Studies on Contamination Levels of Chlorinated Trihalomethanes (Thms) Disinfection By-Products (Dbps) In A Community Drinking Water Supply In Hossana Town, Ethiopia
Demis Zelelew1*, Habdelo Esatu2 and Hadush Gebrehiwot1
1College of Natural and Computational Sciences, Wachemo University, Hossaina, Ethiopia
2Hossana water supply and sewerage Autority, Hossana, Ethiopia
Submission: March 05, 2018; Published: April 18, 2018
*Corresponding author: Demis Zelelew, College of Natural and Computational Sciences, Wachemo University, Hossaina, Ethiopia, Email: demisl921zelelew@gmail.com
How to cite this article: Demis Z, Habdelo E, Hadush G. Studies on Contamination Levels of Chlorinated Trihalomethanes (Thms) Disinfection By-Products (Dbps) In A Community Drinking Water Supply In Hossana Town, Ethiopia. Int J Environ Sci Nat Res. 2018; 10(1): 555779. DOI: 10.19080/IJESNR.2018.10.555779
Abstract
The disinfection of water using chlorine is most commonly used practice all over the world because of its low cost, easy to use, efficient against a broad spectrum of microorganisms and enables the maintenance of a residual protection. Disinfection of municipal water using chlorine has a strong drawback, which is the generation of several Disinfection By-products (DBPs). These THMs are formed due to reaction of chlorine with natural organic matter and other precursors found in water. Total Trihalomethanes (THMs) content can vary greatly, depending both on water quality and on treatment conditions, in the same water supply system. This study aims at establishing the concentration range of THMs in drinking water supplies from treatment plant and distribution systems in Hossana town. Concentration levels of trihalomethanes in water were analyzed by solvent Extraction followed by GC- ECD detection. The concentrations of THMs (34.19-135.72^g/l) found to be lower than the prescribed USEPA standards and WHO guidelines. The study also revealed that amongst various THMs, contribution of chloroform was highest followed by other THMs.
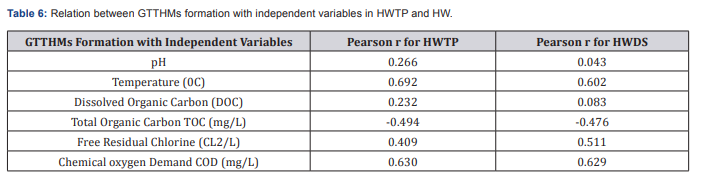
Correlation study delineated that pH, Temperature, TOC, DOC, free residual chlorine and COD are the main organic precursors responsible for the formation of THMs in drinking water. Some independent variables including pH, temperature, TOC, DOC, free residual chlorine and COD were measured by Pearson method to find a relation between THMS formation and these variables. In the case of pH, Pearson method showed a correlation of r = 0.266 and r = 0.043, r = 0.692 and r = 0.602, r = 0.232 and r = 0.083, r = -0.494 and r = -0.476, r = 0.630 and r = 0.629, r =0. 409 and r = 0.511for pH, temperature, DOC, TOC, COD, and free residual chlorine for HWTP and HWDS respectively. Pearson correlation matrix revealed that pH and DOC have weak and TOC have negative moderate correlation with THMs formation but Temperature, COD and FRC are significant and definite correlation with the THMs. A general correlation prediction is in good agreement with the observed results regardless of the fact that it is restricted to this study.
Keywords: Disinfection By-Products; Drinking water; Trihalomethanes; Chlorination; Pearson Correlation Matrix
Introduction
Protection of drinking water quality goes back several hundred years. Scientific and medical advances in the 1800s, along with the need to provide basic sanitation in the rapidly urbanizing cities, laid the foundation for today's drinking water field [1]. There are various methods to make water safe and attractive to the consumer, which depends on the character and source of the raw water (surface water, groundwater). Surface water coagulation/flocculation followed by sedimentation, filtration and disinfection, often done by addition of chlorine, used worldwide in water treatment industry before distribution of the treated water to consumers [2]. Disinfection has been accepted and appreciated as an indispensable process in the treatment of drinking water. This is because it serves as the major barrier against the transmission of water borne diseases, the occurrence of which has been linked to many deaths in developing countries. In 1993 alone cholera outbreak in developing countries was of the magnitude of 950,000 reported cases with more than 9,000 deaths [3]. As a result, disinfection is considered the most important process in drinking water treatment and one of the major public health triumphs of the 20th century. Because of this, microbial safety has taken precedence over associated health risks of disinfecting drinking water in most developing countries [4].
Almost 97.5% of all water found in the oceans, from the remaining fresh water, only 1% is accessible for extraction and use. The current worldwide growth in population and affluence is putting global resources under increasing pressure. Specifically, in most developing countries like Ethiopia, there is a growing public health concern, which directly or indirectly related to contamination of water sources. Due to limited alternatives, surface water either from rivers or from rain fed ponds has become one of the main sources of water supply. Surface waters used for drinking purposes can vary markedly in their organic and inorganic content. High levels of variation occur in a range of water quality parameters such as turbidity, alkalinity, color, and natural organic matter (NOM), algae and microorganisms [5]. Many communities have problems with DBPs in their water that comes from organics present in the water reacting with the disinfectant, which used to reduce the occurrence of waterborne disease. The Organic substances that originate from soil, decaying of vegetable matter or contamination by domestic and industrial wastewater, are commonly present in lake or surface water. They presence not only imparts taste and color to water, but also form organohalodes like THM with chlorine [6].
In the disinfection process in water treatment, chlorine and other disinfectants react with organic matter or its intermediates such as humus, fulvic acids and amides that are dissolved in water. Subsequently, potentially harmful disinfection by-products (DBPs) are produced. Among these, the trihalomethanes (THMs) are the first group of DBPs to be recognized. These groups of compounds have been implicated in liver and kidney defects, central nervous system problems and increased risk of carcinogenicity and mutagenicity as Class B carcinogens (USEPA, 1990). Other studies have linked the THMs to reproductive problems, including low birth weight, miscarriages, prematurity, still birth, intra-uterine growth retardation.
Four of these THMs are being regulated in drinking water. Among these, bromo dichloromethane- CHCl2Br is known to be most toxic followed by dibromochloromethane-CHClBr2 and tribromomethane-CHBr3 while trichloromethane,-CHCl3 is least toxic even though it is the most common and dominant compound of these regulated THMs in drinking water. Apart from these four regulated THMs, others are considered in this study as emerging trihalomethanes disinfection by-products (Emerg THM-DBPs). The concentrations of the THMs and HAAs can be used as indicators of the total loading of all chlorinated disinfections byproducts, which may be found in water. The formation of different DBPs depends on the chemical disinfectant used, the quality of raw water, the disinfectant dose, the pH, the temperature and the time available for reaction (contact time). Regulated DBPs only capture a fraction of all by-products and may miss many others often referred to as emerging DBPs. Moreover, although the use of chlorine may produce high concentrations of chlorination byproducts such as trihalomethanes (THMs) and haloacetic acids (HAAs), many other DBPs can be formed at lower concentrations (i.e., < 0.01 mg/L and often < 0.001 mg/L). The principal route of human exposure to THMs is the consumptions of chlorinated drinking water (Figure 1).
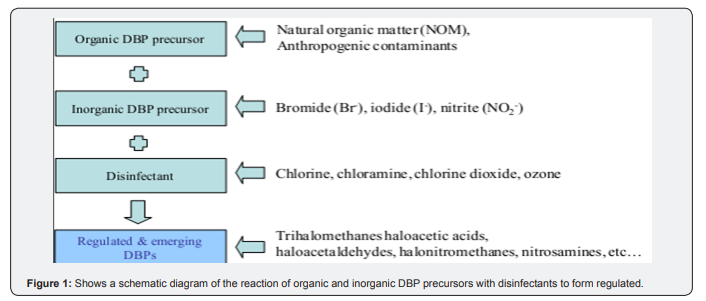
Trihalomethanes are volatile, they vaporize during water usage and as a result, inhalation therefore becomes an important exposure route in addition to ingestion. Upon ingestion THMs are metabolized primarily to carbon dioxide and carbon monoxide and then exhaled. However due to their lipophilicity, THMs accumulate mostly in tissues with highest lipid content in the following order: adipose tissue, brain, kidney and blood. Long-term exposures will eventually result in the negative health effects. Due to the risks posed by these substances, various countries have set limits for THMs [7]. The main goal of this research is to assess the proportion of Chlorinated Trihalomethanes (THMs) Disinfection By-Products (DBPs) in drinking water supply available to the community in view of the use of chlorine based disinfectant in drinking water from the Treatment and Distribution Systems in Hossana town; Ethiopia. And to show the relation between THMs formation and some of independent variables including TOC, residual chlorine, pH, temperature, DOC and COD was be investigated.
Materials And Methods
Description of Study Are
Hossana, the capital city of Hadiya zone of North West Ethiopia, is 235 km south from capital city, Addis Ababa, Located at 7°33'32" N Latitude and 37°51’58" E Longitude, the climate of the city is subtropical, with a mean annual temperature of 17.10C and annual average rainfall ranging from 920.4 mm tO 1436.5 mm. The highest temperature is experienced between January and March and the lowest between July and September. Based on 2007 census results the regional average growth rate of 2.9%, the population of Hossana town was estimated to be 83,046 in 2013.
Sampling Methods
Fifty four (54) samples were being collected in duplicates longitudinally along the treatment and distribution system of the Hossana town Water Plant. These samples was taken from five points from treatment system (HWTP) including: the source (raw) water canal, the sedimentation tanks (before coagulation units), out-let pipe of Hossana water treatment plant after chlorination, at the booster station (downstream of filtration) and at residential house level. Also five locations were selected at different times for sampling from Hossana town water distribution systems (HWDS). In HWDS, before sample collecting, the faucet was turned on for about5 minutes, to ensure that water was coming directly from the public distribution system rather than building's plumbing system. Glass bottles with ground-glass stoppers which was washed with phosphatefree detergent and rinsed with de-ionized water and placed in an oven at 400°C for 1 hr was be selected for THMs sampling.
For sampling, bottles were filling to zero head space to prevent THM volatilization. Sampling was undertaken in the afternoon and before sampling, a sodium thiosulfate solution was added to the bottles to remove residual chlorine and to prevent additional chlorination DBPs formation during transportation to the laboratory. Samples were transported in an ice box to the laboratory. This test lasted for 6 months (November- to April). The samples were collected every 2 weeks. Care was taken to avoid overtopping or spilling of sampled water from the vials. The samples were subsequently stored at temperature of about 40C or less in ice filled jugs in accordance with USEPA method 551.1. In addition, three (3) duplicate samples were taken longitudinally along the treatment and distribution systems. These samples were used for some physico-chemical analysis.
Experimental and Analytical Methods
All the reagents used were of analytical grade. TOC was analyzed by means of a TOC analyzer according to standard method for the examination of water and wastewater was used to determine the DBPs in the sample extracts. Water pH and temperature were measured on site by using a solid selective electrode. Free chlorine was measured by the DPD titrimetric method with a colorimeter according to standard method for the examination of water and wastewater. Samples for physicochemical analysis was first subjected to in situ (at sampling) measurement of their temperature with a thermometer. They were further subjected to chemical analyses for pH, total dissolved solids, total carbon, residual chlorine, Chemical oxygen demand in accordance with standard methods APHA [8]. Due to rapid chemical changes that occur in water samples during transit and storage, certain parameters were measured on site, once the samples were collected. These parameters were residual chlorine, temperature and pH. Other parameters including TOC, DOC and different THM compounds: chloroform, BDCM, DBCM, and bromo form were measured at the lab. The reliability of any analytical measurements is directly coupled to the uncertainties of the sampling process, sample storage, preservation and pretreatment prior to analysis. In organic matter residues analysis basically there are three steps. Under the sample preparation step also there are sub-steps, like homogenisation, extraction, concentration and clean up. Each of them was sequentially done in the experimental work.
Florisil and Anhydrous Sodium Sulphate Activation
Florisil was purified by soxhlet extraction using hexane and activated by baking in the oven at 200°C for 24 hours. The anhydrous sodium sulphate dried in oven for 24 hours at 200°C before use.
Extraction Procedure
Sample (5mL) was transferred into a 100 mL volumetric flask and double distilled n-hexane (3mL) added. Extraction was completed by mechanically shaking the flask for 2 hours, storing for 24 hours and then transferring the organic phase into the vials.
The Cleanup Procedure
To remove some organic macro pollutants and elemental sulphur, which normally interfere in the GC analysis, the extract was poured into a chromatographic column containing activated florisil (7g) and anhydrous sodium sulphate (2g) that had been prewetted with 20mL of double distilled n-hexane. The column was first eluted with 5 mL of double distilled n-hexane collecting the first fraction. The second elution was with 3mL of 10% methanol in hexane. No concentration step was involved as this would result in loss of the volatile halocarbons. The extracts were kept in sample bottles for GC analysis.
Preparation of Composite Standar
10 mL of each trihalomethane standard were taken from individual stock solutions of concentration 1x 10-3 g/mL into a 100mL volumetric flask. The standards were dissolved by shaking and sonicating in hexane and then made up to the mark with hexane to give a solution with a concentration of 1 x l0-4 g/ mL. These stock solutions were diluted and mixed to produce working standards with the following concentrations (Table 1). Sample (100mL) was placed in a round-bottomed flask and dichromate solution (25mL) was added. Concentrated sulphuric acid (75mL) was then added carefully and the mixture will be mixed after each addition. Glass beads were added to the reflux mixture to prevent bumping. After refluxing for 2 hrs, the condenser was cooled and then washed with demonized water (25mL). The refluxed mixture was transferred to a 500mL Erlenmeyer flask, and the reflux flask was washed 4 times with demonized water. The sample was diluted to 350mL and the excess dichromate was titrated with standard ferrous ammonium sulfate. A blank sample consisting of 50mL demonized water and reagents was refluxed in the same manner.
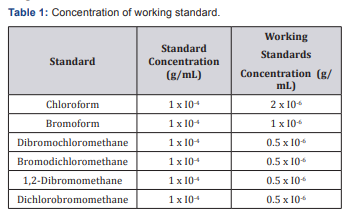
Sodium sulfate (25g) was dissolved in 1 liter of distilled water and this was standardized against potassium dichromate. Water sample (100mL) was placed into a conical flask and an excess of potassium iodide crystal was added to the sample. The mixture was heated whilst stirring to completely dissolve the potassium iodide crystals. The mixture was cooled and titrated with sodium thiosulphate using starch as an indicator. pH was determined using a pH meter. The pH meter was calibrated using pH 4 and 7 buffers. After calibration, the glass pH electrode was immersed into the water samples and the pH was recorded.
Samples for experiment were extracted by means of hexane and were analyzed within 14 days by the liquid-liquid extraction method. Liquid-liquid extraction was performed according to standard method for the examination of water and wastewater. For analyze THMs concentration in the samples, a rapid and simple method by purge and trap coupled with capillary column GC with ECD was used [9]. For extraction of THM compounds, the inert gas was bubbled through the sample and afterward THMs were trapped in a tube that contains sorbent materials. After completion of extraction, sorbent tuber was heated and back flushed with an inert gas to desorb trapped sample components in a GC column. The temperature characteristics of GC set and flow rate of gases for THMs analysis were as follows: Injector temperature: 150°C, detector temperature: 250°C, flow of carrier gas He: 20cm/s, flow of supportive gas N2: 40 cm/s, temperature program: 32°C (5 min), 32-120°C (107 min) and 120°C (10 min). The retention times of the trihalomethanes were determined by running standards. Each trihalo methane was identified in the sample chromatogram by comparing the retention of the suspect peak against that of the standard. The concentration of each trihalo methane was calculated by comparing the peak areas of the samples to those of the standards.
Results And Discussion
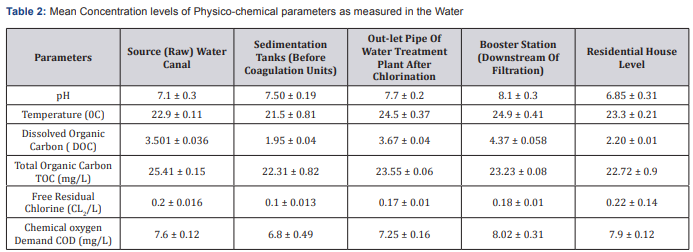
The previous researches demonstrated that the major factors which affect TTHM formation are residual chlorine dose; concentration and type of Natural Organic Material (NOM), contact time, pH and temperature of water. The water quality characteristics measured in raw and treated water during investigation period are presented in (Table 2). The water samples had pH values that were within the neutral pH range of 6.85 and 8.1, which indicated that the pH value has met the drinking water standards and the temperature ranged between21.5 ± 0.81and 24.9 ± 0.410C. The chloride concentrations in the raw water samples were relatively lower in water treatment plants. It ranged from 0.1±0.013 to 0.22±0.14mg/L in samples. WHO recommends that the residual chlorine in drinking water takes 0.6-1.0mg/L as standard .The TOC levels in raw water samples were so high, ranging from 22.31 ± 0.82to 25.41 ± 0.15mg/L. In the case of TOC, THMs formation rose by increasing soluble humic material content in natural water. The rate of THM formation was equal to the TOC consumption. Indeed in the higher available TOC more THM was occurred. The COD ranges from 6.8 ± 0.49 to 8.02 ± 0.31. In treated water, the organic matter was generally low but enough to interact with chlorine to form trihalomethanes. The low values of the chemical oxygen demand in treated water are because of the treatment processes carried out at Works Plant which include coagulation, flocculation and sedimentation.
Samples from the Treatment and Distribution Systems of the Hossana town.
(Table 3) shows the mean values of GTTHM at each sampling station. Generally, the mean concentrations of the trihalomethanes as determined for the water samples were found to vary at different treatment stages. According to this table, the mean value of TTHM in water treated at DBWTP is between 56.4 to 124.67|ig/L and in HWDS respectively. Concentration of Trichloromethane is higher than others this may be due to levels of chloroform, the most common THM, are generally high in chlorinated water originating from surface water, because of higher organic matter present [10], also Chlorine is also more reactive than bromine, hence chlorine substitution is more likely to occur, and thus chloroform is in higher concentration. And there is an increment of levels of THMs formation after flocculating/ coagulation. Since, THMs are as a result of the reaction between organic matter in water and chlorine used for the disinfection, chlorine is consumed whilst the concentration of TTHMs increases (Figure 2).
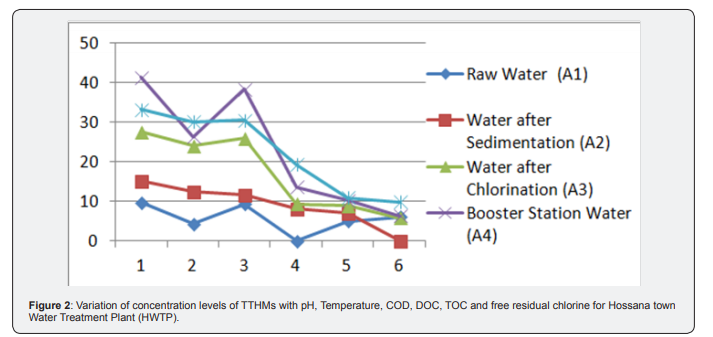
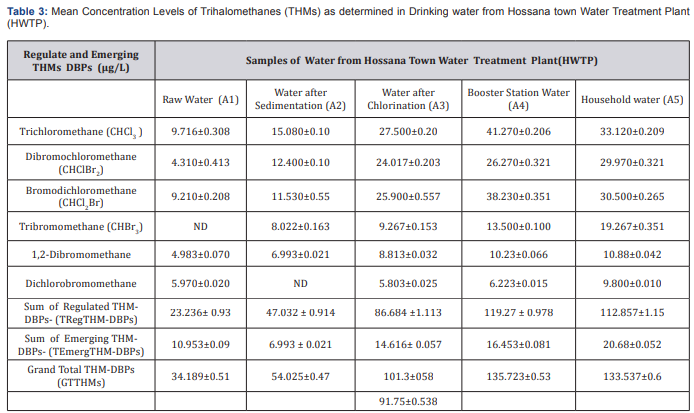
The residence time in the storage tank had a remarkable effect on THMs occurrence [11]. Subsequently, THMs concentration was expected to be higher in the downstream of HWDS. Rather than HWTP outlet pipe. Results showed that lower value of THMs concentration was in distribution system that proved relation between resident time and chlorination DBP formation. But, the results of this study point out that maximum decrease of THMs concentration in DBWDS is after coagulation & flocculation and filtration units. Hence, the low concentrations obtained can be attributable to evaporation of the THMs whilst in the lake levels of THMs increase as the chlorinated water moves from the water treatment plant through the distribution system, because of the continued presence of residual chlorine. According to Abdullaha there is a relationship between level of TTHM and distance from treatment plant in the distribution system. Results of levels of THMs in HWDS this study confirmed a direct relation between THMS compounds and distance of chlorination point (Table 4).
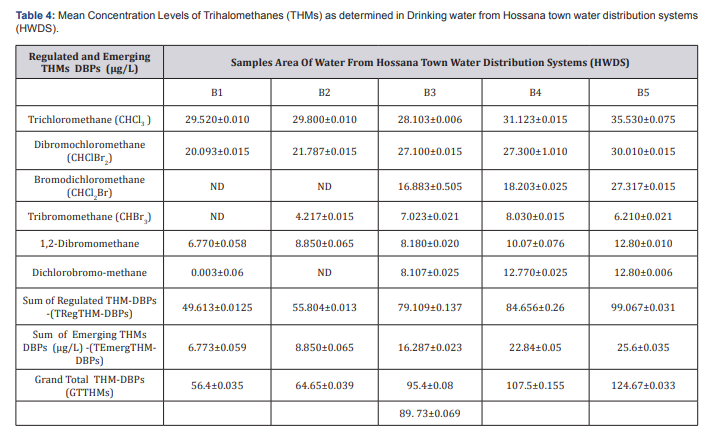
Generally, Water samples collected from the treatment plant had TTHMs concentrations ranging form34.189 to 135.723|ig/L. These concentration levels are higher than those obtained in the residential areas which range from 56.4 to 124.67|ig/L. Hence, the low concentrations obtained can be attributable to evaporation of the THMs whilst in the lake levels of THMs increase as the chlorinated water moves from the water treatment plant through the distribution system, because of the continued presence of residual chlorine. The reaction between chlorine and organic matter is not instantaneous. The formation of trihalomethanes is dependent on the contact time between chlorine and organic matter. Due to this direct relation is because of enough contact time for reaction between chlorine and TOC (Figure 3). Variation of concentration levels of TTHMs with pH, Temperature, COD, DOC, TOC and free residual chlorine for Hossana town water distribution systems (HWDS). Due to this effect, some characteristics of water quality were measured in this research and Pearson correlation coefficient was used to measure the strength of relation between these variables with formation of THMs compounds. The Person Correlation Matrix showing the correlation of various water quality parameters on THM formation for Hossana town Water Treatment Plant and distribution systems is given in below (Table 5).
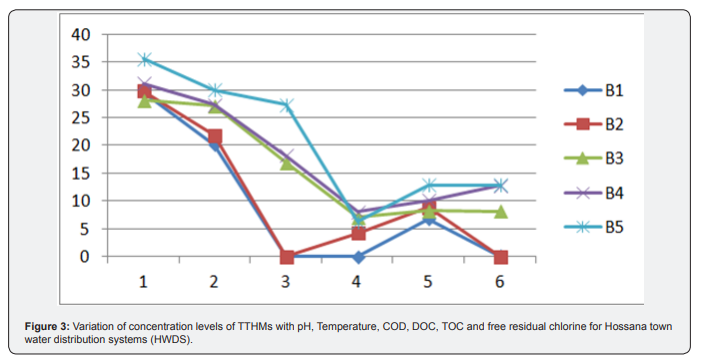
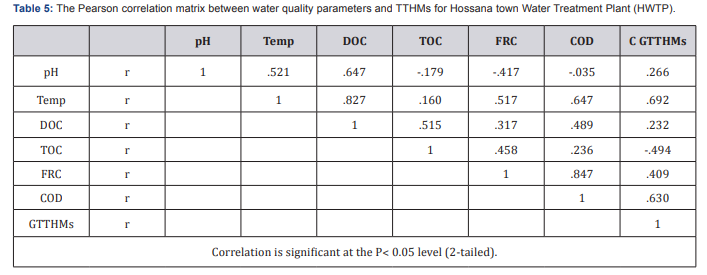
Some independent variables including pH, temperature, dissolved organic carbon (DOC), Total organic Carbon (TOC), free residual chlorine and residual chlorine were measured by Pearson method to find a relation between THMS formation and these variables. In the case of pH, Pearson method showed a lower correlation of r = 0.266 for HWTP and r = 0.043 for HWDS between THM formation and pH. This could be because the treatment process controls the pH within acceptable limits. Also the medium relationship for HWTP was r = 0.692and r = 0.602 for HWDS between THM formation and temperature. Correlation between THM formation and DOC, Pearson method showed r = 0.232 for HWTP and r = 0.083for HWDS and there is a negative moderate correlation between THM formation and TOC (Table 6). The Pearson correlation matrix between water quality parameters and TTHMs for Hossana town water distribution systems (HWDS). Pearson method showed r = -0.494 for HWTP and r = -0.476 for HWDS this may be TOC affects the concentration levels of total trihalomethanes and Possible reasons for negative correlations are including: not all of the organic compounds necessarily result in the formation of disinfection byproducts. TOC does not differentiate between the various chemical compounds that make up the precursor compounds. Pearson correlation method, showed a moderate relationship between THM formation and COD. Indeed the correlation coefficient was r = 0.630for HWTP and r = 0.629 for HWDS. This indicates that the dissolved organic matter is consumed as the THMs are being formed. In the case of residual chlorine, Pearson method showed a relationship of r = 0.409 for HWTP and r = 0.511for HWDS which is moderately correlated. Since THMs are as a result of the reaction between organic matter in water and chlorine used for the disinfection, chlorine is consumed whilst the concentration of TTHMs increases.
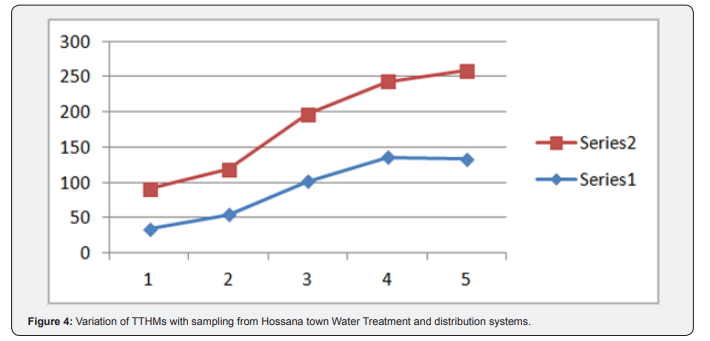
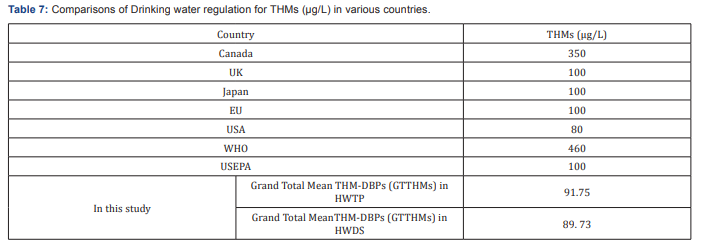
The results was assessed, and compared with national and international standards. The total trihalomethanes concentrations studied are not exceeding the maximum permissible concentration levels of 460|ig/L set by the World Health Organization. In the samples analyzed, none of the exceeded these individual trihalomethane set standards 100|ig/L maximum permissible limits set by the EPA. The above figure shows that the level of trihalomethanes detected depends upon a number of variables. Firstly, the status of the raw water prior to the treatment influence the levels detected (Table 7). The process of coagulation, flocculation, sedimentation and filtration in the water treatment plant do not totally eliminate the dissolved organic matter. Thus, THMs concentration increased in the outlet pipe upon addition of chlorine into treated water. Levels of TTHM can increase while the chlorinated water discharges from water treatment plant through water distribution system, due to continued presences of residual chlorine Golfinopoulos et al. [12] (Figure 4).
Statistical Data Analysis
The data obtained was be recorded, organized and summarized in simple descriptive statistics methods using SPSS- PC statistical package (SPSS version 20 for windows). Results were presented in tables and ANOVA for mean comparison among parameters. Moreover, interpretation of data was be done by the Pearson correlation matrix between water quality parameters and TTHMs for their magnitudes such as concentration of the level of variation of concentration levels of THMs with pH, temperature, Chemical oxygen demand, total dissolved solids, Dissolved Organic Carbon and free residual chlorine [13-26].
Conclusion
The analysis of THMs in drinking water revealed the presence of moderate concentration of THMs. (34.19-135.72|ig/ l) which is lower than the USEPA standards and WHO guidelines. The study depicted that amongst various THMs, the contribution of chloroform was highest followed by DBCM, CBDM & bromo form. The study revealed that pH and DOC have weak and TOC have negative moderate correlation with THMs formation but Temperature, COD and FRC are significant and definite correlation with the THMs this also indicate the predominance of aromatic content of organic matter in the water which are normally not removed from the conventional treatment processes and lead to the formation of THMs in drinking water. A general correlation for the prediction of THMs as a function of these parameters is proposed. Its predictions are in good agreement with the observed results regardless of the fact that it is restricted to this study. Also, there is an increase in concentration levels of trihalomethanes with increasing distance from the chlorination point. Thus the result provide an understanding of the probable health related risks faced by those consuming the treated water and provide empirical basis for the managers of the treatment works to modify their drinking water treatment methods. The knowledge from this study can generate and help water experts and chemists to deliver effective water treatment at micro and macro level especially for Hossana town Water Treatment Plant.
Acknowledgement
The project was financed by Wachemo University and the authors are grateful for the financial support. The authors appreciate Wachemo University research and community service vice president office, and Wachemo University administration for necessary facilities and technical support. We thank the Department of Chemistry and Department of Environmental Science, Addis Ababa University, for the support they offered for detection of THMs and the Hossana water supply and sewerage Authority of Hadiya Zone for providing us with the necessary guidance and resource to conduct the study.
References
- Edzwald JK (2011) Water Quality & Treatment Handbook on Drinking.Water American Water Works Association, (6th edn.); Mc graw-Hill,New York, USA.
- Baruth EE (2005) Water Treatment Plant Design. American Water Works Association and American Society of Civil Engineers (4th edn.); Mc graw-Hill, New York, USA.
- USEPA (1995) Methods for the Determination of Organic Compounds in Drinking Water. Supplement III. EPA/600/R-95/131, Office of Research and Development, Washington, USA.
- Schoeny R (2010) Disinfection by-Products; A Question of balance. Environ Health Perspect 118(11): a466-a467.
- Leeuwen J, Daly R, Holmes M (2005) Modeling The Treatment Of Drinking Water To Maximize Dissolved Organic Matter Removal And Minimize Disinfection By- Product Formation. Elsevier: Desalination 176(1-2): 81-89.
- Trusell R, Umphrfs MD (1978) The formation of THMs J AWWA 70(11): 609-612.
- Jackson P (2000) Trihalomethanes in Drinking Water. Background Document of Development of WHO Guidelines for Drinking Water Quality 82(88): 1-24.
- APHA (1998) Standard Methods for the Examination of Water and Wastewater (20th edn.); American Water Works Association, Water Environment Federation, Washington, USA.
- Abdullaha MP, Yewa CH, Ramlib MS (2003) Formation, modeling and validation of trihalomethanes in Malaysian drinking water: a case study in the districts of Tampin, Negeri Sembilan and SabakBernam, Selangor, Malaysia. Water Research 37(19): 4637-4644.
- Stevens AA (1976) Chlorination of organics in drinking water. J American Water Works Association 68(11): 615-620.
- Rodriguez MJ, Serodes JB, Levallois P (2004) Behavior of trihalomethanes and haloacitic acids in a drink-ing water distribution system. Water Research 38(6): 4367-4382.
- Golfinopoulos SK (2000) The occurrence of trihalomethanes in the drinking water in Greece. Chemo-sphere 41(11): 1761-1767.
- APHA (1998) Standard Methods for the Examination of Water and Wastewater (20th edn.); American Water Works Association, Water Environment Federation, Washington, USA.
- Baghoth S (2012) Characterizing Natural Organic Matter in Drinking Water Treatment Processes. Delft University of Technology, Netherlands.
- Bagwell T, Barrett KM Benjes, HH, Chang Y (2001) Handbook of Public Water Systems (2nd edn.); John Wiley & Sons, New York, USA.
- David P, Gilkey DC (1998) Water Disinfection By-Products: Trihalomethanes and Carcinogenity. Naturopathy Digest 18(18): 855863.
- Ding L (2010) Mechanisms of Competitive Adsorption between Trace Organic Contaminants and Natural Organic Matter on Activated Carbon. Dissertation. University of Illinois at Urbana-Champaign, Urbana, Illinois, USA.
- Kenny C (2010) Optimization of Chemical Dosing in Water Treatment for Enhanced Coagulation/Softening as It Pertains to DBPs Removal. University of Missouri, Columbia, New York, USA.
- Krasner SW (1989) The Occurrence of Disinfection By-Products in US Drinking Water. J American Water Works Association 81(8): 41-53.
- Latifoglu A (2003) Formation of trihalomethanes by the disinfection of drinking water. Indoor Built Environ 12(6): pp. 413-417.
- Morris RD, Audet AM (1992) Chlorination By-Products and Cancer: A Metal Analysis. American Journal of Public Health 82(97): 955-963.
- Munson AE (1982) Toxicology of organic drinking water contaminants: trichloromethane, bromodichloromethane, dibromochloromethane, and tribromomethane. Environmental Health Perspectives 46: 117126.
- Najm I, Patania NL, Jacangelo JG, Krasner SW (1994) Evaluating surrogates for disinfection by-products. AWWA 86(6): 98-106.
- Hodgeson JW, Cohen AL (1990) Method 551.1: Determination of chlorination disinfection byproducts, chlorinated solvents, and halogenated pesticides/herbicides in drinking water by liquid-liquid extraction and gas chromatography with electron-capture detection.
- Pilot LS (1995) Disinfection of Drinking Water, Disinfection ByProducts. Australian Journal of Public Health 19(1): 89-93.
- Vara S (2012) Screening and Evaluation of Innate Coagulants for Water Treatment: International Journal of Energy and Environmental Engineering: Springer Open 3: 29.
- Yan M, Wang D, Qu J (2008) Enhanced coagulation for high alkalinity and micro-polluted water: The third way through coagulant optimization. Science 42(8-9): 2278-2286.






























