Application of Tigernuts (Cyperus Esculentus) as Biosorbent for Cr6+ in Aqueous Solution
Sanusi KA*1, Santuraki AH1, Abdus Salam N2 and Akpan SE1
1Department of Chemical Sciences, Federal University Kashere, Nigeria
2Department of Chemistrty, University of Ilorin, Nigeria
Submission: January 21, 2019; Published: February 26, 2019
*Corresponding author: Sanusi KA, Department of Chemical Sciences, Federal University Kashere, Gombe State, Nigeria
How to cite this article: Sanusi KA, Santuraki A, Abdus S N, Akpan S. Application of Tigernuts (Cyperus Esculentus) as Biosorbent for Cr6+ in Aqueous Solution. Int J Environ Sci Nat Res. 2019; 17(3): 555965. DOI:10.19080/IJESNR.2019.17.555965
Abstract
Biosorption of Cr6+ ions onto raw biomass of the tigernut (Cyperus esculentus) was studied using the batch equilibrium adsorption method. The sorption characteristics of the adsorbent was investigated under various experimental conditions, such as pH, contact time, initial concentration of Cr6+ and sorbent mass. The FT-IR analysis showed that likely functional groups responsible for the adsorption are carboxyl, carbonyl, amides and hydroxyl groups. The pH for optimum adsorption is 3.0 when adsorbent dose is 2.0g at contant time of 15min. Equilibrium data fit well to the Langmuir isotherm. The estimated maximum adsorption capacity was found to be 63.58mg/g at 308°K and 90 .95mg/g at 328°K. Taking into account its good adsorption capacity, ease of sample treatment, as well as availability, the biomass of tigernut is a promising cost-effective biosorbent for Cr6+ removal from aqueous environment
Keywords: Biosorption; Tigernut; Cadmium; Equilibrium; Isotherm; Adsorption capacity
Introduction
Heavy metals ions have been a major problem faced by modern society that leads to ecological disequilibrium and health hazards. Excessive releases of pollutants into the environment especially water bodies often pose severe threats to humans and the entire ecosystem [1].
Heavy metal ions such as cadmium, lead, copper, chromium and zinc found in industrial waste water present acute toxicity to all forms of life including humans. Thus, the discharge of effluents into the aqueous environment is a major concern for environmental scientists [2].
The release of these pollutants into the environment, especially water bodies has been on increase as a result of urbanization and industrialization including pharmaceutical, leather, textile, circuit board printing metal finishing, mining, battery manufacturing and electroplating. Waste generated from these industries contain heavy metals which is the cause of environmental contamination (Demirbas, 2004).
The waste water from industries like, metal plating, mining operations, tanneries and radiator manufacturing, smelting, alloy industries, textile industry, dye industries effluents need the treatment [3]. The heavy metals are poisonous, persistent and, thus they are non-biodegradable. Most heavy metal ions are toxic or carcinogenic and hence present a hazard to human health and the environment when they are discharged into various water bodies [4].
Chromium is one of priority metal pollutants and it is non-biodegradable like other toxic pollutants, introduced into water bodies from many industrial processes such as dyeing, tanning, metal processing, paint manufacturing and steel fabrication. When introduced into the soil, it contaminates ground water which is a source of water for drinking; causing toxic effect in human beings and animals. Cr occurs in the aquatic environment as both trivalent and hexavalent states. Hexavalent chromium (Cr6+) which is primarily present in the form of chromate (Cr2O42-) and dichromate (Cr2O72-), possesses significantly higher levels of toxicity than the other valency states [5].
The metal cannot be degraded or readily detoxified biologically and have tendency to accumulate in living organism. Besides that, heavy metals discharge in the wastewater can be toxic to aquatic life and make water unsuitable for human consumption. Thus, effluents from municipal and industries need to be treated before repository into the ecosystem [6].
A number of conventional treatment technologies have been utilized for removal of heavy metals and from industrial effluents. Some of the conventional methods for the removal of metals from industrial waste water includes chemical precipitation, coagulation, solvent extraction, electrolysis, membrane separation, ion-exchange, adsorption, and membrane filtration [7]. However, these methods have been found to have some drawbacks, since they often involve high capital and operational costs and may be associated with the generation of secondary waste, which are the present treatment problems.
Because of its high efficiency and low cost, adsorption is by far the most effective and widely used (the preferable) technique for the removal of toxic heavy metals from wastewater [8]. Activated carbon is the most popular and widely used adsorbent in waste water treatment but it remains an expensive material, because the better quality of activated carbon costs high. Therefore, this situation makes it no longer attractive to be widely used in small-scale industries because of cost inefficiency.
Through many investigations, low-cost adsorbents have been evaluated due to their high adsorption efficiency of heavy metals including copper, lead, chromium, zinc ions [9]. In the last several decades, various agricultural wastes have been explored as low-cost adsorbent. Some of them include the shells or stones of fruits like nuts, peanuts, olive wastes, almonds, apricots stones and cherries and wastes resulting from the production of cereals such as rice, maize and corn as well as sugarcane bagasse and coir pith [10].
Like other, biosorbents previously used by other researchers, Cyperus esculentus (Tigernut) should be among the best candidates as alternative adsorbent of heavy metal ions from aqueous solution. However, no previous study has been reported on the application of tigernut for waste water treatment purpose towards removal of Cr6+ ions, this research was geared towards the investigation of tigernut as an alternative adsorbent for the removal of Cr6+ from aqueous solution.
The aim of this work is to investigate the adsorption capacity of Tigernuts (Cyperus esculentus) for the removal of Cr6+ in aqueous solution. Adsorption characteristics of the biosorbent for Cr6+ are studied under various operation parameter such as contact time, pH, Adsorbent dose, and sorbent concentration.
Materials and Methods
Preparation of adsorbate solutions (synthetic solutions)
A stock solution of 1000mg/L of Cr6+ was prepared by dissolving 2.8287 g of 99.9% K2Cr2O7 (Analytical Grade) in 1L distilled water in 1000ml volumetric flask. Solution of varying concentrations of 10mg/L, 20mg/L, 30mg/L, 40mg/L and 50mg/L were prepared by serial dilution from the stock solution. pH adjustment of the solutions was done by adding the required amounts of 0.1M HCl and 0.1M NaOH.
Preparation of the adsorbent materials (Tigernut)
The tigernuts (TN) were obtained from Kashere market, Kashere, Akko local government of Gombe State. The TN were washed with tap water first, then distilled water several times to remove dirt, dust and other impurities. The juicy parts were extracted, and residual waste materials were then dried, ground and sieved then sun dried for weeks. The powdered form of TN was stored and used for the study. No other physical or chemical treatments was employed prior to adsorption experiments.
Characterization of the adsorbent
pH of the adsorbents was determined by weighing 2g of the TN sample into a beaker containing 50cm3 of distilled water and agitated for 5minutes using a magnetic stirrer. Then the pH value was determined using a well calibrated electrical pH meter (pHS- 3C). FTIR spectra of adsorbent was obtained using Perkin Elmer infrared spectrometer. For FTIR spectra of the adsorbent, 0.1g of the adsorbent was ground and mixed with 0.5g of KBr salt and pressed into pellets. This pellet was introduced into the Perkin Elmer infrared spectrometer (Nexus 870 FT-IR) which showed peaks corresponding to the spectra of surface functional groups of the adsorbent. Philip XL30 Scanning Electron Microscope/EDAX was used to obtain microgram scan of TN samples.
Adsorption experiments
Adsorption experiments were carried out by adding 20mL solution of specified concentration of Cr6+ metal into vials containing 2g of the adsorbent. The adsorbent/metal mixture was then equilibrated by shaking at 100rpm in a temperature (27±1°C) controlled shaker until equilibrium; where necessary during the experiments, the pH of the working solutions was adjusted by adding drops of either HCl or NaOH. Effect of pH on adsorption of the Cr6+ metal was carried out by varying pH from 2±0.2 to 8±0.2 using 50mg/L of metal ion and equilibrating for 180min, effect of time was investigated by using 50mg/L of the Cr6+ metal and time varied from 10 to 180min, equilibrium experiments were studied at solution pH of 3±0.2 and room temperatures of 27±1°C, while varying the concentrations from 10 to 50mg/L at solution pH of 3±0.2 and equilibrating for 180min. At equilibrium, the vials were centrifuged at 1000rpm for 10min and the concentrations of Cr6+ metal left in solution was determined using the UV-visible spectrometer (T60) on absorbance mode. The experiments were conducted in duplicate and the mean of the values were used for analytical result.
Data treatment
Calculation of the removal of metal ions by tiger nuts adsorbent
The amount of metal ion sorbed was calculated using equation 3.1.
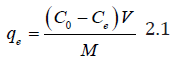
Where; C0 and Ce are the initial and final sorbate concentrations in solutions, qe, V and M are the amount of sorbate sorbed (mg/g), volume of the solution (mL) used for incubation and mass (g) of TN sample, respectively.
Calculation of the percentage metal ions removed by the adsorbent (TN)
The percentage removal of metal ions was calculated using the following equation;
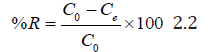
Where; R is the removal efficiency of the metal ions adsorbent studied; C0 is the initial metal ions concentration in solution (mg/L); Ce is the metal ions concentration removed or adsorbed by adsorbent at equilibrium (mg/L).
To obtain insights into the surface properties and degree of affinity of TN, sorption data were tested against the Langmuir and Freundlich equilibrium isotherms using the linear forms of these models in Eqs. (2.2) and (2.3).
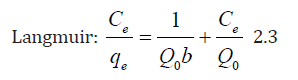
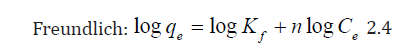
where; Q0 is the maximum adsorption capacity per unit weight of sorbent, b is a solute–surface interaction energy-related parameter, while qe and Ce are the amount of solute adsorbed per unit mass of the adsorbent (mg/g) and equilibrium metal concentrations in the solution (mg/L); where Kf and n are the Freundlich model capacity factor and the isotherm linearity parameter, respectively.
Results and Discussion
Characterization result

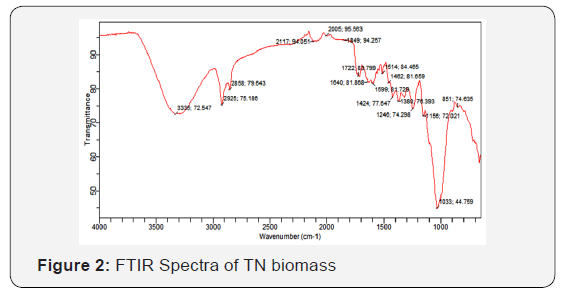
FT-IR spectra of unloaded TN biomass showed several absorption peaks between the scanning frequency ranges of 4000- 500cm-1 (Figure 2). The broad band around 3335cm–1 is indicative of the stretching band of the carbonyl double bond from carboxylic acids and their salts, a stretching vibration of free hydroxyl functional groups of the aromatic and aliphatic origin and possibly N-H stretch of amides. Also, the numerous free hydroxyl groups in the polysaccharide structure of its wall may explain the presence of the band. The two sharp absorption bands at 2925 and 2858cm–1 are indicative of C–H stretch likely from aliphatic (alkane and methylene) groups. The sharp band at 1722cm-1 is characteristic of the C=O stretch of carbonyl double bond either from free or esterified carboxyl groups. However, a distinct absorption band at 1640cm–1 have been assigned to amide-I of protein secondary structures. The strong band around 1156 and 1033cm-1 is due likely to the C−OH bond and –C-C− stretching, which are characteristic for polysaccharides. Similar observations have been reported by [11,12].
Effect of pH on adsorption
pH is an important adsorption parameter because it influences the activities of functional groups present on the surface of the adsorbents which enhance its adsorption capacity, and subsequently competitive adsorption of ions onto active sites of the adsorbent (Olu-Owolabi et al., 2012b) [11]. Establishing the capacity of Tigernuts (Cyperus esculentus) as novel adsorbent materials for Cr6+ ions requires knowing the optimum pH for adsorption. This was determined by equilibrating the adsorbent-metal mixture containing 50mg/L of Cr6+ ions at pH values between 2 and 8. As shown in Figure 3, the uptake of Cr (VI) is pH dependent. The results showed that at low pH of 1 and 3, the adsorbents adsorbed higher amount of Cr6+ onto their active adsorption sites.
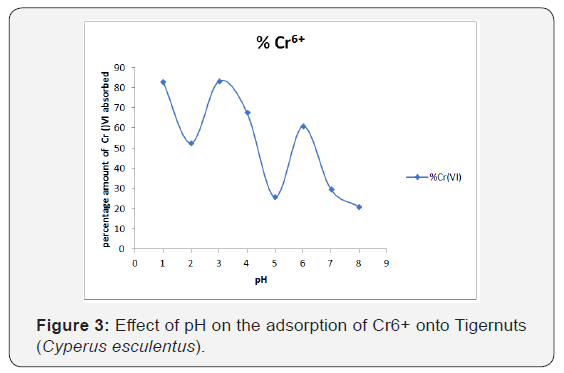
Precisely, for uptake of Cr6+ ions by TN, results (Figure 3) showed that the maximum adsorption was attained at a pH value of 3 with (qmax) of 0.200mg/g and then drops considerably from pH higher than 4. The amount of Cr6+ removed by TN was 0.17mg/g, respectively, at pH 3. Then the amount uptake decreases slightly in the pH range of 4-8. The highest adsorption of Cr6+ ions at pH 3 may be explained by the electrostatic attraction between the adsorbent and the two different adsorbate species in aqueous solution at the lower pH. Cr exists mostly in two oxidation states which are Cr6+ and Cr3+ and the stability of these ionic forms is dependent on the pH of the system. At acidic pH, the predominant species of Cr6+ are Cr2O7 2−, HCrO4 − and CrO4 2− while the surface of the adsorbent becomes protonated and attracts anionic species i.e HCrO4 −. Increasing the pH will shift the concentration of HCrO4 - to other forms, CrO4 2- and Cr2O7 2-. Maximum adsorption at pH 2 indicated that the HCrO4 - form of Cr6+ is the dominant species at lower pH values which is preferentially adsorbed onto the TN adsorbent surface. Similar observation was reported by Demirbas et al. (2004).
Effect of adsorbent dosage
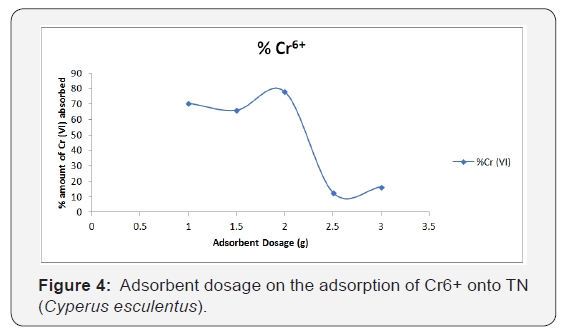
Figure 4 shows the effect of adsorbent dose on the percentage removal of Cr6+ respectively. The percentage of Cr+6 ions adsorbed by Tigernut decreases with an increase in the weight of adsorbent from 1 to 3g of Tigernut.
From the analysis of the experimental data obtained for the metal ions, it was observed that the adsorption efficiency decreases progressively with increase in adsorbent dose for Cr6+ ion. It was observed that the adsorption efficiency increased with increase in adsorbent dose up to a particular dosage (2g) above which the efficiency reduced. An increase in biomass dosage generally increases the amount of biosorbed metal ion because of an increase in surface area of the biosorbent, which consequently increases the number of available binding sites as also reported by [13]. However, adsorption capacity (qe) of Cr6+ decreases continuously up to a particular dosage (1.5g) above which efficiency increased slightly as the biosorbent was increased to 2g and later decreased again as the biomass dosage increases to 2.5g. The highest Cr6+ uptake by TN was 83%.
Effect of initial metal concentration on adsorption
Figure 5 shows the effect of initial metal concentration on the percentage removal of Cr6+ respectively. The percentage of Cr6+ ions adsorbed by tigernut increases with an increase in the initial metal concentration.
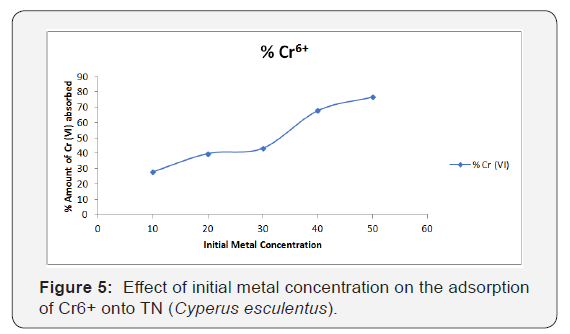
From the analysis of the experimental data obtained for metal ions, it was observed that the adsorption efficiency increases progressively with increase in initial metal concentration for Cr6+ ion. The Optimum Cr6+ uptake by the TN adsorbent was achieved at 50mg/l with maximum Cr6+ ions uptake of 79.89%. [13].
Effect of contact time on biosorption onto tigernut
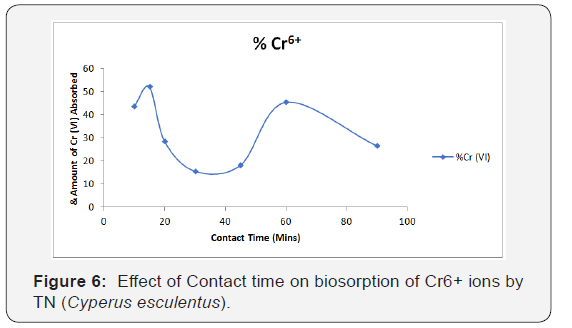
Figure 6 shows the effect of contact time on the percentage removal of Cr6+ respectively. The percentage of Cr6+ ions adsorbed by TN was at optimum at 15mins after which the absorption rate becomes inconsistent as the Cr6+ uptake decreased, later increased at 60 minute and then further decreased at more contact time. It is therefore possible for negative ions at the surface of the adsorbent to diffuse into and block the pores. As a result, Cr6+ ion may not be able to diffuse further into the pores and this will enhance inconsistency of the overall rate of adsorption. It could also be as a result of precipitation of the Cr6+ ions as also observed in the study by Demirbas et al. (2004).
Antifungal resistance
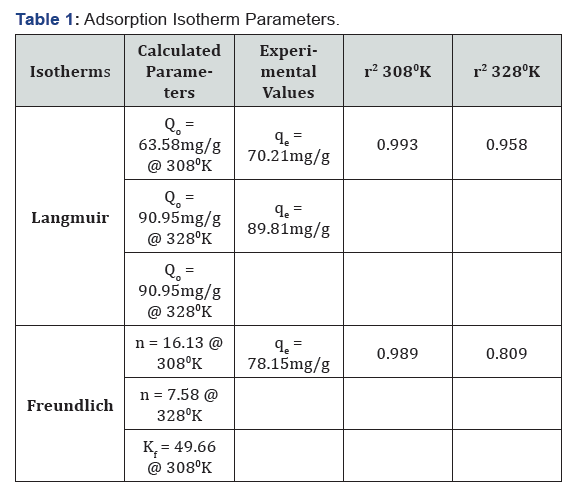
Langmuir and Freundlich adsorption isotherms (Table 1) were used to assess the TN adsorption capacity and surface sorption properties. Langmuir isotherm, which is based on the assumption that all sites possess equal affinity for the sorbate while forming a sorbate monolayer on the biosorbent surface at saturation, gave better fit to the sorption data than the Freundlich model which assumes sorption on heterogeneous surfaces [14]. The estimated monolayer adsorption capacity (Qo) is 63.58mg/g and 90.95mg/g at 308°K and 328°K respectively. This fact could be further established by comparative result and r2 values for the two isotherm models as shown in table 1 below.
Conclusion
The application of tigernut (TN) as biosorbent to remove Cr6+ from aqueous solution was investigated in batch experimental conditions. The results obtained showed that the biomass of TN has an optimum pH for Cr6+ adsorption at 3.0, nearly attains maximum adsorption within 1hr at 2.0g adsorbent dose, and 50mg/l initial sorbate concentration. The Langmuir isotherm described the equilibrium data better than other isotherms indicating adsorption is monolayer with monolayer adsorption capacity of 63.58mg/g at 308°K and 90.95mg/g at 328°K. The FT-IR analysis showed that possible functional groups responsible for metal adsorption are carboxyl, carbonyl, amides, hydroxyl and possibly other smaller groups that cannot be identified using only FT-IR spectra. Taking into account its good adsorption capacity, ease of sample treatment, as well as availability, the biomass of Tigernut is a promising cost-effective biosorbent for Cr6+ removal from aqueous environment.
References
- Allury HK, Ronda SR, Settalluri VS, Bondili JS, Suryanarayana V, et al. (2007) Biosorption: an eco-friendly alternative for heavy metal removal. African Journal of Biochemical Engineering Journal 6(25): 184-191.
- Sanusi KA, Umar BA, Abdullahi S (2015) Biosorption of Cd2+ from aqueous solution using Papaya (Carica papaya) seed: Equilibrium and Kinetic studies. Merit Research Journal of Environmental Science and Toxicology 3(8): 083-090.
- Geetha KS, Belagali SL (2013) Removal of Heavy Metals and Dyes Using Low Cost Adsorbents from Aqueous Medium- a Review. IOSR Journal of Environmental Science, Toxicology and Food Technology (IOSRJESTFT) 4(3): 56-68.
- Jassal PS, Raut VP, Anand N (2010) Removal of Chromium (VI) ions from Aqueous solution onto Chitosan and Cross-linked Chitosan Beads. Proc Indian Natn Sci Acad 1(2): 1-6.
- Wang J, Chen C (2009) Biosorbents for heavy metals removal and their future. Biotechnol Adv 27(2): 195-226.
- Sanusi KA, Umar BA, Sani IM (2016) Evaluation of the Application of Carica papaya Seed Modified Feldspar Clay for Adsorption of Pb+2 and Cu+2 in Aqueous Media: Equilibrium and Thermodynamic studies. Journal of Environmental & Analytical Toxicology 6(2): 1-9.
- Jeon P (2003) Impact of Heavy Metals on Forest Trees from Mining Areas. In: International Conference on Mining and the Environment III, Sudbury, Ontario, Canada, 12(6): 46-52.
- Chand S, Aggarwal VK, Kumar P (2004) Removal of hexavalent chromium from the wastewater by adsorption. Indian Journal of Environ Health 36(3): 151-158
- Saad O, Al-Jalil P (2001) Studies on Dietary Fiber: A Method for the Analysis and Chemical. Swedish journal of agricultural research 2(6): 46-52.
- Pollard SJT, Fowler GD, Sollars CJ, Perry R (2002) Low-cost Adsorbents for waste and waste water treatment; a review. Sci Environ and Toxicology 116(1-2): 31-35.
- Olu-Owolabi BI, Diagboya PN. Ebaddan WC (2012a) Mechanism of Pb (II) removal from aqueous solution using a non-living moss biomass. Chemical Engineering Journal 195-196: 270-275.
- Gupta VK, Gupta M, Sharma S (2001) Process development for the removal of lead and chromium from aqueous solution using red mud - an aluminum industry waste. Water Res 3(5): 1125-1134.
- Razmosvki KR, Sciban TV (1997) Dye removal using low coast adsorbents. Water Science and Technology 36(2-3): 189-196.
- Khan NA, Ibrahim S, Subramaniam P (2004) Elimination of heavy metals from wastewater using agricultural wastes as adsorbents. Malaysian Journal of Science 23: 43-51.






























