Investigations of Physico-Chemical Composition of Groundwater in Otuoke and Environs, Bayelsa State, Nigeria
Abadom DC* and Nwankwoala HO
Department of Geology, University of Port Harcourt, Nigeria
Submission: January 05, 2017; Published: February 26, 2018
*Corresponding author: HO Nwankwoala, Department of Geology, University of Port Harcourt, Nigeria; Email: drkhalezakaria@gmail.com
How to cite this article: Abadom DC, Nwankwoala HO. Investigations of Physico-Chemical Composition of Groundwater in Otuoke and Environs, Bayelsa State, Nigeria. Int J Environ Sci Nat Res. 2018; 9(1): 555752. DOI: 10.19080/IJESNR.2018.08.555752.
Abstract
The study was designed to investigate the groundwater quality in Federal University of Otuoke and environs, Bayelsa State. A total of fourteen groundwater samples were acquired and analyzed for their physico-chemical and heavy metal parameters. Heavy metals were all analyzed with the Atomic Adsorption Spectrometer. The result revealed that for the heavy metals, iron was most predominant, ranging from 0.004 to 10mg/L with mean and SD of 0.95±2.63mg/L. Manganese ranged from 0.01 to 0.91mg/L, with mean and SD of 0.18±0.21mg/L. Copper was below the machine detectable limit in six samples, but ranged from 0.006 to 0.013mg/L, with a mean of 0.01mg/L. All other heavy metal including lead, arsenic, cobalt, boron and barium were below the detectable limit of the machine (< 0.001mg/L).In order of decreasing magnitude, the average cationic concentrations in the groundwater are in the order; Na (101.71mg/L) >Ca (29.04mg/L) > Mg (10.01mg/L) > K (2.31mg/L); and anionic concentrations; Cl (25.88mg/L) > HCO3 (1.65 mg/L) > PO4 (0.42 mg/L) > NO3 (0.08mg/L) > SO4 (0.05mg/L).Assessment of groundwater in the area for drinking purposes revealed that the water is predominantly acidic (pH=6.37), with iron (0.95mg/L) and manganese (maximum =0.91mg/L) contents exceeding regulatory guidelines (WHO, 2011; NSDWQ, 2007) for potable drinking water in most locations. This shows that the groundwater sources are unsafe for consumption purposes. All other measured chemical parameters were within regulatory requirements. Constant monitoring and quality assessment is necessary to ensure that groundwater in the area is within regulatory requirements.
Keywords: Physico- chemical; Groundwater quality; Heavy metals
Abbreviations: GPS: Geographic Positioning System; AAS: Atomic Absorption Spectrophotometer; SD: Standard Deviation; TDS : Total Dissolved Solids; APHA: American Public Health Association
Introduction
Groundwater quality is determined by the solutes, flow paths and soil gases dissolved in the water, as well as the matter suspended in and floating on the water. Hence, groundwater quality is a consequence of the natural physical and chemical state of the water as well as any alteration factors that may have occurred as a consequence of human activity and microbial activities in soils [1]. The quality of groundwater is of vital concern, since it is directly linked with health and human welfare. Ranjana 2010 clearly stated that the quality of public health depends greatly on the quality of groundwater. Groundwater in the preferred source of potable water in the Niger delta, because it is less prone to contamination as a result of its natural filtration [2]. Contamination of groundwater from heavy metals may occur due to factors including irrigation with contaminated water, transportation, industrial emissions, the use of fertilizers and metal based pesticides, etc. [3]. The presence of heavy metal, even at small concentrations in water is an indication of contamination and the persistent consumption of such water could result to adverse health effects.
Although groundwater quality is more preffered when compared to surface water, its quality is the sum of natural and anthropogenic influences [4]. Water quality parameters reflect the level of contamination in water resources and show whether water is suitable for human consumption, irrigation and/or industrial usage. Drinking contaminated water is unacceptable because of its adverse health effects [5]. There are two basic contaminant indicators whose presence or absence helps determines the quality of water in any given area (elemental and microbial). Microbial contaminants in water includes; fungi, pathogenic bacteria and viruses. Elemental contaminants include; physicochemical parameters, metals as well as organic chemical contaminants such as; pesticides and radioactive contaminants Akunobi and Chibuzor 2012. The quantity of water may not be an issue in a terrain such as Niger Delta but its quality is of utmost importance. A substantial part of the study area is motor able and is close to Yenagoa the State capital. This has led to continuous influx of people and increased business activities in the area over the last decade with strong dependence on groundwater. Predominant anthropogenic activities in the area which can pose severe risk to the groundwater resources includes gas flaring from exploitation of oil and gas resources, leakages and corrosion of pipelines, septic tanks and possible effluents from industries, open dumping, etc. Therefore, it becomes obligatory to undertake a groundwater hydro chemical survey in the area to ascertain its quality for drinking, domestic use and other purposes.
Description of the Study Area
The study area Federal University Otuoke and environ is located within the lower section of the upper flood plain deposits of the sub-aerial Niger Delta [6]. Geographically, it lies between latitudes 40 46'N and 50 51'N and longitudes 60 15'E and 60 2 3'E (Figure 1). The area is bounded on the North by Yenagoa, the capital of Bayelsa State and on the south by Brass and Nembe Local Government Areas of Bayelsa State, to the West by southern Ijaw and Ahoada-west local government areas of Bayelsa State and Rivers State respectively. The area can be accessed from the north by the Mbiama-Yenagoa road and on the south by the Nembe and Brass Rivers [7]. Most part of the area is motor-able; hence there is a network of roads that links the different parts of the area.
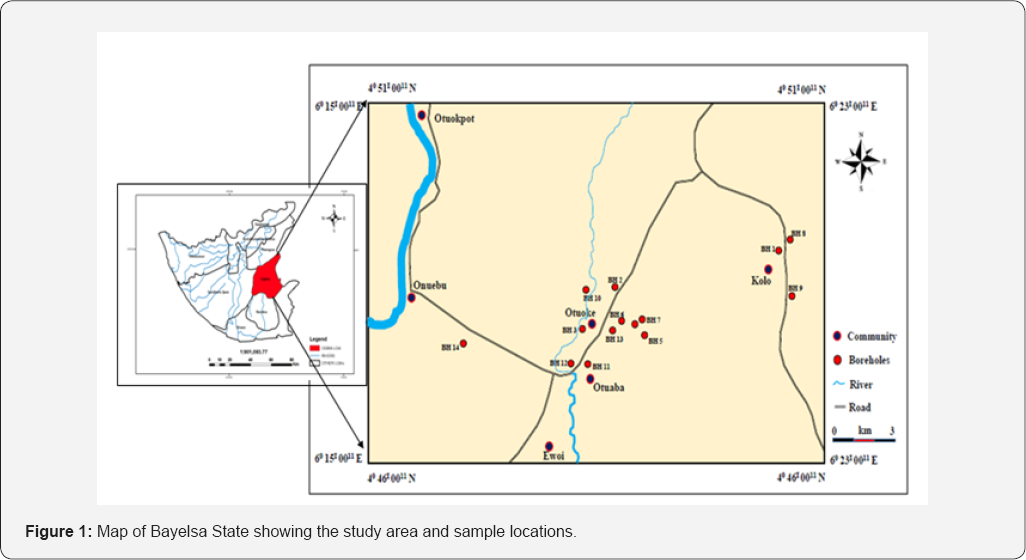
Brief Geology and Hydrogeology of the Area
The study area lies in the coastal Niger Delta sedimentary basin. The geology of the Niger Delta has been described in details by various authors. The formation of the Delta started during Early Paleocene and resulted mainly from the buildup of fine grained sediments eroded and transported by the River Niger and its tributaries. The Tertiary Niger Delta is a sedimentary structure formed as a complex regressive off-lap sequence of clastic sediments ranging in thickness from 9,000-I2,000m [8]. Starting as separate depocenters, the Niger Delta has coalesced to form a single united system since Miocene. The Niger Delta is a large and ecologically sensitive region, in which various water species including surface and sub-surface water bodies exist in a state of dynamic equilibrium [8]. Stratigraphically, the Niger Delta is sub-divided into Benin, Agbada and Akata Formations in order of increasing age. The Benin Formation is the water bearing zone of the area (Table 1). It is overlain by Quaternary deposits (40-I50m thick) and generally consists of rapidly alternating sequence of sands and silty clays with the latter becoming increasingly more prominent seawards [9]. The clayey intercalations within the Benin formation have given rise to multi-aquifer system in the area [9]. The first aquifer is commonly unconfined while the rest are confined. The study area has been noted to have poor groundwater quality due to objectionable high concentration of certain groundwater parameters and encroachment of saltwater or brackish water into the freshwater aquifers [10-12]. The static water level in the area ranges from 0-2m during the rainy season and I-3m during the dry season [13]. The main source of recharge is through direct precipitation where annual rainfall is as high as 3000mm [14,15]. The water infiltrates through the highly permeable sands of the Benin Formation to recharge the aquifers [16,17]. Groundwater in the area occurs principally under water table conditions [18].
Materials and Methods
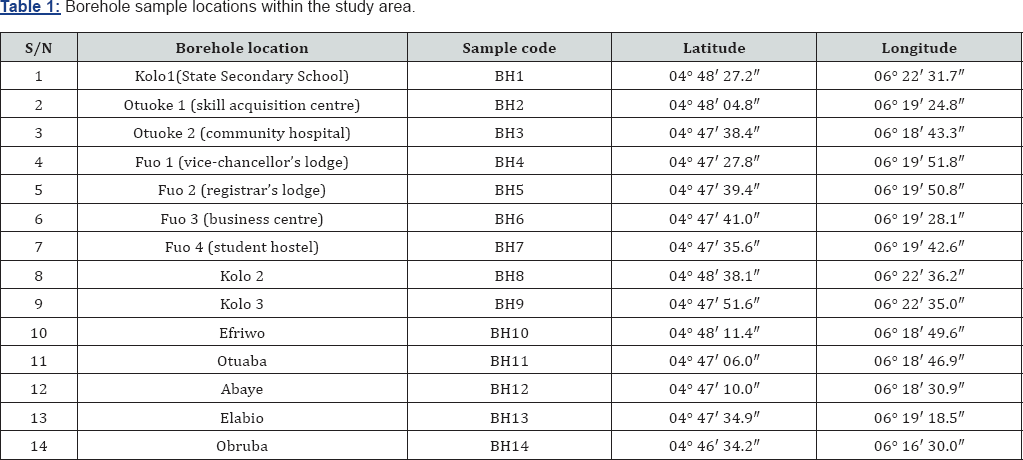
Groundwater samples were collected from fourteen boreholes in Federal University of Otuoke and its environs during the rainy season. The boreholes utilized for this study were selected from eight communities at random. Both private and public water sources were sampled in this study. Sterilized water bottles were used to collect representative water samples to prevent contamination. At each borehole location, the sample bottles were washed and rinsed thoroughly with the sample water before being sampled. The samples were collected close to the well head to maintain the water integrity. The boreholes were allowed to flow for about 3 minutes to ensure stable conditions before samples were collected. The bottle was filled to the brim with the sample water, and the lid immediately replaced to minimize oxygen contamination and escape of dissolved gases. Sampling was done using two sets of prelabelled bottles of one litre capacity for ionic and heavy metals analysis respectively Water samples for the determination of cations were stabilized by adding few drops of diluted HCl to them after collection. To maintain the integrity of the water samples, physico-chemical parameters sensitive to environmental changes such as pH, conductivity and temperature were measured and recorded in- situ using portable digital meters. The co-ordinates of all the sampling locations were recorded using a Garmin 78 model Geographic Positioning System (GPS). The samples were later transported to the laboratory in an ice chest for chemical analysis. Table 1 shows the borehole sampling locations along with the geographic cordinates. Heavy metals were determined using an Atomic Absorption Spectrophotometer (AAS) as described in APHA 3111B and ASTM D3651 [19-22]. This involved direct aspiration of the sample into an air/acetylene or nitrous oxide/acetylene flame generated by a hollow cathode lamp at a specific wavelength peculiar only to the metal programmed for analysis. For every metal investigated, standards and blanks were prepared and used for calibration before samples were aspirated. Concentrations at specific absorbance displayed on the data system monitor for printing. The equipment limit of detection is <0.001mg/L. Table 2 shows the equipment and analytical methods used for groundwater samples analysis.
Results and Discussion
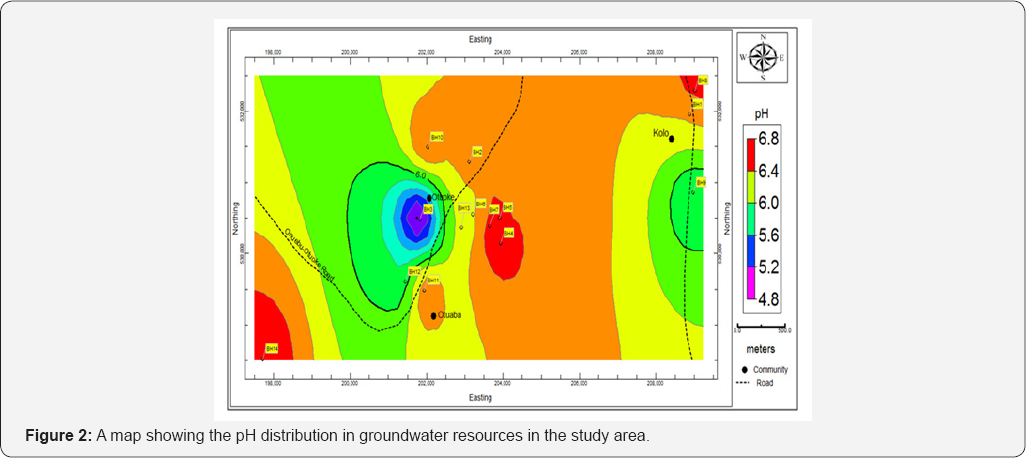
Groundwater temperature in the study area ranges from 24.60 to 29.5oC with mean of 27.31±1.12oC and variance of 1.25 (Table 3). Groundwater pH which is a measure of acidity or alkalinity, ranges from 4.78 to 7.01 with mean, standard deviation (SD) and variance of 6.27±0.56 and 0.32. The highest pH values were obtained from BH7 (7.01) and BH8 (6.83) whereas the lowest pH was obtained at BH3 (4.98) (Figure 2); (Table 3). This shows that the water in the area is predominantly acidic. The EC ranges from 53.20 - 130.30 |iS/cm with mean of 94.81±22.12|iS/cm and variance of 489.13. The high standard deviation and variance shows that there is wide degree of variability in the EC of the groundwater resources in the area. Groundwater turbidity ranges from 3.71 - 5.14 NTU, with mean, SD and variance of 4.37±.46 NTU and 0.21 respectively (Tables 3 & 4). Total soluble solids ranged from 4.72 to 13.02mg/L while hardness ranged from 32.0 to 61.0mg/L. Water is said to be hard when it contains large amount of dissolved salts, such as calcium and magnesium ions. Total Dissolved Solids ranges from 4.11 to 92.10mg/L with mean, SD and variance of 36.02±23.30 and 542.92 respectively (Table 4). Alkalinity ranged from 10.34 to 12.01mg/L with mean and SD of 11.19±0.35mg/L
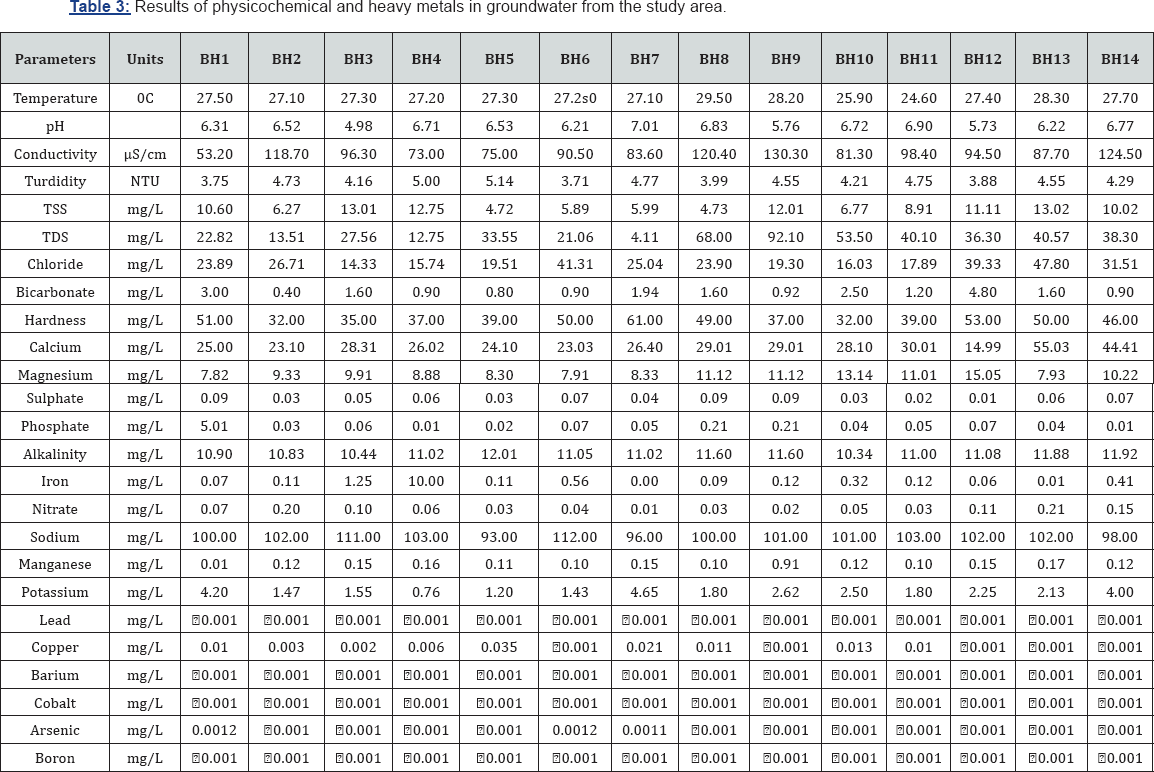
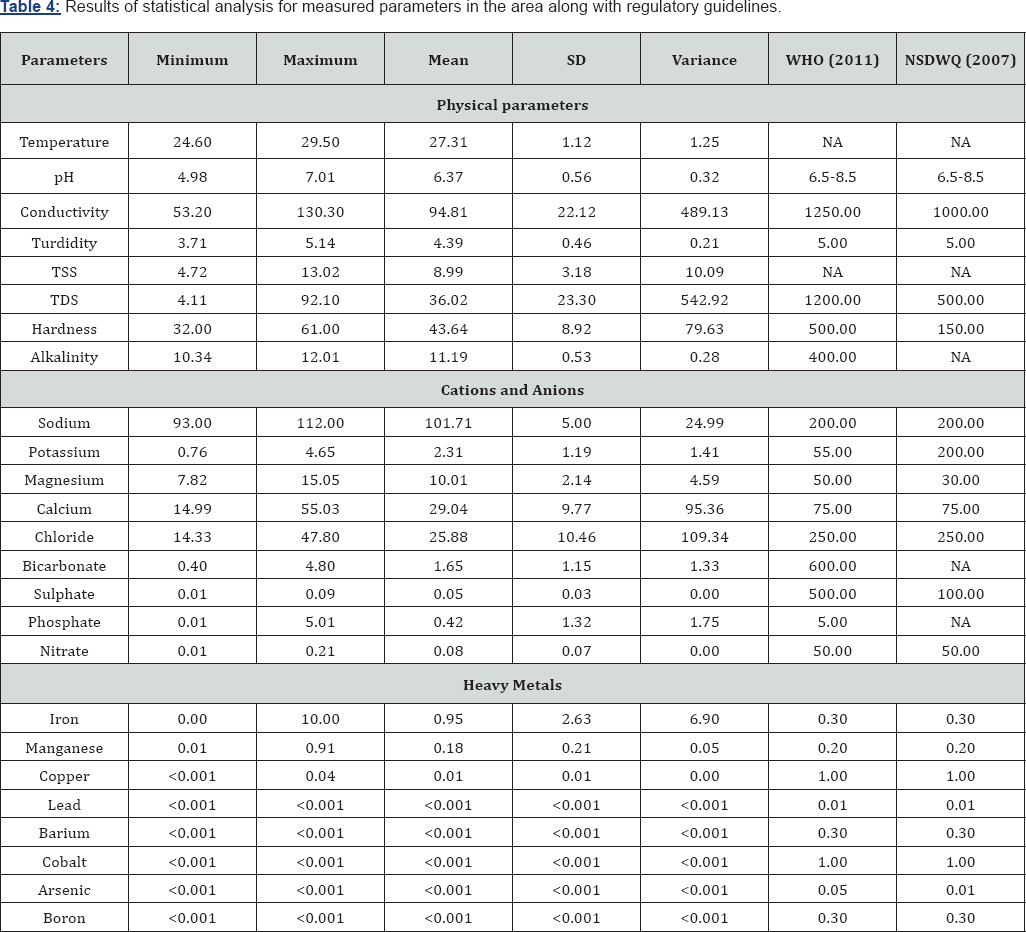
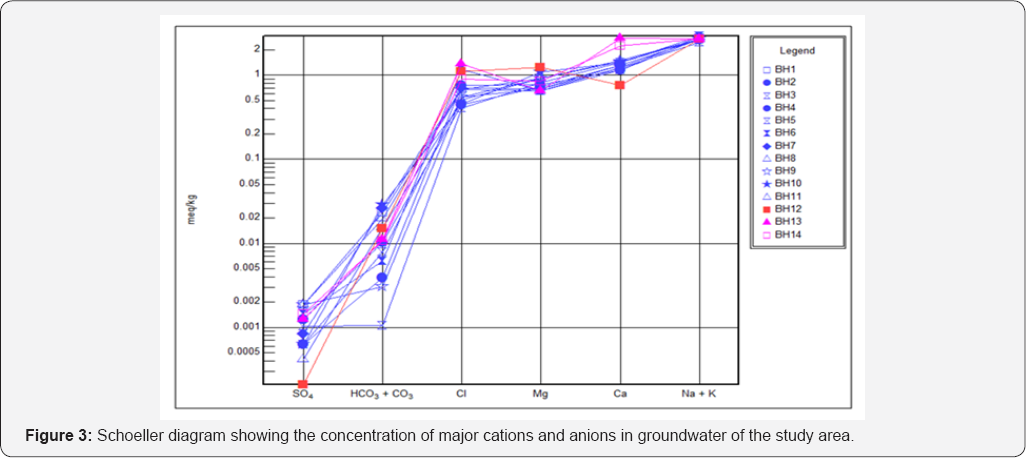
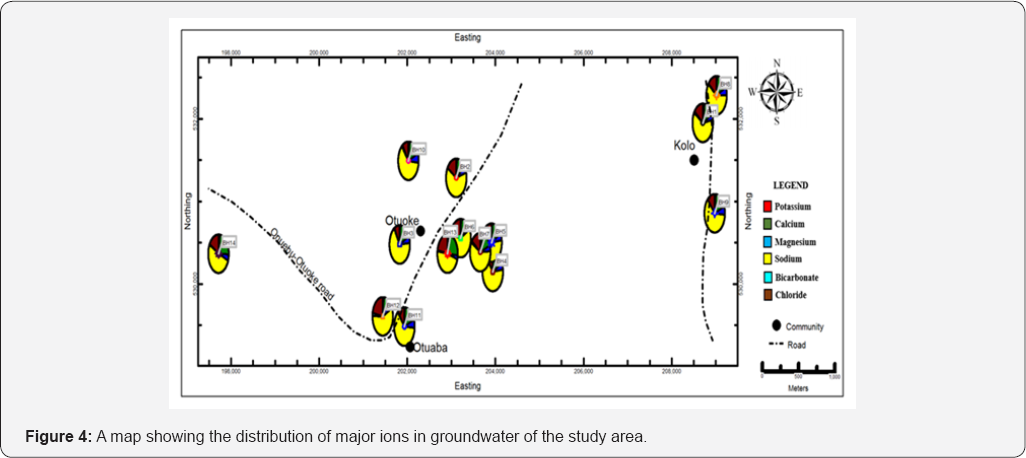
The ionic concentrations in were in the order; Na >Ca> Mg> K, and Cl> PO4> HCO3> SO4> NO3 (Table 4). Schoeller diagram which is a graphical presentation of cations and anions shows that cations predominate over anions in the groundwater (Figure 3). For cations, sodium ranged from 93 to 112mg/L with mean and SD of 101.70±5.0mg/L. Calcium which is the second most dominant cation in groundwater in the area ranged from 14.99 to 55.03mg/L, with mean, SD and variance of 29.04±9.77mg/L and 95.36mg/L respectively. The highest calcium concentration was obtained from BH13 and the lowest was from BH12. Magnesium and potassium ranged from 7.82 to 15.05mg/L and 0.76 to 4.65mg/L, with mean and SD of 10.01±2.14 and 2.31±1.19mg/L respectively (Table 4). Chloride was the most dominant cation in the groundwater, ranging from 14.33 to 47.80mg/L, with mean and SD of 25.88±10.46mg/L. The highest chloride concentration was obtained from BH13 while the lowest was obtained from BH3. Although the concentrations of bicarbonate were relatively low, it was the second most dominant anion in the groundwater, ranging from 0.40 to 4.80mg/L with mean and SD of 1.65±1.15mg/L. Phosphate concentration ranges from 0.01 to 5.01mg/L, with mean and SD of 0.42±1.32mg/L while Nitrate ranged from 0.01 to 0.21mg/L with mean and SD of 0.08±0.07 mg/L. Sulphate had the lowest anionic concentration, ranging from 0.01 to 0.09mg/L with mean and SD of 0.05±0.03mg/L. A map showing the distribution of cations and anions in the study is presented in Figure 4. The map shows sodium is the most predominant cation while chloride is the most predominant anion in the groundwater.
For the heavy metals, iron was most predominant, ranging from 0.004 to 10mg/L with mean and SD of 0.95±2.63mg/L. Manganese ranged from 0.01 to 0.91mg/L, with mean and SD of 0.18±0.21mg/L. Copper was below the machine detectable limit in six samples, but ranged from 0.006 to 0.013mg/L, with a mean of 0.01 mg/L. All other heavy metal including lead, arsenic, cobalt, boron and barium were below the detectable limit of the machine (< 0.001mg/L) (Table 4).
Suitability for Drinking Purposes
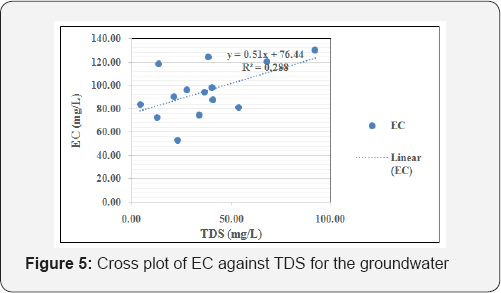
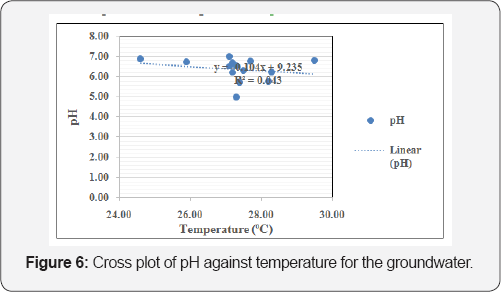
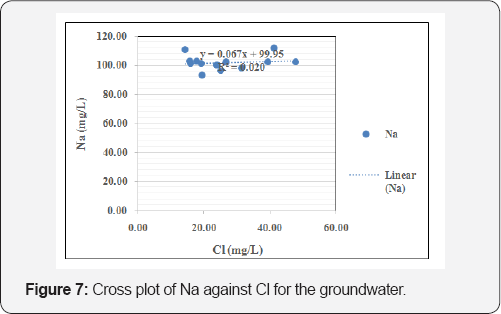
The suitability of groundwater for drinking purpose was achieved by comparing the acquired groundwater geochemical results with regulatory guidelines for potable water. The average pH of the groundwater shows that the water is acidic (pH=6.37) and deviates from both WHO (2011) and NSDWQ (2007) guidelines of 6.5-8.5 [23,24], and hence is unfit for drinking. Prolonged consumption of acidic water over long periods of time may result in derangement of the balance of acid to base in the human body, which results in metabolic acidosis [25]. Also, the average concentration of iron (0.95mg/L) exceeds both WHO 2011 and NSDWQ 2007 guidelines of 0.30 mg/L,concentration can kill organisms directly, and while continued exposure over long periods of time to lower concentrations can lead to stunted growth, lower reproduction rates, deformities, and even mortality (Lewis and Clark, 1996). Based on Freeze and Cherry [26] classification scheme, the groundwater samples are classified as soft, with hardness values < 100 in most of the boreholes. Based on total dissolved solids (TDS), Davis, Deweist [27] classify groundwater in the study area as 'desirable for drinking' having TDS values all below 500mg/L. Figure 5 is the cross plot of EC against TDS for the groundwater while Figure 6 is the cross plot of pH against temperature for the groundwater. Figure 7 is the cross plot of Na against Cl for the groundwater in the area [28-31].
Summary and Conclusion
In order of decreasing magnitude, the average cationic concentrations in the groundwater are in the order; Na (101.71mg/L) >Ca (29.04mg/L) > Mg (10.01 mg/L) > K (2.31mg/L); and anionic concentrations; Cl (25.88mg/L) > HCO3 (1.65mg/L) > PO4 (0.42mg/L) > NO3 (0.08mg/L) > SO4 (0.05mg/L). For the heavy metals, iron was most predominant, ranging from 0.004 to 10mg/L with mean and SD of 0.95±2.63 mg/L. Manganese ranged from 0.01 to 0.91mg/L, with mean hence the water is unfit for drinking, unless treated for iron. Manganese is relatively higher than WHO (2011) and NSDWQ (2007) of 0.2mg/L in BH8 (0.91mg/L), and must be treated before consumption. Because of the toxic nature of heavy metals, exposure to a high concentration can kill organisms directly, and while continued exposure over long periods of time to lower concentrations can lead to stunted growth, lower reproduction rates, deformities, and even mortality (Lewis and Clark, 1996). Based on Freeze and Cherry [26] classification scheme, the groundwater samples are classified as soft, with hardness values < 100 in most of the boreholes. Based on total dissolved solids (TDS), Davis, Deweist [27] classify groundwater in the study area as 'desirable for drinking' having TDS values all below 500mg/L. Figure 5 is the cross plot of EC against TDS for the groundwater while Figure 6 is the cross plot of pH against temperature for the groundwater. Figure 7 is the cross plot of Na against Cl for the groundwater in the area [28-31]. and SD of 0.18±0.21 mg/L. Copper was below the machine detectable limit in six samples, but ranged from 0.006 to 0. 013.g/L, with a mean of 0.01mg/L. All other heavy metal including lead, arsenic, cobalt, boron and barium were below the detectable limit of the machine (< 0.001mg/L). Assessment of groundwater in the area for drinking purposes revealed that the water is predominantly acidic (pH = 6.37), with iron (0.95mg/L) and manganese (maximum = 0.91mg/L) contents exceeding regulatory guidelines WHO, 2011; NSDWQ, 2007 for potable drinking water in most locations. This shows that the groundwater sources are unsafe for consumption purposes. All other measured chemical parameters were within regulatory requirements. Constant monitoring and quality assessment on the groundwater is necessary to ensure that groundwater in the area is within regulatory requirements.
References
- Hwang JY, Park S, Kim HK, Kim MS, Jo HJ et al. (2017) Hydrochemistry for the Assessment of Groundwater Qualityin Korea. Journal of Agricultural Chemistry and Environment 6(1): 1-2.
- Agbalagba OE, Agbalagba OH, Ononugbo CP, Alao AA (2011) Investigation into the Physico-Chemical Properties and Hydrochemical Processes of Groundwater from Commercial Boreholes in Yenagoa,Bayelsa State, Nigeria. African Journal of Environmental Science and Technology 5(7): 473-481.
- Duran A, Tuzen M, Soylak M (2007) Trace Element levels in some Dried Fruit samples from Turkey. International Journal of Food Science and Nutrition 59(7-8):581-589.
- Chapman D (1996) Water Quality Assessments. A guide to use biota, sediments and water in environmental monitoring (2nd edn), University Press, Cambridge, London.
- Suthra S, Bishnoi P, Singh S, Mutiyar PK, Nema AK (2009) Nitrate contamination in groundwater of some rural areas of Rajasthan. Department of Civil Engineering, Indian Institute of Technology New Delhi India, Pp. 111-135.
- Allen JRL (I965) Coastal Geomorphology of the Eastern Nigerian Beach Ridges and Vegetated Tidal Flats. Geology and Mining, Mijabonw 44: 2-20.
- Tariwari CN, Ohimain EI, Mieyepa CE (2015) The Potability of Groundwater in Bayelsa State, Central Niger Delta, Nigeria: A Review Journal of Environmental Treatment Techniques 3(2): 127-162.
- Abam TKS (1999) Dynamics and Quality of Water Resources in the Niger Delta proceedings of IUGG 99 Symposium HSS Birmingham. IAHS Publ 259: 429-437.
- Etu Efeotor JO, Akpokodje EG (1990) Aquifer Systems of the Niger Delta. Journal Mining and Geology 26: 279-284.
- Nwankwoala HO (2011) Assessment of Groundwater Quality in parts of Bayelsa State, Eastern Niger Delta, Nigeria. Journal of Soil and Water Sciences 4 (4): 7-23.
- Nwankwoala HO, Udom GJ, Ugwu SA, (2011) Some heavy metal investigations in groundwater sources in Yenegoa, Bayelsa State, Nigeria. Journal of Applied Technology in Environmental Sanitation 1(2): 163-170.
- Nwankwoala HO (2013) Evaluation of Hydrochemical Characteristics of Groundwater in Port Harcourt, Nigeria. Unpublished PhD Dissertation, University of Port Harcourt, Nigeria, pp. 294.
- Nwankwoala HO and Daniel TE (2016) Physico-Chemical Evaluation of Groundwater in Ogbia, Bayelsa State, Nigeria. Journal of Groundwater Research 5(2): 1-15.
- Amajor LC, Ofoegbu CO (1988) Determination of Polluted Aquifers by Stratigraphically Controlled Biochemical Mapping; Eastern Niger delta, Nigeria. Groundwater and Mineral Resources of Nigeria, pp. 62- 73.
- Amajor LC (1986) Geochemical Characteristics of Groundwater in Port Harcourt and its Environs. Proc Int'l Symposium on Groundwater Resources of Nigeria, Lagos, pp. 358-375.
- Nwankwoala HO, GJ Udom (2011) Hydrochemical evaluation of groundwater in parts of Eastern Niger Delta, Nigeria. Journal of Academic and Applied Sciences 1(2): 33-58.
- Oborie E, Nwankwoala HO (2014) Analysis of major ions constituents in Groundwater in Amassoma and Environs, Bayelsa state, Nigeria. Journal of Applied Chemistry 2(5): 1-13.
- Udom GJ, Amah EA (2006) Quality Status of Groundwater in Yenagoa and its Environs, Bayelsa State, Nigeria. Journal of Scientific and Industrial Studies 4(1): 45-51.
- ASTM (1998) Standard Practice for Calculation and Adjustment of the Langelier Saturation Index for Reverse Osmosis. D3739-D3794.
- American Public Health Association (APHA), American Water Works Association (AWWA) and Water Pollution Federation (WPCF) (1985) Standard Methods for the examination of Water and Waste water (16th edn), Washington, USA, pp. 1260-1268.
- APHA (1995) American Water Works Association and Water Environment Federation. Standard Methods for the Examination of Water and Wastewater (21st edn), American Public Health Association, Washington, USA.
- American Society Testing Materials (ASTM) (1969) Manual on Water. (4th edn), Hamilton, Philadelphia, Pennsylvania, USA. pp. 171-227.
- World Health Organization (2011) Guidelines for drinking water quality criteria (4th edn), Geneva, Switzerland, pp. 307-441.
- NSDWQ (2007) Nigerian Standard for drinking water Quality guidelines. (2nd edn), Nigerian Industrial standard, Nigeria, p. 14-21.
- Ofoma AE, Omologbe D, Aigberua P (2005) Physic0-Chemical Quality of Groundwater in parts of Port Harcourt City, Eastern Niger Delta. Water Resources, Nigeria 16:18-24.
- Freeze RA, Cherry AJ (1979) Groundwater. Prentice-Hall, Englewood Cliffs, New Jersey, USA. pp. 604.
- Davis SN, DeWiest RJM (1966) Hydrogeology. Wiley, New York, USA.
- Amadi PA, Morrison T, Ofoegbu CO (1989) Hydrogeochemical Assessment of Groundwater Quality in Parts of the Niger Delta, Nigeria. Environmental Geology 14(3): 195-202.
- American Water Works Association (1971) Water quality and treatment. Mc Graw-Hill. New York, USA. pp. 654.
- Rajana A (2010) Physico-Chemical Analysis of some Groundwater Samples of Kotputli Town Jaipur. Rajasthan India 1(2): 111-113.
- Short KC, Stauble AJ (1967) Outline Geology of the Niger Delta. AAPG Bull 51:761-779.






























