Assessment of Ground Water Quality in Tiruvannamalai District-Random Study in Periyakallapadi Village
B Sundararaman1* and K Muthuramu2
1SKP Engineering College, Tamilnadu, India
2Principal, Shanmuganathan Engineering College, India
Submission: February 18, 2018; Published: March 12, 2018
*Corresponding author: B Sundararaman, SKP Engineering College, Tamilnadu, India; Email: sundararaman.b@gmail.com
How to cite this article: B Sundararaman, K Muthuramu . Assessment of Ground Water Quality in Tiruvannamalai District-Random Study in Periyakallapadi Village. Int J Environ Sci Nat Res. 2018; 9(2): 555758. DOI: 10.19080/IJESNR.2018.09.555758
Abstract
Innovative methods of cultivation of food crops, unplanned rapid growth in urbanization to cater the needs of the growing population, the chemical process either by effluent from industries or by the application of chemical pesticide and manures to the plant have created negative impact on the ground water aquifer. Random samples were collected from open wells in periyakallapadi village. Where the contamination of ground water is largely reported and analysis done. The physico-chemical parameter which contaminate the ground water such as total dissolved solid (TDS), Electrical conductivity(EC), pH, total hardness (Na+), SO4, NO3, Cl- have been analyzed by standard method. The study reveals that the quality of ground water is not suitable both for irrigation and drinking, as most of the values are beyond the acceptable limits prescribed by WHO and irrigation standards.
Keywords: Water Quality; Water; Hydro Geological
Abbreviations: TDS: Total Dissolved Solid; EC: Electrical conductivity; SAR: Sodium Adsorption Ratio; TH: Total Hardness
Introduction
Water is the basic requirement for any development. It is a source for not only drinking and irrigation, but also for various other sectors like Industrial and power generation. India is a developing country. The WHO report states that 80% of diseases are caused due to poor water quality [1]. The pollution of ground water is mostly from the industries and by the non- treatment of the effluent. There are other areas where the ground water is polluted. Agriculture soil lost its fertility without any external influence factors such as industries or sewage. Depending upon the general geology, degree of chemical weathering of rocks, quality of recharged water and source input other than water - rock interaction, the water quality is differentiated [2]. Several reports states agricultural activities and urban development affect the groundwater quality directly or indirectly and also by fertilizers, pesticides, industrial growth [3]. In ground water, major problem is that once it is contaminated, it is very difficult to restore it Jothivenkatachalam [4].
Quality of water is getting changed by interaction between soil and water, soil-gas, rocks, residence time and reactions that take place within the aquifer [5,6] Fetter, Appelo, Postma. The chemical analysis of water quality was studied in Sanganer area, Jaipur to observe whether the water is suitable for drinking and for irrigation [7]. Quality of ground water varies from place to place, depth of water table, from season to season and by dissolved solids. The decreasing trend in availability of fresh water resources poses a danger to human population, plants and animals. So, the quality of the water has to be maintained at all cost for sustainability of human population and environment. Physico-chemical characteristics of water is needed to assess the suitability of water for drinking and irrigation. The objective of this paper is to study the hydro geo- chemical characteristic of ground water analysis for drinking and for irrigation purpose. For drinking water suitability, the experiments such as pH, electrical conductivity (EC), total dissolved solids (TDS), Ca++, Mg++, Na, Cl-, SO4, NO3, and for irrigation (pH), EC, sodium adsorption ratio (SAR), and Total Hardness (TH) along with the effects of specific ions need to be studied [8]. For irrigation, based on amounts of salts present in the water and their effects on crop growth and development. The qualities differs with salt concentrations at higher level influence of osmotic pressure of the soil solution and affect the ability of plants to absorb water through their roots [9].
Background Data
The district is divided into seven taluks Arani, Chengam, Cheyyar, Polur, and Thandrampet. Physiographical, the district formed an undulating terrain dotted with cluster of hillocks,particularly in western and north-western parts. Jawadhu hills are the major hilly regions and other smaller hillocks are scattered in Tiruvannamalai, Chengam, Polur, Arani, and Kalasapakam blocks. The area under forest was 1,53,318ha. The important hills in the district are jawadhu and gouthy hills. There are no perennial rivers in the districts. The district was mostly drained by Cheyyar River, a major tributary of river palar, originating from the forest area of jawadhu hills and traveling through Chengam, Polur, Arani and Cherry taluks before. It empties into river Cheyyar. Tanks and dug well were the major sources of irrigation in the district. There were 1,050 private bore wells, 200 dug-cum bore wells and 1,54,415 open well in the district. Sathanur reservoir is built across the Thenpennai River with an ayacut of 18,882ha. Agriculture is the main occupation in the district. The Gross and Net cultivated areas are 3,04,929 and 2,42,387ha. The cropping intensity was 126 percent and the ratio of net sown area to cultivable area 0.87% (indicating extent of use of cultivable area) Paddy, sugarcane and groundnut were the major crops grown in the district.
Materials and Methods
Sampling and Methodology
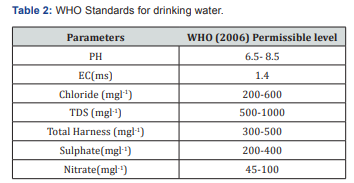
Water samples were collected in four open wells in Periyakallapadi village, Tiruvannamalai. The samples were drawn in sterilized bottles and analyzed immediately for physicochemical characteristics. PH, electrical conductivity (EC), total dissolved solids (TDS), Ca++ and Mg++, Total Hardness (TH), Sodium (Na) and Sulphate (SO4), Nitrate (NO3) was determined by standards procedure within 48 hrs. Of collection and sodium absorption ratio were calculated (Figure 1).
Result and Discussion
PH Suitability for Drinking and Irrigation Purpose
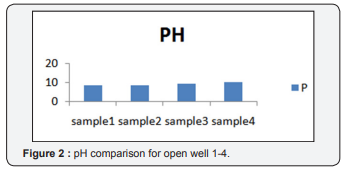
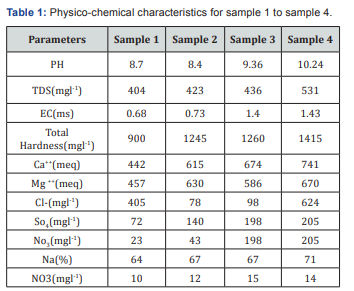
pH is determined from pH meter. From the Figure 2 it depicts the value 8.7,8.4,9.36,10.24. Out of four samples, fourth sample occupies the highest range of value 10.24. It clearly indicates that the value is quite higher than the standards for drinking and for Irrigation.Suitability for drinking and irrigation purpose are not attained from the well 3 and well 4.
TDS Suitability for Drinking and Irrigation Purpose
TDS was analysed by standard method. The value for sample 1-4 contain 35, 130, 250 and 261(mgl-1)respectively. Out of which sample 3 and sample 4 attained the highest TDS value by comparing with other two samples (Figure 3).

Evaluation of Ec Suitability for Drinking and Irrigation Purpose
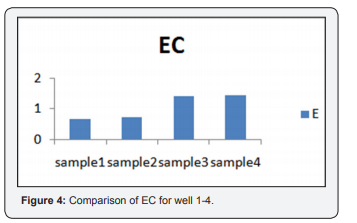
1.4 and sample 4 is 1.43. These experiments indicate ionic concnetration and its dependency on concentration, temperature and types of ions present (Figure 4).
Evaluation of Hardness Suitability for Drinking and Irrigation Purpose
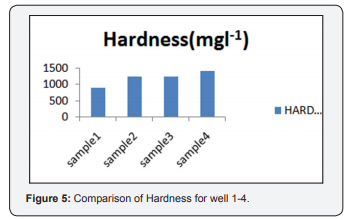
Figure 5 depicts Hardness of the water and it is measured by EDTA method. The sample1 contains 900mgl-1,sample 2 contains 1245mgl-1, sample 3 contains 1260(mgl-1) and for sample 4 contains 1415(mgl-1). Hence all the sample values are obtained above the permissible standards based on WHO and also irrigiation standards, All the four water samples are not suitable for drinking and for irrigation purposes.Hardness is due to interaction between water and geological formation [10].
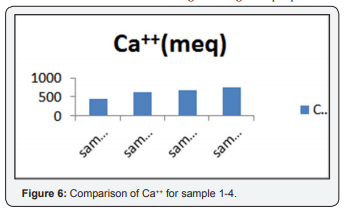
Evaluation of Ca++ Suitability for Drinking and Irrigation Purpose
Figure 6 shows the ca++ interaction in water. The value of ca++ sample(1,2,3,4,) was 442, 615, 674, 741 (meq). It shows the higher ions involved to cause hardness of water. So, it depicts that water is doubtful to use for drinking and irrigation purposes.
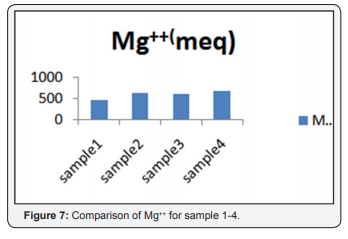
The value of mg++ in sample (1,2,3,4) was 457, 630, 586, 670 (meq). Based on heavy interaction of ions, water cannot be used for both driniking and for irrigation. The value shown in Table 1 and Figure 7.
Evaluation of Sulphate Suitability for Drinking and Irrigation Purpose
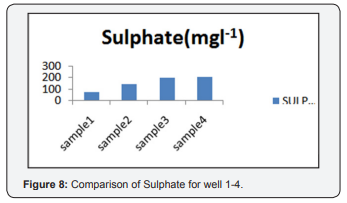
For sulphate sample contains 72(mgl-1), for sample 2 contains 140(mgl-1), sample 3 contains 198(mgl-1) and for sample 4 which contains 205(mgl-1). Sulphate is precipitated in hydrochloric acid medium with BaCl2 to form BaSO4. The turbidity of BaSO4 suspension is measured in Turbidity meter. All the value depicted in Table 1 shown it's below than the permissible limit, but it's nearer to the permissible value (Figure 8).
Evaluation of SAR Suitability for Drinking and Irrigation Purpose
For irrigation the quality of water based on dissolved salts and if salt concentration is high it form saline and leads to alkaline soil. Sodium Absorption Ratio (SAR) which influence the water quality and its suitability for irrigation. SAR is to calculate degree to which irrigation water tends to enter into cation exchange section in the soil. The main problem with high sodium concentration is its effect on soil permeability. Based on the result shows the water is not fit for drinking and for irrigation purpose (Tables 2 & 3).
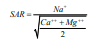
Evaluation of Sodium Percentage Suitability for Drinking and Irrigation Purpose
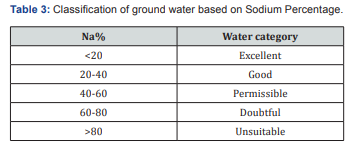
Sodium concentration can reduce the soil permeability and soil structure and this help little or no plant growth. Excess sodium in water leads to change in soil property and reducing soil permeability and affects soil nutrient up taking capacity Palanisamy [11]. Because of the above reason sodium play important factor for determining groundwater suitability for irrigation purposes. Presents of sodium is usually expressed in terms of sodium percentage. The entire four sample attained in the range of 64% to 71%. It indicates in doubtful zone based on the standards [12-14].
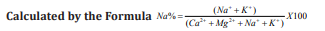
Evaluation of Nitrate Suitability for Drinking and Irrigation Purpose
For nitrate, Presence of nitrate indicates organic pollution of biological decomposition of nitrogenous organic matter by animal waste. Presences of nitrate in irrigation water leads salt cake deposition and spoil the soil texture. From the sample 1 to sample 4 all the sample is come under permissible limits and it ranges from 10(mgl-1) to 14 (mgl-1).The study reveals that all the water samples collected from the wells taken from the irrigated area of the village called periyakallapadi, Tiruvannamalai, are found to have a higher influencing parameters such as in chlorides, Sodium, TDS, SAR ratio, Total Hardness, Ca++ ions, Mg++ ions. Because of the above, and also by comparing the WHO standards all the groundwater is not suitable for drinking and also for irrigation purposes. So groundwater in the village Periyakallpadi is the problematic aspects with respect to drinking purposes and for irrigation purposes.
Acknowledgement
The authors are grateful to the management and staff of SKP Institute of Technology, Tiruvannamalai, and Tamilnadu for conducting the lab tests and supporting the research.
References
- Olajire AA, FE Imeokparia (2001) Water Quality Assessment of Osun River: Studies on Inorganic Nutrients. Environ Monitor Assess 69(1): 17-28.
- Domenico P (1972) A Concepts and models in groundwater hydrology. Mc Graw Hill, New York, USA.
- Jalali M (2005) Nitrates leaching from agricultural land in Hamadan, western Iran. Agriculture, Ecosystems & Environment 110(2-4): 210218.
- Jothivenkatachalam, A Nithya, Chandra Mohan (2010) Correlation analysis of drinking water quality around perur block of coimabtore district, Tamiladu, India 3(4): 649-654.
- Freeze RA, JA Cherry (1979) Groundwater. Prentice Hall Inc, Englewood Cliffs, New Jersey, USA. 7632: pp. 604.
- Hem JD (1985) Study and interpretation of chemical characteristics of Natural water, U.S. Geol. Survey, water supply paper. 2254: pp. 264.
- Sanjay Gupta, Akhalesh kumar, Ojha CK, Gita Seth (2004) J: Environmental science and Engineering 46(1): 74-78.
- Yidana SM, D Ophori, B Banoeng Yakubo (2008) Hydrogeological and hydrochemical characterization of the Voltaian Basin: The Afram Plains area. Environ Geol 53: 1213-1223.
- Glover CR (1996) Irrigation water classification systems. Cooperative Extension Service, College of Agriculture and Home Economics, New Mexico State University, Mexico.
- Angino EE (1983) Geochemistry and water quality, Applied Environmental Geochemistry, Academic press, London pp. 171-199.
- Palanisamy PN, Geetha A, Sujatha M, Sivakumar P, K Karunakaran (2007) Assessment of ground water quality around gopichettipalyam Town Erode District, Tamilnadu. E Journal Chemistry 4(3): 434-439.
- Andre L, Franceschi M, Pouchan P, Atteia (2002) O: Using geochemical data and modeling to enhance the understanding of groundwater flow in a regional deep aquifer, Aquitain Basin, south-west of France. Journal of Hydrology, APHA, Standard methods for the examination of water and waste water (20th edn). American Public and Health Association 305: 40-62.
- Edmunds W (2003) M: Renewable and non-renewable groundwater in semi-arid regions. Developments in Water Science 50: 265-280.
- Singh AK, Mondal GC, Kumar S, Singh TB, Teqry A (2008) Major ion chemistry, weathering process and water quality assessment in upper catchment of Damodar river basin, Environ Geol, India 54(4): 745-758.






























