Magnesium: A Key Element in Natural Resources
Hussein K Abdel-Aal1*, M Abdel Kareem2 and Khaled Zohdi2
1 Professor Emeritus of Chemical Eng. / Petroleum Refining (retired), NRC, Cairo
2Higher Technological Institute, Tenth of Ramadan City, Egypt
Submission: September 06, 2018; Published: September 12, 2018
*Corresponding author: Hussein K Abdel-Aal, Professor Emeritus of Chemical Eng. / Petroleum Refining (retired), NRC, Cairo, Egypt, Email: habdelaal@link.net
How to cite this article: Hussein K abdelAal, M Abdel Kareem, Khaled Zohdi. Magnesium: A Key Element in Natural Resources. Int J Environ Sci Nat Res. 2018; 14(2): 555882. DOI: 10.19080/IJESNR.2018.14.555882.
Short Communication
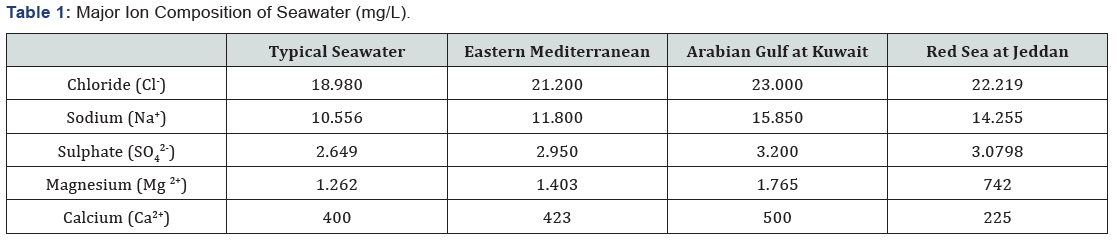
Land and fossil fuels resources are limited in our plant. Therefore, it is significantly important to harness the sun and sea for the creation of new resources. Magnesium recovery from seawater is such one area that represents a potential target. The oceans are vast and contain immense amounts of magnesium. Sea is considered a “store house’ for magnesium, since it contains a minimum of one million tons magnesium per one cubic kilometer. This makes about 1.7x1024tons stored in the oceans, which, in principle, could be recovered without the need of digging, crushing, processing, and all the other complex and energy expensive procedures that we need for land mining.Magnesium is the metal of the future! This post was found on the web from an enthusiastic magnesium believer. It resumes fairly the consensus surrounding the future of the magnesium. Magnesium is 33% lighter than aluminum, 60% lighter than titanium, and 75% lighter than steel. The magnesium content in seawater is shown as given next, in Table 1.
Background
Magnesium is described by having a wide range of uses in industry. In addition, it is an important element in the field of medicines. For industrial applications, it is often used with aluminum in the form of an alloy[1]. Adding magnesium in the form of an alloy lightens the weight of aluminum, while keeping its mechanical, fabrication and welding characteristics intact.Magnesium is well-ranked as:
The eighth most abundant element in the earth’s crust, the fourth most common element in the earth, following iron, oxygen and silicon, the eleventh most abundant element by mass in the human body, the third most abundant element dissolved in seawater, where sodium and chlorineare ranked first and second respectively.
Other additional features that magnesium enjoys are reported as well as follows:
a) About 13% of the planet’s mass is made by magnesium
b) About 2.1 percent of the earth’s crust is due to magnesium, which is a sign of its abundance
c) It is abundant in nature mainly in seawater and brines as positive ions.
d) It is essential to all cells and some 300 enzymes.
e) Also it is abundant in the terrestrial crust in the magnesite form, (MgCO3) and dolomite.
When it comes to the resources of magnesium, it is found in seawater and brines, as well as in deposits in the earth. There are three different types of magnesium ore: magnesite, dolomite and carnallite. It is also found in salt wells, brine and seawater.
Magnesium: The Lamp of Life
The element is a part of green plant chlorophyll. Inside chlorophyll is the lamp of life and that lamp is magnesium. The capture of light energy from the sun is magnesium dependent. Magnesium is bound as the central atom of the porphyrin ring of the green plant pigment chlorophyll. Magnesium is the element that causes plants to be able to convert light into energy and chlorophyll is identical to hemoglobin except the magnesium atom at the center has been taken out and iron put in. The whole basis of life and the food chain is seen in the sunlight-chlorophyllmagnesium chain[2]. Since animals and humans obtain their food supply by eating plants, magnesium can be said to be the source of life for it is at the heart of chlorophyll and the process of photosynthesis.






























