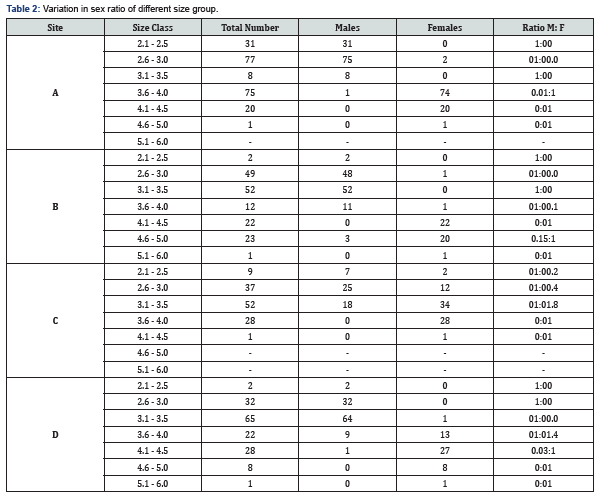
The data on sex ratio in relation to size group is given in Table 2 in details. However, it suggests that in site A, females less than males in all the size groups except in 3.6-4.0cm group (74) and 4.1-4.5cm group (20). In site B, males were more than females in all size group except in 4.1-4.5cm group (22) and 4.6-5.0cm group (20). In site C, males were more than females in 2.1-2.5 (7) and 2.6-3.0cm (25) size groups. While females were more than males in all the rest of size groups (Table 2). Lastly, in site D, males were more than females in 2.1-2.5cm (2), 2.6-3.0cm (32), and 3.1-3.5cm (64) size groups, whereas, females were more than males in 3.6- 4.0cm (13), 4.1-4.5cm (27), 4.6-5.0cm (8) and 5.1-6.0cm (1) size groups. Probability from chi-square test showed that P values were found to be significant differences in all size groups of all sites (P>0.05).
P: probability from χ2-test showed P>0.05 in all sites.
Populations’ growth trends
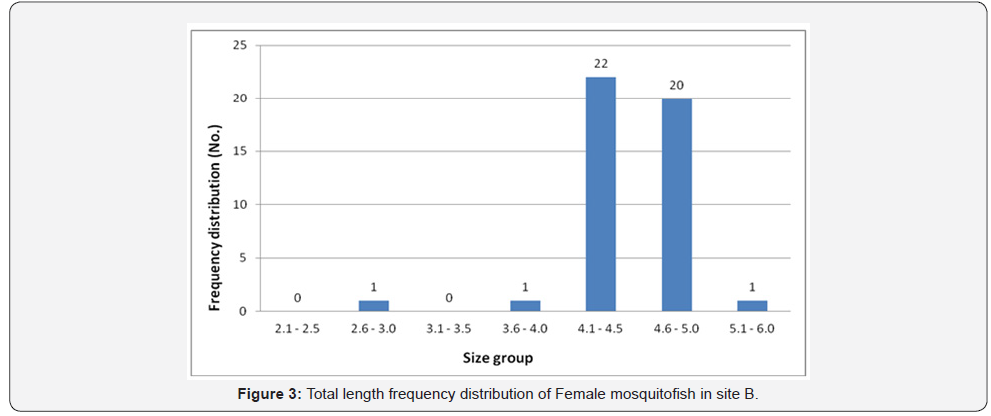
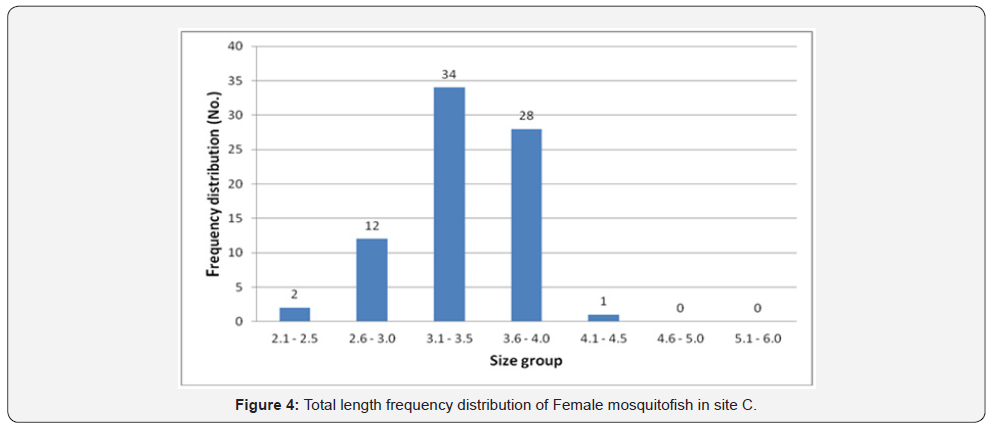
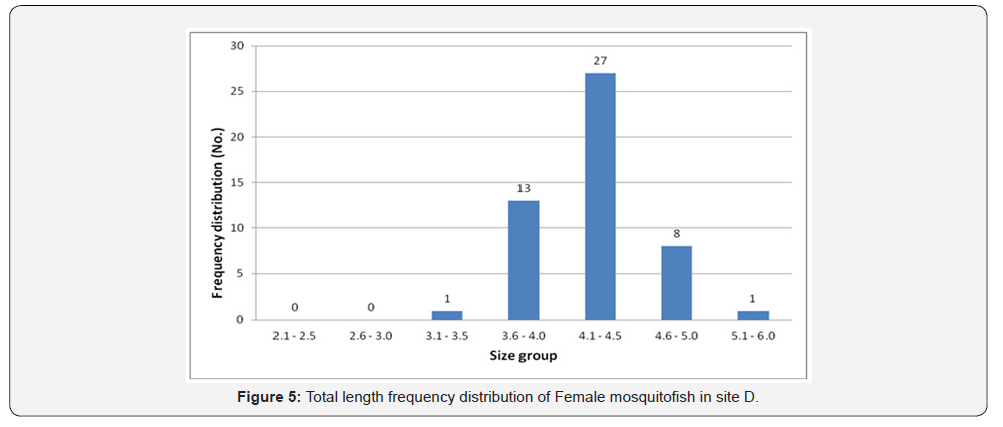
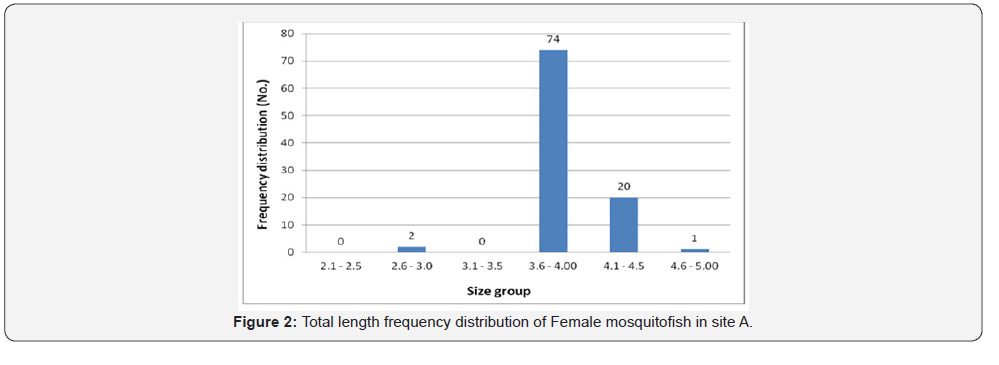
Female mosquitofish collected during this study ranged from 3.0cm to 4.6cm in total length in site A, 3.0 to 5.3 in site B, 2.4 to 4.2 in site C, and ranged from 3.5 to 5.3 in site D. Male mosquitofish collected during this study ranged from 2.2cm to 4.0cm in total length in site A, 2.1 to 5.0 in site B, 2.5 to 3.8 in site C, and ranged from 2.4 to 4.2 in site D. Accordingly, total length frequency distribution within the sites showed that female fish size (TL) ranging between 3.6 and 4.0 was the highest in site A (74 fish) (Figure 2), in site B the highest number was between 4.1-4.5 (22 fish) (Figure 3), in site C was between 3.1 and 3.5 is the highest (43 fish) (Figure 4), and 27 fish in site D between 4.1 and 4.5 (Figure 5). However, there was a significant difference (P>0.05) according to t-test between the sites A, B and C. While site D did not differ significantly with site B.

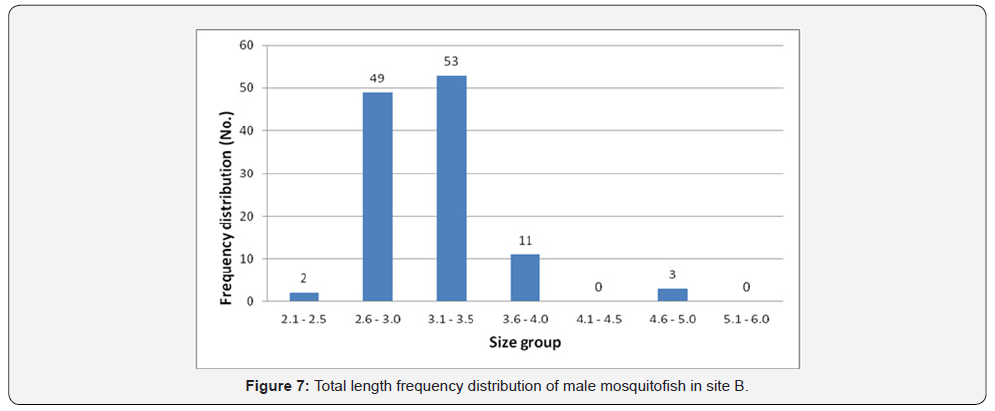
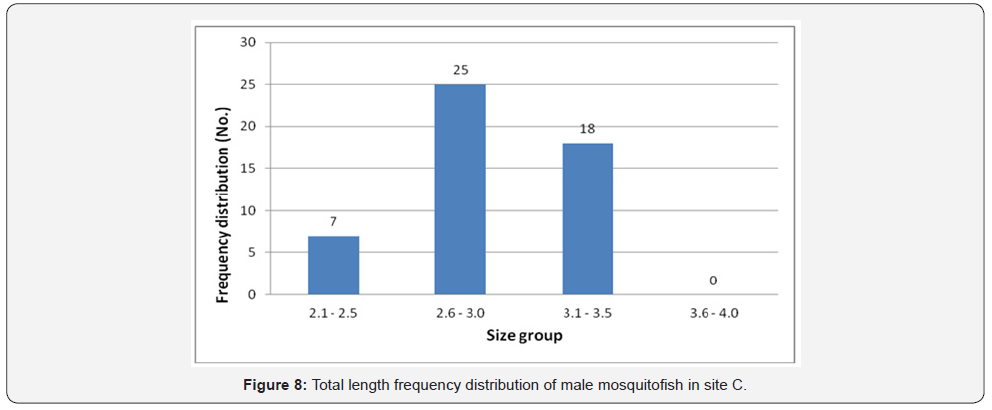
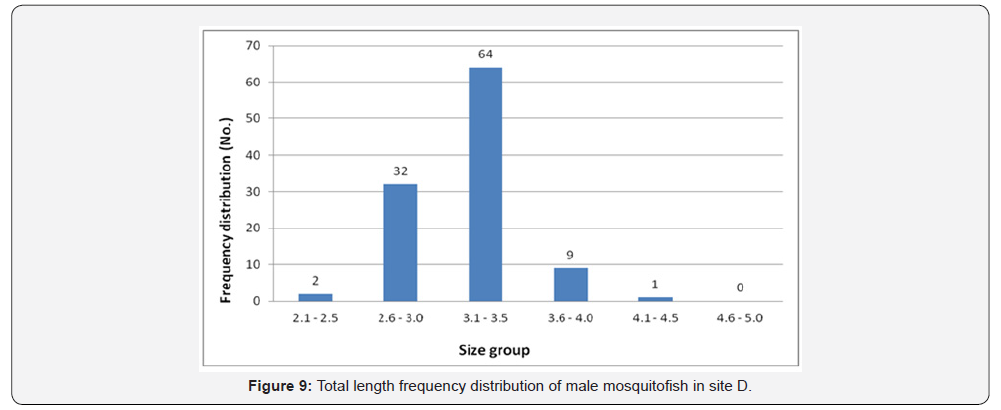
Total length frequency distribution within the sites showed that male fish size (TL) ranging between 2.6 and 3.0 was the highest in site A (75 fish) (Figure 6), in site B the highest number was between 3.1-3.5 (53 fish) (Figure 7), in site C was between 2.6 and 3.0 is the highest (25 fish) (Figure 8), and 64 fish in site D between 3.1 and 3.5 (Figure 9). However, there was no significant difference, (P<0.05) according to t-test, between the sites.
Mean total length of female mosquitofish in site A was 3.95 ± 0.2cm (mean ± SD), in site B was 4.52 ± 0.3cm, in site C was 3.44 ± 0.3cm and in site D was 4.21±0.3cm. While mean total length of male mosquitofish in site A was 2.73 ± 0.2cm (mean ± SD), in site B was 3.14 ± 0.3cm, in site C was 2.94 ± 0.28cm and in site D was 3.19 ± 0.3cm. The statistical analysis, however, showed that there was a significant difference in the total length of females between all the sites (P>0.05). While there were no significant differences only between sites B and D in the total length of male’s fish.
Female mosquitofish weight ranged from 0.53g to 1.44g in site A, 0.1g to 2.1g in site B, 0.1g to 0.8g in site C and 0.3g to 1.6g in site D. Males mosquitofish weight ranged from 0.1g to 0.9g in site A, 0.1 to 2.0g in site B, 0.1 to 0.3 in site C and 0.1g to 1.0g in site D. Moreover, mean female fish weight in site A was 0.86 ± 0.1, in site B was 1.28 ± 0.3, in site C was 0.50 ± 0.1 and in site D was 1.02 ± 0.27. While, mean male fish weight in site A was 0.21± 0.1, in site B was 0.33 ± 0.2, in site C was 0.26 ± 0.06 and in site D was 0.35 ± 0.1. The statistical analysis showed that was a significant differences (P>0.05) in all female weight in all sites, while there was no significant differences in male weight between site A and C, site B and D. Lastly, Mean female length and weight were larger than mean male length and weight in all sites.
Condition factor (K)
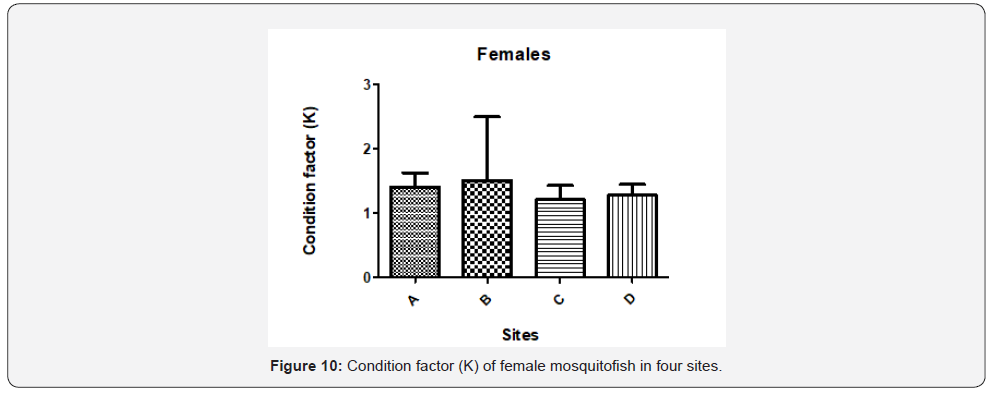
The variation of condition factor (K) in females and males of G. affinis within the sites is shown in Figure 10 and Figure 11. The data showed different trends in both sexes. Furthermore, in males the minimum K value was reported in site A (0.80) and the maximum in site B (0.90). The mean K values of female’s fish of site A was 1.40 ± 0.2, site B was 1.51 ± 0.9, site C was 1.20 ± 0.2, and site D was 1.27 ± 0.1. Moreover, the statistical analysis showed that Sites A and C, B and C differ significantly (P>0.05) while the other sites do not differ significantly. However, in males, K values was not significantly different (P<0.05) between the sites according to Tukey’s Multiple Comparison test.
Discussion
Sex ratio
Sex ratio calculation is very important to understanding the relationship between fishes, their environment and population status [17]. However, it may vary from species to species, or even in the same population at different times, because it may be influenced by several factors such as reproductive behavior, food availability and environmental conditions and genetic factors like sex chromosomes [18].
Environmental conditions are expected to be quite variable in the four studied lakes regarding to genetic variation and its influences on sex ratios could have effect on the equilibrium states in the populations, as has been found in some fishes where the influence of genetics and temperature on sex ratios differs along a latitudinal gradient [19,20]. Therefore, studying the sex ratio between population variations might be of great help to further increase our knowledge of the evolution of sex-determining mechanisms.
Furthermore, sex ratio indicates the proportion of male and female in the population and is expected to be 1:1 in nature, any differences from this ratio may indicate the dominance of one sex over the other [21]. The sex ratio of males to females was 1.1:1 in site A, 2.5:1 in site B, 0.6:1 in site C, and 2:1 in site D (Table 1) and the analysis showed that in sites A and B the ratio of males and females differ significantly (P=0.42 and P=1.08, respectively), While in site C and D the differences was statistically insignificant (P<0.05). However, in sites B and C males were dominance over females. There was not much studies on sex ratio of mosquitofish, therefore results were compared with other fish species such as major Carp where a ratio of 1:1.4 was reported, which did not deviate significantly from the its hypothetical distribution [22].
Growth trends
The highest number of males of the total length was between 2.7-3.2cm in all lakes, and the lowest number of males of the total length was between 4.5-5.0cm in site B. while, the highest total length of females was 4.5-5.0cm, the lowest of total length was 2.7-3.2cm and 5.1-5.6 cm. Gkenas et al. [23], found G. holbrooki females live longer and reach larger size than males where the smallest male was 1.6cm and the largest male was 3.4cm, while the smallest female was 2cm and the largest female was 4.3. However, In this the study observed the highest mean total length of females was 4.50 ± 0.34 in site B, and the lowest mean total length was 3.42 ± 0.35 in site C. while the highest mean total length of males was 3.19 ± 0.29 in site D, and 2.73 ± 0.28 in site A was the lowest mean of males. however, the highest mean weight of females was 1.28 ± 0.33 in site B and 0.49 ± 0.01 in site C, the lowest mean of males was 0.35 ± 0.12 in site D, 0.21 ± 0.09 in site A. Mean total length and weight of females were higher than mean total length and weight of males because females need longer time to mature and continue growing throughout their life [24]. Males stop growing or exhibit a decelerating growth rate after the gonopodium has been completely formed and they do not live as long after reaching maturity [25]. After the completion of the reproductive period the largest individuals (mostly females) become rare due to the high mortality which follows the reproduction [24,26,27]. In other study in turkey, females ware greater in size and weight than males in population studied, furthermore, the total length and weight of males were from 1.0 to 3.3cm (average, 2.34 ± 0.14cm) and from 0.01 to 0.49g respectively, while females from 1.3 to 5.7cm (average, 3.35 ± 0.35cm) and from 0.01 to 1.90g [28].
Condition factor (K)
The condition of fishes is influences by the gonadal development, feeding activity and several other factors [29]. In the present investigation, comparing K within different populations of mosquitofish collected from different lakes. The results suggest that feeding intensity may be the main but not the only factor responsible for the variation in K in mosquitofish. It seems that there is an interrelation between feeding intensity and reproduction and these two factors are the most important that influence the condition factor [30]. These findings corroborate observations by Qasim [31] in Centronotus gunnellus and Das [32] in Mugil cephalus.
Conclusion
The populations structure of the mosquitofish Gambusia affinis collected from the four different lakes in Benghazi, Libya exhibited significant differences between the lakes. The differences could be mainly attributed to degradation in water quality, and lakes productivity. However, more studies need to be done regarding to the water quality parameters as well as genotype differences between the species within the populations in the lakes.
References
- Hoese HD, Moore RH (1977) Fishes of the Gulf of Mexico. Taxas, Louisiana, and Adjacent waters. Texas A and M University press, College station Tx, p. 327.
- Robins CR, Ray GC, Douglas (1986) A field Guide to Atlantic Coast Fishes. The Peterson Field Guide Series. Houghton Mifflin Co., Boston, p. 354.
- Gerberick JB, Laird M (1966) An annotated bibliography of papers relating to the control of mosquitoes by the use of fish. World Health Organization. EBI series, London.
- Arthington AH (1988) Diet of Gambusia affinis holbrooki (baird and Girard) in streams of the Brisbane region, Southeastern Queensland, Australia. Proceedings of the Societe Internationale Limnologie.
- Webb C, Joss J (1997) Does predation by the fish Gambusia hobrooki (Atheriniformes: poeciliidae) contribute to declining frog populations? Australion zoologist 30(3): 316-324.
- Arthington AH, Loloyd LN (1989) Introduced poeciliidae in Australia and New Zealand. In: Meffe GK, Snelson FF (Eds.), Evolation and Ecology of Livebearing Fishes (poeciliidae), prentice-Hall, New york, USA, pp. 33-348.
- Ivantsoff W, Aran (1999) Detection of predation on Australian native fishes by Gambusia holbrooki. Marine and Freshwater Research 50(5): 467-468.
- Specziar A (2004) Life history pattern and feeding ecology of the introduced eastern mosquitofish, Gambusia holbrooki, in a thermal spa under temperate climate, of lake Hévíz, Hydrobiologia 522(1-3): 149-260.
- Weihrauch F (2006) Eastern mosquitofish Gambusia holbrooki as a predator of dragonfly eggs (Teleostei: poeciliidae; Odonata: Libellulidae). Libellua 25(3-4): 209-214.
- Froese R, Pauly D (2013) Fishbase.
- Kurmholz LA (1948) Reproduction in the Western mosquito fish Gambusia affinis affinis (Baird and Girard), and its use in mosquito control. Ecological Monographs 18(1): 1-43.
- Haas RC, Thomas MV, Towns GL (2003) An assessment of the potential use of Gambusia for mosquito control in Michigan. Michigan department of natural resources fisheries technical report (2).
- Nordlie FG, Mirandi A (1996) Salinity relationships in Fresh water population of eastern mosquitofish. Journal of Fish Biology 49(6): 1226-1232.
- Carles A, Emili GB (2007) Life history variation of invasive mosquitofish (Gambusia holbrooki) along a salinity gradient. Biological Conservation 39(1-2): 83-92.
- Sokal RR, Jamesrohlf F (1987) Introduction to biostatistics. (2nd edn) Freeman and Co. New York, USA, p. 363.
- Pauly D (1983) Some simple methods for the assessment of tropical fish stocks. FAO Fisheries Tech. Pap. 234, FAO. Rome, p.
- Oliveira MR, Costa EFS, Araújo AS, Pessoa EKR (2012) Sex Ratio and Length-Weight Relationship for Five Marine Fish Species from Brazil. Journal of Marine Biology and Oceanograph 1: 2.
- Baroiller JF, D’Cotta H, Saillant E (2009) Environmental effects on fish sex determination and differentiation. Sexual Development 3(2-3): 118-135.
- Lagomarsino IV, Conover DO (1993) Variation in environmental and genotypic determining mechanisms across a latitudinal gradient in the fish, menidia menidia. Evolution 47(2): 487- 494.
- Pen I, Uller T, Feldmeyer B, Harts A, While GM, et al. (2010) Climate driven population divergence in sex-determining systems. Nature 468(7322): 436-438.
- Bal DV, Rao KV (1984) Marine Fisheries; McGraw – Hill Publishing company Limited, p. 457.
- Chatterji A, Ansari ZA (1982) Fecundity of Dolphin Fish, Coryphaena hippurus L. Mahasagar, Bull National Institute. Oceano 15(2): 129-133.
- Gkenas C, Oikonomou A, Economou A, Kiosse F, Leonardos I (2012) Life history pattern and feeding habits of the invasive mosquitofish, Gambusia holbrooki, in Lake Pamvotis (NW Greece). Journal of Biological Research 17: 121-136.
- Fernándeƶ- Delgado C, Hernando JA, Herrera M, Bellido M (1989) Age and growth of yellow eels, Anguilla anguilla, in the estuary of the Guadalauivir River (south- westspain). Journal of Fish Biology 34(4): 561-570.
- Thresher RE, Canning M, Bax NJ (2013) Demographic effects on the use of genetic options for the control of mosquitofish, Gambusia holbrooki. Ecol Appl 23(4): 801-814.
- Vargas MJ, De Sostoa A (1996) Life history of Gambusia holbrooki (Pisces, Poeciliidae) in the Ebro delta (NE Iberian Peninsula). Hydrobiologia 341(3): 215- 224.
- García-Berthou E (1999) Food of introduced mosquitofish: ontogenetic diet shift and prey selection. Journal of Fish Biology 55(1): 135-147.
- Erguden SA (2013) Age, growth, sex ratio and diet of eastern mosquitofish Gambusia holbrooki Girard, 1859 in Seyhan Dam Lake (Adana/turkey). Iranian Journal of Fisheries Sciences 12(1): 204-218.
- Doddamani M, Rameshaand TJ, shanbhogne SL (2001) Length-weight relationship and condition factor of stolephorus bataviensis from mangalore area. Indian Journal of Fisheries 48: 329-332.
- Froese R (2006) Cube law, condition factor and weight-length relationship: history, meta-analysis and recommendations. Jou Appl Ichthyol 22(4): 241-253.
- Qasim SZ (1957) The biology of Blennius pholis L. Proceedings the Zoological Society 128(2): 161-208.
- Das HP (1978) Studies on the Grey Mullet, Mugil cephalus (Linnaeus) from the Goa Waters; A Ph.D. Thesis Submitted to the University of Bombay.






























