Can Untangling the Phorectic Association of Insect Vector and Pinewood Nematode Bursaphelenchus Xylophilus Solve the Pine Wilt Disease?
Kamrul Hassan1* Md. Fuad Mondal1 Naymul Hassan2 and Mahfuza Pervin3
1 Department of Entomology, Faculty of Agriculture, Sylhet Agriculture University, Sylhet-3100, Bangladesh
2Department of Agriculture, Noakhali Science and Technology University, Sonapur-3814, Bangladesh
3Faculty of Agriculture, University of Catania, Italy
Submission: June 14, 2018; Published: July 02, 2018
*Corresponding author: Kamrul Hassan, Department of Entomology, Faculty of Agriculture, Sylhet Agriculture University, Sylhet-3100, Bangladesh, Email: khassan.entom@sau.ac.bd
How to cite this article: Kamrul H, Md. Fuad M, Naymul H, Mahfuza P. Can Untangling the Phorectic Association of Insect Vector and Pinewood Nematode Bursaphelenchus Xylophilus Solve the Pine Wilt Disease?. Int J Environ Sci Nat Res. 2018; 12(5): 555848. DOI:10.19080/IJESNR.2018.12.555848.
Abstract
Pine wilt disease (PWD) is the most serious threat for declining the pine forest in worldwide. Pine wood nematode (PWN) Bursaphelenchus xylophilus is responsible for this disease. As a result of successful phorectic association between Insect vector and PWN, the PWD is occurred which subsequently infect and kill the pine trees. Moreover, the chemical signal via volatile compounds produced by beetle in pine trees assist phorectic association. For this reason, our review was attempted to describe well about the infestation biology of PWN, the role of phoretic association for pathogenicity, invasiveness and parasitism of PWN and how such generated knowledge can be incorporated to the field of management of pine wilt disease. There was noticeable declining of the pine forest in the area where it is grown enough and the identification of durable control solution could be needed to reduce the infestation of PWN and safe the pine forest throughout the world. Our review results extrapolated that unravelling the mechanism of phorectic association among the pine trees and insect vector with nematode sway the control of pine wilt disease. Furthermore, comparison studied of several control measures helps to find best strategies for the management of PWN and indicated the pathway for future research .
Keywords: Pine wilt disease; Bursaphelenchus xylophilus; Phorectic association; Insect vector; Control measures
Introduction
Pine wood nematode (PWN) B. xylophilus is an invasive species causing serious damage to pine tree (Futai, [1]). It is a native species of North America but not a serious pest of local pine species (Futai, [1]; Bergdahl, [2]). However, it is causes serious destruction of pine forest in Asian countries such as China, Korea, and Taiwan, and also some European countries such as Portugal and Spain (Togashi and Shigesada, Vicente et al., Futai, [1]) due to import infested wood packaging materials from North America. It destroyed the pine forest in Europe while pine timber and wood packaging materials were imported from East Asia (Vieira et al.,). Hence, it is now a serious threat for the reduction of pine forest both in Asia and Europe. The life style of Pine wood nematode is quite complex (Futai, [1]; Kulinich et al., [3]). They mainly feed on pine trees. But it is very difficult to PWN to reach the pine trees. Because, they are approximately 0.85mm long and 2μm wide (Mamiya and Kiyohara, [4]) and can disperse only few kilometers per year (Fujishita, [5]). However, this organism solves the problem by using an insect vector Monochamus spp. and transmitted very successfully from weak and dead tree to healthy pine trees (Alves et al, [6]). This transmission process is occurred due to the synchronization between insect and pine wood nematode for same resources [7] such as oviposition (Vicente et al [8]). The association between B. xylophilus and Pine swayer beetle Monochamus spp result the devastating pine wilt disease in the pine forest via phoretic association (Yang and Wang). Phoretic association means insect used as a vehicle to transfer plant parasitic nematode into pine tree. Indeed, the development of pine wilt disease is the result of tritrophic interaction. Tritrophic interaction (insect-nematode-pine trees) means insect acts as a vector for nematode to reach their host plant. This interaction could be explained as insect vectored plant parasitism (Davis et al.). We already know that the rapid declining the susceptible species of pine tree due to PWN. But until 1960 most of the scientist believed that the rapid destruction of pine forest due to the severe attack of beetle because, the dead pine trees always attacked by beetle. But at later stage it was confirmed that pine beetle never attacks healthy pine trees. Only dead and weak pine trees are attack by pine beetle (Iwasaki and Morimoto, [7]) and pinewood nematode B. xylophilus is responsible for huge destruction of pine forest. Generally, the vector Pine swayer beetle Monochamus spp can be found throughout the pine forest especially North America, Europe and Asia (Futai, [1]). At early history of the pine wilt disease, they considered as minor pests until the discovery of the association with pine wood nematode. Later on, it was considered as major pest (Vieira et al. [8]). In 2003 United States Department of Agriculture reported that, annual estimated agricultural losses due to plant parasitic nematode were $125 billion USD worldwide. Pinewood nematode B. xylophilus has contribution to these huge losses due to rapid wilting and dead of pine trees. Infact, the recent report of devastating infestation of pine wood nematode B.xylophilus in Asia and Europe stimulate more scientific interest to untangling the phorectic association of insect vector and pinewood nematode B. xylophilus to solve the pine wilt disease. The control method primarily depends on application of synthetic chemical because the vector Monochamus spp is responsible to spread pine wilt diseases. But application of synthetic chemical causes huge environmental pollution due to drifting of chemical in the environment which later on causes human health hazard. So, searching non-chemical solution is judicious for reducing the pest attack. Recently, the possible solution was reported by several scientific literatures. The quarantine measure during importation of wood packaging materials, application of botanicals, induced host plant resistant by avirulent microbes, trunk injection of emamectin benzoate, and application of bio-control agents are available to reduce PWN infestation. In this review we will unravel how using the knowledge of pine wood nematode interaction with plants and beetles, their epidemiology and parasitism can be incorporated with possible appropriate control methods that cited in the recent scientific literature. So identically, we will try to find the way of future research strategies on pine wilt disease by understanding; Does pine beetle solely responsible for phorectic association with PWN? Does PWN alternatively ameliorate the process of association via chemical signaling while infestation with pine trees? Does such biological knowledge can be applicable with current knowledge for reducing PWN infestation and if then what could be the future research trajectories?
Phorectic association between B. xylophilus and Monochamus spp. The evolutionary ecology of pine wilt disease is on the basis of phoretic association between pine wood nematode B. xylophilus and wood boring beetle, Monochamus spp (Futai, [1]; Kulinich et al. [3]). It depends on flight phenology, sex ratio and size of the vector and also the climatic factor. When the beetle flight longer distances it indicates longer transportation of pine wood nematode (Robinet et al. [9]). This phenomenon helps pine wood nematode for successful invading the Euroasian pine forest. Indeed, the phoretic interaction is species specific and also synchronized the life cycle of among three species (Krishnan et al., [10]). However, in insect nematode phoretic system dauer larvae are more attracted by female than male beetle. Because, the female beetle is larger than the male and also flight more distance in the pine forest. It provides most reliable transport of pine wood nematode to new resource patches (Krishnan et al., [10]; Yoshigia et al., [11]). So it is extrapolated that female afford more efficient transmission than male. Climatic factor especially temperature has significant influence on the speed up the pine wilt disease. As, the phoretic association is modified by the temperature variation. Besides, it initiates the rapid growth and development of nematode population inside the pine tree (Naves et al. [12]). It also helps to faster degradation of phloem tissue of pine trees. It was reported that the pine wilt disease is lower in Finland than Iberia due to the temperature variation. Generally, Finland has lower warm than Iberia. As a result, the population of beetle is lower in Finland than Iberia. The lower beetle population indicated that the less dispersal of PWN. So, for the management of this pest the emphasis should be given on the temperature of the environment (Tomminen [13])
Box 1
Relationship of pine swayer beetle and pine wood nematode Pine swayer beetle Monochamus spp is the vector of PWD which feed on twigs of pine trees. They prefer to lay eggs on bark of the dead pine trees. The newly hatched larvae feed on phloem and late larvae feed on xylem and make a U-shaped gallery in xylem where the pupation takes place. Surprisingly, this activity of beetle does not kill the pine trees. But such activity helps pine wood nematode to enter the xylem tissue of pine trees. Subsequently, pine wood nematode blocks the movement of water and nutrient from root to plant body. This result causes the development of pine wilt diseases of pine trees (Linsley and Chemsak, [14]; Akbulut et al, [15]).
Pine wood nematode interaction with plants and beetles
The pine wood nematode interacts with plants and beetles through chemical stimuli. Infact the chemo-ecological response between pine wood nematode B. xylophilus and its vector Monochamus spp helps to find the host pine trees (Necibi and linit, [16,17]). Generally, third stage dispersal juvenile (JIII) aggregated around the pupal chamber of beetle and this chamber are formed by mature larvae of beetle around the outer xylem of dead pine trees (Mamiya [18]; Linit, [16]). The main mechanism of this dispersal is synchronization of the behavior of beetle and nematode. Before entering the tracheal system of beetle, the Juvenile (JIII) larvae molt to dauer larval stage (JIV). At last, this dauer larval stage (JIV) transported from infected pine trees to healthy trees by the beetle for successful infection of pine trees (Figure 1) (Lint [16]; Necibi and Linit [17]; Yang et al. [19]). This transportation mechanism of pine wood nematode is the result of volatile chemical compound namely linoleic and oleic acid, 1-monoolein, and toluene produced by beetle (Miyazaki et al. [20]; Bolla et al. [21]; Necibi and linit, [17]; Stamps and Linit, [22]).

Mechanisms in parasitism
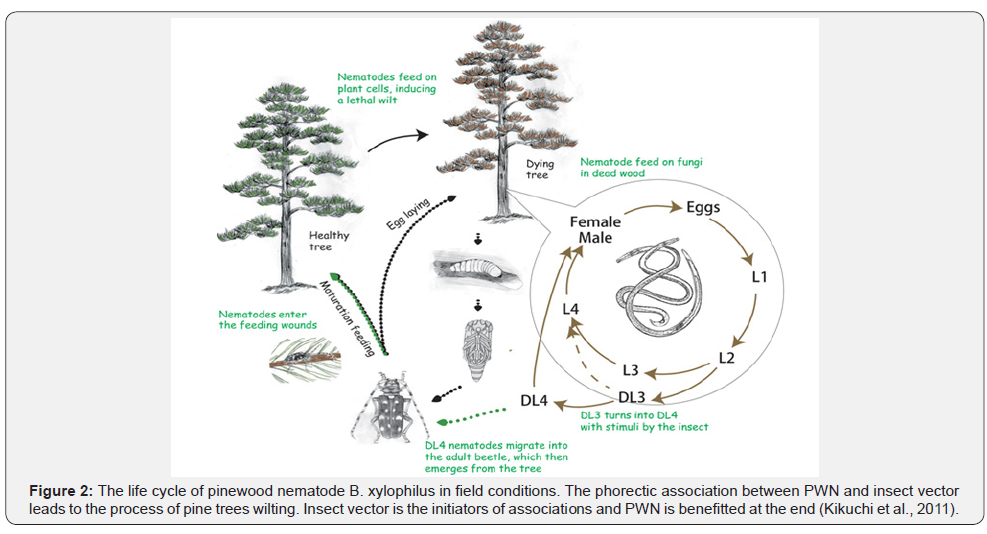
Beetles prefer to mate on dead pine trees instead of healthy trees. During mating dauer larval stage (JIV) entered the respiratory system of beetle. After oviposition and pupation when newly developed beetle emerged from dead pine trees they start to find out and feeding on healthy pine trees (Son et al; [23]). Through feeding pine wood nematode get entry into healthy pine trees. It is called portal entry of pine wood nematode (Mamiya, [24]). Then it migrates through cortical and xylem axial resin canals and destroying the epithelial cells through their stylet. Afterwards, they move horizontally from both resin canals to surrounding tissue and destroy the tissue cells including cortex, phloem, xylem, pith and cambium (Mamiya, [24], and [25]; Ishida et al; [26]). They take three or four weeks for developing wilting symptom in the healthy plant and finally cause the death of pine trees. At the time of infection, oleoresin flow decrease and plant produce volatiles namely ethanol and terpines (McGawley and Winchell, [27]). This volatiles act as attractant to beetles to mate with their partner in disease or dead pine trees. However, when insect going to pupation the dauer juveniles of the nematode again aggregated around the pupal chamber and afterwards enter the insect body. After one month later newly, emerged beetle again migrates the healthy trees by carrying pine wood nematode in their respiratory system. So, this mechanism involved three organism pine trees as host, pine wood nematode, and vector Monochamus spp. (Figure 2) (Mahera and Futai, [28]; Mamiya and Enda, [18]; McGawley and Winchell, [27]).
How Pine wood nematode infestation could be reduced in pine forest?
The reduction of PWN in the pine forest is now a most crucial issue for the plant protection scientists. Due to its spreading nature quarantine measure considered as most promising solution. On the other hand, application of synthetic chemical likes emamectin benzoate also an available solution of PWN. But application of botanicals like ethyl acetate layer of the root of Stellera chamaejasme is more impressive than chemical insecticide. Furthermore, application of biocontrol agents for instance, Esteya vermicola and induced host resistant by avirulent microbes also impressive for controlling PWN. We were compared the each of the possible solution of PWN infestation with limitation, so future research may focus on the combination of few approaches to sustainable control of nematode.
Wood materials and quarantine measures
The presence of PWN into an alien area is the result of less quarantine measures during importation of wood packaging material. It entered into Europe and Asia through wood packaging materials (Mota,). For this reason, it is listed as quarantine pest in many countries like Japan, South Korea, and also in Europe (Rautapaa, [29] and Dwinell, [30]). It causes huge economic losses if they disperse through the imported wood packaging material (Bergseng et al; [31] and Vicente et al; [8]). Indeed, once pine wood nematode attack pine trees it’s very difficult to eradicate of pine wilt disease. Hence, strict quarantine measure reduces the dispersal of PWN and safe the pine forest.
Synthetic insecticide and Botanicals
Emamectin benzoate is a semi synthetic insecticide has strong nematicidal activity against B. xylophilus and M. galloprovincialis (Takai et al; [32]). The beetle population is controlled by the trunk injection of Emamectin benzoate. Applications of this insecticide protect pine trees in the field, M. galloprovincialis population attack in the field and B. xylophilus infection in the pine tree. It is currently used as good option in Portugal and also in other country of Europe. Indeed, application of synthetic pesticide like Emamectin benzoate causes environmental pollution and also causes human health hazard. In that case application of botanicals for controlling this pest is a potential option. The ethyl acetate layer extracted from the root of Stellera chamaejasme has strong nematocidal effect on B. xylophilus. The plant S. chamaejasme widely distributed in grassland of North and West China. It was primarily believed that ethanol extract from plant S. chamaejasme was highly effective against B. xylophilus. Eight purified compound were tested in vitro for nematocidal activity against B. xylophilus where combination with the roots extract from S. chamaejasme showed significant activity for the mortality of B.xylophilus and B. mucronatus (Cui et al; [33]). In a nutshell, reducing the application of chemical pesticide and increase the applications of botanicals significantly reduce the population of B. xylophilus and B. mucronatus.
Temperature and Biocontrol agent Esteya vermicola
Nematophagus fungus (Esteya vermicola) is a promising biocontrol agent against pine wilt disease. They produce two types of conidia bacilloid and lunate where lunate conidia can infect pine wood nematode by cuticle penetration, immobilization and digestion of the internal contents (Wang et al. [34,35]). But the efficacy of E. vermicola is influenced by temperature and water factor (Gock et al. [36]; Teshler et al. [37]). It was reported that at low temperature (40C) E. vermicola showed highest infectibility against pine wood nematode (Trivedi et al. [38]). On the other hand, the population of pine sawyer beetles also decreasing with low temperature and high altitude. As a result, the spread of pine wilt disease is tremendously reduced in that area. At temperature 110C with 850m altitude the beetle population increases whereas at 9. 10C with 950m the growth and development and also movement of pine swayer beetle limit effectively (Enda, 1970). These results revealed that less movement of beetles carry lower number of nematode in their body and slow up the spread of pine wilt disease. Besides at low temperature availability of moisture also increases that influence the viability and longevity of conidia. Because E. vermicola could not tolerate and survive under dry condition. So, the progress of biocontrol agent E. vermicola is largely influenced by the temperature and moisture factor and the movement of vector also limited by the temperature variation. The correlation between the movement of vector and application efficacy of E. vermicola could be minimized the pine wilt disease effectively.
Botanicals and avirulent microbes
Indeed, when pine wood nematode is devastating to pine trees application of botanicals is quite difficult because it works very slowly. The pre-inoculation of avirulent microbes into host plant is called induced resistant then, quite promising to control of pine wilt disease. The two avirulent microbes C14-5 and fungus Botrytis cinera were reported most potential against pine wilt nematode (Hunt et al [39]; Heil and Bostock, [40]). The seedling treatments with these two avirulent microbes gave lower mortality than without pre-inoculation of microbes of susceptible Japanese black pine (Yuko et al [41]). Like, chemical insecticide application of botanicals required more labor and could be reduced efficacy by wind and rain after application of botanicals. As a result, the application of pre-inoculation of avirulent microbes is more effective than botanicals. The avirulent microbes B. cinera was least effective than C14-5 because it became weak with the progress of time. C14-5 is isolated from pine wilt nematode and it can survive pine trees for a long period of time without developing any symptom. For this reason, induced host resistant by two avirulent microbes in compare to the synthetic chemical and botanicals could be considered as good option against B. xylophilus.
Discussion
The scientific literature described well enough about the development of pine wilt disease mechanism in pine forest. Most of the paper described about the devastating effect of PWN. Indeed, the literature provides a comprehensive understanding of the mechanism used by pine wood nematode for successful dispersal and invasion mechanism. The growing body of literature described the nature of phoresy of PWN and beetle. Different factors are described well enough for successful phoretic association. For instance, female beetle is larger than male and they attracted by mostly female during phoretic association. But how large body size of beetle influences the dispersal of PWN need to answer in the future research? Phoretic association is influenced by temperature because low temperature reduces the movement of beetle. But how variation of temperature influences the life cycle of PWN need to be explored? Successful phorectic association between PWN and beetle is the result of pine wilt disease. Recent scientific findings described well that beetle is attracted by volatile compound produced by pine trees and by this way beetle find the mating partner in dead or weak trees. But no description about the volatile compound has any impact on the PWN and how it could be linked for the control measures of PWN. The chemio-ecological signal or stimuli between the PWN and beetle through volatile compound produced by beetle attracts the PWN. But no knowledge why actually it attracted by PWN (Zhao et al; [42]). However, it is clear that chemical signal influences the aggregation of JIII and JIV around the pupal chamber of the beetle. But the description is superficial that’s why profound knowledge about the chemical stimuli could be needed further research so that the knowledge can be correlated into control measures. Studies of infestation biology facilitate the prevention and management of PWN. Indeed, lack of quarantine measure facilitates the longer dispersal of PWN. Recent findings, gave some promising solution of this pest. As vector beetle is key responsible for spreading the disease so application of synthetic insecticide like Emamectin benzoate quite promising. But use of synthetic insecticide is now banned worldwide to safe the existing ecosystem services. The Nematophagus fungus (Esteya vermicola) is now a promising bio-control agent for the management of PWN. The conidia are responsible to infect the PWN. But temperature and water factor reduce the shelf life and infectibility of conidia (Zian et al; [43]) which is opposite to some previous study where reported that temperature and water has no impact on viability of conidia (Pavone et al. [44]; Sabuquillo et al. [45]; Ya´nez-Mendiza´bal). So how impact of temperature and water on shelf life and infectibility of conidia reduces the biocontrol of PWN need to be addressed in upcoming research. The root of Stellera chamaejasme has effect on the mortality of PWN and this technique is better in compare to chemical insecticide but not like Nematophagus fungus. Indeed, efficacy of botanicals reduced with the progress of time because field stability of botanicals is difficult due to climatic variation. However, induced host resistant by C14-5 and fungus Botrytis cinera is more effective in compare to chemical and botanicals. So, attention on using induced host resistant by C14-5 and fungus Botrytis cinera in future research strategy will be safe solution for proper control of pine wilt disease [46-55].
Conclusion and Direction of Future Research
It is clear that PWN biology especially chemical signaling, phoretic association and mechanism of parasitism is described well in the article. But knowledge gap how this chemical signal could be linked for the management of PWN. Uses of botanicals, bio-control agent and induced host resistant are promising but still have huge limitation. So further investigation could be needed for linking the chemical signal to management of PWN and also overcome the limitation of the possible solution for better control solution of PWN.
References
- Futai K (2013) Pine wood nematode, Bursaphelenchus xylophilus. Annual Review of Phytopathology 51: 61-83.
- Bergdahl DR (1988) Impact of pine wood nematode in North America: present and future. J. Nematol 20(2): 260-265.
- Kulinich OA, Zao B, Ryss AY, Kozyreva NI (2010) Pine wood nematode. Zashchita Karantin Rasteniĭ (7): 36-38.
- Mamiya Y (1972) Aggregation of the pinewood nematode around the pupal chamber of the vector beetle. Jpn For Soc 24:30.
- Fujishita A (1978) Pine wilt disease damage in Shizuoka prefecture Jap. For. Soc. 26:193-98.
- Alves M, Pereira A, Matos P, Henriques J, Vicente C, et al. (2016) Bacterial community associated to the pine wilt disease insect vectors Monochamus galloprovincialis and Monochamus alternatus. Scientific reports 6: 23908.
- Iwasaki A, Morimoto K (1971) Host conditions suitable for oviposition by pine beetles. Trans. Meet. Kyushu Branch Jap For Soc 25:168-69.
- Vicente C, Espada M, Vieira P, Mota M (2012) Pine Wilt Disease: a threat to European forestry. Eur. J Plant Pathol 133 (1): 89-99.
- Robinet C, Van Opstal N, Baker R, Roques A (2011) Applying a spread model to identify the entry points from which the pine wood nematode, the vector of pine wilt disease, would spread most rapidly across Europe. Biological Invasions, 13(12): 2981-2995.
- Krishnan A, Muralidharan S, Sharma L, Borges RM (2010) A hitchhiker’s guide to a crowded syconium: how do fig nematodes find the right ride? Functional Ecology 24(4): 741-749.
- Yoshiga T, Ishikawa Y, Tanaka R, Hironaka M, Okumura E (2013) Species-specific and female host-biased ectophoresy in the roundworm Caenorhabditis japonica. Naturwissenschaften, 100 (2): 205-208.
- Naves PM, Sousa E, Rodrigues JM (2008) Biology of Monochamusgalloprovincialis (Coleoptera, Cerambycidae) in the Pine Wilt Disease affected zone, Southern Portugal. Silva Lusit. 16 (2): 133- 148.
- Tomminen J (1993) Development of Monochamusgalloprovincialis Olivier (Coleoptera: Cerambycidae) in cut trees of young pines (Pinussylvestris L) and log bolts in southern Finland. Entomol Fenn 4 (3): 137-142
- Linsley EG, Chemsak JA (1984) The Cerambycidae of North America, Part VII, No. 1: Taxonomy and Classification of the Subfamily Lamiinae, Tribes Parmenini through Acanthoderini. University of California Publications in Entomology 102: 11-258.
- Akbulut S, Stamps WT (2012) Insect vectors of the pinewood nematode: a review of the biology and ecology of Monochamus species. Forest Pathology 42(2): 89-99.
- Linit MJ (1988) Nematode-vector relationships in the pine wilt disease system. J. Nematol. 20(2): 227-235.
- Necibi S, Linit MJ (1998) Effect of Monochamus carolinensis on Bursaphelenchus xylophilus dispersal stage formation. J Nematol 30(2): 246-254.
- Mamiya Y, Enda N (1972) Transmission of Bursaphelenchus lignicolus (Nematoda: Aphelenchoididae) by Monochamus alternatus (Coleoptera: Cerambycidae). Nematologica 18:159-62.
- Yang BJ, Pan HY, Tang J, Wang YY, Wang LF, et al. (2003) Bursaphelenchusxylophilus Chinese Forestry Press, Beijing.
- Miyazaki M, Oda K, Yamaguchi A (1977) Behavior of Bursaphelenchus lignicolus to unsaturated fatty acids. J Jpn Wood Res Soc 23: 254-261
- Bolla A, Bramble J, Bolla RI (1989) Attraction of Bursaphelenchus xylophilus, Pathotype MPS-1, to Monochamus carolinensis larvae. Japanese Journal of Nematology 19:32-37.
- Stamps WT, Linit MJ (2001) Integration of intrinsic and extrinsic chemical cues in the behavior of Bursaphelenchusxylophilus (Aphelenchida: Aphelechoididae) in relation to its beetle vectors. Nematology 3:295-301.
- Son JA, Komatsu M, Matsushita N, Hogetsu T (2010) Migration of pine wood nematodes in the tissues of Pinusthunbergii. Journal of Forest Research 15 (3): 186-193.
- Mamiya Y (1980) Inoculation of the first year pine (Pinusdensiflora) seeding with Bursaphelenchuslignicolus and the histopathology of diseased seedlings. Journal of the Japanese Forest Society 62: 176-183.
- Mamiya Y (1985) Initial pathological changes and disease development in pine trees induced by the pine wood nematode, Bursaphelenchusxylophilus. Annals of the Phytopathological Society of Japan 51: 546-555.
- Ishida K, Hogetsu T, Fukuda K, Suzuki K (1993) Cortical responses in Japanese black pine to attack by the pine wood nematode. Canadian Journal of Botany 71(11): 1399-1405.
- McGawley EC, Winchell KL, Jones JP, Birchfield W, Berggren GT (1985) Population development and influence of Bursaphelenchus xylophilus on Gliocladium virens. J Nematol 17(1):69-76.
- Maehara N, Futai K (1997) Effect of fungal interactions on the numbers of the pinewood nematode, Bursaphelenchusxylophilus (Nematoda: Aphelenchoididae) carried by the Japanese pine sawyer, Monochamusalternatus (Coleoptera: Cerambycidae). Fundam Appl Nematol 20(6): 611-617.
- Rautapaa J (1986) Experiences with Bursaphelenchus in Finland. Conference on Pest and Disease Problems in European Forests. EPPO Bull.16: 453-456.
- Dwinell LD (1997) The pinewood nematode: regulation and mitigation. Annu Rev Phytopathol 35: 153-166.
- Bergseng E, Økland B, Gobakken T, Magnusson C (2012) Combining ecological and economic modelling in analyzing a pest invasion contingency plan: The case of pine wood nematode in Norway Scand J For Res 27(4): 337-349.
- Takai K, Soejima T, Suzuki T, Kawazu K (2000) Emamectin benzoate as a candidate for a trunkinjection agent against the pine wood nematode, Bursaphelenchusxylophilus. Pest Management Science, 56: 937-941.
- Haiyan Cui, Hui Jin, Quan Liu, Zhiqiang Yan, Lan Ding, et al. (2013) Nematicidal metabolites from roots of Stellera chamaejasme against Bursaphelenchus xylophilus and Bursaphelenchus mucronatus. Pest Management Science 70(5): 827-835.
- Wang CY, Fang ZM, Sun BS, Gu LJ, Zhang KQ,et al. (2008) High infectivity of an endoparasitic fungus strain, Esteyavermicola, against nematodes. J Microbiol 46(4):380-389.
- Wang CY, Fang ZM, Wang Z, Gu LJ, Sun BS et al. (2009) High infection activities of two Esteyavermicola isolates against pinewood nematode. Afr J Microbiol Res 3(10): 581-584.
- Gock MA, Hocking AD, Pitt JI, Poulos PG (2003) Influence of temperature, water activity and pH on growth of some xerophilic fungi. Int J Food Microbiol 81(1):11-19.
- Teshler MP, Ash GJ, Zolotarov Y, Watson AK (2007) Increased shelflife of a bio-herbicide through combining modified atmosphere packaging and low temperatures. Biocontrol Sci Tech17(4):387- 400.
- Trivedi P, Pandey A, Palni LMS (2005) Carrier-based preparations of plant growth-promoting bacterial inoculants suitable for use in cooler regions. World J MicrobiolBiotechnol 21(6–7): 941-945
- Hunt MD, Neuenschwander UH, Delaney TP, Weymann KB, Friedrich LB, et al. (1996) Recent advances in systemic acquired resistance research – a review. Gene 179(1): 89-95.
- Heil M, Bostock RM (2002) Induced systemic resistance (ISR) against pathogens in the context of induced plant defenses. Annals of Botany 89(5): 503-512.
- Yuko T, Natsumi K, Kazuyoshi F (2006) How different is induced host resistance against the pine wood nematode, Bursaphelenchusxylophilus, by two avirulent microbes? Nematology 8(3): 435-442.
- Li Lin Zhao, Wei Wei, Le Kang, Jiang Hua Sun (2007) Chemotaxis of the Pinewood Nematode, Bursaphelenchus xylophilus, to Volatiles Associated with Host Pine, Pinus massoniana, and its Vector Monochamus alternates. J Chem Ecol (2007) 33(6): 1207-1216.
- Xue JJ, Hou JG, Zhang YA, Wang CY, Wang Z, et al. (2014) Optimization of storage condition for maintaining long-term viability of nematophagous fungus Esteya vermicola as biocontrol agent against pinewood nematode. World J Microbiol Biotechnol 30(11): 2805-2810.
- Pavone D, Dıaz M, Trujillo L, Dorta B (2009) A granular formulation of Nomuraearileyi Farlow (Samson) for the control of Spodopterafrugiperda (Lepidoptera: Noctuidae) Interciencia 34(2):130-134.
- Sabuquillo P, De Cal A, Melgarejo P (2010) Development of a dried Penicilliumoxalicumconidial formulation for use as a biological agent against Fusarium wilt of tomato: selection of optimal additives and storage conditions for maintaining conidial viability. Biol Control 54(3):221-229.
- Chitwood DJ (2003) Research on plant parasitic nematode biology conducted by the United States Department of Agriculture- Agricultural Research Service. Pest Manag Sci 59(6-7): 748-753.
- Futai K (2003) Role of asymptomatic carrier trees in epidemic spread of pine wilt disease. Journal of Forest Research 8(4): 253-260.
- Giblin Davis RM, Davies KA, Morris K, Thomas WK (2003) Evolution of parasitism in insect-transmitted plant nematodes. Journal of Nematology 35(2): 133-141.
- Enda N (1976) Life cycle of Monochamus alternatus. For Pest 25: 182- 185.
- Kikuchi T, Cotton JA, Dalzell JJ, Hasegawa K, Kanzaki N, et al. (2011) Genomic Insights into the Origin of Parasitism in the Emerging Plant Pathogen Bursaphelenchus xylophilus. PLOS Pathogens 7(9): 1002219.
- Quimby PC, Zidack NK, Boyette CD, Grey WE (1999) A simple method for stabilizing and granulating fungi. Biocontrol Sci Tech 9(1): 5-8.
- Shabana YM, Muller Stover D, Sauerborn J (2003) Granular pesta formulation of Fusariumoxysporum f sp orthoceras for biological control of sunflower broomrape: efficacy and shelf-life. Biol Control 26(2):189-201.
- Togashi K, Shigesada N (2006) Spread of the pinewood nematode vectored by the Japanese pine sawyer: modeling and analytical approaches. Popul Ecol 48 (4): 271-283.
- Yang B, Wang Q (1989) Distribution of the pinewood nematode in China and susceptibility of some Chinese and exotic pines to the nematode. Canadian Journal of Forest Research, 19 (2): 1527-1530.
- Giblin Davis RM, Davies KA, Morris K, Thomas WK (2003) Evolution of parasitism in insect-transmitted plant nematodes. Journal of Nematology 35(2): 133-141.






























