Air Pollutants and Their Possible Health Effects at Different Locations in Dhaka City
Martinez-Toledo A*1, Rodriguez-Vazquez R2 and Ilizaliturri-Hernandez CA3
1Universidad Aut�noma de San Luis Potos�, Facultad de Ingenier�a, Zona Universitaria, San Lu�s Potos�, M�xico
2Departamento de Biotecnolog�a y Bioingenier�a, Centro de Investigaci�n y de Estudios Avanzados del IPN, Col. San Pedro Zacatenco, M�xico
3Facultad de Medicina, Coordinaci�n para la Aplicaci�n de la Ciencia y la Tecnolog�a-Centro de Investigaci�n Aplicada en Ambiente y Salud (CIACyT-CIAAS), México
Submission: April 18, 2018; Published: April 26, 2018
*Corresponding author: Martinez-Toledo A, Universidad Autonoma de San Luis Potosf, Facultad de Ingenieria, Zona Universitaria, CP. 78290, San Lui's Potosf, Mexico, Tel: +52-444-8262330; Email: anmartin_70@hotmail.com
How to cite this article: Mart�nez-Toledo A, Rodr�guez-V�zquez R, Ilizaliturri-Hern�ndez CA. Culture Media Formulation and Growth Conditions for Biosurfactants Production by Bacteria. Int J Environ Sci Nat Res. 2018; 10(3): 555790. DOI: 10.19080/IJESNR.2018.09.555790
Abstract
At present, several industrial cities are showing a lot of pollution problems due to xenobiotics in soil. Most of them are recalcitrant to the environment dueto their low 1-octanol-water partitioning coefficient (hydrophobic organic contaminant),in addition they are toxic to the biota. Nowadays, the technique that is most compatible with the environment is bioremediation, which is used to remove these kinds of toxics from different matrixes. For this reason, a number of microorganisms where selected not only because they produce changes in the molecular structures of the toxics (through their enzymatic activity), but also because of the extra and intra cellular surface active compounds (biosurfactants)that they generate, containing tensoactive properties which are very similar to synthetic surfactants, that are involve in micelles the toxic from the soil, improve their desorption or their biodegradation by the same or by other microorganisms, and in this case the most studied where bacterial microorganisms. In order to induce the production of these substances in microorganisms, vast research had to previously be done; the aim of this work was to generate an overview of some of these important research projects, to be able to understand the growth conditions and nutritional requirements of the different microorganisms that produce biosurfactants as well as to outline the possibility of using them in the bioremediation process.
Keywords: Bacteria; Biosurfactants; Cell culture; Nutrients; Environmental conditions
Introduction
There are various microorganisms capable of synthesizing a wide spectrum of biosurfactants (bio-emulsifiers)-of either a high or a low molecular weight class. Low molecular weight biosurfactants are mostly glycolipids (trehalolipids, sophorolipids and rhamnolipids), however, they could also be lip peptides such as Surfactant, Gramicidin and Polymyxin. Heavier molecular biosurfactants include amphiphilicpolysaccharides, proteins, lip polysaccharides, lipoproteins or complex mixtures of these polymers [1]. The lighter ones reduce surface tension while the heavier ones, produce stable emulsions of water in oil [2]. Rhamnolipidsare made up of either one or two rhamnose molecules linked to one or two molecules of te-hydroxypentanoic acid. Rhamnose with glycolipids was first described in Pseudomonas aeruginosa Trehalolipids, it is formed by trehalose disaccharides linked by mycolic acids onto C-6 and C-6'and it is associated with most species of Mycobacterium, Nocardia and Corynebacterium; threalose lipids from Rhodococcuserythropolis and Arthrobactersp. Lowered the surface and the interfacial tension. Sophorolipids contain dimeric sophorose carbohydrates linked to long chains of aliphatic (hydroxyaspartic) acids which are mainly produced by yeast (Torulopsisbombicola, T. petrophilum and T. apicola) [1]. Biosurfactants have better surface and interface activity than synthetic surfactants, these properties result in strong tolerance to high temperatures and salinity. Biosurfactants are biodegradable in soil or water, and their emulsion can be broken easily [3].
At present, bacteria are the focus of researchers investigating and working on the production of biosurfactants. Among those microorganisms that are being deeply studied we find Pseudomonas aeruginosa which consistently producer hamnolipids from various sources of carbon as shown by numerous studies. Its most negative aspect is the fact that it is inconvenient for use given its pathogenicity for humans. There are numerous studies about secondary metabolism biosynthetic pathways through which biosurfactants are produced. It is possible to establish that bacteria metabolism may shift toward sources of carbon in order to produce biosurfactants. In order to obtain energy and to produce biosurfactants from carbohydrates, the catalytic cycle of tricarboxylic acid (CTA) must occur. One of the enzymes from the CTA cycle is dehydrogenate is ocitrate which correlates to the production of biosurfactant.
It has been proven that this enzyme is of vital importance in the production of polihydroxibutirate (PHB) produced by the Azotobacterbiejerinckii.
To be able to redirect the metabolism of microorganisms towards the production of biosurfactants in a liquid culture, various nutritional and environmental conditions must be administered. Some of the most popular variables are the carbon source, nitrogen source, and environmental variables [4-12]. In the following paragraphs are presented some research with bacterial that produce biosurfactants.
Nutritional Factors
Carbon Sources: In order to produce biosurfactants from Pseudomonasspp the following water soluble carbon sources have been deployed: glycerol, glucose, mannitol and ethanol. Production levels ofbiosurfactants fall when using carbon sources which are immiscible in water such as olive oil and alkanes. Also, it has been shown that the various sources of carbon used by microorganisms modify the composition of biosurfactants in its polar fraction while keeping the glycolipid chain's length at the non-polar fraction unchanged (fatty acids). Nonetheless, a qualitative variation exists among biosurfactant production by Acinetobactersp strains H13-A and H01-N as is evidenced by the number of alkane carbons [13]. Bacillus subtilis ATCC 21332 was used as a carbon source potato (60 g/L) for the production of Surfactin (decreased surface tension of the medium was 41.8 mN/m) [14]. Linhardt et al. [15], observed than Pseudomonas aeruginosa strain U129791, used corn oil as a carbon source and produced rhamnolipids, the highest production of rhamnose (5.4 g/L) was obtained when the concentration in culture medium was 40 g/L of corn oil. Cellulomonascellulans produced glycolipids (8.9 g/L, expressed as glucose) when it grew in liquid medium with 30 g glycerol/L [16].
P. aeruginosa 44T1 used hydrocarbons with carbon chains C12 and olive oil as carbon sources to produce rhanmolipids. However, in the presence of fructose, production is inhibited [17,18]. Benincasa et al. [19] proved that P.aeruginosa LB1 uses oapstock (3g/l) as carbon source to produce rhamnolipids (12 g/L). Torulopsismagnolie employslong-chain fatty acids, hydrocarbons and glycerin to produce So phorolipids [20]. When Arthrobacterparaffineus ATCC 19558 grows in D-glucose and when hexadecane is added during its latent growth phase, biosurfactant performance has a significant peak. Patel and Desai [21] reported that Pseudomonas aeruginosa GS3 increased their biosurfactant production with molasses (48%w/v of carbohydrates) present in the medium at 7% as a carbon source, which generated 0.24 g/L of rhamnose after 96h of incubation. Pruthi and Cameotra [22] reported that Serratiamarcescens produced biosurfactant (30 mN/m) using minimal medium containing sucrose (2% w/v) at a wide pH (2 - 12) and temperature (10-120°C) range.n-hexadecane (2% v/v) was used as the sole carbon source by Arthrobacterprotophormiae MTCC 688 to produce biosurfactant at different salt concentrations (10-100 g/L), where the highest concentration (5.3 g/L) was obtained using 20 g/L of salt (NaCl) at psychrophilic conditions (10°C) [23]. Chayabutra et al. [24] examined different carbon substrates (palmitic acid, stearic acid, oleic acid, linoleic acid, glycerol, vegetable oil, hexadecane and glucose) for P. aeruginosa in anaerobic and aerobic conditions. All of these carbon sources were able to support cell growth, and synthesis of rhamnolipids was only effective when using hexadecane, palmitic and stearic acid.
Hartoto and Mangumwidjaja (2002) studied the biosurfactant production (lipopeptides) ability of Bacillussp BMN14 using different sugars (glucose, fructose, sucrose, etc.) at differ concentrations (1, 2, 3, 4, 5, y 6%, in the liquid medium) as carbon sources, and observed that the highest biomass (6.35 g/L) and biosurfactant production (2.23 g/L), the maximum specific growth (0.065/h), and the lowest surface tension (29 mN/m) were obtained with glucose at 4%. Yateem et al. [25] investigated the biosurfactant production by two different strains of Pseudomonas aeruginosa (KISR C1 and KISR B1) using crude oil, olive oil, hexadecane, diesel, kerosene and oil sludge, the results showed that the highest reduction in surface tension was recorded using de strain C1 with olive oil (10 mg/L) as a sole carbon sources. George and Jayachandran [25] conducted a research on rhamnolipid production from Pseudomonas aeruginosa MTCC 2297 and studied the cost-effectiveness of several waste material supplies (arrange peelings, carrot peel waste, lime peelings, coconut oil cake, and banana waste). The results reported that the orange peelings were the best substrate, producing 9.18 g/L of rhamnolipids with a surface tension reduction of up to 31.1 mN/m. Xia et al. [27] enhanced rhamnolipid production (80g/L) by Pseudomonas aeruginosa WJ-1using sunflower oil.
Corynebacterium lepusc an grow in glucose but if hexadecane is added it becomes able to synthesize large amounts of biosurfactants [13]. The way Torulopsisbombicola handles its carbon sources allows us to observe an increase in production of glycolipids [28]. Tuelva et al. [29] reported that P. Putida 21BN is able to produce rhamnolipids utilizing hexadecane carbon as its only source. A study done by Martinez-Toledo et al. [30] reported that P. Putida CB-100 was able to increase production of rhamnolipids when phenanthrene (200 mg/L) was added to the culture. Oliveira et al. [21] reported that P. aeruginosa FR produces biosurfactants when palm oil is used as a carbon source. Other studies have shown that Pseudomonas sp. Strain PP2, could produce biosurfactants growing on either phenanthrene or dextrose as carbon sources, but phenanthrene showed the highest production levels compared to dextrose [31]. de Lima, et al. [32] observed the rhamnolipid production due to Pseudomonas aeruginosa PACL growth on different waste frying soybean oils, where the maximum rhamnose production (4.3 g/L) was obtained using the NUSO soybean oil. Pseudomonas aeruginosa SP4 used palm oil as carbon source and produced biosurfactant at oil loading rates of 2 kg/m3 24h, the surface tension of the liquid medium was reduced by 58% Pornsunthorntawee et al. [33]. Pseudomonas sp. LP1 produced biosurfactants in a culture where motor oil was the only available carbon source [34]. Rahman et al. [35] reported that P. aeruginosa DS10-129 produced 4.31 g/L of rhamnolipids in a mineral salts medium (MSM) with 2 g/L of glucose and soybean oil. Lima et al. [36] reported that Bacillus subtilis ICA56 produces a biosurfactant (120 mg/L) in mineral salts medium (MSM) with glycerol (20 g/L), which results in a reduction of the surface tension from 72 to 28 mN/m. In another report [37] a bacterial strain Bacillus sp. MTCC 5877 produces a biosurfactant in a MSM with 1% of glycerol added and 1% of cooking oil, the reduction in the surface tension was of 42 mN/m and the emulsification index (E24) was 76%. A bacterium Bacillus subtillus R14914 produces 200 mg/L in a mineral medium with 20 g/l of sucrose Fernandes et al. [38]. Bacillus tequilensis ZB10 in a MSM containing hemicellulosic (50%) and cellulose (50%) hydrolysates yielded 1.52 g /L of biosurfactant that reduced surface tension to 38.6 mN/M. Hang el at. [39], tested a petroleum-degrader bacterium (Bacillus sp BS-8) at different concentrations in order to produce biosurfactant. Their results revealed that the best carbon source was glucose (20g/L) which reached the best oil displacement (62 mm). Bacillus pseudomycoides BS6 is a bacterium strain that was isolated from an oil-contaminated soil, with the capacity of producing biosurfactant that reduces surface tension from 76 mN/m to 30.2 mN/m, and produces an emulsion activity between 62.8% to 94.2% using waste soybean oil (20 g/L) as a carbon source. Bacillus pumilus 2IR is another bacterium strain which was isolated from an Iranian oil field as a potent biosurfactant- producing bacterium. The maximum biosurfactant production (30 nN/m) was observed in a medium formulated with 30.31 g/L of glucose, 0.8% of crude oil, as carbon sources.
Nitrogen and Phosphorus Sources
The effect of other macro nutrients added to the environment has been investigated as part of the production of biosurfactants. Apart from carbon (C), the most widely studied macro nutrients are Nitrogen (N) and Phosphorous (P). From these, N is of greater relevance. Arthrobacterparaffineus utilizes ammonium and urea salts as sources of nitrogen for the production of biosurfactants [20]. P. aeruginosa and Rhodococcusspp are able to use nitrates in order to increase the production of biosurfactants. A. paraffineus increases its production of biosurfactants when adding L-amino acids such as spastic acid, glutamic acid, asparagine and glycine to the environment. The structure of Surfact in produced by Bacillus subtilis modifies with concentrations of L-amino acids in the environment, producing Surfactin Val-7 or Leu-7. It has been observed, that Nitrate is the best source of nitrogen for the production of biosurfactants for P. aeruginosa strain 44T1 and for Rhodococcus strain ST-5 grown in olive oil and paraffin, respectively. Its production of biosurfactants started after 30 hours of growth, when the culture reached limiting concentrations of nitrogen and continued its production for up to 58 hours of fermentation. Patel and Desai (1997) introduced cornsteep liquor (0.5%, v/v) in the medium as a source of nitrogen to induce P. aeruginosa GS3 for the production of biosurfactants after 96 h of incubation. The produced rhamnolipid had the ability to emulsify various hydrocarbons (n-heptane, n-decane, n-hexadecane, benzene, toluene, n-paraffin, 2-methylnapthalene + n-hexadecane (1:1%, v/v, crude oil and olive oil). Xia et al. [27] used a culture medium with a N limit concentration (C/N of 8) and with NaNO3 as a source of N; under these conditions in a fermenter (3000 L) with Pseudomonas aeruginosa WJ-1 50.2 g/L of rhamnolipids were produced and the surface tension decreased 43.09 mN/m.
In general, it has been observed that limiting concentrations of nitrogen favors the production of biosurfactants by various microorganisms. Davis et al. [40] observed that Bacillus subtilis ATCC 21332 increased its production of biosurfactants only when nitrate decreased in the culture medium. An increase in the production of rhamnolipids by P. aeruginosa LBI was observed following nitrogen depletion [19]. Corynebacterium lepus increased its production of biosurfactants when it used nitrate as its single source of nitrogen in the culture [41]. Some authors have observed that the synthesis of rhamnolipids by P. aeruginosa takes place when the source of nitrogen has run out and when its growth cycle is at its latency phase, and that when nitrogen sources are added production of rhamnolipids by Pseudomona ssp DSM 2874 [42] is inhibited. It has been reported that genes are activated to produce P. aeruginosa biosurfactants of P. fluorescents and P Putida only under limited nitrogen conditions. Arino et al. [16] reported that glycolipid production by Cellulomonascellulans began after nitrogen sources (such as sodium nitrate) were exhausted in the medium and increased when their growth had stopped.
There is evidence of an existing connection between rhamnolipid synthesis and glutamine synthetase activity in P. aeruginosa. This enzyme delivers maximum activity levels when its exponential growth phase ends, and at that time its production of biosurfactants begins. Furthermore, Mulligan et al. [28] reported that the shift in metabolism to phosphate coincides with the production of biosurfactants. P limitation in the culture media was more effective for biosurfactant production by P. aeruginosa than N limitation, only when hexadecane or palmitic acid were used as carbon sources in anaerobic conditions [24]. In studies done by Martinez-Toledo and Rodriguez-Vazquez [43] it was also noted that P. Putida CB- 100 is able to produce rhamnolipids with variations in its polar fraction due to the relationship changes among C:N:P, where glucose and phenanthrene were used as carbon sources in the culture medium.
Micronutrients
Several papers report on the great importance of micronutrients in the culture media for bacteria (Table 1). Guerra-Santos et al. [44] proved that under limited ion conditions Mg, Ca, K, Na, Fe and other trace elements, a high rate Pseudomonasspp is obtained when it reaches its latency growth of biosurfactant production by P aeruginosa is delivered. Various stage, under limited nitrogen and iron conditions. authors have shown that an overproduction of biosurfactants by Pseudomonasspp is obtained when it reaches its latency growth stage, under limited nitrogen and iran condition.
Phosphate, iron, magnesium and sodium are all elements of importance to the production of biosurfactants by Rhodococcussp. but potassium and calcium are the elements of greatest importance. Iron also has an important impact on the production of rhamnolipids by P. aeruginosa. The concentration of manganese and iron affects the production of Surfactin by Bacillus subtilis. A report discusses how B. subtilis has an active transportation system for both ions, manganese may act as a cofactor to many enzymes involved in the metabolism of nitrogen, even in reactions from glutamate and ammonium catalyzed by the glutamine synthetase enzyme, this being its main assimilation mechanism of inorganic nitrogen, observed that with a concentration of 0.5 mg/L of FeSO4 7H2O (C/Fe ratio of 72 400) in the culture medium rhamnolipids production by Pseudomonas aeruginosa increased (500mg/L). Bacillus subtilis ATCC 21332 increases its production of Surfactin in the presence of 1.7 mM of iron sulfate in the culture medium [45]. The iron concentration of 50-100 |ig/L was the optimal concentration in the liquid medium for rhamnolipid production by Pseudomonas aeruginosa strain U129791 [38]. Amezcua-Vega et al. [17] observed an increase in the production of rhamnolipids by P. Putida CB-100 at a low C/Fe ratio (26000) in the culture medium, where the carbon source was corn oil at 2% (w/v). Similar results were observed with Rhodoccocussp. 51T7 that produced glycolipids (3g/L) at a low C/Fe ratio of 12000 [18].
Environmental Conditions
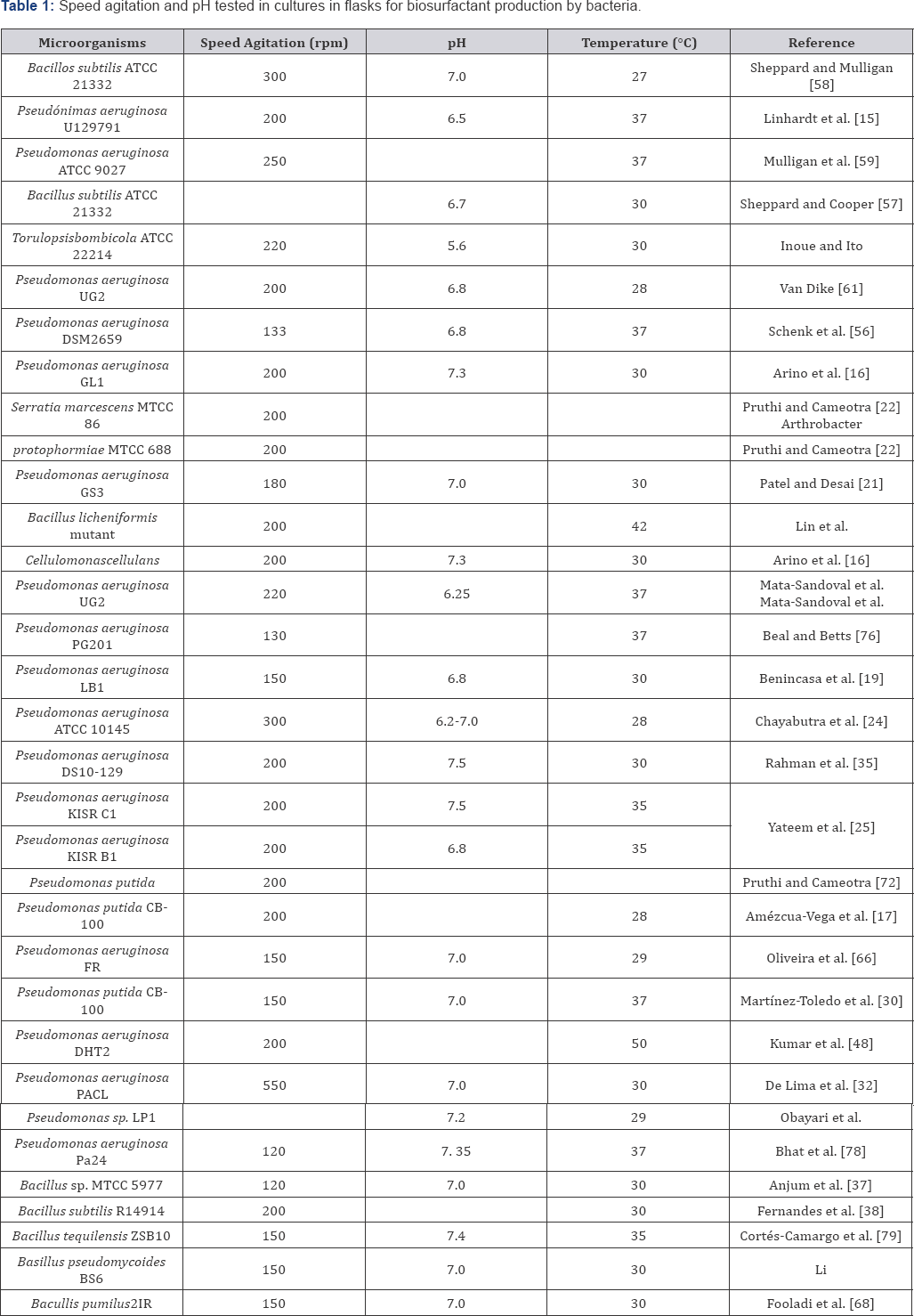
Environmental factors and growth conditions (Table 2) such as pH, temperature, agitation, availability of oxygen, etc., also affect the production of biosurfactants, via effects on both growth and cell activity. Production of rhamnolipids by Pseudomonasspp maximizes with a pH range between 6 and 6.5 and rapidly decreases when pH is higher than 7. It is also noted that the production of amphiphatic and disaccaride lipids by N. corynbacteroides does not undergo any changes when pH ranges from 6 to 8. It is known that surface tension as well as critical micelle concentration or CMC remain stable in a wide range of pH, however the emulsification has a limited pH range. Guerra-Santos et al. [44] determined that within a pH range from 6.2 to 6.4 Pseudomonas aeruginosa augments its production of rhamnolipids. On the other hand, A. paraffinues and the Peudomonasp strain DSM-2874, the temperature provokes an increase in biosurfactants production. A thermophilic Bacillus sp grew and produced a biosurfactant at temperatures above 40°C. Some biosurfactants do not alter either one of their properties: low surface tension or emulsifying efficiency, which remain stable even after sterilizing at 120°C for 15 minutes. Within a temperature range of 32 to 34°C Pseudomona aeruginosa increases production of rhamnolipids. Javaherin et al. [46] reported that Bacillus licheniformis produces biosurfactants at 50°C temperature. It has been observed that salt concentration affects production of biosurfactants which in turn have a direct effect over cell activity. Desai and Banat reported that salt concentrations above 10% (weight/volume) do not affect production of biosurfactants while a slight reduction in the CMC has been noted. Kumar et al. [47] reported that "Planococcusmaitriensis Anita I" (marine bacterium), produced biosurfactant in a medium with 0.5-12% of salt. A strain of P. aeruginosawas capable of growing in an environment which had a high degree of salinity (10%) together with high temperatures (45°C), using hexadecane as carbon source [48]. Elazzazzy reported that VirgiBacillussalarius KSA-T was the best bacterium in terms of biosurfactant production (30 mN/m) in a MSM with glucose (2%) and waste frying oil, urea and NaNO3 were the best N sources in biosurfactant production (29.5 mN/m) by KSA-T (V salarius), besides, this strain works at extreme conditions: pH 9, 45 -50°C and 4% of NaCl.

Biosurfactant producers are mainly aerobic microorganisms that grow in liquid media, which is why aeration plays an important role in the biosurfactant yield (Table 2). The production of biosurfactants by Nocardiaerythopolis decreases while agitation speed increases in the culture environment.
It has been reported that in order for some yeasts to produce biosurfactant, performance is enhanced at both high agitation speeds and a high degree of aeration. A paper reports that the transference of oxygen is an important parameter in optimization processes for large scale production of Surfactin by B. subtilis. For the strain P. aeruginosa PACL, aeration was the principal variable that affected their biosurfactant production, and the maximum rhamnose concentration (3.3g/L) was obtained at 300 rpm.
Perspectives
Production costs are often an obstacle for biotechnological processes, particularly those involving biosurfactants. Success in the production of biosurfactants hinges on the development of economic processes and on the use of low cost raw materials which represents between 10 and 30% of production costs. Biosurfactants must compete with surfactants of petrochemical origin in three key areas: costs, functionality and production capacity. High production costs m ay be afforded on a low- demand biosurfactant or on products of high commercial value such as cosmetics, medicines, etc. However, for applications aimed at the recovery of contaminated soils in which large volumes of biosurfactants are needed, its high production cost is incompatible with its use. Makkar and Cameotra [49] have suggested four factors which may cut back production costs. Microorganisms (choose, adapt or modify to obtain peak production performance), the process (choose, adapt or design for low capital investments and operating costs), the substrate for microorganisms and the feeding processes (choose to reduce costs), and the sub-products from the production process (minimize residue) [50-58].
Some research on the choice of adequate substrates has focused mostly on tropical agro-industrial plantations and on residues [59-65]. These include plantations like tapioca, soy, sugar beet, sweet-potato, potato, tender sorghum, harvest residues like bran, straw, wheat and rice, husk of soybean, corn and rice, sugarcane bagasse, and tapioca; residues from the coffee industry such as coffee pulp, coffee grain husks, residual ground coffee; residues from fruit processing like grapefruit, apple and grapes; residues from pineapple and carrot processing, banana residues, residues from the processing of milling to obtain oil, such as coconut paste, soy paste, peanut paste, canola, palm, and others such as the shavings from corn cobs, sheath of carob trees, tea residues, etc [65-74]. Additional substrates have been suggested for the production of biosurfactants such as water miscible residues, molasses, milk serum, or the residual distiller. Therefore, it is very important to select the microorganisms and know their different metabolic capabilities, in order for them to be used as potential tools in the bioremediation of polluted matrixes [75-80]. In addition, saving costs and likely reducing the time of the biological process, as well as their potential applications in environmental restauration, food, cosmetics, agriculture and pharmaceutical industries.
References
- Muthusamy K, Gopalakrishnan S, Ravi TK, Sivachidambaram P (2008) Biosurfactants: Properties commercial production and application. Curr Sci 94(6): 736-747.
- Rosenberg E, Ron EZ (1999) High- and Low- molecular- mass microbial surfactant. Appl Microbiol Biotechnol 52(2): 154-162.
- Bognolo G (1999) Biosurfactantsas emulsifying agents for hydrocarbons. Colloid Surface A 152: 41-52.
- Rodrigues RL, Teixeira JA, Oliveira R (2006) Low cost fermentative medium for biosurfactant production by probiotic bacteria. Biochem Eng J 32: 135-142.
- Rodrigues L, Teixeira J, Oliveira R, van der Mei (2006) Response surface optimization of the medium components for the production of biosurfactants by probiotic bacteria. Process Biochem 41: 1-10.
- Joshi S, Jadav S, Desai AJ (2008) Application of response-surface methodology to evaluate the optimum medium components for the enhanced production of lichenysin byBacillus licheniformisR2. Biochem Eng J 41: 122-127.
- Mukherjee S, Das P, Sen CSR (2008) Enhanced production of biosurfactant by a marine bacterium on statistical screening of nutritional parameters. Biochem Eng J 42: 254-260.
- Mutalik SR, Vaidya BK, Joshi RM, Desai K M, Nene SN (2008) Use of response surface optimization for the productionof biosurfactant from Rhodococcus spp. MTCC 2574. Bioresource Technol 99: 7875-7880.
- Najafi AR, Rahimpour MR, Jahanmiri AH, Roostaazad R, Arabian D, et al. (2010) Enhancing biosurfactant production from an indigenous strain of Bacillus mycoides by optimizing the growth conditions using a response surface methodology. Biochem Eng J 163: 188-194.
- Kiran GS, Thomas AT, Selvin J, Sabarathnam B, Lipton AP (2010) Optimization and characterization of a new lipopeptidebiosurfactant producedby marine Brevibacteriumaureum MSA13 in solid state culture. Bioresource Technol 101: 2389-2396.
- Najafi AR, Rahimpour MR, Jahanmiri AH, Roostaazad R, Arabian D (2011) Interactive optimization of biosurfactant production by Paenibacillus alveiARN63 isolated from an Iranian oil well. Colloid Surface B 82: 32-39.
- Vaz DA, Gudina EJ, Alameda EJ, Teixeira JA, Rodrigues RL (2012) Performance of a biosurfactant produced by a Bacillus subtilis strain isolated from crude oil samples as compared to commercial chemical surfactants. Colloid Surface B 89: 167-174.
- Desai JD, Banat IM (1997) Microbial production of surfactants and their commercial potential. Microbiol MolBiol R Mar 61(1): 47-64.
- Fox SL, Bala GA (2000) Production of surfactant from Bacillus Subtilis ATCC 21332 using potato substrates. Bioresource Technol 75: 325240.
- Linhardt RJ, Bakhit R, Daniels L (1989) Microbially produced rhamnolipid as a source of rhamnose. Biotechnol Bioeng. 33: 365-368.
- Arino S, Marchal R, Vandecasteele JP (1998) Production of new extracellular glycolipids by a strain of Cellulomonascellulans (Oreskoviaxanthineolytica) and their structural characterization. Can J Micro Biol 44: 238-243.
- Amezcua Vega C, Ferrera Cerrato R, Esparza Garcia F, Ri'osLeal E, Rodriguez Vazquez R (2004) Effect of combined nutrients on biosurfactant produced by Pseudomonas putida. J Environ Sci Heal A 39 (11-12): 2983-2991.
- Robert M, Mercad ME, Bosch MP, Parra JL, Espuny MJ (1989) Effect of the carbon source on biosurfactant production by Pseudomonas aeruginosa 44T1. Biotech Lett 11(12): 871-874.
- Benincasa M, Contiero J, Manresa MA, Moraes JO (2002) Rhamnolipid productions by Pseudomonas aeruginosa LBI Growing on soap stock as the sole carbon source. J Food En 54: 283-288.
- Cameotra SS, Makkar RS (1998) Synthesis of biosurfactants in extreme conditions. Appl Microbiol Biotechnol 50: 520-529.
- Patel RM, Desai AJ (1997) Biosurfactant production by Pseudomonas aeruginosa GS3 from molasses. Lett Appl Microbiol 25: 91-94.
- Pruthi V, Cameotra SS (1997) Short Communication: Production of a biosurfactant exhibiting excellent emulsification and surface active properties by Serratiamarcescens. World J Microb Biot 13:133-135.
- Pruthi V, Cameotra SS (1997) Short Communication: Production and properties of a biosurfactant synthesized by Arthrobacter protophormiae an Antarctic strain World. J Microb Biot 13: 137-139.
- Chayabutra C, Wu J, Ju LK (2000) Rhamnolipidproducton by Pseudomonas aeruginosa under denitrification: effects of limiting nutrients and carbon substrates. Biotechnol Bioeng 72(1): 25-33.
- Yateem A, Balba MT, Al-Shayji Y, Al-Awadhi N (2002) Isolation and characterization of biosurfactant-producing bacteria from oil- contaminated soil. Soil Sediment Contam 11(1): 41-55.
- George S, Jayachandran K (2009) Analysis of rhamnolipid biosurfactant produced trough submerged fermentation using orange fruit peelings as sole carbon source. Appl Biochem Biotechnol 158(3): 694-705.
- Xia WJ, Luo ZB, Dong HP, Yu L (2013) Studies of biosurfactant for microbial enhanced oil recovery by using bacteria isolated form the formation water of a petroleum reservoir. Petrol Sci Technol 31(21): 2311-2317.
- deRoubin MR, Mulligan N, Gibbs BF (1989) Correlation and enhanced surfact in production with decreased isocitratedehydrogenase activity. Can J Microbiol 35: 854-859.
- Tuelva KT, George R, Cristovaa I, Christovaa NE (2002) Biosurfactant production by a new Pseudomonas putida Strain. Z Naturforsch 57(3- 4): 356-360.
- Martinez Toledo A, Ri'os Leal E, Vazquez Duhalt F, Gonzalez-Chavez, M Del C (2006) Role of phenanthrene in rhamnolipid production by P. putida in different media. Environ Technol 27: 137-142.
- Prabhu Y, Phale PS (2003) Biodegradation of phenanthrene by Pseudomonas sp. Strain PP2: novel metabolic pathway, role of biosurfactant and cell surface hydrophobicity in hydrocarbons assimilation. Appl Microbiol Biotechnol 61: 342-351.
- de Lima CJB, Ribeiro EJ, Servulo EFC, Resende MM, Cardoso VL (2009) Biosurfactant production by Pseudomonas aeruginosa grown in residual Soybean Oil. Appl Biochem Biotechnol 152:156-168.
- Pornsunthorntawee O, Maksung S, Huayyai O, Rujiravanit R, Chavadej S (2009) Biosurfacatnt production by Pseudomonas aeruginosa SP4 using sequencing batch reactors: Effects of oil loading rate and cycle time. Bioresource Technol 100: 812-818.
- Obayori OS, Ilori MO, Adebusoye SA, Oyetibo GO, Omotayo AE, et al. (2009) Degradation of hydrocarbons and biosurfactant production by Pseudomonas sp Strain LP1 World J Microbiol Biotechnol 25: 16151623.
- Rahman KSM, Banat IM, Thahira J, Thayumanavan T, Lakshmanaperumalsamy, P (2002) Bioremediation of gasoline contaminated soil by bacterial consortium with poultry litter, coir pith and rhamnolipid biosurfactant. Bioresource Technol 81: 25-32.
- Lima de Franca IW, Lima AP, Lemos JAM, Lemos CGF, Melo VMM (2015) Batista de San'ana Goncalves, LRB. Production of a biosurfactant by Bacillus subtilis ICA56 aiming bioremediation of impacted soils. Catal Today 255: 10-15.
- Anjum F, Gautam G, Edgard G, Negi S (2016) Biosurfactant production through Bacillus sp. MTCC5877 and multifarious application in food industry. Bioresource Technol 213: 262-269.
- Fernandes PL, Rodrigues EM, Paiva FR, Ayupe BAL, McInerney MJ (2016) Biosurfactan, solvent and polymer production by Bacillus subtilis RI4914 and their application for enhanced oil recovery. Fuel 180: 551-557.
- Chang H, Xing W, Xia T, Fu R, Tao L (2016) Biological characteristics of biosurfactant producing Petroleum degrader bacterium Bacillus BS-8. Agricultural Science and Technology 17(1): 1-3.
- Davis DA, Lynch HC, Varley J (1999) The Production of surfactanin batch culture by Bacillus subtilis ATCC 21332 is strongly influenced by the conditions of nitrogen metabolism. Enzyme Microb Tech 25: 322329.
- Gerson DF, Zajic JE (1978) Surfactant production from hydrocarbons by Corynebacteriumlepus sp N and P asphaltenicus. Deev Ind Microbiol 19: 577-599.
- Lang S, Wullbrandt D (1999) Rhamnoselipids biosynthesis, microbial production and application potential. Appl Microbiol Biot 51(1): 22
- Martinez Toledo A, Rodriguez Vazquez R (2010) Response surface methodology (Box-Behnken) to improve a liquid media formulation to produce biosurfactant and phenanthrene removal by Pseudomonas putida. Ann Microbiol 61: 605-613.
- Guerra Santos L, Kappeli O, Fiechter A (1986) Dependence of pseudomonas aeruginosacontinuous culture biosurfactantproduction on nutritional and environmental factors. Appl Microbiol Biotechnol 24: 443-448.
- Wei Y-H, Chu I-M (1998) Enhancement of Surfactin production in iron- enriched media by Bacillus subtilis ATCC 21332. Enzyme Microb Tech 22(8): 724-728.
- Javaherin M, Jennema GE, Mclnerney MJ, Knapp RM (1985) Anaerobic Production of a Biosurfactant by Bacillus licheniformis JF-2. Appl Environ Biotechnol 50: 690-700.
- Kumar AS, Mody K, andJha B (2007) Evaluation of Biosurfactant/ Bioemulsifier production by a marine bacterium. Bull Environ Contam Toxicol 79: 617-621.
- Kumar MJ, Leon V, Materano ADe S, Ilzins OA, Luis L (2008) Biosurfactant production and hydrocarbon-degradation by halotolerant and thermotoleran Pseudomonas sp World J. Microbiol Biotechnol 24: 1047-1057.
- Makkar RS, Cameotra SS (2002) An update the use of unconventional substrates for biosurfantant products and their new application. Appl Microbiol Biotechnol 58: 428-434.
- Maier RM, Pepper IL, Gerba CP (2000) Environmental microbiology. Academic Press, USA, pp 371-372.
- Mercade ME, Manresa MA (1994) The use of agroindustrial byproducts for biosurfactant production. JAOCS 71(1): 61-64.
- MataSandoval JC, Karns J, Torrents A (2000) Effect of rhamnolipids produced by Pseudomonas aeruginosa UG2 on the solubilization of pesticides. Environ Sci Technol 34: 4923-4930.
- Mata Sandoval JC, Karns J, Torrents A (1999) High performance liquid chromatography method for the characterization of rhamnolipid mixtures produced by Pseudomonas aeruginosa UG2 on corn oil. J Chromatogr A 864: 211-220.
- Sharma D, Ansari MJ, Gupta S, Ghamdi AA, Pruthi P, et al. (2015) Structural characterization and antimicrobial activity of a biosurfactant obtained from Bacillus pumilus DSVP18 on potato peels. Junsdishapur J Microbiol 8(9): e21257.
- Sharma D (2016) Biosurfactant in food. International Publishing Switzerland, Europe.
- Schenk T, Schuphan I, Schmith B (1995) High-performance liquid chromatographic determination of the rhamnolipids produced by Pseudomonas aeuroginosa. J Chromatogr A 693: 7-13.
- Sheppard JD, Cooper DG (1991) The response of Bacillus subtilis ATCC 21332 to manganese during continuous-phase growth. Appl Microbiol Biotechnol 35: 72-76.
- Sheppard JD, Mulligan CN (1987) The production of surfaction by Bacillus subtilis grown on peat hydrolysate. Appl Microbiol Biotechnol 27(2): 110-116.
- Mulligan CN, Mahmourides G, Gibbs BF (1989) The influence e of phosphate metabolism on biosurfactant production by Pseudomonas aeruginosa. J Biotechnol 12: 199-210.
- Mulligan CN, Young RN, Gibbs BF, James S, Bennett HPJ (1999) Metal removal from contaminated soil and sediments by the biosurfactant Surfactin. Environ Sci Technol 32: 3812-3820.
- Van Dike MI, Couture P, Brauer M, Lee H, Trevors JT (1993) Pseudomonas aeruginosa UG2 rhamnolipidbiosurfactants: structural characterization and their use in removing hydrophobic compounds from soil. Can J Microbiol 39(11): 1071-1078.
- Varvaresou A, Iakovou K (2015) Biosurfactants in cosmetics and biopharmaceuticals. Lett ApplMicrobio 61(3): 214-223.
- Khan A, Butt A (2016) Biosurfactants and their Potential Applications for Microbes and Mankind: An Overview. Middle East Journal of business 11(12): 9-18.
- Khire JM (2010) Bacteria biosurfactant, and their role in microbial enhanced oil recovery (MEOR) In Ramkrishna S Biosurfactants, Springer Science + Business Media, LLC Landes Bioscience. West Bengal, India.
- Li J, Deng M, Wang Y, Chen W ( 2016) Production and characteristics of biosurfactant produced by Bacilluspseudomycoides BS6 utilizing soybean wastes. IntBiodeterBiodegr., 112: 72-79.
- Oliveira FS, Vazquez L, De Campos NP, De Franca FP (2006) Biosurfactants production by Pseudomonas aeruginosa FR using Pal oil. Appliochem Bioctech 131(1-3): 727-37.
- Pereira JFB, Gudine EJ, Costa R, Vitorino R, Teixeira JA, et al. (2013) Optimization and characterization of biosurfactant production by Bacillus subtilis isolates towards microbial enhanced oil recovery applications Fuel 111: 259-268.
- Fooladi T, Moazami N, Abdeshahian P, Kadier A, Ghojavand, et al. (2014) Characterization, production and optimization of lipopeptide biosurfactant by new strain Bacillus pumilus 2IR isolated by an Iranian oil field. J Petrol Sci Eng 145: 510-519.
- Guerra Santos L, Kappeli O, Fiechter A (1984) Pseudomonas aeruginosabio surfactant production in continues culture with glucose as carbon source. Appl Microbiol Biotechnol 48(2): 301-305.
- Hartoto L, Mangunwidjaja D (2002) Production of lipopeptide biosurfactant by indigenous isolated of Bacillus sp. BMN 14 in a Batch bioreactor, 2nd World Engineering Congress Sarawak, Malaysia, p. 22
- Itho S, Honda H, Tomita F, Suzuki T (1971) Rhamnolipid produced by Pseudomonas aeruginosa grown on n paraffin (mixture C12, C13 and C14 Fractions). J Antibiot 24(12): 855-859.
- Pruthi V, Cameotra SS (2003) Effect of nutrients on optimal production of biosurfactants by Pseudomonas putida A Gujarat oil field isolate. J Surfactants Deterg 6(1): 65-68.
- Ries RS, Pacheco GJ, Pereira AG, Freire DMG (2013) Biosurfactants: Production and Applications. INTECH.
- Abalos A, Pinazo A, Infante MR, Casals M, Garci'a F (2001) Physicochemical and antimicrobial properties of new rhamnolipids produced by Pseudomonas aeruginosa AT10 from soybean oil refinery wastes. Lagmuir 64: 1367-1371.
- Banat IM (1995) Biosurfactants Productions and possible uses in microbial enhanced oil recovery and oil pollution remediation: a review. Bioresource Technol 51: 1-12.
- Beal R, Betts WB (2000) Role of rhamnolipidbiosurfactants in the uptake and mineralization of hexadecane in Pseudomonas aeruginosa. J Appl Micro biol 89: 158-168.
- Bezza FA, Chirwa EMN (2016) Pyrene biodegradation enhancement potential of lipopeptide biosurfactant produced by PaenibacillusdendritiformisCN5 strain. J Hazardous Materials 321: 218-227.
- Bhat R, KJD Hathwar S, Hegde R, Kush A (2015) Exploration on production of rhamnolipidbiosurfactants using native Pseudomonas aeruginosa strains. J Bio Sci Biotechnol 4(2): 157-166.
- Cortes Camargo E, Perez Rodriguez N, Pinheiro de Souza Oliveira R, Barragan Huerta BR, Dominguez JM (2016) Production of biosurfactant from vine-trimming shoots using the halotolerant strain Bacillus tequilensis ZSB10. Ind Crop Prod 79: 258-266.
- Elazzazy AM, Abdelmoniem TS, Almagharabi OA (2015) Isolation and Characterization of biosurfactant production under extreme environmental conditions by alkali halo thermophilic bacteria by Saudi Arabia. Saudi J Biol Sci 22: 466-475.






























