Feasibility of Bioethanol Production Potential and Optimization from Selected Lignocellulosic Waste Biomass
Demis Zelelew1*, Hadush Gebrehiwot1, Wondimu Fikre2
1College of Natural and Computational Sciences, Wachemo University, Ethiopia
2Ministry of Mining & Energy of Hadiya Zone, Ethiopia
Submission: March 01, 2018; Published: March 27, 2018
*Corresponding author: Demis Zelelew, College of Natural and Computational Sciences, Wachemo University, Hossaina, Ethiopia, Email:
How to cite this article: Demis Zelelew, Hadush Gebrehiwot, Wondimu Fikre. Feasibility of Bioethanol Production Potential and Optimization from Selected Lignocellulosic Waste Biomass. Int J Environ Sci Nat Res. 2018; 9(3): 555765. DOI: 10.19080/IJESNR.2018.09.555765
Abstract
renewable energy sources that substitute the fossil fuels. Bioconversion of lignocelluloses based biomass to ethanol is significantly hindered by the structural and chemical complexity of biomass, which makes these materials a challenge to be used as feed stocks for cellulosic ethanol production. Lignocelluloses waste has a content of cellulose and hemicelluloses, which make it suitable as fermentation substrate when hydrolyzed. The objective of this work was to evaluate the feasibility of ethanol production and optimization from Lignocelluloses waste by using commercial bakery yeast, i.e., S. cereviciae. To conduct this study, Hydrolysis of lignocelluloses waste was carried out with dilute acid and the optimum condition was examined. Fermentation was held at temperature 30 °C and pH 5, and treated using different acid concentrations and residence times and temperature. Those all three factors were significant variables for the yield of ethanol. The optimization study showed that the highest bioethanol concentration of 4.4, 5.5 and 5.1 g/ml was observed under the optimum conditions of with distilled water hydrolysis for 12 h by keeping boiling temperature with reflux, and hydrolysis temperature of 1200C held at 30 °C with backer yeasts, which is appreciable.
Keywords: Lignocelluloses waste; Bioethanol; Fermentation; Hydrolysis; Optimization
Introduction
As a number of population increases demand for energy increases, while fossil fuel supplies are depleting and oil prices are rising. Reliance on fossil fuel energy has also resulted in pollution of the atmosphere and accelerated global warming. This speeds up the development of renewable fuels like bioethanol. Bioethanol is a vital energy source that contributes to sustainable economic, agricultural and rural development and environmental protection. Most of the developing countries are suffering from what many call the energy crisis, which is characterized by depletion of locally available energy resources and dependence on imported fuel. In the 20th century, the world economy has been dominated by technologies that depend on fossil energy, such as petroleum, coal, or natural gas to produce fuels, chemicals, materials and power [1]. The continued use of fossil fuels to meet the majority of the world's energy demand is threatened by increasing concentration of CO2 in the atmosphere and concerns over global warming [2]. The combustion of fossil fuel is responsible for 73 % of the CO2 emission. Interest in alternative transportation fuels are growing due to oil supply insecurity and its impending peak, and the imperative to lower GHG emissions from fossil fuel use in order to stave off adverse global climatic changes. The main contributors to air pollution are vehicular emissions of GHG and particulate matter. Renewable energies are essential contributors to the energy supply portfolio as they contribute to world energy supply security, reducing dependency of fossil fuel resources, and providing opportunities for reducing emissions of GHG (IEA, 2006). Energy security and climate change imperatives require large scale substitution of petroleum-based fuels as well as improved vehicle efficiency. Replacing petroleum with biofuel can reduce air pollution, improve rural economies by creating job opportunities and raising farm incomes, diversify energy portfolios, minimize dependence on foreign oil and improve trade balances in oil-importing nations. To reduce the net contribution of GHGs to the atmosphere, bioethanol has been recognized as a potential alternative to petroleum derived transportation fuels and cooking fuels [2].
One of the main economic factors in ethanol process is the cost of raw material which can be substantially reduced by utilization of industrial lignocelluloses wastes. However, due to structural complexity, pretreatment is required to disrupt the recalcitrant structure of lignocelluloses materials and to increase the accessibility of hydrolytic enzymes to the carbohydrates polymers. Until now, several types of pretreatment are used, including steam explosion, acid, alkali, organic solvent, alkaline hydrogen peroxide, ammonia and liquid hot water treatments. The choice of pretreatment process is possibly the most important factor in the economics of ethanol production process because it influences waste treatment, cellulose conversion rates and mainly hydrolytic enzymes performance.
Bio-ethanol feedstock's can be divided into three major groups: sucrose-containing feedstock's (e.g. sugar cane, sugar beet, sweet sorghum and fruits), starchy materials (e.g. corn, Milo, wheat, rice, potatoes, cassava, sweet potatoes and barley), and lignocelluloses biomass (e.g. wood, paper mill sludge straw, and grasses). In the short-term, the production of bio-ethanol as a vehicular fuel is almost entirely dependent on starch and sugars from existing food crops [4].The drawback in producing bio-ethanol from sugar or starch is that the feedstock tends to be expensive and demanded by other applications as well. Any bio-ethanol project attacks seven major national issues: sustainability, global climate change, biodegradability, urban air pollution, carbon sequestration, national security, and the farm economy. Lignocelluloses biomass is envisaged to provide a significant portion of the raw materials for bio-ethanol production in the medium and long-term due to its low cost and high availability.
Lignocellulosic biomass appears to be an attractive feedstock for three main reasons, it is a renewable resource that could be sustainably developed in the future, it appears to have formidably positive environmental properties resulting in no net releases of carbon dioxide and very low sulfur content, and it appears to have significant economic potential provided that fossil fuel prices increase in the future. There is a growing interest worldwide to find out new and cheap carbohydrate sources for production of bio-ethanol [5]. Though it may seem beneficial to use renewable plant materials for bio-fuel, the use of crop residues and other biomass for bio-fuels raises many concerns about major environmental problems, including food shortages and serious destruction of vital soil resources. The aim of this work was to study the optimum hydrolysis of selected lignocelluloses biomass with diluted sulfuric acid and distilled water, and determining the influence of acid concentration, temperature and retention times. Also to evaluate the feasibility of bioethanol production by fermentation of lignocelluloses biomass by using commercial bakery yeast such as Saccharomyces cereviciae [6].
Materials and Methods
Description of the Study Area
Hossana, the capital city of Hadiya zone of North West Ethiopia, is 235 km South from Addis Ababa, Located at 7 ° 33' 32" N Latitude and 37 ° 51 ’ 58" E Longitude, the climate of the city is subtropical, with a mean annual temperature of 17.10C and annual average rainfall ranging from 920.4 mm to 1436.5 mm [7]. The highest temperature is experienced between January and March and the lowest between July and September. Based on 2007 census results the regional average growth rate of 2.9 %, the population of Hossana town was estimated to be 83,046 in 2013.
Sampling Technique
For this study, purposive sampling and stratified random sampling in selected three Kebeles of the three sub-cities i.e. Addis reteam, Sichuan and Gofer mead sub-cities, Stratified sampling technique was used considering the variability of the nature of the population. Heterogeneous units were divided into non-overlapping groups to have a representative sampling [8]. The number of samples within each stratum was determined based on respective proportional percentages of each stratum. Sample size of households that participate was determined by using a sample technique. This was done in the three selected Kebeles and the total sample size for each strata or group was determined based on their respective percentage proportion in the town.
Experimental Methods
Sample Preparation: In this study lignocelluloses biomass wastes sawdust, coffee husks, and waste papers was utilized as substrates. The solid particles of substrates were made into particle sizes of 1.5 x 1.0 mm for sawdust, 1.0 x 2.4 mm for coffee husk and 2.0 x 3.0 mm for paper. The lignocelluloses biomass will be oven-dried at 60 °C for 48 h (to moisture content of 15 %), grinded by using grinder and sieved [9]. The sample was stored in hermetically closed plastic containers at room temperature, until required for treatments. Five kilograms of the biomass waste was air dried on the laboratory bench for 3 weeks. 900 g of the dried biomass was ground in order to increase the biomass surface area prior to pretreatment.
Pretreatment of Substrates
Steam Pretreatment: The purpose of pretreatment is to remove lignin, reduce cellulose crystalline, and increases the porosity of the materials. Pretreatment must meet the following requirements: improve the formation of sugar, avoid the degradation or loss of carbohydrate, avoid the formation of by-product inhibitors and must be cost effective. The powders of lignocelluloses biomass were treated inside autoclave; every different sample was treated separately [10]. Steam pretreatment uses steam at 1210C temperatures. Flow through processes steam at temperature of 1210C through the hemicelluloses and cellulosic material. First, the lignocelluloses biomass powders was treated and it feed as batches, every batch contains 100 g (equal proportion, 33.3g) of screened lignocelluloses biomass powder with 10:1(v/w) ratio of water to the sample. The temperatures were applied at 1210C; then released the pressure until the pressure became 0 bars [11]. The retention time for every batch was 15 min. Finally the samples were kept in autoclave for the given pretreatment time and temperature and allowed to cool.
Determination of Moisture and Volatile Matter Content
5.00g of sawdust, coffee husk and waste paper was separately weighed using the analytical balance and put separately into two dry beakers. The biomass was dried to constant mass in an air-oven at a temperature of 10 60C. It was cooled in desiccators and weighed again after one hour; the desiccants used were anhydrous calcium chloride. Constant mass were considered attained when successive heating for one hour period showed a difference of not more than 5 mg in the net loss in mass [12].
Hydrolysis
Dilute Acid Hydrolysis: The carbohydrate polymers in lignocelluloses materials need to be converted to simple sugars before fermentation, through a process called hydrolysis. Various methods for the hydrolysis of lignocellulosic materials have recently been described. The most commonly applied methods can be classified in two groups: chemical hydrolysis and enzymatic hydrolysis. Even though there are many types of hydrolysis types, dilute acid hydrolysis is an easy and productive process and the amount of alcohol produced in case of acid hydrolysis is more than that of alkaline hydrolysis. This process is conducted under high temperature and pressure, and has a reaction time in the range of seconds or minutes, which facilitates continuous processing [13]. The lignocellulosic biomass was hydrolyzed with dilute sulfuric acid (H2SO4) at different concentrations (1 to 5 % H2SO4). In order to break down the cellulose and hemicelluloses into simple sugar the ground lignocellulosic biomass (i.e. sawdust, coffee husks, and waste papers) samples was maintained at solid to liquid ratio of 1:10, in 250 mL round bottom flask, and refluxed, retaining samples of 15, 30, 40, 50 and 60 min for subsequent fermentation experiments. Similarly, the hydrolysis experiment was repeated with distilled water without using dilute sulfuric acid. After hydrolysis the liquid fraction of the hydrolysate samples was cooled, filtered, collected, and their sugar composition determined by Fehling method [14,15]. The distilled water and dilute sulfuric acid hydrolysate was adjusted to pH 5 by adding concentrated sulfuric acid and 2N Sodium hydroxide, and the solutions were prepared for fermentation.
pH Determination (adjustment)
Before addition of any microorganism to the diluted hydrolyzed sample, pH of these samples had to be adjusted. Otherwise the microorganism was dying in hyper acidic or basic state. The pH meter Jenway type 3071(CE UK) was used to determine the pH. The acid-sugar mixture pH was adjusted from the original pH of 1.46 to pH 3.94 using 360 cm3 of lime or NaOH solution prior to fermentation. The pH was raised because the ideal pH for yeast (Saccharomyces cerevisiae) is around 4 to 5 [16].
Fermentation
The yeast S. cereviciae, purchased from local market was used in these experiments. After hydrolysis, the flasks containing the hydrolyzed samples was covered with cotton wool, wrapped in aluminum foil, autoclaved for 15 minutes at 121 °C and allowed to cool at room temperature [17]. Fermentation was carried out in 250 mL Erlenmeyer flask with 3 g/L of yeast (S. cereviciae) at incubation temperature of 300C. Ethanol concentration was analyzed by Dichromate method at different fermentation times (06 to 50 hr). Samples was withdrawn every 6 hr and the fermentation was carried out for 50 hr.
Fractional Distillation Separation Process
A distillation system was used to separate the bioethanol from water in the liquid mixture. All distillation experiments were carried out at a temperature of 850C and a distillation time of 3 hours by rotary evaporator. Distillation was the last step in the production of ethanol from lignocellulosic biomass (i.e. sawdust, coffee husks, and waste papers) experiments. Distillation is the method used to separate two liquid based on their different boiling points. However, to achieve high purification, several distillations are required. In this experiment separation were conducted by rotary evaporator at a temperature of 850C for 3hrs. The decanted solution from the mash was poured in a 250 ml round bottomed flask placed in a mantle [18-20]. A fractionating column was connected to the flask. The boiling point of absolute ethanol at standard pressure is 78.30C. The heating mantle temperature was set at 750C for the fractional distillation of ethanol from the mash. Since ethanol-water azeotrope (95 %) has a lower boiling point compared to that of water, the azeotrope turned into the vapour state before water, it condensed and was successfully separated. Samples were withdrawn regularly for analysis at 48hr intervals. All the fermented solutions were centrifuged at 10,000 rpm for 5 minutes to separate supernatant. After centrifugation, the supernatant was filtered and then analyzed for ethanol concentration by Dichromate method
Determination of Sugar Content
The amount of sugar in the hydrolyzed samples was determined by Fehling method. 50 mL of hydrolyzed sample solution was dissolved in 10 mL of distilled water and 2 mL of concentrated HCl were added and boiled. The obtained sample were neutralized with NaOH and the solution will be made up to a volume of 300 mL and taken into the burette. The 5 mL of Fehling A and 5 ml of Fehling was taken and mixed with 90 mL of distilled water in 250 mL Erlenmeyer flask and Methylene blue indicator was added [21-23]. The solution in the flask was titrated with burette solution in boiling conditions until disappearance of blue color and the volume at which brick red color observed were recorded. For each sample the sugar content was calculated by using the formula given below.
Sugar Content ( %) = (300mlxf / V) x100
Where, f is Fehling factor and V is volume used in the titration.
Ethanol Concentration by Dichromate Method
Identification of Bioethanol: About 5ml fermented sample was taken and pinch of potassium dichromate and a few drops of H2SO4 were added. Color change from pink to green indicated the presence of bioethanol.
Determination of Ethanol Concentration: The ethanol concentration of the samples collected every 3 hours interval by rotary evaporator of fermented solution was measured by alcoholmeter. An alcoholmeter is a hydrometer which is used for determining the alcoholic strength of liquids. It only measures the density of the fluid. Alcohol meters have scales marked with volume percent of potential alcohol, based on a pre-calculated specific gravity.
Results and Discussion
The process consists of four parts: pretreatment to remove lignin, reduce cellulose crystallinity, sterilize the lignocellulosic waste biomass and increase the porosity of the materials, dilute acid hydrolysis and fermentation to produce ethanol, distillation to remove the ethanol.
The Effect of Acid Concentration on Sugar Content
In this section, the results of the experiment carried out on lignocellulosic waste biomass for bioethanol potential through distilled water and acid hydrolysis, the effect of acid concentration on the amount of glucose formed was investigated and discussed here under. The sugar content of the lignocellulosic waste biomass is presented in Table 1 below.
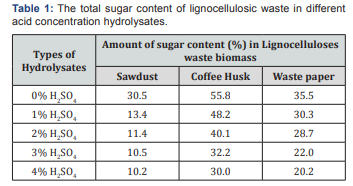
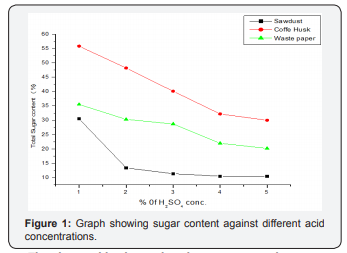
The above table shows that the maximum reducing sugar concentration of 30.5, 55.8 and 35.5 % was produced from distilled water hydrolysate of the selected lignocellulosic waste followed by the production of decreasing of sugar % from 1,2,3 and 4 % dilute sulfuric acid treated hydrolysate, respectively. This shows that distilled water hydrolysis is more effective in simple sugar production than dilute sulfuric acid of different concentrations (1, 2, 3 and 4 %) hydrolysis. The result showed that the amount of sugar obtained decreases as the acid concentration increases and reaches maximum in acid free hydrolysis [24-27]. The decrease of sugar content in acid treated samples with increasing of acid concentration may be due to degradation of monomeric sugars (xylose, glucose) to furfural and HMF or may be derived from dehydrating or oxidizing by sulfuric acid on glucose or it could be attributed from the conversion of glucose to levulinic and formic acid which leads to decrease in glucose yield. These substances are toxic substances for yeast and can inhibit the yeast growth (Figure 1).
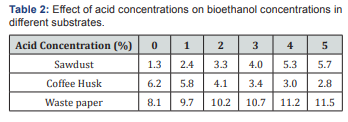
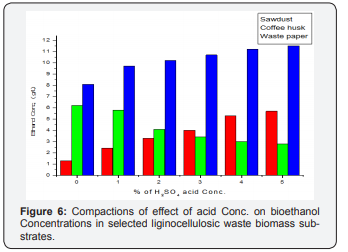
Optimization of acid concentration for maximum amount of bioethanol production
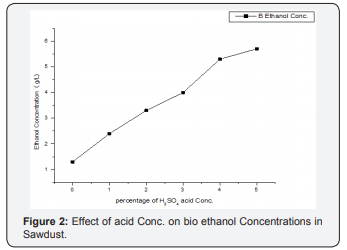

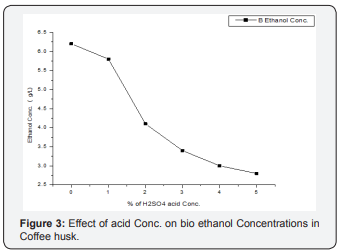
The experiment was conducted for 24 h hydrolysis, using acid concentrations (0, 1, 2, 3, 4 and 5 % H2SO4) and 48 h fermentation, as shown in Figures 2-5 (Table 2). The effect of acid concentration on the production of ethanol was carried out 24 h hydrolysis time is shown in Figures. It was found that with the increasing the percentage of acid the concentration of ethanol was decreases in coffee husk substrate. The maximum concentration ethanol was 6.2 g/ml was obtained when acid percentage was zero hydrolysis time 24 h and fermentation time 48 h respectively [28]. After that ethanol concentration 5.8, 4.1, 3.4, 3.0, 2.8 g/ml decreases with increase in acid 1, 2, 3, 4, 5 % (percentage). This condition is similar with the work of Negusu Teheran where maximum amount of ethanol from distilled water hydrolysate than acid hydrolysate of Prosopisjuliflora. This decrease in bioethanol concentration may account for the further sugar degradation that occurred under the severe acidity. Overall, these results indicate that extreme acidity had an unfavorable effect on sugar conversion of coffee pulp. But there is an increment of ethanol concentration production may bedew to degradation of nonnumeric sugars (xylems, glucose) to furfural and HMF or may be derived from dehydrating or oxidizing by sulfuric acid on glucose or it could be attributed from the conversion of glucose to levulinic and formic acid which leads to decrease in glucose yield.

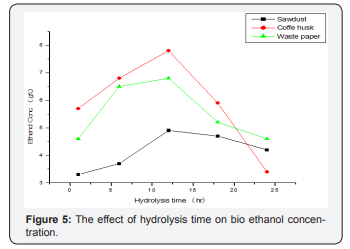
Optimization of Hydrolysis Time for Maximum Amount of Bioethanol Production
Based on the acid optimization for bioethanol production, distilled water hydrolysis was selected as an optimum condition and employed for the second series of the experiment. The effect of hydrolysis times (1,6,12,18 and 24h) on bioethanol yield was investigated under the constant conditions of distilled water and 48 h fermentation (Figure 6). The effect of hydrolysis time of selected lignocellulosic waste feedstock ’s on the bioethanol yield was significantly different from that of the acid concentration. Slightly prolonging the hydrolysis time significantly increased bioethanol concentration and then started to decline after 12 h hydrolysis. The effect of hydrolysis on ethanol concentration was carried out zero percentage of acid at 48h fermentation time, which is shown in Figure 6 (Table 3).
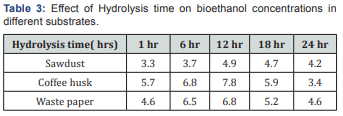
It maximum ethanol concentration 4.9, 7.8 and 6.8 g/ml of ethanol from sawdust, coffee husk and waste paper respectively as found at 12h hydrolysis time. When hydrolysis times are 1 and 6 h the ethanol concentration was observed an increment in ethanol production. Further increase in hydrolysis times 18, and 24 h the bioethanol concentration was show a decrement in concentration [29]. This attribute to that longer residence time makes the sugars degraded to form inhibitors (furfural and HMF). Therefore, distilled water hydrolysis and 12h residence time were selected as the optimum conditions for hydrolysis of selected lignocellulosic waste feed stocks for bioethanol production (Figure 6).
Optimizationof Hydrolysis Temperature for Maximum Amount of Bioethanol Production
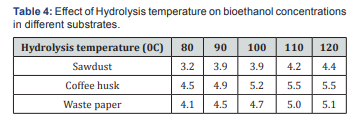
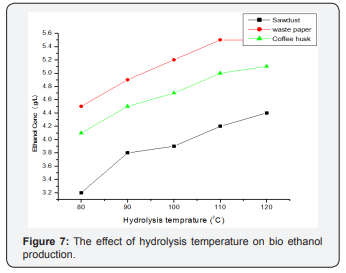
The effect of hydrolysis temperature on bioethanol concentration was carried out at 12h hydrolysis time, which shown in Fig below. It was observed that 1200C is the optimum temperature for maximum yield of 4.4, 5.5 and 5.1 g/ml for sawdust, coffee husk and waste paper respectively of bioethanol concentration (Table 4) (Figure 7). This increasing in ethanol concentration with increase in hydrolysis temperatures might be due to the consumption of sugar by the microorganisms for ethanol production or the hydrolyzed does contain significant levels of metabolic inhibitors (e.g., furfural and HMF) that can interfere with fermentation [30-32].
Conclusion
Lignocelluloses waste biomass is promising feed stocks for bioethanol production. One of the most important factors in the acid treatment of lignocelluloses is the determination of optimal conditions required to provide the maximum yield of fermentable sugars. In this study, the feasibility of ethanol production from Lignocelluloses waste biomass by means of dilute acid and distilled water hydrolysis techniques and ethanol fermentation time by S. cerevisiae was investigated. Dilute acid and distilled water hydrolysis was applied to produce simple sugars from lignocelluloses waste biomass which followed by fermentation for production of bioethanol. The bioethanol production from lignocelluloses waste biomass and optimization test have shown that distilled water is preferable than dilute acid hydrolysis. Hydrolysis of lignocelluloses waste was carried out with dilute acid and the optimum condition was examined. Those all three factors were significant variables for the yield of ethanol. The optimization study showed that the highest bioethanol concentration of 4.4, 5.5 and 5.1 g/ml was observed under the optimum conditions of with distilled water hydrolysis for 12 h by keeping boiling temperature with reflux, and hydrolysis temperature of 1200C held at 30 °C with backer yeasts, which is appreciable. Based on these facts, lignocellulosic waste which is an agricultural waste is a promising alternative feedstock for bioethanol production. The use of this waste as an alternative can also reduces the environmental impacts arising from dumping of the waste directly to the nearby rivers, burning in the atmosphere and could also contribute to the solution of fossil fuel replacement in Ethiopia. Very high and low acid concentration, low temperature and Very high and low retention time have negative effect on the yield of ethanol. The plant uses raw material from the municipality waste. Thus it brings no wastes that are dangerous to the environment and health. The by-product is also biodegradable and we can use it for the production of fertilizer. The waste also generates financial revenue in addition to protect the environment from disposed lignocellulosic waste biomass in open yard landfill areas.
References
- Cardona C, Sanchez O (2007) Fuel ethanol production process design trends and integration opportunities. Biores Technol 98(12): 24152457.
- Chandel A, Rudravaram R, Narasu M, Rao L, Ravindra P (2007) Economics and environmental impact of bioethanol production technologies an appraisal. Biotechnol Molec Biol Rev 2: 14-32.
- Demirbas F, Bozbas K, Balat M (2004) Carbon dioxide emission trends and environmental problems in Turkey Energy Explor. Exploit 22(5): 355-365.
- Demirbas A (2006) Hazardous emissions, global climate change and environmental precautions Energy Sources 1: 75-84.
- Dawson L, Boopaty R (2008) Cellulosic Ethanol production from sugarcane bagase without enzymatic saccharification. Bioresb 3: 452460.
- Farrell A, Plevin R, Turner B, Jones A, O Hare M et al. (2006) Ethanol can contribute to energy and environmental goals. Science 311: 506508.
- Gaur K (2006) Process optimization for the production of ethanol via fermentation. Master thesis Thapar Institute of Engineering and Technology Patiala.
- Govindaswamy S, Vane L (2007) Kinetics of growth and ethanol production on differentcarbon substrates using genetically engineered xylose fermenting yeast Biores. Technol 98(3): 677-685.
- Galbe M, Zacchi G (2002) A review of the production of ethanol from softwood. Appl Biochem Biotechnol 59: 618-628.
- NegusuTefera (2009) Potential of ProsopisJuliflora for Bioethanol Production. Master thesis Addis Ababa University Ethiopia.
- Malca J, Freire F (2006) Renewability and life-cycle energy efficiency of bioethanol andbio ethyl tertiary butyl ether (bioETBE) assessing the implications of allocation. Energy 31: 3362-3380.
- Hahn Hagerdal, B Galbe, M Gorwa Grauslund, M Liden, G Zacchi (2006) Bioethanol the fuel of tomorrow from the residues of today Tre. Biotechnol 24(12): 549-556.
- Lombardi L (2003) Life cycle assessment comparison of technical solutions for CO2 emissions reduction in power generation. Energy Convers Manag 44(1): 93-108.
- Lynd L, Lyford K, South C, van Walsum P, Levenson K (2001) Evaluation of papersludge for amenability to enzymatic hydrolysis and conversion to ethanol. TAPPI J 84: 50-55.
- Kim S, Dale B (2004) Global potential bioethanol production from wasted crops and crop residues.Biomass Bioenergy 26(4): 361-375.
- Malca J, Freire F (2006) Renewability and life cycle energy efficiency of bioethanol and bio ethyl tertiary butyl ether (bioETBE) assessing the implications of allocation Energy 31: 3362-3380.
- Ministry of Finance and Economic Development (MoFED) (2010) Ethiopia: Country Report on the Implementation of the Brussels Program of Action (BPOA) Addis Ababa.
- Sun Y, Cheng J (2002) Hydrolysis of lignocellulosic materials for ethanol production areview Biores. Technol 83(1): 1-11.
- Subramanian K, Singal S, Saxena M, Singhal S (2005) Utilization of liquid biofuels in automotive diesel engines An Indian perspective. Biomass Bioenergy 29: 65-72.
- Paster M (2003) Industrial bioproduct Today and tomorrow US Department of Energy, Office of Energy Efficiency and Renewable Energy, Office of the Biomass Program Washington DC.
- Wildenborg T, Lokhorst A (2005) Introduction on CO2 Geological storage classificationof storage options. Oil Gas Sci Technol Rev 60: 513-515.
- Paster M (2003) Industrial bioproduct today and tomorrow US Department of Energy Office of Energy Efficiency and Renewable Energy. Office of the Biomass Program, Washington, USA.
- Subramanian K, Singal S, Saxena M, Singhal S (2005) Utilization of liquid biofuels in automotive diesel engines An Indian perspective. Biomass Bioenergy 29: 65-72.
- Shapouri H, Salassi M, Nelson J (2006) The economic feasibility of ethanol production from sugar in the United States. Department of Agriculture (USDA), Washington, USA.
- Linon N, Kumar V, Dhavala P, Goswami A, Maithel S (2006) Liquid biofuels in South Asia resources and technologies. Asian Biotechnol Develop Rev 8: 31-49.
- Ohgrem K, Bura R, Lesnicki G (2007) A comparison between simultaneous saccharificationand fermentation and separate hydrolysis and fermentation using steam pretreated corn stover. Process Biochem 42(5): 834-839.
- Oliveria M, Vaughan B, Rykiel J (2005) Ethanol as fuel energy carbon dioxide balances and ecological footprint. BioScience 55: 593-602.
- Olfert R, Weseen S (2007) Assessing the Viability of an Ethanol Industry in Saskatchewan. The Saskatchewan Institute of Public Policy, p. 48.
- Taherzadeh M, Karimi K (2007) Acid based hydrolysis processes for ethanol from lignocellulosic materials A Review. BioRes 2: 472-499.
- Thuesombat P, Thanonkeo P, Laopaiboon L, Laopaiboo P, Yunchalard (2007) The batch ethanol fermentation of jerusalem artichoke using S cerevisiae. J sci Technol 7: 93-96.
- Uma V, Polasa H (2000) cerevisiae of palm wine enhanced ethanol production by using mutagens. J Indust Microbiol Biotechnol 5: 1-4.
- Wyman C, Dale B, Elander R, Holtzapple M, Ladisch M (2005) Coordinated development of leading pretreatment technologies .Biores Technol 96: 1959-1966.






























