Application of Lab-Scale MBBR to Treat Industrial Wastewater using K3 Carriers: Effects of HRT, High COD Influent, and Temperature
Ahmadlouydarab Majid* and Mehrazaran Mahna
Faculty of Chemical & Petroleum Engineering, University of Tabriz, Iran
Submission: April 22, 2019; Published: June 28, 2019
*Corresponding author: Ahmadlouydarab Majid, Faculty of Chemical & Petroleum Engineering, University of Tabriz, Iran, Po Box: 51666-16471
How to cite this article: Ahmadlouydarab M, Mehrazaran M. Application of Lab-Scale MBBR to Treat Industrial Wastewater using K3 Carriers: Effects of HRT, High COD Influent, and Temperature. Int J Environ Sci Nat Res. 2019; 20(2): 556031. DOI:10.19080/IJESNR.2019.20.556031
Abstract
In current study, the efficiency of a lab-scale MBBR was studied using K3 Kaldnes carriers. COD containing contaminant of 1000 to 3500mg/l was used to examine the system performance by measuring BOD and COD at different HRTs, temperature and inlet CODs. It should be noted that K3 Kaldnes carriers was not used in previous studies. To determine optimal HRT, BOD and COD of the effluent were examined at different HRTs of 3, 5, 8, and 12 hours. The optimum HRT was 8 hours with COD removal efficiency more than 80%. Despite of HRT=12 hours with the highest COD removal efficiency ~86%, HRT=8 hours were chosen as the optimal HRT due to the time-consuming refinement process with only a slight difference of 3% in removal efficiencies. Additionally, the effects of temperature in the range of 19 to 32°C were studied at optimal HRT. The results indicate that there is a sharp change in COD removal efficiency slope at a temperature range between 20 to 25°C due to the high activity of microorganisms, leading to an increase in the COD removal efficiency from 70% to ~90%. By increasing the temperature beyond 25°C, the rate of increase in COD removal efficiency is reduced by as much as 94%. According to the results, 27°C may be considered as the optimum temperature. Studying the effects of different inlet CODs up to 3500mg/l reflects a non-monotonic behavior in MBBR’s performance. In general, for all inlet CODs, MBBR system shows more than 80% removal efficiency.
Keywords: K3 carriers; MBBR; Wastewater; Lab-scale; High COD; MLSS
Abbrevations: ACC: Activated Carbon Concentration; MFR: Media Filling Ratio; TPH: Total Petroleum Hydrocarbon; TSS: Total Suspended Solid; BOD: Biological Oxygen Demand; COD: Chemical Oxygen Demand; ASP: Activated Sludge Process; MBR: Membrane Bioreactor System
Introduction
With industrial development in most countries, industrial wastewater effluents are also increasing. On the other hand, nowadays, the problem of water scarcity of the world has become one of the major concerns of the societies. Therefore, it is important to find an optimal and cost-effective way to recycle wastewater effluents. Industrial wastewater is made by water consumption in industrial activities and during various stages of production. Sometimes, they constitute the most dangerous type of wastewater. The bulk of industrial wastewater also contains contaminants that are characterized by organic matter (soluble or insoluble) which are the most important contaminates. The most important compounds of industrial wastewater are arsenic, cadmium, mercury, and lead. If they are not properly collected and routed to the wastewater treatment plant, these materials will enter flowing water streams and so to the environment, which will have irreversible harmness to the environment and human beings. The quantity and quality of wastewaters or industrial effluents in different factories and industries vary depending on the type of production, which lead to have variable treatment processes.
In the treatment of domestic and industrial wastewaters, physical and biological treatment methods are used to obtain environmental certification. The most important issue in the treatment of industrial wastewater is to understand its nature and the quality of elements and factors such as toxic compounds, decomposing factors, Total suspended solid (TSS), (biological oxygen demand) BOD, (chemical oxygen demand) COD and color. With solid knowledge about these factors, it will be possible to design appropriate treatment methods for industrial wastewater and effluents.
Among the available techniques, scavenging, flotation with soluble air, adsorption, and membrane filtration are physical methods. Also, chemical methods include chemical oxidation, electrochemical oxidation, and coagulation. However, physical and chemical methods are expensive because of the high cost of chemicals, equipment, and the need to remove excess sludge. Therefore, biological methods are preferred due to simplicity, affordability and environmental compatibility [1].
The biological methods involve two types of aerobic and anaerobic systems. Different biological aerobic systems for wastewater treatment such as activated sludge process (ASP), membrane filtration system (MBR), (up flow anaerobic sludge bioreactor) UASB system are utilized. However, all these systems have drawbacks that cannot be ignored [2]. In the activated sludge process (which is currently one of the most widely used methods for industrial wastewater treatment), there should be sludge returning cycle, which is costly. In addition, the removal efficiency of organic matters from wastewater is not satisfactory due to the complexity of chemicals in the wastewater industrial. Moreover, activated sludge system is continuously exposed to changes in pH, temperature, solids concentration, erosion, and other parameters. Overall, these parameters decrease the system’s life and efficiency [3]. Compared to the membrane bioreactor system (MBR) with the same apparent volume, (moving bed biofilm reactor) MBBR provides more specific volume (high capacity) for biological treatment. In MBR systems, membranes should also be cleaned regularly, which results in waste of time. The rest of the systems are not recommended due to the costs and low efficiency over long periods. Indeed, these are two main factors of being a non-optimal system. Thus, MBBR can be considered as an innovative and cost-effective with high removal efficiency [4-6].
In 1980, for the first time in history, the MBBR system was used in Norway for wastewater treatment. These bioreactors have advantages such as low-pressure drop. Their resistance to factors such as temperature changes, pesticides, pH changes are also very good. Today, more than 500 MBBR systems are used to treat wastewaters and remove BOD, COD, nitrification, etc. all around the world [7,8]. It is also an optimal system for processes such as organic matters removal, nitrification, and denitrification since it has high efficiency in eliminating and reducing biodegradable organic and inorganic matters. Basically, the MBBR system is an evolved type of fixed bed and activated sludge system defined based on biofilms so that, the microorganisms can stick to polyethylene carriers and start the process of filtration. In aerobic processes, the movement of carriers is due to the aeration while in anaerobic reactors, using a mixer (with a horizontal or vertical shaft) helps carriers to move. MBBR system does not require secondary treatment. Additionally, the whole volume of the reactor can be used for microorganisms’ growth [9]. Todays, MBBR systems are used in textile wastewater domestic wastewater, sewage, livestock, poultry, refineries, and petrochemicals [10], industrial wastewaters [11] and so on.
In 2006, Xiao et al. [12] conducted a test to present the structural features of biomass in the hybrid MBBR aeration tank [12]. The experiment took place in two subsequent periods, which differed in hydraulic and substrate loads. The physical characteristics of attached-growth biomass, such as biofilm thickness, density, porosity, and inner and surface fractal dimensions were studied. Moreover, parameters of suspendedgrowth biomass including floc size distribution, density, porosity, inner and surface fractal dimensions, were investigated and compared to those of attached-growth biofilms. The authors used the activated sludge, and MBBR systems with filling ratio of 70% from Kaldnes carriers in two subsequent periods. The average density of attached-growth biofilm was 5 - 13 times higher than that of activated sludge flocs, though they were coexisting in the same reactor and same ecological environments. The boundary fractal dimension of biofilm was found to be always higher than that of activated sludge flocs.
In 2007, Choi and his colleagues [13] conducted a research on two different systems i.e., MBBR system and an integrated continuous bed activated sludge (ICBAS) system [13]. Researchers used these systems for nitrification and denitrification in both summer and winter seasons. The results showed that the MBBR system performs better than ICBAS in ambient air conditions in removing ammonia. In addition, the limited conditions for refining were not an obstacle for MBBR to have higher performance compared to that of ICBAS. Similar experiment was carried out by Motsch et al. [14] at Water Protection and Prevention Agency in Virginia-USA [14]. During the operation, favorable outcomes were obtained e.g., for the MBBR system the ammonia removal efficiency was higher than that of ICBAS. The tough environmental conditions for treatment process when using ICBAS, resulted in changes in the mixed liquor suspended solid (MLSS) level.
In 2010, Ferrai and colleagues [15] studied the kinetic and stoichiometric parameters by testing MBBR biofilms for urban wastewater treatment [15]. Authors used isolated and nonisolated samples from a large-scale laboratory MBBR, and oxygen uptake rate (OUR) heterotrophic sections of biomass for study. It was then modeled using ASM3 system. Isolated biofilms showed more tendency to sediment inside the bioreactor compared to non-isolated biofilms. Additionally, it was observed that there is a limited growth of biomass inside sediment sludge.
In other experimental research, Stamper and his colleagues studied the effects of combining MBBR system with an anaerobic system for better and stronger treatment [16]. Authors predicted that the anaerobic systems are prone to failure and require a lot of investment.
Also, in 2015, Mr. Barwal and his colleagues [17] investigate the effects of bio-carriers on the oxygen uptake rate in the effluent of the reactor with an aim to provide optimized filling ratio for the practical operation of a MBBR [17]. The value of OUR increased 2-3 times with the augmentation of carrier filling ratio, but it decreased when more carriers were added inside the reactor.
In 2015, Goswami et al. [18] compared ASP and MBBR systems performance in composting wastewater treatment plant made of chrome [18]. The plant contained desensitizable substances such as phosphorus, sulfur, chromium, and other toxic substances. Authors concluded that the higher the number of biofilms was, the higher the concentration of biomass was.Additionally, the results indicated that COD removal rate in MBBR was 80%, which was much higher than that of ASP (60- 70%). However, there was no difference in nitrogen removal for two reactors.
In 2016, Sayyah-Zadeh and colleagues [19] tried to improve the efficiency of the MBBR system in order to treat hydrocarbons using active carbon monoxide carriers [19]. Researchers monitored the performance of the system by filling the carrier’s holes with activated carbon granules made of almonds and walnuts. Parameters i.e., activated carbon concentration (ACC), COD, and media filling ratio (MFR) were measured. The results showed that the removal efficiency of COD and total petroleum hydrocarbon (TPH) in the MBBR using carriers filled by almond and walnut is higher than that of polyethylene carriers.
In the same year, Young et al. [20] conducted research to study effects of low temperatures on the ammonia removal efficiency in a MBBR bioreactor [20]. The process was carried out between 1 and 20°C. Various biofilms were observed inside the carriers in 20°C rather than 1°C. Authors could thicken the biofilm thickness at 1°C. Results indicated that under hard conditions (low temperature, chocks, and so on) the removal efficiency of ammonia gets lower mainly due to low mass transfer.
Recently, Haung and his colleagues [21] prepared a report on the importance of attached biomass in IFAS, MBBR and MBR systems [21]. The results showed that MBBR had better performance in naphthenic acids removal. Most recently, a study was done on the removal of organic solids from the wastewater using MBBR system. As an important result, it became clear that the MBBR system had the power to break down the drug substances [22-24].
The main objectives of this study, considering literature review, can be summarized as follow:
a) In previous studies, effects of a limited range of temperature on the amount of COD removal has been addressed. Studied rages are 1 to 20 ºC and 30 to 50 ºC [20,25]. This means that there is not a comprehensive understanding about the temperature effect on the MBBR system behavior. In this experimental study to enhance the current knowledge, the removal efficiency of different amounts of COD and the activity of microorganisms on the lab-scale MBBR system where temperature ranges from 19 to 32 ºC will be studied.
b) Literature review shows that the maximum studied COD in the wastewater stream was 2500mg/l. In current research, evaluation of the performance of a lab-scale MBBR system will be investigated using different input CODs up to 3500mg/l.
c) There is lack of comprehensive knowledge about the optimal HRT determination as well as its effects on BOD removal efficiency. Therefore, in this study, while determining the optimal HRT, its effects on the removal of BOD and COD will be addressed.
d) Despite the previous studies in which K1 and K2 carriers have been used, in this research, K3 carriers will be utilized in industrial wastewater treatment process.
Materials and Methods
Generally, in order to build the MBBR reactor, the following steps were considered. Moreover, necessary chemicals were prepared, and mandatory tests were carried out.
At first step, the lab-scale MBBR was designed and made to conduct the tests, which will be described in section 2.1. At second step, industrial wastewater sludge was prepared, and then specific volume of sludge was poured into the MBBR reactor. Consequently, artificial wastewater was added to the primary sludge. After that, the system was aerated. Subsequently, microorganisms were adapted to refine and remove organic matters. After ensuring that the sludge was compatible with artificial wastewater and COD was stable inside the bioreactor, COD and BOD removal rates were investigated at different HRTs and temperatures.
MBBR configuration and operation
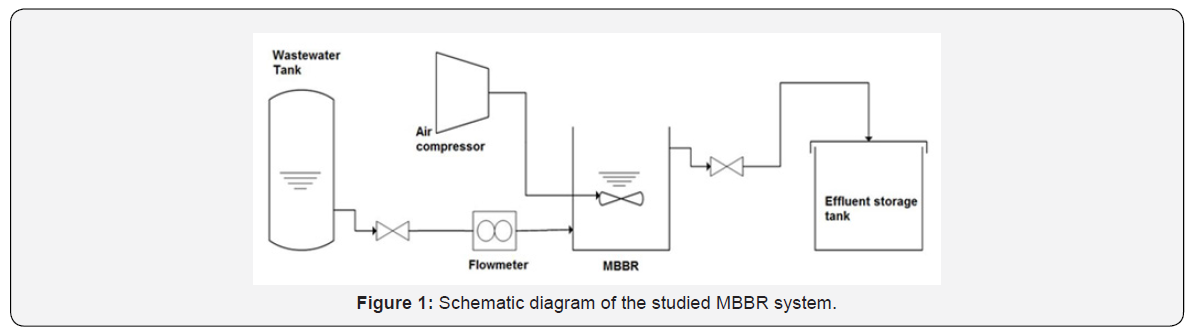
Figure 1 represents the schematic diagram, and Figure 2 shows the experimental setup of the MBBR system studied in this study.
The bioreactor is made of Plexiglas, which has a thickness of 6mm. The length of the bioreactor is 30cm, and its width is 15cm. It’s pure and wastewater filled heights are 40 and 24cm, respectively. In order to control inlet and outlet of the fluids and to sample wastewater from the bioreactor, five valves were connected to the rectors as follow: one valve was connected to the bottom of the reactor and four valves connected to the bioreactor’s walls with distances of 5, 10, 25, and 35cm from bottom of the bioreactor. To run the bioreactor, 12 liters of the bioreactor volume was filled with dense sludge (Return sludge of Pegah Co. East Azerbaijan). Then the total volume of the fluid inside the bioreactor was reached to the 24 liters by adding artificial wastewater. The experimental set-up of the MBBR system is shown in Figure 2.


From standard Kaldnes packing medias, the K3 carrier manufactured by Pakzist Co., was used to fill the bioreactor. Figure 3 shows the different types of Kaldness carriers including K1, K2, and K3. The characteristics of the K3 packing media used in this study are presented in Table 1. In order to suspend carriers inside the bioreactor and supply the required oxygen, three small air pumps with a total aeration rate of 8 liters per minute were used.
Chemicals and measuring devices
All the chemicals utilized in this study were purchased from Merck. The main chemicals used in this research are as follows:
a) H2SO4, K2Cr2O7, HgSO4 and Ag2SO 4 to measure COD,
b) Silica gel to balance solid’s moisture and weight,
c) K2HPO4 and KH2PO4 salts as sources of phosphorus,
d) CH4N2O as nitrogen sources to provide a ratio of P: N: COD of 1: 5: 100 [26],
e) Molasses as a source of nutrition feed for biofilms and COD stabilization.
The following devices were also used to perform measurements and tests:
a) Spectrophotometer (UV-VIS 2800 manufactured by Unico) for COD measurement,
b) Digital scale (AS 220 R2 model manufactured by Rad Wag) with a precision of 0.001gram, BOD measurement device (OXITOP RESPIROMETER),
c) Avon (YCO-500-64 model manufactured by Yaran) for measuring MLSS
d) Air pumps (Air-8000 made by Resun Company, and AT- 703 manufactured by Atman) to supply demanded oxygen.

Preparation of the artificial wastewater

Molasses, potassium dehydrogenase phosphate and urea were utilized to make artificial wastewater. Molasses were used to stabilize the COD of the sewage entering the bioreactor and to fix the COD of the initial wastewater contained within the bioreactor. Also, K2HPO4 was used to stabilize phosphorus level in the initial wastewater contained within the bioreactor. In addition, urea (CH4N2O) was used to stabilize nitrogen level in the initial wastewater inside the bioreactor.
Adaptation of sludge and microorganisms
At first step, to adapt the slurry environment for the activity of microorganisms, synthetic wastewater as feed was injected into the MBBR system daily. Meanwhile the system was continuously aerated to stabilize the MLSS of the wastewater in the bioreactor. After one month of commissioning the system, the MLSS and COD parameters were sampled from the bioreactor for the measurement purpose. During the period of adaptation of the sludge and microorganisms, the temperature of the laboratory was between 16 and 17°C. After 90 days, the system’s MLSS was stabilized at 3200mg/l. Moreover, the COD removal rate was about 45%, indicating the adaptation of the sludge and microorganisms [26]. Finally, the pH of the system was adjusted to about 7, which is very suitable for growth of the microorganisms [26]. It should be noted that at the end of the adaptation period, the very first biofilm layers were observed on the internal surfaces of the K3 carriers. At the same time the external surfaces of the carriers became slippery.
Sampling for analysis
Following procedure was used to sample and analyze. First, after a specific period, the aeration pump was turned off to allow the sludge to be deposited inside the bioreactor. Then, for the COD test, 500ml of wastewater sampled from the bioreactor. According to the results of the COD removal, a certain amount of the same sample selected for BOD measurement. Due to the aerobic nature of the process of microorganism activity inside the system, supply of oxygen for microorganisms is very important. Therefore, by performing regular tests pH control parameters and MLSS were evaluated. When MBBR was running, the pH of the system was kept at an average of 7, and the MLSS was maintained at a range of 3500-3000mg/l [26].
Results and Discussion
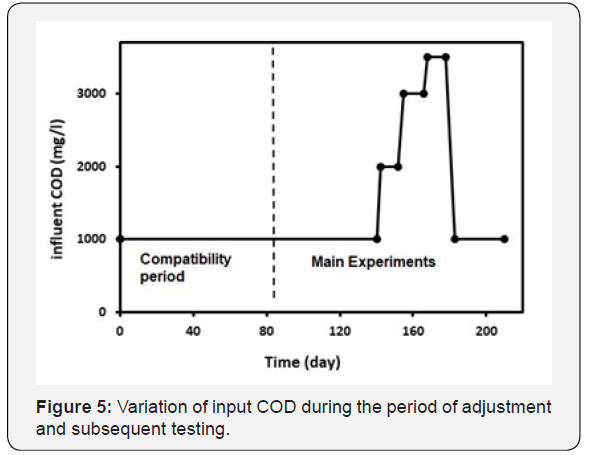
Upon completion of the adaptation period (90 days), the main experiments were carried out. During the tests, the following objectives were studied:
a) Effects of influent wastewater with High COD on MBBR system performance,
b) Effects of HRT on bioreactor operation (removal of BOD and COD) in order to find an optimum HRT,
c) Effects of bioreactor temperature on its performance while using input wastewater with average COD of 1000 mg/l in optimal HRT./p>
Variation of inlet wastewater COD with time during the microorganism’s adaptation and main experiments has been presented in Figure 5.
Biofilm growth
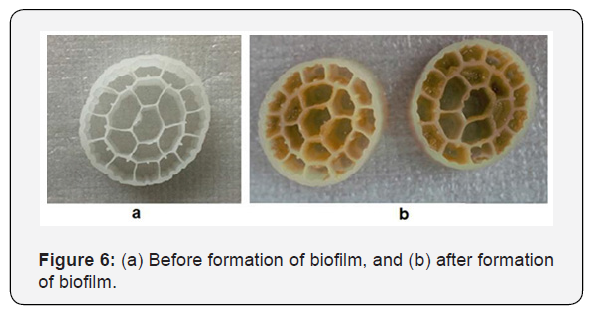
As already mentioned, at the end of the MBBR system compatibility period, biofilm formation was observed on the surface of the carriers and the external surfaces of the carriers got slippery [5]. After approximately one week (since the end of the adaptation period) the number of biofilms increased significantly. Figure 6 shows the inner and outer surfaces of K3 carrier before and after the formation of the biofilm layers. It should be noted that on the outer surfaces of some media packings, only a very thin layer of biofilm is observed. This is due to aeration and collision of the carriers which causes that the biofilm sticking to the outer surface of them is peeled and become a suspended biomass. For this reason, the inner surface of the media packings is considered an effective surface for biofilm formation.
HRT effect analysis
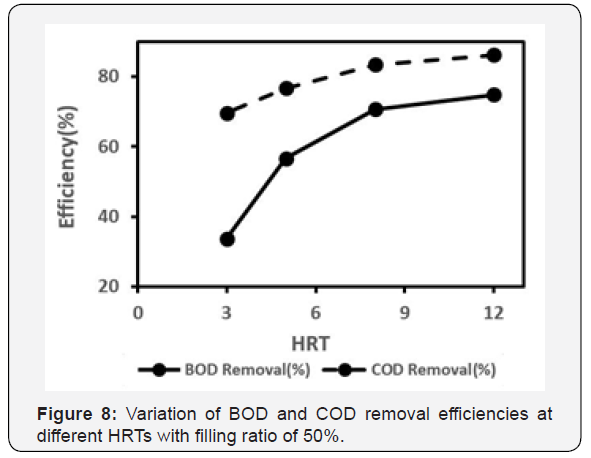
The removal efficiency of COD and BOD at different HRTs for the constant loading of COD=1000mg/l was investigated to find the optimum HRT. It worth mentioning that in previous studies, the MBBR system functionality evaluated at low HRTs, had a maximum COD removal at HRT of 2 hours [4].
Figure 7 shows the amount of effluent COD and BOD at a constant loading rate of COD=1000mg/l and BOD=490 mg/l at different HRTs. As shown in both Figure 7a & 7b, the COD and BOD effluent at HRT of 12 hours is the lowest which means both COD and BOD removals are the highest. In addition, the efficiency of both BOD and COD removals have been compared at different HRTs in Figure 8. As known, the amount of COD removal is always higher than BOD removal as COD represents both organic and non-biodegradable organic compounds, but BOD only contains biodegradable organic compounds. Also Figure 8 indicates that at low HRTs, COD is more eliminated in comparison with BOD. The reason is the lack of required time to start the activity of microorganisms in order to remove organic matter with biological treatments.

By increasing HRT, enough time to start the activity of these microorganisms is provided. On the other hand, as shown in Figure 8, the removal efficiency of COD and BOD at HRT of 12h is more than the others, which is 87% and 75%, respectively. At HRT of 8 hours, these efficiencies were calculated 84% and 71%, respectively. As the biological treatment processes are time consuming, and there is a marginal difference between removal efficiencies at HRT of 8 and 12 hour, someone may choose HRT = 8h as an optimal HRT.
Effects of different input CODs in optimal HRT analysis
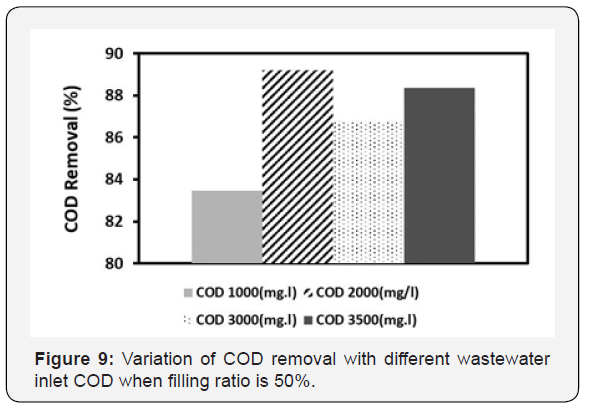
Since industrial wastewater has high CODs, treatment process should be done by a system which saves both time and cost. In the Figure 9, effects of inlet COD up to 3500mg/l on the MBBR system performance (COD removal efficiencies) has been shown. It should be considered that the COD of the system was continuously increased from 1000 to 3500mg/l. As the results indicate, the COD removal follows a non-monotonic behavior along with increasing the amount of inlet wastewater COD. This can be understood considering behavior of the microorganisms. At inlet COD range between 1000 to 2000mg/l, microorganisms still have the capacity to absorb more organic compounds. Therefore, with increasing COD up to 2000mg/l, the removal efficiency also increases. However, beyond COD~2000mg/l microorganism’s capacity for further COD removal is full. Therefore, an increase in inlet COD amount up to ~3000mg/l results in accumulation of COD inside bioreactor. Thus, this ends in reduction of COD removal efficiency. However, when resident time increases the MBBR system gets compatible with high CODs (~3000mg/l). Subsequently, this leads to an increment in both activity and number of microorganisms. Consequently, these result in COD removal increase. As Figure 9 also shows, the MBBR system has higher efficiencies > 80% at high CODs, which can indicate that the MBBR system is suitable for industrial wastewater treatment processes.
Temperature effect analysis
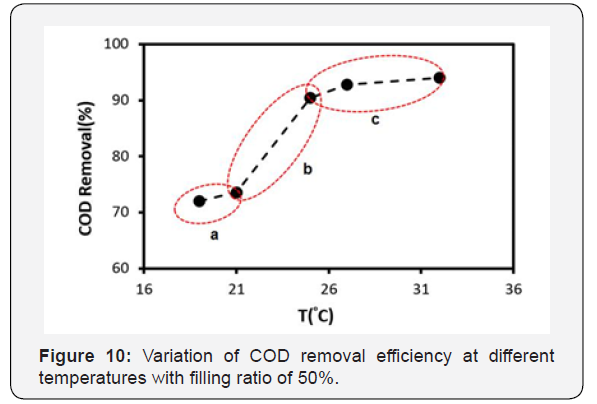
As already mentioned, temperature is one of the most important parameters in biofilm growth rate and compatibility during system stabilization for testing [27]. Due to the lack of sufficient knowledge about the effects of temperature on the performance of the MBBR system, and in order to enhance current knowledge, the system performance was studied at a temperature range of 19 to 32°C. Effects of temperature variation on the COD removal efficiency has been shown in Figure 10. According to the results, the COD removal efficiency diagram can be divided into three phases. As shown in phase (a), the COD removal efficiency is increases very slowly to ~72% between 19 to 21°C. In phase (b), there is an abrupt increase in the removal efficiency reaching up to 90%. In phase (c), the removal efficiency is increased slightly.
Overall, this behavior of removal efficiency can be rationalized as follow: In phase (a) (approximately less than 20°C) due to the limited ability of microorganism’s adaption with low temperature conditions; the COD removal efficiency increases very slowly up to ~72%. By increasing the temperature of the system from 20°C up to about 25°C (phase b), due to the sudden rise in the microorganism’s activity, sharp increase in COD removal efficiency from ~72% to ~92% happens. At the same time, the thickness of biofilms also increases. The highest COD removal efficiency of 94% occurs at a temperature of 32°C in phase (c), which is much better than the results reported in literature [25]. However, from 25 to 32, as mentioned, the removal efficiency rate increases slowly. For example, the COD removal efficiency is 92% at 27°C. The biofilm thickness observed in inner surfaces of the carriers at 32°C is higher than the biofilm thickness at 27°C. In general, increasing the thickness of the biofilm reduces the mass transfer and decreases the rate of increment in removal efficiency. Hence, it is expected that due to the reduction in mass transfer, the COD removal efficiency will grow very slowly by increasing temperature from 25 to above 32°C.
Conclusion
MBBR is one of the modern and innovative systems for urban and industrial wastewater treatment. Many studies have been done to prove that this system is suitable for wastewater treatment process in comparison with others. MBBR does not have common problems such as sludge bulking and rising, foaming, poor sludge settling, and carriers clogging. Some features such as strong resistance to impact, and no need to return the sludge make the system much easier to operate [4]. Besides, it is tenacious at high temperatures and CODs, as well as against shocks such as pH [28] etc.
In this study, after adaptation of microorganisms and their accumulation on K3 carriers, system performance was studied by examining the removal efficiency of BOD and COD at different HRTs, temperature, and wastewater inlet CODs. Studied COD range was 1000 to 3500mg/l. It was for first time that K3 carriers were used to study the MBBR system.
The BOD and COD parameters were determined at HRTs of 3, 5, 8, and 12 hours with a filling ratio of 50%. Although the HRT of 12 hours had a COD removal efficiency of 86%, but because of a slight difference of 3% and shortening the treatment process, HRT of 8 hours was selected as an optimum HRT.
In addition to choosing the optimal HRT, the effects of different temperatures ranging from 19 to 32°C were also investigated. The results indicate that there is an abrupt change in the COD removal efficiency at temperature range between 20 to 25°C because of high activity of the microorganisms. By raising the temperature to 25°C, the activity of microorganisms is relatively reduced, and the rate of COD removal efficiency increment is decreased. According to the results, the optimum operating temperature was suggested to be 27°C.
In MBBR systems, high level of inlet wastewater COD is also an essential and important factor in system’s performance detection, since in industrial and urban wastewaters the number of organic compounds is high. Therefore, the effects of different inlet CODs up to 3500mg/l were studied. The results indicate a non-monotonic behavior of the MBBR system, which can be understood considering the behavior of microorganisms in different CODs. In general, for all COD values, the MBBR system always has a removal efficiency more than 80%.
References
- Costa Lopes AM, Joao KG, Morais ARC, Lukasik EB, Lulasik RB (2013) Ionic liquid as a tool for lignocellulosic biomass fractionation. Sustainable Chemical Processes 1:3.
- Brandt A, Grasvik J, Hallett JP, Welton T (2013) Deconstruction of lignocellulosic biomass with ionic liquids. Green Chemistry 15(3): 550-583.
- Zhang YP (2008) Reviving the carbohydrate economy via multiproduct lignocellulose biorefineries. J Ind Microbiol Biotechnol 35(5): 367-375.
- Ragauskas AJ, Williams CK, Davison BH, Britovsek G, Cairney J, et al. (2006) The path forward for biofuels and biomaterials. Science 311(5760): 484-489.
- Clark JH (2007) Green chemistry for the second generation biorefinery sustainable chemical manufacturing based on biomass. Journal of Chemical Technology and Biotechnology 82(7): 603-609.
- McKendry P (2002) Energy production from biomass (part1): overview of biomass. Bioresour Technol 83(1): 37-46.
- Lynd LR, Weimer PJ, van Zyl WH, Pretorius IS (2002) Microbial cellulose utilisation: Fundamentals and biotechnology. Microbiol Mol Biol Rev 66(3): 506-577.
- Mai C, Kües U, Militz H (2004) Biotechnology in the wood industry. Appl Microbiol Biotechnol 63(5): 477-494.
- Hon DNS, Shiraishi N (1991) Wood and cellulosic chemistry, second edition, M. Dekkar Inc, New York, p. 1020.
- Whetten RW, MacKay JJ, Sederoff RR (2003) Recent Advances in understanding lignin biosynthesis. Annu Rev Plant Physiol Plant Mol Biol 49: 585-609.
- Li Q, He YC, Xian M, Jun G, Xu X, et al. (2009) Improving enzymatic hydrolysis of wheat straw using ionic liquid 1-ethyl-3-methyl imidazolium diethyl phosphate pretreatment. Bioresour Technol 100(14): 3570-3575.
- Arora R, Manisseri C, Li C, Ong MD, Scheller HV, et al. (2010) Monitoring and analyzing process streams towards understanding ionic liquid pretreatment of switchgrass (Panicum virgatum). BioEnergy Research 3(2): 134-145.
- Stark A (2011) Ionic Liquids in the biorefinery: a critical assessment of their potential. Energy Environmental Science 4(1): 19-32.
- Swatloski RP, Spear SK, Holbrey JD, Rogers RD (2002) Dissolution of cellulose with ionic liquids. Journal of American Chemical Society 124(18): 4974-4975.
- Lee SH, Doherty TV, Linhardt RJ, Dordick JS (2009) Ionic liquid mediated selective extraction of lignin from wood leading to enhanced enzymatic cellulose hydrolysis. Biotechnol Bioeng 102(5): 1368-1376.
- Zhang H, Wu J, Zhang J, He JS (2005) 1-Allyl-3-methylimidazolium chloride room temperature ionic liquid: A new and powerful non-derivatizing solvent for the cellulose. Macromolecules 38(20): 8272-8277.
- Fort DA, Remsing RC, Swatloski RP, Moyna P, Moyna G, et al. (2007) Can ionic liquids dissolve wood? Processing and analysis of lignocellulosic materials with 1-n-butyl-3-methylimidazolium chloride. Green Chemistry 9: 63-69.
- Olivier-Bourbigou H, Magna L, Morvan D (2010) Ionic liquids and catalysis: Recent progress from knowledge to applications. Applied Catalysis A: General 373(1-2): 1-56.
- Dadi AP, Schall CA, Varanasi S (2007) Mitigation of cellulose recalcitrance to enzymatic hydrolysis by ionic liquid pretreatment. Appl Biochem Biotechnol 137-140(1-12): 407-421.
- Li C, Knierim B, Manisseri C, Arora R, Scheller HV, et al. (2010) Comparison of dilute acid and ionic liquid pretreatment of switchgrass: biomass recalcitrance, delignification and enzymatic saccharification. Bioresource Technology 101(13): 4900-4906.
- Flieger J, Grushka EB, Zelazko AC (2014) Ionic liquids as solvents in separation processes. Austin journal of analytical and pharmaceutical chemistry.
- Neves CM, Ventura SP, Freire MG, Marrucho IM, Coutinho JA (2009) Evaluation of cation influence on the formation and extraction capability of ionic liquid based aqueous biphasic systems. J Phys Chem B 113(15): 5194-5199.
- Tadesse H, Luque R (2011) Advances on biomass pretreatment using ionic liquids: An overview. Energy Environment Sci 4(10): 3913-3929.
- Kilpeläinen I, Xie H, King A, Granstrom M, Heikkinen S, et al. (2007) Dissolution of wood in ionic liquids. J J Agric Food Chem 55(22): 9142-9148.
- Lan W, Liu CF, Sun RC (2011) Fractionation of bagasse into cellulose, hemicelluloses, and lignin with ionic liquid treatment followed by alkaline extraction. Journal of Agricultural Food Chemistry 59(16): 8691-8701.
- Yang D, Zhong LX, Yuan TQ, Peng XW, Sun RC (2013) Studies on the structural characterization of lignin, hemicelluloses and cellulose fractionated by ionic liquid followed by alkaline extraction from bamboo. Industrial Crops and Products 43: 141-149.
- Fu D, Mazza G, Tamaki Y (2010) Lignin extraction from straw by ionic liquids and enzymatic hydrolysis of the cellulosic residues. J Agric Food Chem 58(5): 2915-2922.
- Sun N, Mafizur R, Qin Y, Maxim ML, Rodriguez H, et al. (2009) Complete dissolution and partial delignification of wood in the ionic liquid 1-ethyl-3-methylimidazolium acetate. Green Chemistry 11(5): 646-655.
- Singh S, Simmons BA, Vogel KP (2009) Visualization of biomass solubilization and cellulose regeneration during ionic liquid pretreatment of switchgrass. Biotechnol Bioeng 104(1): 68-75.
- Doherty TV, Mora-Pale M, Foley SE, Linhardt RJ, Dordick JS (2010) Ionic liquid solvent properties as predictors of lignocellulose pretreatment efficacy. Green Chemistry (11): 1967-1975.
- Brandt A, Ray MJ, To TQ, Leak DJ, Murphy RJ, et al. (2011) Ionic liquid pretreatment of lignocellulosic biomass with ionic liquid-water mixtures. Green Chemistry 13(9): 2489-2499.
- Shamsuri AA, Abdullah DK (2010) Isolation and characterization of lignin from rubber wood in ionic liquid medium. Modern Applied Science 4(11): 19-27.
- Lynam JG, Toufiq Reza M, Vasquez VR, Coronella CJ (2012) Pretreatment of rice hulls by ionic liquid dissolution. Bioresource Technology 114: 629-636.
- Dadi AP, Varanasi S, Schall CA (2006) Enhancement of cellulose saccharification kinetics using an ionic liquid pretreatment step. Biotechnol Bioeng 95(5): 904-910.
- Zavrel M, Bross D, Funke M, Büchs J, Spiess AC (2009) High throughput screening for ionic liquids dissolving (ligno-) cellulose. Bioresour Technol 100(9): 2580-2587.
- Vo HT, Kim CS, Ahn BS, Kim HS, Lee H (2011) Study on dissolution and regeneration of poplar wood in imidazolium based ionic liquids. Journal of Wood Chemistry and Technology 31: 89-102.
- Yoon LW, Ngoh GC, Chua M, Seak A, Hashim M (2011) Comparison of ionic liquid, acid and alkali pretreatments for sugarcane bagasse enzymatic saccharification. Journal of Chemical Technology and Biotechnology 86(10): 1342-1348.
- Simmons BA, Singh S, Holmes BM, Blanch HW (2010) Ionic liquid pretreatment. Chemical Engineering Progress 106: 50-55.
- Fukaya Y, Sugimoto A, Ohno H (2006) Superior solubility of polysaccharides in low viscosity, polar, and halogen free 1,3-dialkylimidazolium formates. Biomacromolecules 7(12): 3295-3297.
- Kamlet MJ, Taft RW (1976) The solvatochromic comparison method I. Beta-scale of solvent hydrogen-bond acceptor (Hba) basicities. Journal of American Chemical Society 98(2): 377-383.
- Taft RW, Kamlet MJ (1976) Solvatochromic comparison method II. Alpha-scale of solvent hydrogen-bond donor (Hbd) acidities. Journal of American Chemical Society 98(10): 2886-2894.
- Brandt A, Hallett JP, Leak DJ, Murphy RJ, Welton T (2010) The effect of the ionic liquid anion in the pretreatment of pine wood chips. Green Chemistry 12(4): 672-679.
- Wang X, Li H, Cao Y, Tang Q (2011) Cellulose extraction from wood chip in an ionic liquid 1-allyl-3-methylimidazolium chloride (AmimCl). Bioresource Technology 102(17): 7959-7965.
- Shill K, Padmanabhan S, Xin Q, Prausnitz JM, Clark DS, et al. (2011) Ionic liquid pretreatment of cellulosic biomass: enzymatic hydrolysis and ionic liquid recycle. Biotechnol Bioeng 108(3): 511-520.
- Casas A, Palomar J, Alonso MV, Oliet M, Omar S, et al. (2012) Comparison of lignin and cellulose solubilities in ionic liquids by COSMO-RS analysis and experimental validation. Industrial Crops and Products 37(1): 155-163.
- Remsing RC, Swatloski RP, Rogers RD, Moyna G (2006) Mechanism of cellulose dissolution in the ionic liquid 1-n-butyl-3-methylimidazolium chloride: a 13C and 35/37 Cl NMR relaxation study on model systems. Chem Commun (Camb) (12): 1271-1273.
- Feng L, Chen ZI (2008) Research progress on dissolution and functional modification of cellulose in Ionic liquids. Journal of Molecular Liquids 142(1-3): 1-5.
- Zhang J, Zhang H, Wu J, Zhang J, He J, Xiang J (2010) NMR spectroscopic studies of cellobiose solvation in EmimAc aimed to understand the dissolution mechanism of cellulose in ionic liquids. Phys Chem Chem Phys 12(8): 1941-1947.
- Kosan B, Michels C, Meister F (2008) Dissolution and forming of cellulose with ionic liquids. Cellulose 15(1): 59-66.
- Ning S, Mustafizur R, Ying Q, Mirela LM, Hector R, et al. (2009) Complete solution and partial delignification of wood in the ionic liquid 1-ethyl-3-methylimidazolium acetate. Green Chemistry 11: 646-655.
- Janesko BG (2011) Modelling Interactions between lignocelluloses and ionic liquids using DFT-D. Physical Chemistry Chemical Physics 13(23): 11393-11401.
- George A, Tran K, Morgan TJ, Benke PI, Berrueco C, et al. (2011) The effect of ionic liquid cation and anion combinations on the macromolecular structure of lignins. Green Chemistry 13(12): 3375-3385.
- Pu YQ, Jiang N, Ragauskas AJ (2007) Ionic liquids as a green solvent for lignin. Journal of Wood Chemistry and Technology 27: 23-33.
- Liu H, Sale KL, Holmes BM, Simmons BA, Singh S (2010) Understanding the interactions of cellulose with ILs: A molecular dynamics study. J Phys Chem B 114(12): 4293-4301.
- Binder JB, Gray MJ, White JF, Zhang ZC, Holladay JE (2009) Reactions of lignin model compounds in ionic liquids. Biomass and Bioenergy 33(9): 1122-1130.
- Kimon KS, Leslie Alan E, Sinclair DWO (2011) Enhanced saccharification kinetics of sugarcane bagasse pretreated in 1-butyl-3-methylimidazolium chloride at high temperature and without complete dissolution. Bioresour Technol 102(19): 9325-9329.
- Tan SSY, MacFarlane DR, Upfal J, Edye LA, Doherty WOS, et al. (2009) Extraction of lignin from lignocellulose at atmospheric pressure using alkylbenzenesulfonate ionic liquid. Green Chemistry 11(3): 339-345.
- Pezoa R, Cortinez V, Hyvarinen S, Reunanen J, Linenqueo ME, et al. (2010) Use of ionic liquids in the pretreatment of forest and agricultural residues for the production of bioethanol. Cellulose Chemistry and Technology 44(4-6): 165-172.
- Xu F, Shi YC, Wang D (2012) Enhanced production of glucose and xylose with partial dissolution of corn stover in ionic liquid, 1-Ethyl-3-methylimidazolium acetate. Bioresour Technol 114: 720-724.
- Wyman CE, Dale BE, Elander RT, Holtzapple M, Ladisch MR, et al. (2005) Coordinated development of lignocellulosic biomass pretreatment technologies. Bioresour Technol 96(18): 1959-1966.
- Mosier N, Wyman C, Dale B, Elander R, Lee YY, et al. (2005) Features of promising technologies for pretreatment of lingocellulosic biomass. Bioresour Technol 96(6): 673-686.
- Bahcegul E, Apaydin S, Haykir NI, Tatli E, Bakir U (2012) Different ionic liquids favor different lignocellulosic biomass particle sizes during pretreatment to function efficiently, Green Chemistry 14: 1896-1903.
- Nguyen TA, Kim KR, Han SJ, Cho HY, Kim JW, et al. (2010) Pretreatment of rice straw with ammonia and ionic liquid for lignocelluloses conversion to fermentable sugars. Bioresour Technol 101(19): 7432-7438.
- Fu D, Mazza G (2011) Aqueous ionic liquid pretreatment of straw. Bioresour Technol 102(13): 7008-7010.
- Hou XD, Li N, Zong MH (2013) Significantly enhancing enzymatic hydrolysis of rice straw after pretreatment using renewable ionic liquid-water mixtures. Bioresource Technology 136: 469-474.
- Zhang Z, O'Hara IM, Doherty WO (2012) Pretreatment of sugarcane bagasse by acid-catalysed process in aqueous ionic liquid solutions. Bioresour Technol 120: 149-156.
- Wei L, Li K, Ma Y, Hou X (2012) Dissolving lignocellulosic biomass in a 1-butyl-3-methylimidazolium chloride-water mixture. Industrial Crops and Products 37: 227-234.
- Zhang YT, Du HB, Qian XH, Chen EYX (2010) Ionic liquid-water mixtures: enhanced Kw for efficient cellulosic biomass conversion. Energy Fuels 24(4): 2410-2417.
- Ma YC, Wei LG, Li KL, Wang SJ, Yu JY, et al. (2009) Separating lignin and cellulose from lignocellulosic biomass using an aqueous ionic liquid solution (Chinese). Patent, 200910012002.
- Zhang ZY, O’Hara IM, Doherty WO (2013) Effects of pH on pretreatment of sugarcane bagasse using aqueous imidazolium ionic liquids. Green chemistry 15(2): 431-438.
- Uloth VC, Wearing JT (1989) Kraft Lignin Recovery: Acid Precipitation Versus Ultrafiltration, Part II: Technology and economics. Pulp and Paper Canada 90: T357-T360.
- Uloth VC, Wearing JT (1989) Kraft Lignin Recovery: Acid Precipitation Versus Ultrafiltration, Part I: Laboratory Test Results. Pulp Paper Can 90: T310-T314.
- Satyanarayana SV, Bhattacharya PK, De S (2000) Flux decline during ultrafiltration of kraft black liquor using different flow modules: A comparative study. Separation and Purification Technology 20(2-3): 155-167.
- De S, Bhattacharya PK (1996) Flux prediction of black liquor in cross flow ultrafiltration using low and high ejecting membranes. Journal of Membrane Science 109: 109-123.
- Bhattacharjee S, Datta S, Bhattacharjee C (2006) Performance study during ultrafiltration of Kraft black liquor using rotating disk membrane module. Journal of Cleaner Production 14(5): 497-504.
- Padilla V, Rangel MG, Bullon J, Rodríguez-Malaver A, Gonzalez AM, et al. (2002) Surface Activity of Lignin Fractions Obtained by Membrane-Separation Technologies of Industrial Black Liquors. Iberoam Congr Pulp Pap Res 10: 10-12.
- Bhattacharya PK, Todi RK, Tiwari M, Bhattacharjee C, Bhattacharjee S, et al. (2005) Studies on ultrafiltration of spent sulfite liquor using various membranes for the recovery of lignosulphonates. Desalination 174(3): 287-297.
- Ringena O, Saake B, Lehnen R (2005) Isolation and fractionation of lignosulfonates by amine extraction and ultrafiltration: A comparative study. Holzforschung 59(4): 405-412.
- Area MC, Martos MS, Felissia FE, Venica AD, Valade JL (2000) Upgrading Spent Liquors from Nssc Process III. Separation of Spent Liquors Components by Ultrafiltration. Cellul Chem Technol 34: 173-182.
- Wallberg O, Jonsson AS (2006) Separation of lignin in kraft cooking liquor from a continuous digester by ultrafiltration at temperatures above 100˚C. Desalination 195(1-3): 187-200.
- Giummarella N, Lindgren C, Lindstrom M, Henriksso G (2016) Lignin prepared by ultrafiltration of black liquor: Investigation of solubility, viscosity, and ash content. Bio Resources 11: 3494-3510.
- Toledano A, Serrano L, Garcia A, Mondragon I, Labidi J (2010) Comparative study of lignin fractionation by ultrafiltration and selective precipitation. Chemical Engineering Journal 157(1): 93-99.
- Wallberg O, Jonsson AS, Wimmerstedt R (2003) Fractionation and concentration of kraft black liquor lignin with ultrafiltration. Desalination 154(2): 187-199.
- Niemi H, Lahti J, Hatakka H, Karki S, Rovio S, et al. (2011) Fractionation of organic and inorganic compounds from black liquor by combining membrane separation and crystallization. Chemical Engineering and Technology 34(4): 593-598.
- Pateraki C, Ladakis D, Stragier L, Verstraete W, Kookos I, et al. (2016) Pretreatment of spent sulphite liquor via ultrafiltration and nanofiltration for bio-based succinic acid production. Journal of Biotechnology 233: 95-105.
- Keyoumu A, Sjodahl R, Henriksson G, Ek M, Gellerstedt G, et al. (2004) Continuous nano- and ultra-filtration of kraft pulping black liquor with ceramic filters. Industrial Crops and Products 20(2): 143-150.
- Liu G, Liu Y, Ni J, Shi H, Qian Y (2004) Treatability of kraft spent liquor by microfiltration and ultrafiltration. Desalination 160: 131-141.
- Dafinov A, Font J, Valls RG, (2005) Processing of black liquors by UF/NF ceramic membranes. Desalination 173(1): 83-90.
- Ebrahimi M, Busse N, Kerker S, Schmitz O, Hilpert M, et al. (2015) Treatment of the Bleaching Effluent from Sulfite Pulp Production by Ceramic Membrane Filtration. Membranes (Basel) 6(1): E7.
- Olivares M, Guzmán JA, Natho A, Saavedra A (1988) Kraft lignin utilization in adhesives. Wood Sci Technol 22(2): 157-165.
- Sevastyanova O, Helander M, Chowdhury S, Lange H, Wedin H, et al. (2014) Tailoring the molecular and thermo-mechanical properties of kraft lignin by ltrafiltration. J Appl Polym Sci 131(18): 40799(1)-40799(11).
- Toledano A, Garcia A, Mondragon I, Labidi J (2010) Lignin separation and fractionation by ultrafiltration. Separation and Purification Technology 71(1): 38-43.
- Weinwurm F, Drljo A, Friedl A (2015) Lignin concentration by nanofiltration and precipitation in a lignocellulose biorefinery. Chemical Engineering Transactions 45: 901-906.
- Krockel J, Kragl U (2003) Nanofiltration for the separation of non-volatile products from solutions containing ionic liquids. Chem Eng Technol 26(11): 1166-1168.
- Fernández JF, Jastorff B, Störmann R, Stolte S, Thöming J (2011) Thinking in terms of structure-activity-relationships (T-SAR): A tool to better understand nanofiltration membranes. Membranes (Basel) 1(3): 162-183.
- Wu B, Zhang YM, Wang HP (2009) Non-equilibrium thermodynamic analysis of transport properties in the nanofiltration of ionic liquid-water solutions. Molecules 14: 1781-1788.
- Abelsa C, Redepenninga C, Mollb A, Melina T, Wesslinga M (2012) Simple purification of ionic liquid solvents by nanofiltration in biorefining of lignocellulosic substrates. Journal of Membrane Science 405- 406:1–10.
- Hazarika S, Dutta NN, Rao PG (2012) Dissolution of lignocelluloses in ionic liquids and its recovery by nanofiltration membrane. Separation and Purification Technology 97: 123-129.
- Avram AM, Ahmadiannamini P, Qian X, Wickramasinghe SR (2017) Nanofiltration membranes for ionic liquid recovery. Separation Science and Technology 52(13): 2098-2107.






























