Treatment of Highly Concentrated Effluent Generated from Intermediate Chemical and Bulk Drug Industries Renovated with Tertiary Removal
Atun Roy Choudhury1* and Arutchelvan Veeraraghavan2
1 IEISL R&D Lab, Fab City, Maheshwaram, Ranga Reddy District, Telangana, India
2 Department of Civil Engineering, Faculty of Engineering & Technology, Annamalai University, Annamalai Nagar, India
Submission: May 31, 2018; Published: June 13, 2018
*Corresponding author: Atun Roy Choudhury, Annamalai University, Tamil Nadu, India, Tel: +91+8838092641; Fax: +91-4144239731; Email: atnroy10@gmail.com
How to cite this article: Atun Roy Choudhury, Arutchelvan Veeraraghavan. Treatment of Highly Concentrated Effluent Generated from Intermediate Chemical and Bulk Drug Industries Renovated with Tertiary Removal. Int J Environ Sci Nat Res. 2018; 12(2): 555831. DOI: 10.19080/IJESNR.2018.12.555831.
Abstract
Effluent generated from the bulk drug industries is attributed with potential risk to the biotic and abiotic environment. The safe disposal of the same requires multiple stages of standard operation and stringent purification. In order to get rid of the ill impacts associated with the unscientific disposal of the bulk drug industry wastewater common effluent treatment plant (CETP) can be installed as a safe, inexpensive, and wholesome treatment unit. The present study, paid minute attention from the primary collection of the potentially hazardous wastewater to the reuse of the tertiary treated reverse osmosis (RO) process permeate. Initially, the collected effluent was stored in a bottom lined collection tank facilitate with a scrubber system and top enclosure to suppress the odour nuisance. Then effluent was immediately taken to the equalization tank and once homogenization got achieved the same was sent to the flocculation unit, where chemical coagulants were added with the effluent and forwarded to the primary clarifiers to remove the flocculants in the form of solid sludge.
Thereafter, the effluent was taken to the MEE feed tank and the bottom sludge was sent to the dewatering unit. The permeate was sent to the stripping column and the volatile organics were remove. In order to ensure optimal recovery the effluent of the stripper column was fed to the 5 stage multiple effect evaporator (MEE) system and successively followed by agitated thin film dryer (ATFD) set-up. Ultimately, the condensate was sent to the secondary treatment unit incorporated aeration facility and microbial digestion. The supernatant was conveyed to the secondary clarifier and successively to the tertiary treatment facility which comprises pressure sand filter, ultra filtration (UF), and RO. The end product was explicitly analysed to meet the prescribed standards and found to be feasible for reuse in the boiler. Both the primary and secondary sludge was safely discarded into the secured landfill. Thus, an absolute treatment system was delineated above which ensured optimal environmental safety
Keywords:CETP; COD; High TDS treatment; Pharmaceutical wastewater; Tertiary removal
Introduction
Two states located in the southern peninsula of Indian subcontinent namely, Telangana and Andhra Pradesh are considered to be the medicinal drug capital of the country which yields approximately 42% of the annual gross pharmaceutical production. Recently, these two states acting as a pioneer in the above delineated sector and showed a promising potential by having a continuous reciprocation with the market demand, technology up gradation, and modernized synthesis mechanisms. Its steady emergence in the pharmaceutical field has given it an international acclamation and plethora of foreign reputed pharma companies started capitalizing their investment into it [1,2]. Though drugs are often treated as life saving elements and trusted partner in health care industry, the production of it is always attributed to generation of highly toxic and complex wastewater. This aspect found to be contradictory to the progress pathway of the industry and thus, seeks a separate attention. Due to the complexity of the generated effluent it requires extensive treatment to bring down to the disposal or reusable limits. A single system consisting of a singular purification approach can not satisfy the prescribe figures due to high TDS and COD values (i.e. often ranging 1,00,000 mg/l and 45,000 mg/l respectively) [3-6]. Therefore a wholesome treatment system comprising multidimensional treatment facility is of utmost importance in order to treat the bulk drug industry wastewater. Furthermore centralization of the entire facilities under a single platform ensures the minimal environmental intervention with minimization of the land requirement. Hence, common effluent treatment plant is the ultimate solution to optimize the treatment expenditure attributed to waste treatment [7-10]. The treatment with the help of CETP requires similarity of the generated effluent. This is a feasible solution for those small scale industries, clustered together who can’t afford to install a separate individual treatment units at their disposal. It prevents them from any additional burden and extends opportunity to focus on the desired output without posing much additional cost to the stack holders [11,12].
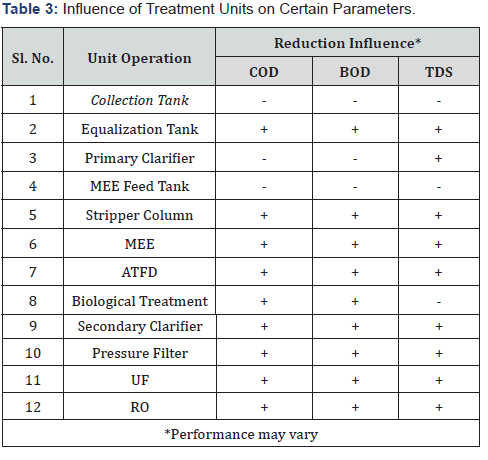
The CETP capable of treating pharmaceutical wastewater should comprise the units such as equalization unit, flocculation unit, biological treatment unit, high TDS treatment unit, and tertiary treatment unit [13-17]. The polished effluent can be further reused into the system as a boiler feed or also for greenbelt development. Each facility play a significant role in reduction of a certain parameter and the malfunction of any unit can successively upset the performance of the further units and thus, the plant operation is a delicate issue which needs to be performed under extreme supervision [18]. Despite all those facts execution of CETP can face significant protest from the nearby residents due to the obnoxious smell generation during the ATFD operation and powder precipitation. Therefore, keeping all the social constraints in mind successful operation of the CETP is a challenging task [19,20]. A study conducted by Popuri [21] reported an ultimate contaminant removal of 98% or above can be achieved by a complete CETP. But, contradictorily, a study carried by Salunke et al. [22] reported the inefficiency of such plants under certain circumstances. Thus, in limelight of the above delineated context the primary objective of this study was to explore the feasibility of the industrial CETP towards the treatment of the bulk drug industry wastewater and application of the same to minimize the environmental hazard.
Study Area
The entire research work was carried out within the premises of the Common Effluent Treatment Plant (CETP) designed and maintained by IL&FS Environment in Fab City. The plant is located in an isolated industrial estate and ensures minimal human and environmental interventions. The influence topography and the geological location optimized the safety with constant progress and changes. The approximate coordinates of the plant include, North West Corner (17°12’5.70”N, 78°30’18.54”E); North East (17°12’5.64”N, 78°30’24.08”E); South West (17°12’1.99”N, 78°30’17.87”E); South East (17°12’1.65”N, 78°30’23.85”E).
Materials and Methods
Collection of Effluent from Industries
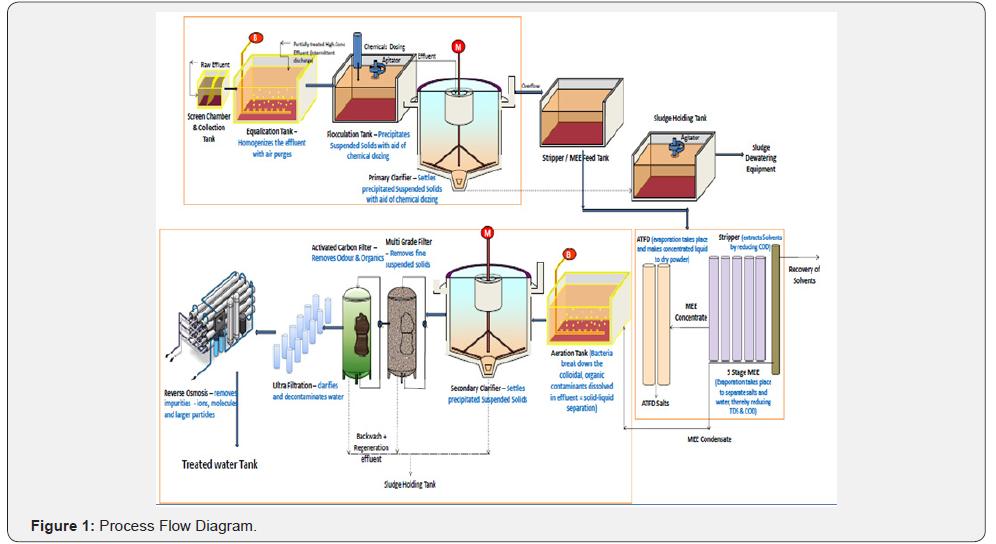
Process effluent from the pharmaceutical industries is transported to the designed location by means of 10m3 capacity GPS mounted closed tankers. The collection chamber was constructed with a design capacity of 150m3 connected to scrubber for control of odour pollution Thus, the collection tank can withhold approx. 15 truck unloading on regular basis.
Equalization of the Influent
Uniformity of the influent is the prime objective for the efficient working and thus, in order to maintain the minimal variation in the characteristics of the influent equalization tank was introduced. The influent mass was withheld in equalization tank over a period of 20 to 30 min and the tank has a capacity of 400m3. The influent in the tank gets homogenized due to air purges located at the bottom of the tank.
Flocculation
Flocculation tank is the set-up which serves as the platform for mixing. It is attributed with an agitator and has a volume holding capacity of 50m3. The agitator has a maximum working speed of 98 RPM, but 60 RPM was generally used for field application. Polyelectrolyte was used as a chemical coagulant and its efficacy was analysed against total solid removal.
Primary Settling
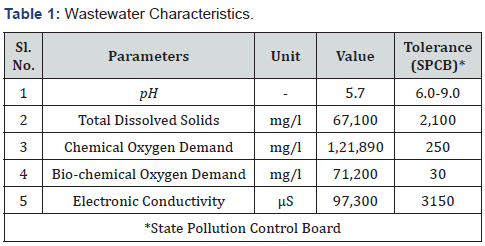
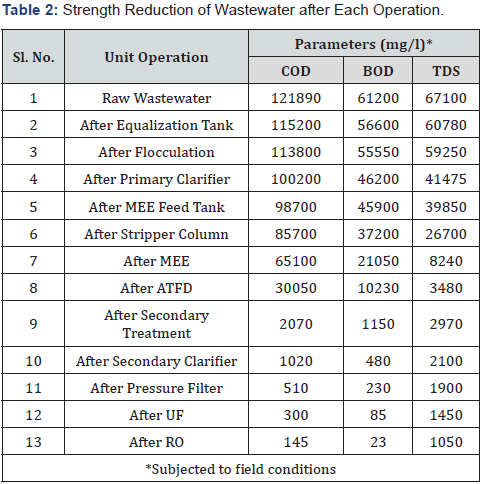
Once the agitation begins, the wastewater was brought into complete rest to ensure optimal precipitation. Settling of the particles with higher specific gravity often ensured in the primary clarifier, which indirectly released the burden of the high TDS treatment units. The facility comprises one primary clarifier with a volumetric capacity of 150m3 and the effluent from the primary clarifier use to be directly sent to the MEE feed tank for further purification (Tables 1-3).
Sludge Dewatering
The process sludge generated from both the clarifiers found to be highly humid in nature and thus, a moisture removal operation was quite mandatory. Excessive moisture was removed in sludge dewatering unit and the slurry got converted into bulky solid mass. The same was discarded in the secure landfill and environmental safety was ensured.
Removal of Volatile Compounds
The supernatant effluent from the MEE feed tank was directly conveyed to the stripping column via pumping station for further removal of volatile organic compounds (VOC). Contaminated water flows downward by gravity action and the air was forced through it. Thereafter, the contaminated air was purified by passing it through a demister before releasing it to the open atmosphere.
Effluent Densification
The end product of stripper column was further sent to the MEE unit for densification and volume reduction. The installed MEE setup has 5 nos. of calandrias and works in a forward flow mode. The raw juice from stripper column was introduced to the first calandria from the bottom and brought in contact with steam. Heat exchange, gasification, and condensation were few of the operation that was performed simultaneously. The condensate from the facility was sent to the tertiary filtration units; whereas, the concentrate was forwarded to the agitated thin film dryer for further solidification.
Secondary Treatment
The condensate from the high TDS treatment system was conveyed to the biological treatment unit for further polishing and removal of the organic contaminants. This facility incorporates aeration and aerobic microbial digestion. An optimum air supply rate of can be achieved in this system which is subjected to the influent BOD concentrations. A mixed consortium of microbial culture was used in the system by mixing the cow dung into the HTDS condensate with a ratio of 10:90.
Secondary Settling
The effluent from the biological treatment unit was retained in the secondary clarifier over a period of 1 hr to remove the settable solids from the stream before sending it to the advanced treatment unit. Once, the retention time got completed supernatant was sent to the tertiary treatment unit whereas, bottom sludge was conveyed to the sludge dewatering facility and discarded in a similar fashion.
Tertiary Treatment
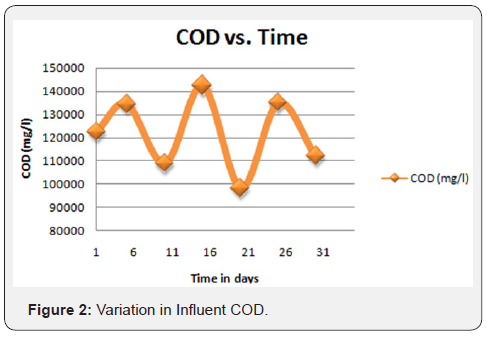
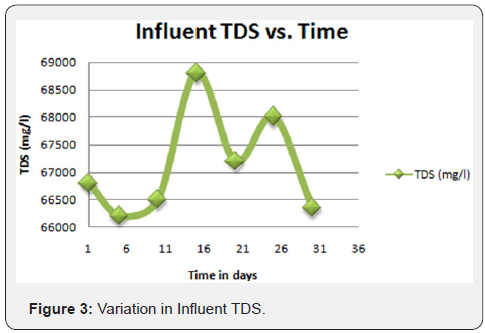
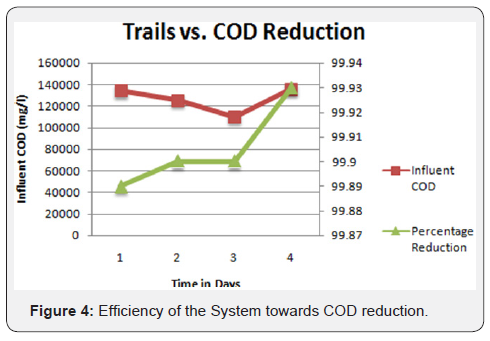
The semi-polished effluent from the physicochemical treatment unit was supplied to the tertiary unit as an influent. The same was initially introduced to the pressure sand filter and successively followed by Ultra Filtration (UF) and reverse osmosis process. The permeate of RO yielded a satisfactory quality of effluent fit for reuse/recycle in the plant and industries for utilities and RO rejects is again sent back to the MEE. The process flowchart of the plant operations has been portrayed in Figures 1-4.
References
- Gohary EI, Abou-Eleha FA, Aly HI (1995) Evaluation of biological technologies for wastewater treatment in the pharmaceutical industry. Water Science and Technology 32(11): 13-20.
- Garcia A, Rivas HM, FigueroaJL, Monroe AL (1995) Case history: Pharmaceutical wastewater treatment plant upgrade, Smith Kline Beecham Pharmaceuticals Company. Desalination 102(1-3): 255-263.
- Hashmi I (2005) Wastewater monitoring of pharmaceutical industry: Treatment and reuse options. Electron J Environ Agric Food Chem 4(4): 994-1004.
- Junico M, Shelef G (1994) Design operation and performance of stabilization reservoir for waste water irrigation in Israel. Wat Res 28: 175-186.
- Khan MA, Ahmad SI (1992) Performance evaluation of pilot waste stabilization ponds in subtropical region. Wat Sci Tech 26: 1717-1728.
- NgMiranda WJ, Yap GS, Sivadas M (1989) Biological treatment of a pharmaceutical wastewater. Biological Wastes 29(4): 299-311.
- Puskas K, Essen II, Banat I, Al Daher R (1991) Performance of an integrated ponding system operated in arid zones. Wat Sci Tech 23: 1543-1542.
- Rosen M, Welander T, Lofqvist A, Holmgren J (1998) Development of a new process for treatment of a pharmaceutical wastewater. Water Science and Technology 37(9): 251-258.
- K Sundara Kumar (2010) Performance evaluation of waste water treatment plant. International Journal of Engineering Science and Technology 2: 7785-7796.
- Sahithya DD, Lakshmi MVVC (2016) Performance Evaluation Study of an Effluent Treatment Plant in Pharmaceutical Industry. International Research Journal of Engineering and Technology 3(8): 67-77.
- Ramteke PW, Awasthi S, Srinath T, Joseph B (2010) Efficiency assessment of Common Effluent Treatment Plant (CETP) treating tannery effluents. Environ Monit Assess 169: 125-131.
- Gadipelly C, Pérez-González A, Yadav GD, Ortiz I, Ibáñez R, et al. (2014) Pharmaceutical Industry Wastewater: Review of the Technologies for Water Treatment and Reuse. Ind Eng Chem Res 53(29): 11571-11592.
- Guo Y, Qi PS, Liu YZ (2017) A Review on Advanced Treatment of Pharmaceutical Wastewater. IOP Conf Series: Earth and Environmental Science 63: 012-025.
- Zeng P, Du J, Liu Y, Gao H, Liu R (2015) Pharmaceutical wastewater treatment in China and the world: A bibliometric analysis of research output during 1990-2013. Research & Reviews: Journal of Pharmaceutical Quality Assurance 1(1): 30-37.
- Radjenovic J, Petrovic M, Barceló D (2007) Analysis of pharmaceuticals in wastewater and removal using a membrane bioreactor. Anal Bioanal Chem 387(4): 1365-1377.
- Ng WJ, Yap MGS, Sivadas M (1989) Biological treatment of a pharmaceutical wastewater. Biological Wastes 29(4): 299-311.
- Jain G, Satyanarayan S, Nawghare P, Kaul SN, Szpyrcowicz L (2001) Treatment of pharmaceutical wastewater (herbal) by a coagulation/ flocculation process. International Journal of Environmental Studies 58(3).
- Joss A, Keller E, Alder AC, Göbel A, McArdell CS, et al. (2005) Removal of pharmaceuticals and fragrances in biological wastewater treatment. Water Research 39(14): 3139-3152.
- Miège C, Choubert JM, Ribeiro L, Eusèbe M, Coquery M (2009) Fate of pharmaceuticals and personal care products in wastewater treatment plants - Conception of a database and first results. Environmental Pollution 157(5): 1721-1726.
- Jelic A, Gros M, Ginebreda A, Cespedes-Sánchez R, Ventura F, et al. (2011) Occurrence, partition and removal of pharmaceuticals in sewage water and sludge during wastewater. Water Research 45(3): 1165-1176.
- Popuri AK (2017) Pharmaceutical and Bulk Drug Industry Wastewater - Solutions for Water Treatment. International Journal of ChemTech Research 10(5): 811-816.
- Salunke KA, Bhave PP, Mata MD (2014) Performance Status of Common Effluent Treatment Plant at Dombivali Cetp. International Journal of Research in Engineering and Technology (IJRET) 3(9): 48-52.






























