Status of Edible Fishes of Lower Gangetic Delta In Terms of Heavy Metalss
Tanmay Ray Chaudhuri1, Pallavi Dutta1, Sufia Zaman' and Abhijit Mitra2
1Department of Oceanography, Techno India University, India
2Department of Marine Science, University of Calcutta, India
Submission: September 16, 2017; Published: September 25, 2017
*Corresponding author: Abhijit Mitra, Department of Marine Science, University of Calcutta, India, Tel: 9831269550, Email: abhijit_mitra@hotmail.com
How to cite this article: Tanmay R C, Pallavi D, Sufia Z, Abhijit M. Status of Edible Fishes of Lower Gangetic Delta In Terms of Heavy Metals. Int J Environ Sci Nat Res. 2017;5(3): 555661. DOI:10.19080/IJESNR.2017.05.555661
Abstract
We analyzed concentrations of zinc, copper, lead and cadmium in ten commercially valuable finfish and shellfish species in the lower stretch of the River Ganga in April, 2016 using a Perkin-Elmer Sciex ELAN 5000 ICP mass spectrometer and expressed as mg kg-1 dry weight.In finfish species the concentrations of Zn, Cu, Pb and Cd ranged from 2.05±0.13 - 111.55±1.38, 1.26±0.11 - 53.15±0.60, BDL - 3.05±0.21 and BDL - 1.32±0.11 respectively. Concentrations in shrimp species ranged as follows: Zn: 1.19±0.06 - 141.21±1.50, Cu: 1.02±0.05 - 92.40±0.93, Pb: BDL - 2.01±0.17 and Cd: BDL in all the sampling stations. In crab the concentrations ranged from 11.65±0.10 - 213.66±1.60 for Zn, 6.18±0.11 - 109.23±1.00 for Cu, BDL - 9.99±0.79 for Pb and BDL - 1.48±0.07 for Cd. Metals in the soft body mass of oyster ranged from 21.44±0.28 - 202.78±1.49 for Zn, 8.66±0.09 - 69.14±0.79 for Cu, BDL - 8.02±0.26 for Pb and BDL for Cd. The concentration of metals was significantly affected by the sampling site and fish species. Heavy metals in the edible parts of the investigated fish species were compared with the permissible safety levels for human uses.
Introduction
Pollution of environment represents a major problem in both developed and underdeveloped countries. Among the different categories of pollutants persistent environmental contaminants are the worst in context to human health. Emissions of harmful substances have negative effects on the natural environment and human health [1]. When the consequences of environmental pollution become visible, it is often too late to prevent and chronic toxic effects, impossible to notice at the initial stage of the process, may manifest themselves after many years [2]. That is the main reason why it is imperative to conduct periodic pollution monitoring of aquatic environments. Heavy metals are stable and persistent environmental contaminants of aquatic environments. They occur in the environment both as a result of natural processes and as pollutants from human activities [3,4]. Some metals like Zn and Cu, which are required for metabolic activity in organisms, lie in the narrow "window" between their essentiality and toxicity. Other heavy metals like Cd and Pb, may exhibit extreme toxicity even at low levels under certain conditions, thus necessitating regular monitoring of sensitive aquatic environments [5-7]. From an environmental point of view, coastal zones can be considered as the geographic space of interaction between terrestrial and marine ecosystems that is of great importance for the survival of a large variety of plants, animals and marine species [8]. The coastal zone receives a large amount of metal pollution from agricultural and industrial activity [9]. Adverse anthropogenic effects on the coastal environment include eutrophication, heavy metals, organic and microbial pollution and oil spills [10]. The discharge of these wastes without adequate treatment often contaminate the estuarine and coastal water with conservative pollutants (like heavy metals), many of which accumulate in the tissues of resident organisms like fishes, oysters, crabs, shrimps, seaweeds etc. In many parts of the world, especially in coastal areas and on smaller islands, shellfish is a major part of food, which sulies all essential elements required for life processes in a balanced manner [11]. The shellfish (particularly the shrimp) is also the major exportable item of countries like India and therefore keenly related to economy of the country. Hence, it is important to investigate the levels of heavy metals in these organisms to assess whether the concentration is within the permissible level and will not pose any hazard to the consumers [12].
In Indian sub-continent the mighty River Ganga (~ 2500 km) is the reservoir of a wide spectrum of fishes distributed in different ecological habitats. The lower stretch of the River Ganga (known as Hooghly estuary) sustains about 256 species of finfish and is the breeding ground of several commercially important shellfish like shrimps, crabs and oysters (Penaeus s., Scylla serrata, Saccostrea sp., Crassostrea sp. Etc.). The River Ganga emerges from a glacier at Gangotri, about 7010 m above mean sea level in the Himalayas and flows down to the Bay of Bengal covering a distance of 2525 km. In this length, Ganga passes along 29 class-I cities (population over 1,00,000), 23 class-II cities (population between 50,000-1,00,000) and 48 towns having less than 50,000 population. About 50% of Indian population lives in the Ganga basin, and there are about 100 urban settlements with a total population of about 120 million on its banks [13]. The delta of Ganga may be said to start from Farakka in the maritime state of West Bengal.
The Gangetic delta, at the apex of Bay of Bengal is recognized as one of the most diversified and productive ecosystems of the Tropics. The deltaic lobe is unique for its wilderness, mangrove gene pool and tiger habitat. However due to intense industrial activities in the upstream zone, and several anthropogenic factors, the western part of the deltaic complex is exposed to pollution from domestic sewage and industrial effluents leading to serious impacts on biota [14]. The presence of Haldia port- cum-industrial complex in the downstream region of the River Ganga (also known as the Hooghly River) has accelerated the pollution problem with a much greater dimension [15]. The organic and inorganic wastes released from these industries and urban units contain substantial concentrations of heavy metals. The central part of the delta (encompassing the surroundings of Matla River) is relatively less stressful in terms of industrial discharge. Due to siltation of the Bidyadhari channel the area does not receive any water suly from the Hooghly River in the western sector and is therefore tide-fed in nature receiving the tidal flux from the Bay of Bengal (average salinity = ~32 psu). The present paper aims to highlight the concentration of selective heavy metals (Zn, Cu, Pb and Cd) in the muscle tissue of five common finfish species namely Polynemusparadiseus, Tenualosa ilisha, Liza parsia, Liza tade and Stolephorus commersonii, two commonly edible shrimp species Penaeus monodon and Penaeus indicus, crab species Scylla serrata and soft body parts of two oyster species Saccostrea cucullata and Crassostrea madrasensis collected from four stations distributed in two sectors (western and central Indian Sundarbans) of the lower Gangetic region.
Materials and Methods
Description of the study site
Two sampling sites were selected each in the western and central sectors of Indian Sundarbans, a Gangetic delta at the apex of the Bay of Bengal. The deltaic complex has an area of 9630 sq. Km and houses 102 islands. The western sector of the deltaic lobe receives the snowmelt water of mighty Himalayan glaciers after being regulated through several barrages on the way. The central sector on the other hand, is fully deprived from such suly due to heavy siltation and clogging of the Bidyadhari channel since the late 15th century [16]. The western sector also receives wastes and effluents of complex nature from multifarious industries concentrated mainly in the upstream zone. On this background four sampling stations (two each in western and central sectors) were selected (Table 1) and (Figure 1) to analyze the concentrations of heavy metals in the common edible finfish and shellfish species inhabiting the zone.
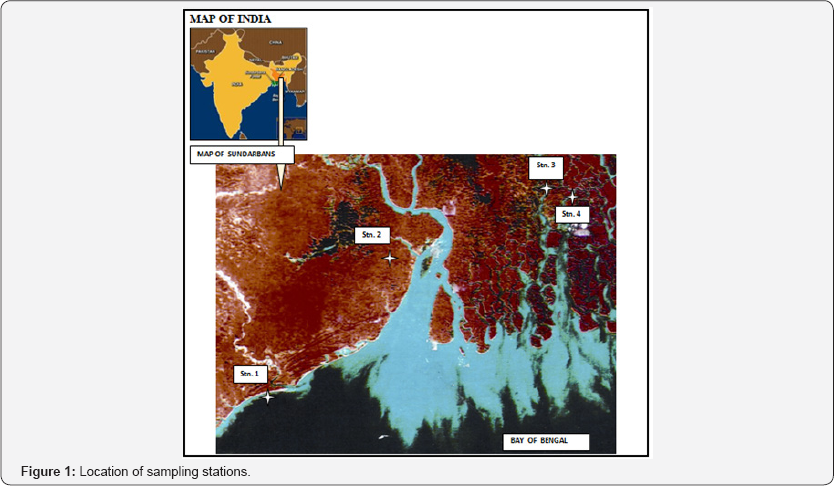
Sampling of Specimen
Five commonly edible finfish species (Polynemus paradiseus, Tenualosa ilisha, Liza parsia, Liza tade and Stolephorus commersonii), two shrimp species (Penaeus monodon and Penaeus indicus), crab species (Scylla serrata) and two oyster species (Saccostrea cucullata and Crassostrea madrasensis) were collected during low tide condition from the selected stations (Table 1) during a rapid EIA study from 10th April to 25th April, 2016. The collected samples were stored in a container, preserved in crushed ice, and brought to the laboratory for further analysis. Similar sized specimens of each species were sorted out for analyzing the metal level in the muscle of finfish, shrimps, crabs and soft body parts of oyster.
Analysis
a) Inductively Coupled Plasma - Mass Spectrometry (ICP-MS) is now - a - day accepted as a fast, reliable means of multi-elemental analysis for a wide variety of sample types [17]. A Perkin-Elmer Sciex ELAN 5000 ICP mass spectrometer was used for the present analysis. A standard torch for this instrument was used with an outer argon gas flow rate of 15 L/min and an intermediate gas flow of 0.9 L/min. The alied power was 1.0 kW. The ion settings were standard settings recommended, when a conventional nebulizer/spray is used with a liquid sample uptake rate of 1.0 mL/min. A Moulinex Super Crousty microwave oven of 2450 MHz frequency magnetron and 1100 Watt maximum power Polytetrafluoroethylene (PTFE) reactor of 115 ml volume, 1 cm wall thickness with hermetic screw caps, were used for the digestion of the collected biological samples. All reagents used were of high purity available and of analytical reagent grade. High purity water was obtained with a Barnstead Nanopure II water-purification system. All glass wares were soaked in 10% (v/v) nitric acid for 24 h and washed with deionized water prior to use.
The analyses were carried out on composite samples of 10 specimens of each species having uniform size. This is a measure to reduce possible variations in metal concentrations due to size and age. 20 mg composite sample from each species of finfish and shellfish were weighed and successively treated with 4 ml aqua regia, 1.5 mL HF and 3 ml H2O2 in a hermetically sealed PIFE reactor, inside a microwave oven, at power levels between 330-550 Watt, for 12 min to obtain a clear solution. The use of microwave-assisted digestion aears to be very relevant for sample dissolution, especially because it is very fast [18-20].
After digestion, 4 ml H2BO3 was added and kept in a hot water bath for 10 min, diluted with distilled water to make up the volume to 50 ml. Taking distilled water in place of biological samples and following all the treatment steps described above the blank process was prepared. The final volume was made up to 50 ml. Finally, the samples and process blank solutions were analyzed by ICP-MS. All analyses were done in triplicate and the results were expressed with standard deviation.
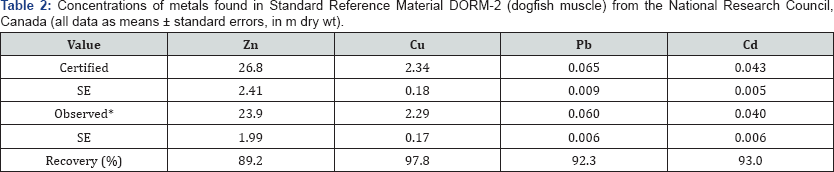
The accuracy and precision of our results were checked by analyzing standard reference material (SRM, Dorm 2). The results indicated good agreement between the certified and the analytical values (Table 2).
b) Statistical analysis
A logarithmic transformation was done on the data to improve normality. Analysis of variance (ANOVA) was performed to assess whether heavy metal concentrations varied significantly between sites and species; possibilities less than 0.01 (p < 0.01) were considered statistically significant. All statistical calculations were performed with SPSS 9.0 for Windows.
Results and Discussion
Heavy Metals in Finfish
The species-wise variation was not uniform for all the metals. Zn accumulated as per the order Liza parsia > Liza tade > Tenualosa ilisha > Polynemus paradiseus > Stolephorus commersonii (Figure 2). Cu accumulated as per the order Liza parsia > Tenualosa ilisha > Polynemus paradiseus > Liza tade > Stolephorus commersonii (Figure 3). Pb accumulated as per the order Liza parsia > Tenualosa ilisha > Polynemus paradiseus > Liza tade > Stolephorus commersonii (Figure 4). In case of Cd the order was Liza parsia > Tenualosa ilisha > Liza tade (Figure 5) [21] pointed out that the affiance of metal uptake from contaminated water and food may differ in relation to ecological needs, metabolism, and the contamination gradients of water, food and sediment, as well as other factors, such a salinity, temperature and interacting agents.
The selected finfish species in the present study have different food preference and different behavioral pattern e.g., Liza parsia, Liza tade, Polynemus paradiseus, and Stolephorus commersonii are resident fish species in the study area, while Tenualosa ilisha exhibit migration from coastal region (~ salinity = 20-30 psu) to freshwater system in the upstream zone of the River Ganga for breeding. These factors may be attributed to species-wise variation of heavy metals in the study zone.
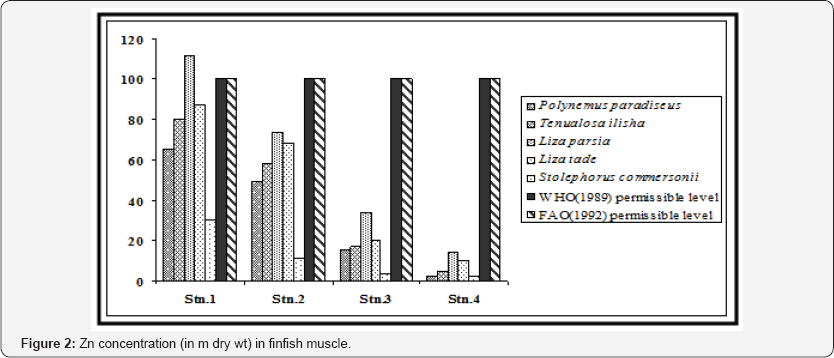
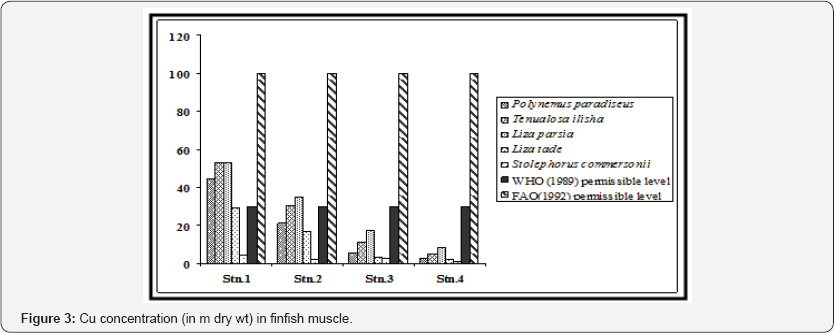
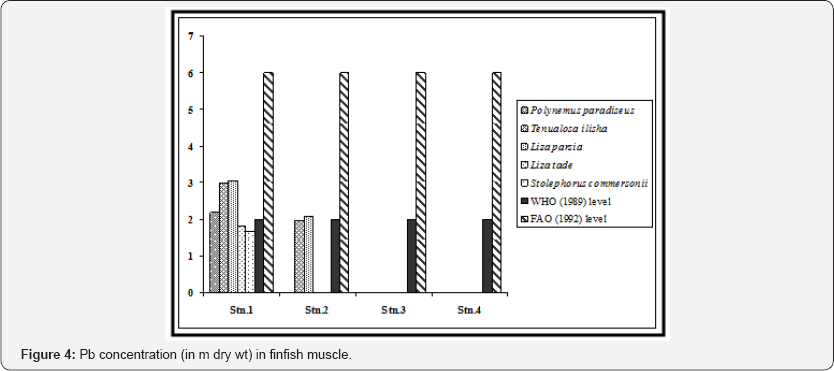
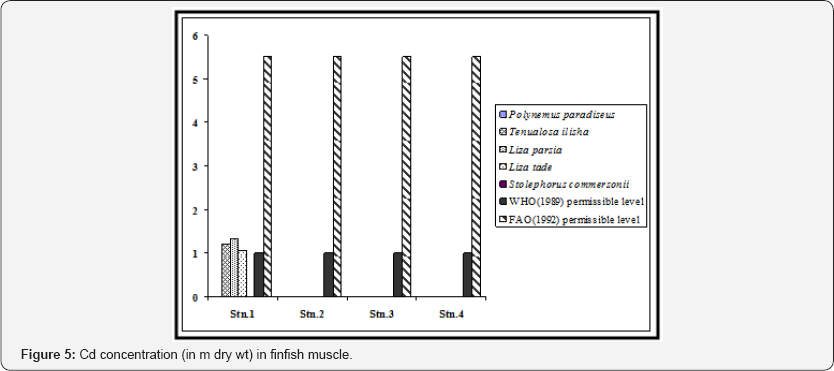
The spatial variation of bioaccumulation followed the order station 1 > station 2 > station 3 > station 4, which may be related to different degree of contamination in different location. The Zn concentration in all the finfish species (except Liza parsia in station 1) were found to be lower than the recommended maximum level allowed in food as prescribed by the World Health Organization [22]. Furthermore the selected heavy metals in finfish muscle (except Zn in Liza parsia in station 1) were also within the permissible limits for human consumption as indicated by the Food and Agricultural Organization [23]. In case of Cu, the concentration in Polynemus paradiseus (in station 1), Tenualosa ilisha (in stations 1 and 2), Liza parsia (in stations 1 and 2) were higher than the WHO (1989) limit, but the Cu concentration in all the fish samples were within the FAO (1992) limit. Pb concentration in Polynemus paradiseus, Tenualosa ilisha and Liza parsia in station 1 and Liza parsia in station 2 exceeded the WHO (1989) limit, but they were all within the limit of FAO (1992). Cd concentration in Tenualosa ilisha, Liza parsia and Liza tade in station 1 exceeded the WHO (1989) limit, but remained within the 0.05 - 5.5 m range of FAO (1992) for Cd in fish.
Heavy metals in shrimp
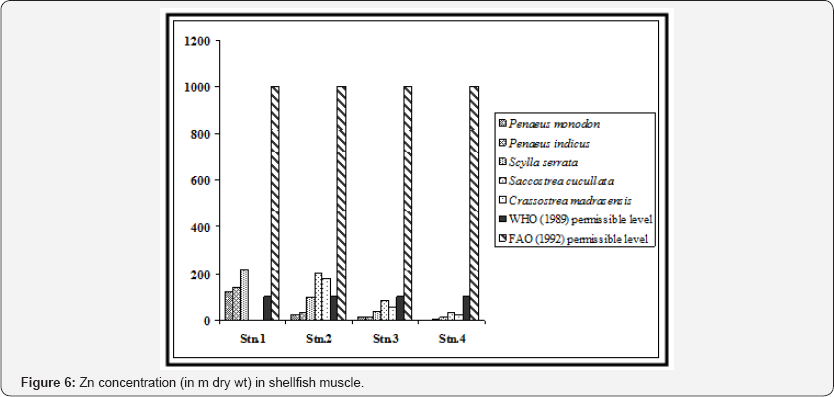
Zn being an essential element for normal growth and metabolism of animals, exhibited highest accumulation in the shrimp muscle when compared with the other three metals. According to the results obtained, the zinc levels in the shrimps from station 1 (Nayachar Island) facing the Haldia port-cum- industrial complex were highest, which were higher than the permissible level, i.e., 400 m in crustacean tissue [24].
These levels also exceeded the recommended maximum level of Zn allowed in food by World Health Organization which is 100 m [22]. The concentrations of Zn in shrimps from other three stations were much below the permissible limit for human consumption (Figure 6). We observed significant variations of Zn in shrimp muscle between the stations and also between the shrimp species (p < 0.01).
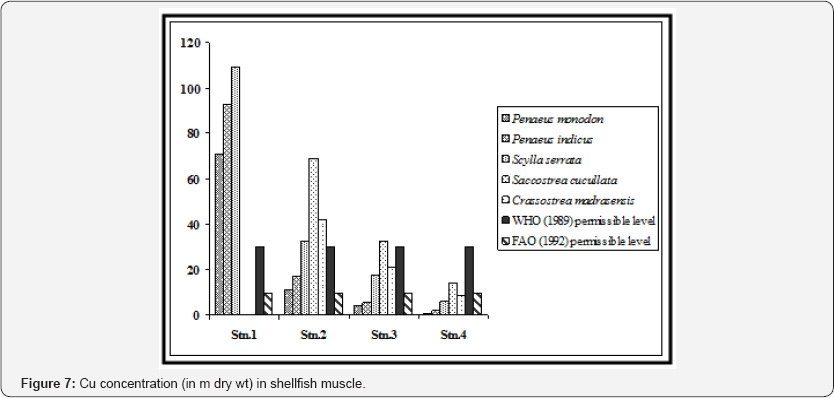
Levels of Coer in shrimps from the selected stations (except for station 1) ranged from 1.02±0.05 to 17.02±0.31 ng/g, far below the normal permissible range, i.e., 120 m as recommended for the crustacean tissue (Franklin, 1987). In case of station 1 the value ranged from 70.88±0.89 to 92.40±0.93 ng/g (Figure 7). These values are higher than the recommended value of Cu in food, which is 30 m as prescribed by WHO (1989).
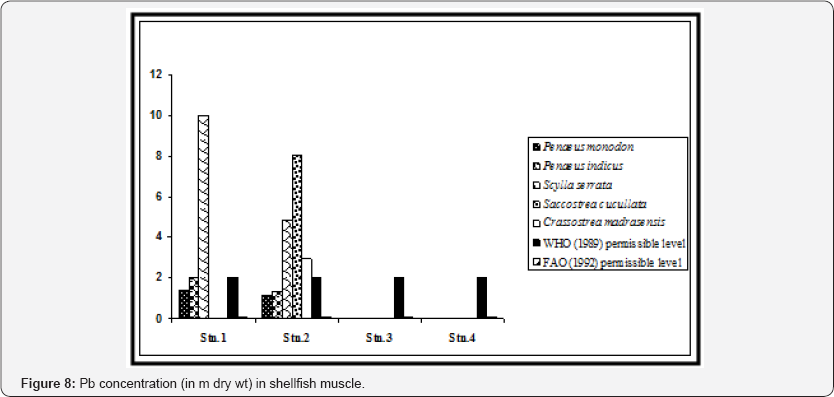
High concentration of Pb was observed in shrimp muscle from station 1 (1.38±0.10 in Penaeus monodon and 2.01±0.17 in Penaeus indicus). Station 2 is within the navigational route of the ships and tankers in the Hooghly channel through which the wastes of the upstream region find way to Bay of Bengal. Hence shrimps sampled from this station exhibited considerable concentrations of Pb in the muscle. Stations 3 (Gosaba) and 4 (Satjelia island) are in the central sector of Gangetic delta, almost without any industrial activities which may be attributed to low concentrations of lead in the shrimp samples collected from these areas (Figure 8).
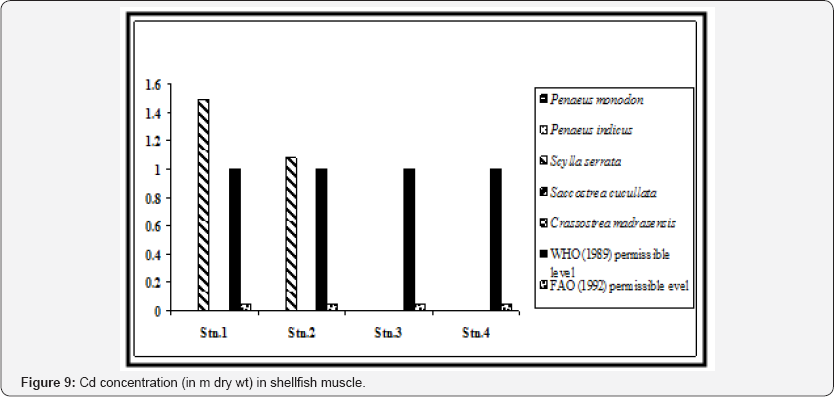
We also observed significant statistical differences in Pb concentrations in shrimp muscle between the stations and also between the selected shrimp species (p < 0.01). Except for station 1, levels of lead in shrimp muscle from three locations were below the permissible level which is 4.0 μg/g for crustacean tissue [24]. When compared with the recommended value of WHO (1989) in context to consumption of shrimp as food (2 m for Pb), the concentrations in Penaeus indicus in station 1 was above this level (Figure 4). All the shrimp species of stations 1 and 2 exhibited higher value than recommended by FAO (1992). Concentration of Cd was below detectable level (BDL) in shrimp tissue in all the sampling stations (Figure 9).
Heavy metals in crab
In Scylla serrata heavy metals accumulated as per the order Zn > Cu > Pb > Cd. The metal level in this group of crustacean is influenced by moulting as the process is known to play a major role in metal mobilization. Hence in the present study we considered marketable sized crab (aroximately 120 gm in weight) to avoid the interference of the ecdysis process. The concentrations of Zn, Cu, Pb and Cd in the muscle ranged from 11.65±0.10 - 213.66±1.60 for Zn, 6.18±0.11 - 109.23±1.00 for Cu, BDL - 9.99±0.79 for Pb and BDL - 1.48±0.07 for Cd respectively. These values are much higher (except stations 3 and 4) in most cases than the recommended levels of WHO (1989) for heavy metals in food (Figure 6-9). Reports on metal concentration in crabs under natural conditions for coastal waters of India are limited [25-27] and the present data therefore can serve as a baseline figure for the species particularly in the Gangetic delta region.
Heavy metals in oyster
The trace metal in soft mass of oyster results from the net balance between the process of metal uptake and metal loss [28]. The distribution of trace metals followed the order Zn > Cu > Pb> Cd. Oyster species could not be collected from station 1 because of their absence due to low aquatic salinity (~ 6.50 psu). The low salinity of station 1 due to freshwater discharge from the Farakka barrage has made the habitat unsuitable for the survival of oysters (that normally require salinity between 10 - 32 psu).
In the present study area oyster is not consumed as food by the local inhabitant, but considering its wide use as seafood in other parts of the world the soft body tissue was examined for Zn, Cu, Pb and Cd whose values ranged from 21.44±0.28 - 202.78±1.49 mg kg-1, 8.66±0.09 - 69.14±0.79 mg kg-1, BDL - 8.02±0.26 mg kg-1 and BDL respectively. The concentrations of Zn, Cu and Pb in oysters of station 2 exceeded the permissible limits of WHO (1989).
Spatial variation of heavy metals in finfish and shellfish
Heavy metal contamination of the environment has been occurring for centuries, but its extent has increased markedly in the last fifty years due to technological developments and increased consumer use of materials containing these metals. Pollution by heavy metals is a serious problem due to their toxicity and ability to accumulate in the biota [29]. There is still a general concern about the impact of metals in the aquatic environment [30]. Heavy metals have contaminated the aquatic environment in the present century due to intense industrialization and urbanization. The Gangetic delta is no exception to this usual trend. The rapid industrialization and urbanization of the city of Kolkata (formerly known as Calcutta), Howrah and the newly emerging Haldia complex in the maritime state of West Bengal has caused considerable ecological imbalance in the adjacent coastal zone [31]. The Hooghly estuary, situated on the western sector of the Gangetic delta receives drainage from these adjacent cities, which have sewage outlets into the estuarine system. The chain of factories and industries situated on the western bank of the Hooghly estuary is a major cause behind the gradual transformation of this beautiful ecotone into stinking cesspools of the megapolis [31]. The lower part of the estuary has multifarious industries such as paper, textiles, chemicals, pharmaceuticals, plastic, shellac, food, leather, jute, tyres and cycle rims [32]. In addition to industrial discharges, proliferation of tourism units has also contaminated the environment to a great extent particularly around Digha (station 2).
These units are point sources of heavy metals in the estuarine and coastal waters. Due to toxic nature of certain heavy metals, these chemical constituents interfere with the ecology of a particular environment and on entering into the food chain they cause potential health hazards, mainly to human beings. It was reported by several workers that the discharge of heavy metals into the sea through rivers and streams results in the accumulation of pollutants in the marine environment especially within shrimps [33]. Thus shellfish and shellfish products can be used for monitoring potential risk to humans because these are directly consumed by a large population [34]. Bioaccumulation patterns of metals in shellfish muscle can be utilized as effective indicators of environmental metal contamination [35,36]. According to many researchers, some shellfishes by virtue of their mobile nature are not fair indicator of aquatic contamination, but their regular consumption by human beings makes it absolutely necessary to monitor their different organs, particularly the muscles. The present study is therefore important not only from the safety point of view of human health, but also from the quality point of view as many of these shellfish species have high export value.
Of the four metals studied in the present work, Zn and Cu are essential elements while Pb and Cd are non-essential elements for most of the living organisms. The concentrations of zinc and coer in all the finfish and shellfish species were relatively higher, compared to the concentration of other metals in same samples. It can be explained because these metals (Cu and Zn) are essential elements required by animals for metabolic process. Zinc and coer aear to diffuse passively (probably as a soluble complex) the gradients created by adsorption of membrane surfaces and are found in blood proteins metallothioneins [37] concluded that different tissues of aquatic animals provide and/or synthesize non-exchangeable binding sites resulting in different accumulation levels.
The primary sources of Zn in the present geographical locale are the galvanization units, paint manufacturing units and pharmaceutical processes, which are mainly concentrated in the Haldia industrial sector (Station 1). Reports of high concentrations of Zn were also highlighted in the same environment by earlier workers [14,15,31].
The main sources of Cu in the coastal waters are antifouling paints [38], particular type of algaecides used in different aquaculture farms, paint manufacturing units, pipe line corrosion and oil sludges (32 to 120 m). Ship bottom paint has been found to produce very high concentration of Cu is sea water and sediment in harbours of Great Britain and southern California [39,40]. In the present study area, the major source of Cu is the antifouling paints used for conditioning fishing vessels and trawlers apart from industrial discharges (that is predominant around station 1). Station 2 (Digha) is not only the site for tourism and beach recreational activities, but it is also a major fish landing station in northeast coast of India, where large number of fishing vessels and trawlers contaminate the water with Cu. This is the reason why Cu was detected in considerable quantity in the fish muscle of station 2. Traces of Cu in the samples of stations 3 and 4 (which is totally an industry- free zone) may also be related to use of antifouling paints to keep the fishing vessels and passenger boats free from biofoulers.
Pb is a toxic heavy metal, which finds its way in coastal waters through the discharge of industrial waste waters, such as from painting, dyeing, battery manufacturing units and oil refineries etc. Antifouling paints used to prevent growth of marine organisms at the bottom of the boats and trawlers also contain lead as an important component. These paints are designed to constantly leach toxic metals into the water to kill organisms that may attach to bottom of the boats, which ultimately is transported to the sediment and aquatic compartments. Lead also enters the oceans and coastal waters both from terrestrial sources and atmosphere and the atmospheric input of lead aerosols can be substantial. Station 1 is exposed to all these activities being proximal to the highly urbanized city of Kolkata, Howrah and the newly emerging Haldia port - cum - industrial complex, which may be attributed to high Pb concentrations in the finfish, shrimp and crab muscle. Because of the low salinity of station 1 (~6.5 psu during the study period) oyster could not be found in this zone.
The main sources of Cd in the present geographical locale are electroplating, manufacturing of Cd alloys, production of Ni- Cd batteries and wielding. No trace of Cd was recorded in the shrimp muscle from stations 3 and 4, which are located almost in industry-free zone surrounded by mangrove vegetation. The accumulation of Cd in the soft body parts of oyster is a cause of concern as many Government departments and NGOs have initiated pilot scale oyster culture in the lower Gangetic region as means of alternative livelihood [41].
Conclusion
Sea foods are a cheap and the most accessible source of animal protein for the coastal inhabitants. However, there is a growing amount of evidence that seafood could be potentially harmful to human health as these are contaminated from heavy metals. In the Gangetic delta region of the tropics few studies have been carried out to assess the magnitude of contamination in commercially important finfish and shellfish species. This study revealed that the commercially important marine organisms in the lower Gangetic delta, at the apex of Bay of Bengal have been contaminated by heavy metals to a greater degree and exceeds the recommended safety levels for consumption at station 1 in the western sector owing to intense industrialization. Antifouling paints used for conditioning vessels and trawlers are also the major sources of heavy metals in the system that is ultimately deposited in the organisms. Strict regulation for the usage of heavy metals and frequent monitoring and controlling programmes are needed to check heavy metal contamination of marine organisms in the area that may potentially cause greater threats to human and ecological health [42].
References
- Gadzala Kopciuch R, Berecka B, Bartoszewicz J, Buszewski B (2004) Some Considerations about Bioindicators in Environmental Monitoring. Polish Journal of Environmental Studies 13 (5): 453-462.
- Alloway BJ, Ayres DC (1998) Chemical principles of environmental pollution. Publishers Stanley Thorens Ltd, London, UK.
- Garcia Montelongo F, Diaz C, Galindo L, Larrechi MS, Rius X (1994) Heavy metals in three fish species from the coastal waters of Santa Cruz de Tenerife (Canary Islands). Scientia Marina 58: 179-183.
- Jordao CP, Pereira MG, Bellato CR, Pereira JL, Matos AT (2002) Assessment of water systems for contaminants from domestic and industrial sewages. Environmental Monitoring Assessment 79: 75100.
- Cohen T, Hee S, Ambrose R (2001) Trace metals in fish and invertebrates of three California Coastal Wetlands. Marine Pollution Zbulletin 42: 232-242.
- Fergusson JE (1990) The heavy elements: Chemistry environmental impact and health effects. 614.
- Peerzada N, McMorrow L, Skiliros S, Guinea M, Ryan P (1990) Distribution of heavy metals in Gove Harbour Northern Territory Australia. Science of the Total Environment 92: 1-12.
- Castro H, Aguilera PA, Martinez JL, Carrique EL (1999) Differentiation of clams from fishing areas an aroximation to coastal quality assessment. Environmental Monitoring Assessment 54: 229-237.
- Usero J, Morillo J, Graccia I (2005) Heavy metal concentrations in molluscs from the Atlantic coast of southern Spain. Chemosphere 59: 1175-1181.
- Boudouresque CF, Verlaque M (2002) Biological pollution in the Mediterranean Sea: invasive versus introduced macrophytes. Marine Pollution Bulletin 44: 32-38.
- Iyengar G (1991) Milestones in biological trace elements research. Science of the Total Environment 1: 100.
- Krishnamurti JA, Nair R (1999) Concentration of metals in shrimps and crabs from Thane Bas sein creek system Maharastra. Indian Journal of Marine Science 28: 92-95.
- Bandhopadhayay J (1990) Natural Concerns. New Delhi: The Economic Times, India.
- Mitra A, Choudhury A (1992) Trace metals in macrobenthic molluscs of the Hooghly estuary India. Marine Pollution Bulletin UK 26 (9): 521522.
- Mitra A (1998) Status of coastal pollution in West Bengal with special reference to heavy metals. Journal of Indian Ocean Studies 5 (2): 135138.
- Chaudhuri AB, Choudhury A (1994) Mangroves of the Sundarbans, India.
- Date AR, Gray AL (Eds) (1988) Alications of Inductively Coupled Plasma Source Mass Spectrometry. (Blackie: Glassgow)
- Nadkarni RA (1984) Allocations of microwave oven sample dissolution in analysis. Analytical Chemistry 56: 22-33.
- Matusiewicz H, Sturgeon RE (1989) Present status of microwave sample dissolution and decomposition for elemental analysis. Progress in Analytical Atomic Spectroscopy 12: 21.
- De la Guardia M (Ed) (1990) Empleo de losHornos de Microondas en Quimica University of Valencia Spain.
- Romeo M, Siaub Y, Sidoumou Z, Gnassia-Barelli M (1999) Science of the Total Environment 232: 169-175.
- World Health Organization (1989) Heavy metals environmental aspects. Environmental Health Criteria , Geneva, Switzerland.
- FAO (1992) Food standard programme. Codex Alimentarius Commission 1: 114-190.?
- Franklin A (1987) Aquatic environmental monitoring report. MAFF Direct Fishery Research Lowestoft 16: 38.
- Zingde MD, Singbal SYS, Moraes CF, Reddy CVG (1976) Arsenic, copper, zinc and manganese in the marine flora and fauna of coastal estuarine waters around Goa. Indian Journal of Marine Science 5: 212.
- Matkar V, Ganpathy S, Pillai KC (1981) Distribution of zinc, coir, manganese and iron in Bombay Harbour bay. Indian Journal of Marine Science 10: 35.
- Qasim SZ, Sen Gupta R (1988) Some problems of coastal pollution in India. Marine Pollution Bulletin 19: 100.
- Rainbow PS (1990) Heavy metal levels in marine invertebrates. In: RW Furness and PS Rainbow (Eds) Heavy metals in the marine environment p. 67-79.
- Islam MD, Tanaka M (2004) Impact of pollution on coastal and marine ecosystems including coastal and marine fisheries and aroach for management: a review and synthesis. Marine Pollution Bulletin 48: 624-649.
- Grosell M, Brix K (2005) Introduction to the special issue on mechanisms in metal toxicology. Aquatic Toxicology 72: 3-4.
- Mitra A, Choudhury A (1993) Seasonal variations in metal content in the gastropod Nerita articulata (Gould). Indian Journal of Environmental Health NEERI 35(1): 31-35.
- UNEP (1982) Pollution and the marine environment in the Indian Ocean. UNEP Regional Seas Programme Activity Centre (Geneva: Switzerland)
- Yusof AM, Yanta NF, Wood AKH (2004) The use of bivalves as bioindicators in the assessment of marine pollution along a coastal area. Journal of radioanalytical and Nuclear Chemistry 259(1): 119-127.
- Subramanian R, Sukumar A (1988) Biological reference materials and analysis of toxic elements. Fresenius Journal of Analytical Chemistry 332: 623-626.
- Atchinson GJ, Murphy BR, Bishop WE, McIntosh AW and Mayes RA (1977) Trace metal concentration of bluegill (Lepomis macrochirus) from two Indiana Lakes. Transactions of the American Fisheries Society 106: 637-640.
- Larsson A, Haux C, Sjobeck M (1985) Fish physiology and metal pollution: Results and experience from laboratory and field studies. Ecotoxicology and Environmental Safety 9: 250-281.
- Carbonell G, Tarazona J (1994) Toxicokinetics of Cu Aquatic Toxicology . 29: 213-221.
- Goldberg ED (1975) The mussel watch - a first step in global marine monitoring. Marine Pollution Bulletin 6: 111.
- Bellinger E, Benhem B (1978) The levels of metals in Dockyard sediments with particular reference to the contributions from ship bottom paints. Environmental Pollution Assessment 15(1): 71-81.
- Young DR, Alexander G, McDermott Ehrlich D (1979) Vessel related contamination of southern California harbours by coer and other metals. Marine Pollution Bulletin 10: 50-56.
- WWF India (2006) Edible Oyster Culture in Indian Sundarban. (In A Mitra and K Banerjee (Eds) Training manual on non-classical uses of mangrove resources of Indian Sundarbans for alternative livelihood programmes. Sunderbans Landscape Project Canning Town West Bengal: India 11-28.
- Enoyer ER (1992) Semi quantitative analysis of environmental materials by laser-sampling inductively coupled plasma mass spectrometry. Journal of Analytical Atomic Spectrometry 7: 11-87.






























