Retrieval of Nickel Affected Phaseolus Mungo L. by Ulva Lactuca
Selvaraj K*
Department of Botany, Sri Kaliswari College, India
Submission: December 18, 2017; Published: January 04, 2018
*Corresponding author: Selvaraj K, Department of Botany, Sri Kaliswari College (Autonomous), Sivakasi, Virudhunagar District, Tamilnadu, Tel: 9789240653; Email: kselvarajphd@gmail.com
How to cite this article: Selvaraj K*. Retrieval of Nickel Affected Phaseolus Mungo L. by Ulva Lactuca. Int J Environ Sci Nat Res. 2017; 8(1): 555729. DOI: 10.19080/IJESNR.2018.08.555729
Abstract
The effect of seaweed Ulva lactuca on germination, growth and biochemical parameters of Phaseolus mungo L was studied. Application of various concentrations from 10% to 30% with optimal nickel chloride solution increased the percentage of growth, pigment content and decreased the content of amino acid and proline. It was found to be increased when compared to the respective control grown with nutrient medium. Thus, the seaweed liquid extract obtained from Ulva lactuca was suitable for alleviating the nickel stressed impact on Phaseolus mungo L.
Keywords: Germination; Biochemical Parameters; Seaweed Liquid Extract
Introduction
Seaweed has valuable properties with numerous applications in food and pharmaceutical industries. India has a vast coastline and more than 800 species of marine algae occur on our coast but not a single species is cultivated on commercial scale [1]. Seaweeds are important marine renewable resources. They are used as food, fodder, fertilizer, agar, alginate and source of various fine chemicals [2]. In recent years, the sea weeds are immensely used as fertilizer [3] and as a substitution in place of conventional synthetic fertilizer. It is also used as auxins [4], as gibberellins [5] as betaines [6], and as macronutrients such as Ca, K, P, and micronutrients like Fe, Cu, Zn, B, Mn, Co and Mo [7], which are necessary for the growth and development of plants. Seaweeds and seaweed extracts also enhance soil health by improving moisture holding capacity and by promoting the growth of beneficial soil microbes.
The use of seaweed as manure is very common in coastal areas throughout the world. Seaweed extracts have been marketed for several years as fertilizer additives and beneficial results from their use have been reported by [8]. Seaweed extracts contain macronutrients, trace elements, organic substance like amino acid and plant growth regulators such as auxins, cytokinins and gibberellins [9]. Plant tolerance to heavy metals depends largely on plant efficiency in the uptake, translocation, and further sequestration of heavy metal in specialized tissues or in trichomes and organelles such as vacuoles. The uptake of metals depends on their bioavailability, and plants have evolved mechanisms to make micronutrients bioavailable. Chelators such as siderophores, organic acids, and phenolics can help to release metal cations from soil particles, and also increasing their bioavailability. For example, organic acids (malate, citrate) excreted by plants act as metal chelators. By lowering the pH around the root, organic acids increase the bioavailability of metal cations. However, organic acids may also inhibit metal uptake by forming a complex with the metal outside the root. Citrate inhibition of aluminum (Al) uptake and resulting Al tolerance in several plant species is an example of this mechanism. Copper tolerance in Arabidopsis is also the result of a similar mechanism. The presence of rhizosphere microbes may also affect plant uptake of in organics. For example, rhizosphere bacteria can enhance plant uptake of mercury and selenium. However, the exact mechanisms of these plant-microbe interactions are largely unknown. It is possible that the microbe mediated enhanced uptake will be either due to a stimulatory effect on root growth or to microbial production of metabolites that could affect plant gene expression of transporter proteins, or to a microbial effect on the bioavailability of the element.
Nickel (Ni) is an essential element that can be toxic and possibly carcinogenic in high concentrations. Ni is ubiquitously distributed in nature. It is found in different concentrations in all soil types of diverse climatic regions [10]. Naturally derived soils from serpentine rocks are rich in Ni, but due to various industrial and anthropogenic activities such as mining, refining of Ni ores, burning of fossil fuels and residual oil and sewage sludge, other areas have also become prone to Ni contamination [11]. The normal range of Ni in soil is 2 to 750ppm, with a critical soil concentration at 100 ppm [12]. Exposure to Ni compounds causes irreversible damage to the central nervous system, cardiovascular system, lungs and gastrointestinal tract. Nickel has been classified among the essential micronutrients and remains associated with some metallo-enzymes, but Ni is toxic at elevated concentrations in plants [10].
Plant subjected to excess supply of nickel accelerates generation of toxic oxygen species leading to oxidative stress [13,14] and induces physiological water stress [15]. Excess nickel was reported to affect a number of biological and physiological processes resulting in an inhibition of plant growth [16,17]. Phytoremediation can be defined as the use of green plants to remove pollutants from the environment or to render them harmless [18,19]. It is also referred to as green technology and can be applied to both organic and inorganic pollutants present in soil (solid substrate), water (liquid substrate) and the air [20]. In this respect, plants can be compared to solar driven pumps capable of extracting and concentrating certain elements from their environment [21]. This technology is being considered as a new highly promising technology for the remediation of polluted sites. Over the last decade, there has been an increase in both industrial activities and urbanization. Both aquatic and terrestrial habitats are becoming progressively polluted due to the discharge of pollutants generated from various industries, transportation and fossil fuel burning.
Materials and Methods
Seeds of Phaseolus mungo L. (Black gram) were procured from local seed centre, Thiruthangal. The healthy and viable seeds of Phaseolus mungo L. were surface sterilized with 0.1% of mercuric chloride for one minute and washed with running tap water followed by distilled water. The seeds were soaked in distilled water for 2 hours. Both control and experimental seeds were allowed to grow in plastic trough containing uniform amount of sandy soil. Seedlings were allowed to grow in halfstrength of Hoagland nutrient solution for seven days. After seven days, the seedling were treated with different concentration of nickel chloride (3mM, 6mM, 9mM, 12mM and 15mM w/v) with half-strength of Hoagland nutrient solution, by keeping one trough without treatment as control. After seven days of metal treatment (on 15th day after germination) various morphometric and biochemical characters were analyzed. The optimum concentration of (9mM) nickel chloride was determined by LSD (Least Significance Difference Test) and it was subjected to treatment with various concentration of seaweed liquid extract (10%, 20% and 30% v/v) along with half-strength of Hoagland nutrient solution and one trough with Hoagland nutrient solution treatment alone kept as control. The seaweed liquid extract in various concentrations was added in 9mM concentration of nickel chloride and was kept in shaker overnight for uniform mixing. Then they were applied to the seedlings for various treatments. After seven days of treatment, various morphometric and biochemical characters were analyzed. Proper control was also maintained for comparison.
For all morphometric characteristics, ten seedlings have been taken from both experimental and control sets and the results indicate the average of ten seedlings along with their respective standard error. The length of root of the randomly selected seedlings was measured with the help of meter scale. The length of the shoot of the randomly selected seedlings was measured for both control and experimental plants with the help of meter scale. The total leaf area of each and every plant was computed and expressed in cm2. The leaf area of the harvested leaves was measured by conventional graphical method. The fresh weight of the seedlings was obtained using an electronic balance soon after harvest. Care was taken to avoid wilting of plant parts. The fresh undamaged seedlings were kept in an oven at 70° C for 24 hours. After complete drying, the seedlings were weighed using an electronic balance.
For all biochemical analysis the average of 5 samples were taken from both control and treated plants separately and the results indicate the average of five seedlings along with their respective standard error. To extract the total chlorophyll from leaves, fresh leaves were deveined and cut into small bits. From the pooled leaf bits, a sample of 100mg was weighed. The leaf bits were homogenized in 100% acetone using a mortar and pestle. The homogenate was centrifuged at 4000rpm for 5 minutes at room temperature. Extraction with 100% acetone was repeated until the pellet becomes pale yellow or white in colour. The supernatant was used for the estimation of photosynthetic pigments. The absorbance was measured at 662nm, 645nm and 470nm for chlorophyll a, chlorophyll b and Carotenoids, respectively using ELICO SL 171 Spectrophotometer. The amount of chlorophyll a,b and total chlorophyll was calculated by using the formula.
Chlorophyll a (mg/L) = 11.75 x A662 - 2.35 x A645
Chlorophyll b (mg/L) = 18.61 x A645 - 3.96 x A662
Total Chlorophyll (mg/L) = 7.79 x A662 + 16.26 x A645
The carotenoid content was calculated using the following formula,
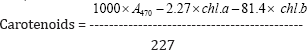
Chl. a = chlorophyll a, Chl. b = chlorophyll b The protein content of the leaf tissue was measure by Lowry's method (1951). Fresh leaf sample (100mg) was ground in 10ml of distilled water with the help of mortar and pestle. The homogenate was centrifuged at 5000rpm for 5 minutes and the supernatant was added with 1 ml of ice cold TCA and again it was centrifuged. The pellet was dissolved with 1ml of 0.1 N NaoH and it was used as test solution. From the test solution, 0.1ml was taken in the test tubes and it was added with 0.5ml of distilled water, 5.5ml of alkaline copper mixture and 0.5ml of Folin phenol reagent. It was mixed thoroughly and kept in condition for 10 minutes to develop blue colour. The absorbance was noted at 650nm using ELICO SL 171 Spectrophotometer. The protein content was calculated from the standard graph of protein constructed with bovine serum albumin as marker protein. Total soluble sugars in leaves were estimated by Anthrone method. 100mg of fresh leaves of both control and treated plants were ground in 10ml of distilled water using mortar and pestle. The homogenate of leaves was centrifuged at 3000rpm for 5 minutes. The supernatant was taken, it was added with 2ml of 10 % TCA and kept in the ice cold condition for 10 minutes, and again it was centrifuged at 5000rpm for 5 minutes. The supernatant was used as test solution. 0.1ml of test solution was taken in test tubes and it was added with 0.9ml of distilled water and 4ml of Anthrone reagent (0.2 %). The test tubes were boiled in water bath for 10 minutes after cooling; the absorbance was measured at 620nm. The amount of sugar present in the extract was calculated from a standard curve using glucose as the standard.
Free amino acids were estimated by Ninhydrin assay method. The leaf material (100 mg fresh weight) was ground in 10ml of ethanol. The homogenate was centrifuged at 5000rpm for 3 minutes. The pellet was discarded and the supernatant was used as test solution. 1 ml of test solution, 3ml of distilled water and 1ml of Ninhydrin reagent were added and mixed thoroughly. After mixing, the test tube was kept in boiling water bath for 10 minutes. Then the tube was cooled down to room temperature and 1ml of 50 % ethanol was added. The absorbance was measured at 550nm using proper blank. Blank solution consisted of 4ml of distilled water, 1ml of Ninhydrin reagent and 1ml of ethanol. The amino acid content was estimated from standard curve prepared with glycine as amino acid source. Proline content was estimated according to. The 100mg leave sample were ground in 3% (w/v) Sulphosalicylic acid. The extract was filtered through Whatmann No.1 filter paper. 2ml of the extract, 2ml of acid Ninhydrin (1.25g of Ninhydrin in a mixture of 30ml of glacial acidic acid and 20ml of 6M Phosphoric acid) and 2ml of glacial acidic acid were added. The contents were shaken well and the tubes were kept in the water bath at 1000 C for 1 hour. After 1 hour the tubes were allowed to cool down to room temperature and then kept in ice for 5 minutes, to terminate the reaction. The 4ml of toluene was added and the tubes were agitated vigorously and then allowed to stand. The proline containing chromophore was aspirated and the absorbance was read at 520nm. The proline content was calculated from a standard curve prepared authentic sample of proline. Statistical analysis Morphometric parameters were determined with ten independent replicates. Biochemical characters were carried out at least five times. The data were reported as mean ± SE and in parentheses represent the percent activity.
Results
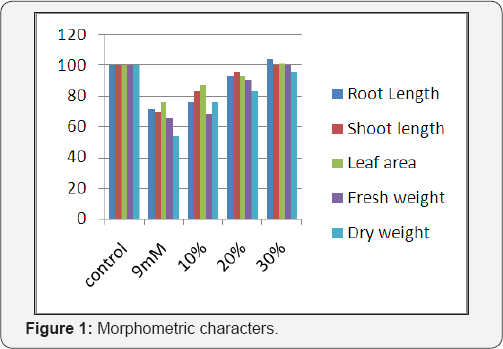
The results obtained on the effect of nickel + seaweed liquid extract treated plants were given as follows. The effect of nickel and Ulva lactucaon the morphometric characteristics of Phaseolus mungo L. is shown in (Figure 1). The treatment of seaweed liquid extract to the seedlings, increased the root length with the increase in the amount of seaweed liquid extract when compared with the optimal nickel chloride solution and the maximum root length (94%) was observed in 30% seaweed liquid extract. Addition of various concentration of seaweed liquid extract has caused significant increase in the shoot length of Phaseolus mungo L. The increase was about 26% in 30% seaweed liquid extract treatment when compared to the 9mM of Nickel chloride solution. In seaweed liquid extract treatment, the leaf area was increased and found to be more than the normal (1% more) in 30% seaweed liquid extract treated seedlings. Seaweed liquid extract treated seedlings showed remarkable increase to about 58% in 30% seaweed liquid extract application when compared to the 9mM Nickel chloride solution. Dry weight was increased with increase in the concentration of seaweed liquid extract. The maximum dry weight more or less normal was observed in 30% seaweed liquid treated plants which were more or less equal to the control.
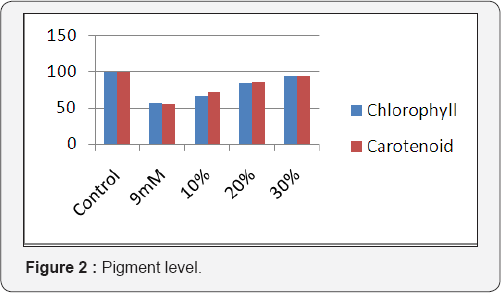
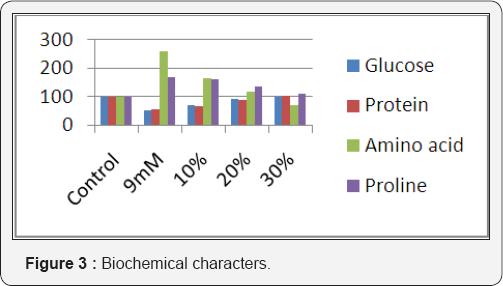
Results obtained on the effect of nickel and Ulva lactuca on the photosynthetic pigments of Phaseolus mungo L. is shown in (Figure 2). The seaweed liquid extract treatment, increased the total chlorophyll and carotenoid contents with the increasing in concentration of seaweed liquid extract. The effect of nickel and Ulva lactuca on the biochemical characteristics of Phaseolus mungo L. is shown in (Figure 3). The seaweed liquid extract application increased the sugar content gradually ranging from 17% to 48 % when compared to the 9mM nickel chloride solution. In the seaweed liquid extract treated seedlings the protein content was significantly increased. The highest range of protein content was found to be in 30% of seaweed liquid extract application where in it was more than normal (2% more) than the control. In the seaweed liquid extract application, the free amino acid content gradually decreased from 14% to 88 % when compared with 9mM nickel chloride solution. In the seaweed liquid extract treated seedlings the proline content decreased ranging from 20% to 59% when compared to the 9mM optimal nickel chloride solution.
Discussion
In the present study, both the leaf area and total chlorophyll are found to increase with the increase in the concentration of seaweed liquid extract. This indicated that, seaweed Ulva lactuca enhanced not only the leaf area but also the content of photosynthetic pigments, which finally led to increase in leaf area and pigment content has been reported after application [22]. As a result of protein degradation during stress condition, the availability of free amino acid is significantly high. The free amino acid content is increased with the increase in nickel supply. It may be due to destruction of protein or to the biosynthesis of amino acid from the nitrate source which were not utilized in the protein synthesis [23]. It is an adaptative mechanism by the plant cell to overcome post stress metabolism [24]. Accumulation of proline has frequently used as a biochemical marker for water stress in plants [25,26]. The reduction of stress in plants has thought to promote the accumulation of proline and to act as a cytoplasmic osmotic solute [27]. L-Proline accumulation may cause by stimulated synthesis from glutamate, slower incorporation of proline into protein and failure in protein synthesis. Proline accumulation is considered a protective device for the plants to preserve water, which is necessary to tide over any internal water deficit situation. The accumulation of proline is also considered as an adaptive response to stress [28]. An increase in protein, sugar and decrease in free amino acid and proline accumulation, after the application of seaweed liquid observed in the present study indicated the promotive nature, on plant growth. This positive response is observed even in low concentration of seaweed. It was also reported by Ramasubramanian and Jeyaprakash [29] that the level of protein increase and proline level decrease after seaweed application. The seaweed application, have caused a reduction in proline accumulation than the stressed plants, indicating the overcome of stress by the application of seaweed, even at low concentration. This result was in accordance with the findings of [30,31] wherein sea weeds were used as bio adsorbents.
Conclusion
The present study clearly shows that the use of green algal extract i.e., the extract of Ulva lactuca can efficiently remove the nickel toxicity. Hence we strongly suggest the Ulva lactuca extract can be used as a liquid fertilizer to remove the toxicity of Nickel polluted environment for sustainable agriculture.
References
- Oza RM, Krishna Kumar GR, Mairh OP, Zaidi SH (2001) Cultivation of Ulvafascicata Delile on the coast of Diau West coast of India. Seaweed Res Utiln 23(1&2): 5-12.
- Sahoo D (2000) Farming the ocean: Seaweeds cultivation and utilization. American Journal of Plant Sciences Aravali, New Delhi. pp. 1-283.
- Hong DD, Hien HM, Son PN (2007) Seaweeds from Vietnam used for functional food, medicine and fertilizer. J Appl Phycol 19(2): 817-826.
- Strik WA, Arthur GD, Lourens AF, Novak O, Strnad M, et al. (2004) Changes in cytokinin and auxin concentrations in seaweed concentrates when stored at an elevated temperature. J Appl Phycol 16: 31-39.
- Wildgoose PB, Blunden G, Jewers K (1978) Seasonal variation in gibberellin activity of some species of Fucaceae and Laminariaceae. Bot Mar 21: 63-65.
- Blunden G, Guiry MD (1991) Agricultural uses of seaweeds and seaweed extracts. Seaweed resources in Europe: Uses and potential. Wiley Chichester p. 65-81.
- Khan W, Rayirath UP, Subramanian S, Jithesh MN, Rayorath P (2009) Seaweed extracts as bio stimulants of plant growth and development. Plant Growth Regul 28(2): 386-399.
- Booth E (1965) The manorial value of seaweed. Bot mar pp. 138-143.
- Williams DC, Brain KR, Blunden G, Wildgoose PB (1981) Commercial seaweed extracts. Proc. (8thedn.), SeaweedSymp, Bango, Marine science laboratories, Menaibridge pp. 761-763.
- Srivastava S, Mishra S, Dwivedi S, Baghel VS, Verma S, et al. (2005) Nickel Phytoremediation Potential of Broad Bean, Viciafabal, and its Biochemical Responses. Bull Environ Contam Toxicol 74: 715-724.
- Nriagu JO (1988) Production and uses of chromium. Chromium in natural and human environment. New York, USA, John Wiley and Sons.
- Gardea Torresdey JL, Peralta-Videa JR, De La Rosa G, Parsons JG (2005) Phytoremediation of heavy metals and study of the metal coordination by x-ray absorption spectroscopy. Coordination Chemistry Reviews 249: 1797-1810.
- Baccouch S, Chaoui A, Ferjani E (1998) Nickel induced oxidative damage and antioxidant responses in Zea maize shoots. Plant Pysiol Biochem 36(4): 689-694.
- Schiclker, Caspi (1999) Responses of antioxidative enzymes to nickel and cadmium stress in hyper accumulator plants of genus Alyssum Physiol Plant 105: 39-44.
- Panday N, Sharma CP (2002) Effect of heavy metals Co2+, Ni2+ and Cd2+ on growth and metabolism of Cabbage. Plant Sci 163: 753-758.
- Jones MD, Hutchinson TC (1998) Nickel toxicity in mycorrhizal birch seedling infected with Lactariusrufus or Scleroderma flavidum. I. Effect on growth, photosynthesis, respiration and transpiration and transpiration. New phytol 108: 451-459.
- Gajeswska E, Slaba M, Andrezejewska R, Sklodowska M (2006) Nickel induced inhibition of wheat root growth is related to H2O2 production, but not to lipid peroxidation. Plant Growth Regul 49: 95-103.
- Berti W, Cunningham SD (2000) Phyto stabilization of metals. In Phyto remediation of toxic metals: using plants to clean-up the environment.
- Salt DE, Kumar PB, Dushenkov S, Raskin I (1994) Phytoremediation: A New Technology for the Environmental Cleanup of Toxic Metals. International Symposium Research on Conservation and Environmental Technology for Metallic Industry. Toronto, Canada.
- Gratao PL, Prasad MN, Cardoso PF, Lea PJ, Azevedo RA (2005) (a) Phytoremediaion: green technology for the clean up of toxic metals in the environment. Braz J Plant Physiol 17(5): 53-64.
- Salt DE, Prince RC, Pickering IJ, Raskin I (1995) Mechanisms of Cadmium Mobility and Accumulation in Indian Mustard. Plant Physiol 109: 1427-1433.
- Yvin JC (1994) New approach to the mode of action of seaweed extracts in agriculture. Proc Int Seaweed 8: 59-63.
- Sharma SK, Srivastava A, Singh VP (1997) Effect of rubber factory effluent on growth in Vignamungo. J Environ Poll 4: 175-177.
- Singh DS, Vijayakumar KP (1974) Carry out the effect of salinity on yield and quality of wheat seed. Seed Res 21: 13-18.
- Alia P, Saradhi PP (1991) Proline accumulation under heavy metal stress. J Plant Physiol 138: 554-558.
- Schat H, Sharma SS, Voojis R (1997) Heavy metal induces accumulation of proline in metal tolerant and non-tolerant ecotypes of Silene Vulgaris. Physiol Plant 101(4): 477-482.
- Levy D (1983) Water deficit enchances praline and alpha - amino nitrogen and its association with susceptibility to drought. Plant Physiol 551: 169-173.
- Lee KG, Gunningam BA, Paulsen GM, Liang GH, Moore RB (1976) Effect of cadmium on respiration rate and activities of several enzymes in soybean seedlings. Physio Plant 36(1): 4-6.
- Ramasubramanian V, Jeyaprakash R, Sumathi M, Ramasamy TN (2006) Amelioration efficacy of Dicyotasps on growth and biochemical characteristics of match industry effluent treated Abelmo schusesculentus (L) Medicus seedlings. Seaweed Res Utiln 28: 139-144.
- Selvaraj K, Jeyaprakash R, Ramasubramanian V (2010) Impact of nickel on growth and biochemical characteristics of Vigna radiate (L.) Wilczek and amelioration of the stress by the seaweed treatment. J Basic & Appl Biol 4(3): 181-187.
- Crouch IJ, Van Staden J (1993) Evidence for the presence of plant growth regulators in commercial seaweed products. Plant Growth Regul 13(2): 21-29.






























