Channel Flow and Depth Relationship in a Forested Stream in the Humid Tropical Environment, Niger Delta Nigeria
PJ Sajil Kumar*
Hydrogeology Group, Institute of Geological Sciences, Freie Universität, Germany
Submission: November 03, 2017; Published: November 20, 2017
*Correspondence author: PJ Sajil Kumar, Institute of Geological Sciences, Hydrogeology Group, Freie Universität Berlin, Malteserstr. 74-100, 12249 Berlin, Germany, Tel: +4917647173950; E-mail: pjsajil@gmail.com; pj.sajil@fu-berlin.de
How to cite this article: PJ Sajil K. Acid Mine Drainage (AMD): Environmental Impacts and Remedies- A Review. Int J Environ Sci Nat Res. 2017; 6(4): 555697. DOI: 10.19080/IJESNR.2017.06.555697
Abstract
Mining is an integral part of the economical sustainability of any country in the world. On the other hand, serious environmental impacts are associated with the mining activities. One of the major problems is Acid Mine Drainage (AMD), which is largely affecting the water resources. Irreversible environmental degradation has occurred in many part of the world through both active and abandoned mining sites. In this paper, we have reviewed the environmental impacts and geochemistry of AMD generation and possible treatment methods were suggested as a measure of mitigation
Keywords: Acid Mine drainage (AMD); Geochemical processes and Treatment of AMD
Introduction
Mining is one of the important factor for the environmental degradation. In the context of Economy, the utilization of natural resources is inevitable. However, sustainable development is needed to protect the environment. There are several problems associated with the mining and related activities, in which acid mine drainage (AMD) is the most common form of environmental contamination. AMD causes damage worth billions of dollars to natural vegetation, silviculture, rivers, watersheds, natural habitats and aquatic life. Among those, Water pollution is one of the serious issue related to the mining activity. This is mainly caused by the metal-rich acid solution, known as Acid Mine Drainage (AMD) [1]. AMD is characterized by contains dissolved solids of high concentration, more than 3g/l of sulphate. Additionally, low pH and presence of heavy metals makes AMD treatment a major concern. Importantly, the generation of AMD will continue even after the Mine is abandoned. In this paper we tried to explain the impacts of AMD on the environment, geochemical processes and the remediation methods.
Geochemical Processes
The basic scenario found at coal and hard-rock sites in relation to environmental concerns are given below. Pyrite (FeS2) is responsible for acid generation and metals dissolution in coal and hard rock sites. When pyrite is exposed to oxygen and water it will be oxidized, resulting in hydrogen ion release - acidity, sulphate ions, and soluble metal cations, (Equation 1). Mining increases the exposed surface area of these sulphur-bearing rocks allowing excess acid generation beyond the water�s natural buffering capabilities.
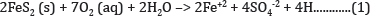
Further oxidation of Fe+2 (ferrous iron) to Fe+3 (ferric iron) occurs when sufficient oxygen is dissolved in the water or when the water is exposed to sufficient atmospheric oxygen.
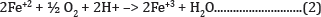
Some acidity is consumed in this process; however, the stage is set for further hydrogen ion release that will surpass these benefits. Ferric iron can either precipitate as ochre (Fe(OH)3 the red-orange precipitate seen in waters affected by acid mine drainage) or it can react directly with pyrite to produce more ferrous iron and acidity.
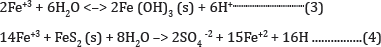
When ferrous iron is produced because of equation 4 and sufficient dissolved oxygen is present the cycle of (Equations 2& 3) is perpetuated [2]. Without dissolved oxygen, equation 4 will continue to completion and water will show elevated levels of ferrous iron. Once the water is sufficiently acidic, acidophilic bacteria - bacteria that thrive in low pH - are able to establish themselves. Microorganisms play a significant role in accelerating the chemical reactions taking place in mine drainage situation. Thiobacillus Ferroxidans, bacteria, is commonly referenced in this case. These bacteria catalyze the oxidation of ferrous iron, further perpetuating equations 2 through 4. Another microbe belonging to the Archaea kingdom, named Ferroplasma Acidarmanus, has recently been discovered to also play a significant role in the production of acidity in mine waters [3]. Though not a major source of acidity, the generation of hydrogen ions when certain metals form precipitates must be considered when considering treatment options.
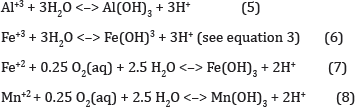
Treatment of AMD
There are two categories of treatment for AMD, mainly active- and passive treatments. Active treatments are alternately known as chemical treatment includes adding chemicals to increase pH, and precipitate metals. By adding the alkaline material to the AMD, oxidation of the ferrous ion taker place and metals precipitate as hydroxide and carbonates [4]. The other active treatment options are adsorption [5], ion exchange [6] and Membrane technologies [7]. However, the most commonly used method in the recent times are Constructed wetlands(biological), sulphate reducing bioreactors and treatment with lime stone drains [8]. Passive treatment options are useful in abandoned mines where there is no regular flow. Passive methods do not need continues chemical inputs and capable of removing large quantities of sludge [4,8]. The most commonly used method is a permeable reactive barrier. This is easy to operate and economically viable when compare to the active methods.
Conclusion
Acid Mine drainage is posing serious threat to the Environment and the Ecosystem Health and causing irreversible damage. It is observed that stopping the mining activities is not at all solving the issue, as abandoned mines are also generating enough drainage which is rich in toxic metals. We have reviewed the geochemical processes that controlling the generation of AMD and Possible treatments were suggested. Active methods such as chemical treatment with lime, adsorption, ion exchange and membrane based techniques and passive techniques such as permeable reactive barrier are suggested. However, the selection of method is strictly based on the characteristic of application site.
References
- Ferguson KD, Erickson PM (1988) Pre-Mine Prediction of Acid Mine Drainage. In: Salomons W, Forstner U Environmental Management of Solid Waste. Springer, Berlin, Heidelberg, p. 24-43.
- Younger, Paul L, Hedin, Robert S, Banwart, Steven (2002) A Mine water: hydrology, pollution, remediation. Kluwer Academic Publishers, Dordrecht; Boston.
- Lauzon, Shelly (2002) New Iron Eating Microbe Major Component of Mining Pollution and Iron and Sulfur Cycling.
- Johnson DB, Hallberg KB (2005) Acid mine drainage remediation options: a review. Sci Total Environ. 338(1-2): 3-14.
- Aprianti T, Afrah BD, Agustina TE (2017) Acid Mine Drainage Treatment Using Activated Carbon Ceramic Adsorbent in Adsorption Column. International Journal on Advanced Science, Engineering and Information Technology 7(4).
- Gaikwad RW, Sapkal VS, Sapkal RS (2010) Ion exchange system design for removal of heavy metals from acid mine drainage. Waste water Acta Montanistica Slovaca Rocni'k 15(2010), ci'slo 4: 298-304.
- Le NL, Nunes SP (2016) Materials and membrane technologies for water and energy sustainability, Sustainable Materials and Technologies. 7: 1-28.
- Kefeni KK, Msagati TAM, Mamba BB (2017) Acid mine drainage: Prevention, treatment options, and resource recovery: A review. Journal of Cleaner Production 151: 475-493.






























