The Ultrasonic of Drug Removal Using Catalysts from Aqueous Solutions
Mohammad Hossein Sayadi*and Najmeh Ahmadpour
Department of Environment, University of Birjand, Iran
Submission: September 27, 2017; Published: October 04, 2017
*Corresponding author: Mohammad Hossein Sayadi, Department of Environment, University of Birjand, Iran, Email: ahmadpour.najme@gmail.com
How to cite this article: Mohammad H S, Najmeh A. The Ultrasonic of Drug Removal Using Catalysts from Aqueous Solutions. 2017;5(4): 555668. DOI:10.19080/IJESNR.2017.05.555668
Abstract
The presence of drug substances in water is a serious environmental problem because many of them are poisonous and may accumulate in organisms and cause illness. The purpose of this study was to review the ultrasonic of drug removal using catalysts from aqueous solutions. Based on the results, optimal conditions for removing ibuprofen at 500 kHz frequency of ultrasonic apparatus were under acidic conditions and reaction time of 90 minutes, which was removed at 99.6%. While at low removal of ibuprofen in the frequencies of 862 kHz for 240 minutes, was 40%. The results of various studies showed that the ultrasonic process alone could not be used as an effective way to remove these compounds. Adding H2O2 and TIO2 catalysts greatly increased the removal process. Therefore, it can be concluded that this combination process is a beneficial way to remove the drug pollution in water and wastewater.
Keywords: Ultrasonic; Environmental pollution; pharmaceutical pollution
Introduction
Medications are a very important and integral part of today’s modern life and are used to treat human and animal illnesses. The presence of medications in the environment is one of the most important issues in the world today. Recently, only a few advanced countries have begun to study the negative effects of these pollutants on the environment [1]. These materials enter the environment in various ways, such as wastewater from pharmaceutical industries, hospitals and human and animal waste [2]. Drugs are detected in surface water, underground, wastewater and even in drinking water in amounts of nanograms to micrograms per liter. Various methods have been used such as active carbon adsorption, reverse osmosis, air buckling and biological methods to remove these drug compounds, but these methods do not eliminate the contaminants but transfer them from one phase to another Slowly [3,4].
Advanced oxidation
Advanced Oxidation Processes (AOPS: Advance Oxidation) are a good way to decompose pharmaceutical compounds, and so far, various processes such as photo electro co catalysis, [5,6] application of photo catalytic processes [7-9]. Photo phenton has been used to remove drugs from the aquatic environment [10]. These processes work on the basis of the production of hydroxyl radicals, which these radicals are almost capable of oxidizing all organic compounds, including these processes, the sono catalyst system. [10,11]. The sono chemical system, coupled with the use of the catalyst, is capable of oxidizing the drug and converting it to harmless or less harmful products for microorganisms [3]. Sono chemical reactions result from high-intensity sound radiation from liquids at frequencies producing cavitation (normally in the range of 1000 - 20 KHz). Therefore, cavitation as an ultrasound energy concentrator is introduced into the micro reactor, which acts simultaneously with the simultaneous release of reactive radicals to any server reactor as a hot spot. Ultrasound waves increase chemical and physical changes in a fluid environment through production and, consequently, the degradation of cavitation bubbles. These bubbles form over a period of a number of cycles and grow to equilibrate to a certain frequency. The destiny of these bubbles is that they collapse in the next intensive (compressed) periods to produce the energy required for chemical and mechanical effects (Michael, 2013). In addition, water sonolysis produces H2O2, which, coupled with the presence of a catalyst and the presence of iron ions, usually increases the decomposition of organic matter [12,13].
The main mechanism of destruction
The two main mechanisms for the destruction of pollutants during the sonolysical bubble are the pyrolysis reaction in cavitation and the other radical reactions by radical water, hydroxyl and hydrogen generated through sonolysis. The two mechanisms are generally as follows: [14]
Titanium dioxide catalyst
Between the various catalysts that can be used as a catalyst in this process, due to high catalytic activity, non-toxicity, stability in aqueous solutions and relatively few costs are commonplace [5]. The sono chemical process with the use of catalysts is relatively new to the process of water purification and AOPs of wastewater processes. Due to its high decomposition power, it can be used as an effective method for decomposition and degradation, such as those that have low biodegradability, such as ibuprofen [12].
Hydrogen peroxide catalyst
One of the strongest (H2O2) hydrogen peroxide is known and stronger oxidants than chlorine, chlorine dioxide, and potassium permanganate. The molecular structure of this material consists of an oxygen bridge, which connects each of these oxygen atoms to a hydrogen atom. This substance can react directly or indirectly with organic matter in water. Hydrogen peroxide acts in the oxidation-reduction reaction and acts as an oxidizing or reducing agent. In indirect reactions, the oxidizing activity is related to the free radicals produced by the decomposition of hydrogen peroxide. Previous studies have shown that the greater the amount of oxidation of substances, due to the fact that the result of a combined system is higher than OH the number of radicals of a separate ultrasonic system, therefore, the combined oxidation velocity will be higher than that of the separate ultrasonic system [15,16].
Removal of drug by ultrasonic process
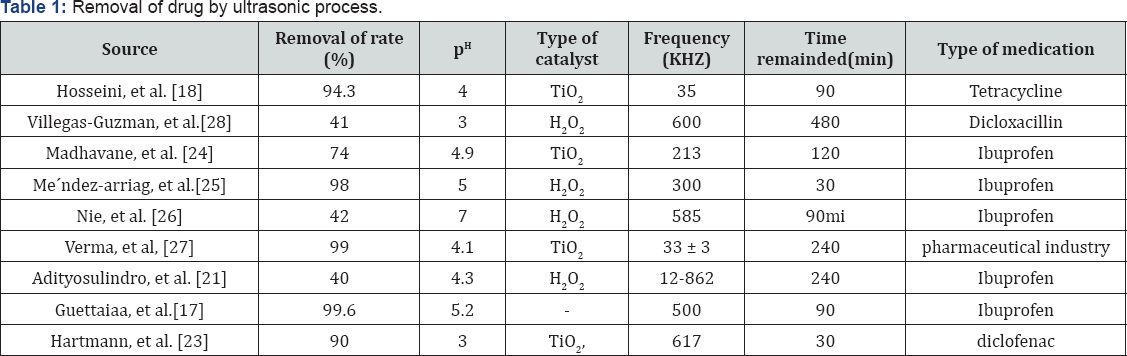
[17] In 2017 examined the sono chemical and photochemical removal of ibuprofen in aqueous solution and concluded that a combination of photochemical and ultrasonic purification resulted in a significant increase in the rate of IBP degradation and the best pH in this study was 5.2 [18] examined the efficacy of removing antibiotic tetracycline from aqueous solutions using a ultrasonic process and evaluating the effect of effective parameters on the process, The results of this study showed that the synolysis process alone cannot be used as an effective way to remove tetracycline antibiotics from aqueous solutions; However, the addition of a TiO2 catalyst improves the removal efficiency considerably, and therefore, this compound process can be used as a useful way to remove this biodegradable pollutant from water and sewage (Table 1)[19].
Examined the destruction of the chemical synthetic cephalexin in aqueous solution and reported that the ultrasonic process is a successful purification method for removing and improving the digestibility of cephalexin in the wastewater. Ultrasonic can effectively decompose cefalexin in aqueous solutions, and its degree of degradation depends on operating conditions, such as ultrasound power and pH value of the environment [20] investigated the ultrasonic degradation of tetracycline by sulfate radicals and reported that the removal efficiency was strongly influenced by the initial concentrations of tetracycline, pH, ultrasound strength, per sulfate concentration, temperature, and ion concentration. The destruction of tetracycline in silver and per sulfate was in the first degree kinetic son chemical process [21-24]. The study has shown that ultrasonic-based chemical oxidation is an effective and applicable technology for the destruction of organic compounds such as antibiotic tetracycline in aqueous solution [24-28].
Conclusion
Several studies have been carried out on the removal of pollutants from water and sewage, which shows that among the methods studied, advanced oxidation techniques based on radical hydroxyl production have the ability to remove and mineralize the drugs. In this review study, the process of drug removal using the ultrasonic process was studied. The results of this study showed that the ultrasonic process alone and individually due to its low efficiency does not require, a long time and has a high energy content and is not usable on large scale, and cannot be used as an effective way to remove drugs from water solutions , but the addition of TiO2 and H2O2 catalyst greatly improves the removal efficiency, and therefore This combined process was used as a useful way to remove these highly degradable biodegradable pollutants from water and wastewater. Therefore, it can be pointed out that this method can be used as an effective way to remove drugs and other similar pollutants.
Acknowledgement
The valuable collaboration of faculty authorities of Natural Resources and Environmental Sciences, University of Birjand to provide necessary facilities for conduction of this study is highly appreciated.
References
- Sayadi MH, Trivedy RK, Pathak RK (2010) Pollution of pharmaceutical in environment. Journal of Industrial Pollution Control 26(1): 89-94.
- Emad SE, Chaudhur M (2011) the feasibility of using combined TiO2 photocatalysis-SBR process for antibiotic wastewater treatment. Desalination 272: 218-224.
- Kemmerer K (2009) Antibiotics in the aquatic environment-A reviewPart I. Chemosphere 75(4): 417-434.
- Elmolla E, Chaudhuri M (2010) Phtocatalytic degradation of amoxicilin ampicillin and cloxacillin antibiotics in aqueous solution using UV/ TiO2 and UV/H2O2/TiO2 photocatalysis. Desalination 252(1-3): 4652.
- Gad-Allah TA, Ali ME, Badawy MI (2011) Photocatalytic oxidation of ciprofloxacin under simulated sunlight. J Hazardous Mater 186(1): 751- 755.
- Hapeshi E, Achilleos A, Vasquez MI, Michael C, Xekoukoulotakis NP, et al. (2010) Drugs degrading photocatalytically Kinetics and mechanisms of ofloxacin and atenolol removal on titania suspensions. Water Res 44(6): 1737-1746.
- Daghrir R, Drogui P, Ibrahim K, Khakani AE (2010) Photoelectrocatalytic degradation of chlortetracycline using Ti/TiO2 nanostructured electrodes deposited by means of a Pulsed Laser Deposition process. Journal of Hazardous Materials 199-200: 15-24.
- BalcIoglu A, Otker M (2003) Treatment of pharmaceutical waste water containing antibiotics by O3 and O3/ H2O2 processes. Chemosphere 50(1): 85-95.
- Bautitz IR, Nogueira RF (2007) Degradation of tetracycline by photo-Fenton process-solar irradiation and matrix effects. Journal of Photochemistry and Photobiology 187(1): 33-39.
- Elmolla E, Chaudhuri M (2009) Improvement of Biode gradability of Synthetic Amoxicillin Wastewater by Photo-Fenton Process. World Applied Science Journal 5: 53-58.
- Homem V, Santos L (2011) Degradation and removal methods of antibiotics from aqueous matrices-A review. J Environ Manage 92(10): 2304-2347.
- Michael I, Rizzo L, McArdell CS, Manaia CM, Merlin C, et al. (2013) urban wastewater treatment plants as hotspots for the release of antibiotics in the environment. Sci Total Environ 447: 345-360.
- Emery RJ, Papadaki M, Freitas dos Santos LM, Mantzavinos D(2005) Extent of sonochemical degradation and change of toxicity of a pharmaceutical precursor (triphenylphosphineoxide) in water as a function of treatment conditions. Environment International 31(2): 207-211.
- Pomati F, Castiglioni S, Zuccato E, Fanelli R, Vigetti D, et al. (2006) Effects of a complex mixture of therapeutic drugs at environmental levels on human embryonic cells. Environ Sci Technol 40(7): 24422447.
- Asgari G, Seid mohammadi A, Chavoshani A, Rahmani AR (2013) Microwave/H2O2 Efficiency in Pentachlorophenol Removal from Aqueous Solutions. J Res Health Sci 14(1): 36-39.
- Shokohi R, Mahvi AH, Bonyadi Z (2009) Comparison efficiency of both sonochemical and sonochemical/hydrogen peroxide processes for cyanide removal from aqueous solutions. J Mazand Univ Med Sic 19(73): 60-67.
- Guettaia D, Mokhtari M, Hihn JY, Stortz Y, Franchi M, et al. (2017) Sonochemical and photochemical elimination of ibuprofen in aqueous solution. JMES 8(9): 3151-3161.
- Hosseini M, Safari GH, Kamani H, Jafari J, Mahvi A (1394) Evaluation of the efficiency of removal of antibiotic tetracycline from aqueous solutions using nanosensory process and evaluation of the effect of effective parameters on the process Journal of Health and Environment. Journal of Health Scientific Research Center of Environmental Health Sciences of Iran 8(2):141-152.
- Guo W, Wang H , Shi Y, Zhang G(2010) Sonochemical degradation of the antibiotic cephalexin in aqueous solution. Water SA 36(5): 651-654.
- Eslami A, Bahrami H, Asadi A , Alinejad A (2016) Enhanced sonochemical degradation of tetracycline by sulfate radicals. Water Science & Technology 73(6): 1293-1300.
- Adityosulindroa S, Barthea L, González-Labradab K, Jáuregui Hazab UJ, Delmasa H, et al. (2017) Sonolysis and sono-Fenton oxidation for removal of ibuprofen in (waste) water. Ultrason Sonochem 39: 889896.
- Emad SE, Chaudhuri M (2010) Comparison of different advanced oxidation processes for treatment of antibiotic aqueous solution. Desalination 256: 43-47.
- Hartmann J, Bartels P, Mau U, Witter M, Tumpling Wv, et al. (2008) Degradation of the drug diclofenac in water by sonolysis in presence of catalysts. Chemosphere 70(3): 453-461.
- Madhavan J, Grieser F, Ashokkumar M (2010) Combined advanced oxidation processes for the synergistic degradation of ibuprofen in aqueous environments. J Hazard Mater 178(1-3): 202-208.
- Me'ndez-Arriaga F, Torres-Palma RA, Pe'trier C, Esplugasd S, Gimenez J, et al. (2008) Ultrasonic treatment of water contaminated with ibuprofen. Water research 42: 4243-4248.
- Nie E, Liu Q, Wang Z, Li J, Yang X, et al. (2015) Degradation of Ibuprofen by Ultrasonic Irradiation and c-Radiolysis Kinetic Studies and Degradation Pathways. Environmental Engineering Science 32(9): 773-780.
- Verma A, Chhikara I, Dixit (2014) Photocatalytic treatment of pharmaceutical industry wastewater over TiO2 using immersion well reactor synergistic effect coupling with ultrasound. Desalination and Water Treatment 52(34-36): 6591-6597.
- Villegas-Guzman P, Silva-Agredo J, Giraldo-Aguirre AL, Flórez-Acosta O, Petrier C, et al. (2015) Enhancement and inhibition effects of water matrices during the sonochemical degradation of the antibiotic dicloxacillin. Ultrason Sonochem 22: 211-219.






























