Cation and Anion Electro-Migration during Electro-Kinetic Treatment for Nickel Chloride Contaminated Soil
Muhammad Naveed Arshad, *Waqas Zulfiqar, Hafiz Muhammad Umair Arshad
Department of Physics, University of Agriculture Faisalabad, Pakistan
Submission: May 22, 2017; Published: June 16, 2017
*Corresponding author: W Zulfiqar, Department of Physics, University of Agriculture Faisalabad, Pakistan, Email: wz.kamoka@gmail.com
How to cite this article: M Naveed A, Zulfiqar W, Arshad M U. Cation and Anion Electro-Migration during Electro-Kinetic Treatment for Nickel Chloride Contaminated Soil. Int J Environ Sci Nat Res. 2017; 2(5): 555597. DOI: 10.19080/IJESNR.2017.02.555597
Abstract
Electro-kinetic (EK) technique is the study of removal of contaminants in soil specimen under the influence of potential gradient. This technique works when the anions and cations get mobilize towards their respective electrodes inside the specimen, called electro-migration. The purpose of this lab-scale study was to discuss the Ni2+ and Cl- ionic movement inside the soil specimen for remediation cause. Two types of electro-kinetic experiments were designed depending upon the catholyte solution. The experimental results show that the maximum amounts of anions were migrated towards/into anode compartment. The statistical results conclude that Cl- ions removal efficiencies for EXP- 1 (distilled water used as catholyte) and EXP-2 (0.1 M of nitric acid HNO3 used as catholyte) were 52% and 73% respectively.
High removal efficiency for EXP-2 shows that 0.1 M of nitric acid (HNO3) separated the anions by breaking their adhesive bonds with soil particles. Moreover the non-toxic chemical agent reduces the solubility of minerals/metals into the soil and new coming minerals are not allowed to make adhesive bonds with soil particles. In contrast, the statistical results for Ni2+ ions concentration did not show desire conclusion. The removal efficiencies of Ni2+ ions were just 0.51% and 0.82% for EXP-1 and EXP-2, respectively, under the used experimental conditions. The effect of 0.1 M of nitric acid in cathode compartment as enhancing agent also did not speed up the cation migration. The less removal efficiency was due to the high adsorption tendency of Ni2+ ions in the form Ni (OH)2 precipitate inside the soil.
Introduction
The presence of heavy metals in soil is one of the major problems for developed as well as under-developed world. These metals have become major concern in present age because of their high toxicity. The excess use of sewage sludge, waste water irrigation, pesticides, and discharge vehicle exhausts, mining, metal casting and smoke have resulted in addition of heavy metals in soil [1,2]. The heavy metal such as nickel (Ni) is the cause of several diseases like lung cancer, cardiovascular disease, neurological deficits and developmental deficits in childhood and high blood pressure [3]. The discharge of heavy metals in polluted soil is an extended source of pollution for groundwater and environment. Their directly or indirectly adverse effect on human, animal health and plant growth are very long term. In order to avoid their harmful effects there is a need to remove these heavy metals from soil [4].
There are several remediation methods have been in use, since twentieth century, from soil including soil washing, solidification and electro-kinetic remediation. Soil washing is a mechanical technique used for removal of heavy metals from soil [5]. However, there are concerns over soil washing including this technique does not immobilize the contaminants. Consequently, it is hard to remove maximum amount of contaminants from soil. Likewise, solidification is another remediation method and done by adding a binding agent to contaminated material. The binding agent reduces the chemical bonding and solubility of the material and gives physical stability to contaminants. However the limitation for solidification is, this technique cannot be useful for oxy-anions metals and the metals have high solubility of hydroxides [6].
The electro-kinetic remediation technique is the only remediation method which is useful for any type of soil and contaminant. Electro-kinetic is the physical and chemical behavior of charge migration and fluid migration through porous medium under the application of potential gradient. In electro- kinetic two types of mechanical flows including electrical and hydraulic are coupled together for the transport of contaminants. There are three major mechanism involved in electro-kinetics i.e. electro-migration, electro-osmosis and electrophoresis [7,8]. Electro-migration is the transport of ionic specie under the application of applied current to pore medium/soil. When electric current is applied across the soil the positive ions move towards cathode and negative ions move towards anode. Electro-migration phenomena depends upon the ionic size i.e. the ions with large size such as potassium, copper and nickel are less mobilize than ions which has smaller size like sodium. Moreover, as the concentration of ions increase the process of electro-migration become fast [9]. Likewise, electro-osmosis is the movement of water under the application of applied current. When electric current is applied to soil, sandwich between two electrodes, the water get mobilize and migrated from anode to cathode part of specimen.
Material and Methods
The soil specimen, with pH 6, was collected from agriculture field of Postgraduate Agriculture Research Station (PARS), University of Agriculture Faisalabad. These samples were placed into the oven at 100oC for one hour. After dry out process the soil was grained with mortar in order to obtained uniformity in soil particles. The larger particles were sieved through mesh of 0.2 cm of pore size and mixed properly. The soil specimen was intentionally contaminated with 5:1000 ratios of nickel chloride (NiCl2) and soil, in grams, and mixed properly in order to get homogenously distribution of contamination. Moreover, 250 ml of distilled water was added to this mixture to create paste like formation. This paste then placed into soil compartment of electro-kinetic cell. Electro-kinetic apparatus was made up of PVC tube of dimensions 30 cm length and 7.5 cm diameter and the ends of tube were attached with two joints. The apparatus consisted of three compartments i.e. soil compartment, anode compartment and cathode compartment. The length of soil compartment was 12cm and anode and cathode compartment of 9 cm each. At cathode compartment there was an outlet pipe which is used to obtain electro-osmotic flow. Two types of electro-kinetic experiments were designed with same procedure. When the soil was sandwiched between two electrodes, the anolyte and catholyte were poured into both anode and cathode compartments, respectively.
In first type of experiment, EXP-1, the equal amount of distilled water i.e. 440 ml was poured in both compartments. In second type of experiment, EXP-2, the 0.1 M of nitric acid (HNO3) solution was used in cathode compartment as enhancing agent. The actual flow of water was from anode to cathode but by reversing the polarities the flow could reverse its direction. The length of soil compartment was 12 cm and 12 V was applied to soil via electrodes. In other words we may say that the potential gradient of 1V/cm was applied to soil specimen. In order to check the current flow inside the specimen the potential difference was measured at both of electrodes within beginning hour. Likewise the variation in potential difference across the soil specimen was measured at six points from anode to cathode, after every 24 hour, in order to check ionic resistance. The small current was measured across the soil with the help of ammeter. The electric field was applied for eight days. The potential across electrode, variation in potential of soil and small current was measured daily.
After eight days treatment the soil sample was separated from electro-kinetic cell. The treated soil specimen was then put in polythene bag and marked anode and cathode on its end. The soil specimen was segmented into five parts each of 2.4 cm in length. The soil was air dried for half an hour and then put into oven at 100oC for one hour. The soil samples as well as reference sample (R) were again grinded by using mortar and sieved through the mesh of for gaining uniformity in samples. The chloride ions were measured by using Argentometry titration method. For Argentometry titration method, take 10 g of segmented piece with 100 ml of distilled water in a beaker and put two drops of 5% of K2Cr2O7 in it. Shake the beaker with 2 Hz frequency for 10 minutes and filtered. Then put the 0.02 N solution of AgNO3 in burette and note the upper reading. Meanwhile the beaker must be under the burette, open the tap in such a way drop by drop solution of AgNO3 0.02 N falls in to the beaker. When solution changing its color into reddish brown then closes the burettes tap and write down the lower reading.
The Ni2+ions were extracted from soil specimen with 1 M of nitric acid (HNO3) solution i.e. 63 ml of nitric acid was added to 1000 ml of water. Then 10 g of soil from each sample from anode to cathode and reference was added in 100 ml of 1M of HNO3. The samples were shacked for half an hour with 3.6 Hz frequency and then filtered through What No. 2 filter paper. In order to find Ni2+ ions concentration the prepared samples were tested by Atomic Absorption Spectrophotometer (Hitachi Polarized Zeeman AAS, Z-8200, Japan).
Results and Discussion
Chloride (Cl") ions Electro-Migration
During electro-kinetic experimental treatment, under the action of potential gradient, the positive and negative ions migrate towards cathode and anode respectively. The movement of anions is due to ionic electro-migration towards anode. However, the direction of electro-osmotic flow is always in the direction of anode to cathode due to negative surface charges of soil. Consequently, the concentrations of chloride ions were high near anode region of soil specimen, as also shown in (Figure 1). The high concentration of chloride ions near cathode was due to absorbance of chloride ions near cathode surface. Initially the Cl- ions contamination in reference sample was 480 mg/ kg for both EXP-1 and EXP-2. However, after the completion of electro-kinetic treatment the concentration of Cl- ions were less than references for both of experiments. (Figure 1) shows the mobility trend of chloride ions concentration in five equal segments of soil for EXP-1. The concentration of chloride ions were greater near anode, as for soil segments near anode the graph was showing a downward trend. In the middle section of soil specimen ionic concentration was at its lowest point, as all anion like Cl“ moved towards anode. The Cl- ions mobility trend then again gradually increased towards cathode portion, it was due to the fact that during electro kinetic experiment some of the chloride ions might be absorbed near cathode instead of migration [10].

Another important factor for high concentration of Chloride ions near cathode was the presence of calcium, sodium and magnesium in soil which form complexes such as [Ca Cl]+& [Mg Cl]+. The formation of these complexes were due to chloride ions wandering by electrolyte or came after repulsion from the anode. These phenomena also attribute for the lower concentration of chloride ions in middle section because chloride ions were attracted by anode or Ca2+ or Mg2+ ions and vacant the middle section moved towards cathode [11]. (Figure 2) is presenting the concentration of chloride ions in EXP-2. The concentrations of ions were greater at anode but some chloride ions were also present at cathode. In the middle section of soil concentration of chloride ions were very less because maximum number of ions moved towards anode or formed complexes with calcium and magnesium and moved towards cathode [12]. The Cl- ions removal efficiencies for EXP-1 and EXP-2 were 52% and 73% respectively.
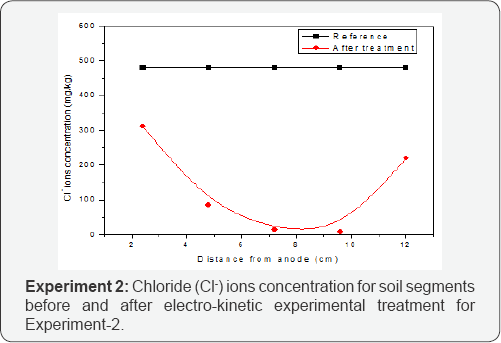
Nickel (Ni2+) Ions Electro-Migration
The nickel ionic concentration after electro-kinetic was analyzed by atomic absorption spectroscopy (AAS). The soil was segmented into five equal parts. These samples were digested with 1M and 3M nitric acid (HNO3). Both, 1M and 3M digested molarities, gave same nickel concentration for treated segments. Hence there was no any concentration difference either digested by 1M or 3M of nitric acid (HNO3). The reference sample was collected from Postgraduate Agriculture Research Station (PARS), Pakistan prior to application of electric current. The initial concentration of Ni in soil was 69.9 mg/kg for EXP-1 and 70 mg/kg for EXP-2. The difference in mass balance was due to in-homogeneity of soil sample which was used for testing. The results conclude that the concentration of Ni ions in soil segments near cathode was high, however not too much, in both experiments because of high pH regions exist near cathode precipitated the metal ions in this section. Therefore low pH of soil was responsible for migration of nickel ions [13]. In both of experiments, with and without use of enhancing agent, the concentration of Ni throughout the soil specimen did not vary with reference as thoughts. The effect of enhancing agent did not speed up the migration. However, the high molarities of enhancing agent may enhance remediation results [11].
The presence of Ni as a single contaminant in the soil also fall the migration rate. When multiple contaminants were present in the soil such as addition of chromium and cadmium the migration rate for Ni ions could climb up in soil [13]. The result shows that the Ni2+ metal ions were travelled at cathode side portion of soil, however, sufficient amount of Ni2+ ions were also observed at anode side portion of soil. (Figure 3) presenting the migration trend of Ni2+ ions from anode to cathode portion of specimen, at end of the EXP-1. The movement of cations such as nickel ions in electro-kinetic experiment was towards cathode and it precipitated in the form of nickel hydroxide Ni (OH)2 towards cathode compartment portion. The nickel hydroxide Ni (OH)2 precipitation was also seen by unaided eyes during the treatment inside the cathode compartment water and also inside soil near to cathode compartment.
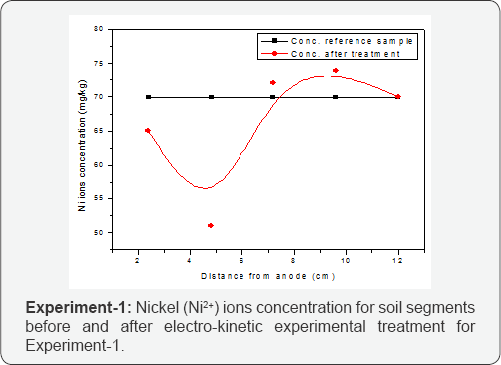
Consequently nickel ions concentration was present in maximum amount at cathode portion of specimen. Furthermore,(Figure 3) is also representing a little amount of metal ions present in soil near anode. This phenomena happened due presence of nitrates and chloride in soil, which form complexes with Ni such as [NiCl]“or [NiNO3]“. The formation of these complexes was for small period of time and moved towards anode. When these complexes reached at anode they dissolved in the soil increasing the amount of nickel and chloride near anode. Another phenomenon that also attributed the migration of nickel from anode to cathode was variation in pH. The low value of pH near anode change the nickel in aqueous form and movement towards cathode started [13]. The removal efficiency of Ni ions was 0.51% for EXP-1 conducted without enhancing agent. The experiment conducted with enhancing agent shows the same results as the experiment conducted without enhancing agent. As shown in (Figure 4) the Ni2+ ions have mobility trend for EXP-2 same as in (Figure 3) for EXP-1. The removal efficiency for Ni2+ ions concentration for EXP-2 using enhancing agent was observed as 0.82%.
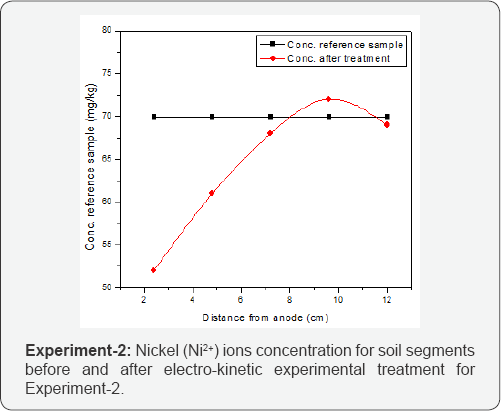
Conclusion
Two types of electro-kinetic experiments were designed in order to study the cation and anion movement under the application of potential gradient inside the soil. Under the action of potential gradient the anion and cation show the mobility trend towards their respective electrodes. The experimental results show that the maximum amounts of anions were migrated towards/into anode compartment. The statistical results conclude that Cl- ions removal efficiencies for EXP-1 and EXP-2 were 52% and 73% respectively. The movement of anions is due to ionic electro-migration towards anode side portion of electro-kinetic cell. Consequently, the concentrations of chloride ions were high near anode region. In the middle section of soil specimen negative charges concentration was at its lowest point, as all anion like Cl“ moved towards anode. A little high concentration of Chloride ions near cathode was due to the presence of calcium, sodium and magnesium in soil which form complexes such as [CaCl]+ and [MgCl]+.
The formation of these complexes were due to chloride ions wandering by electrolyte or came after repulsion from the anode. In contrast, the statistical results for Ni2+ ions migration did not show desire thoughts. The removal efficiencies of Ni2+ ions were 0. 51. and 0.82% for EXP-1 and EXP-2 respectively. In both of experiments, with and without use of enhancing agent, the concentration of Ni2+ ions throughout the soil specimen did not vary with reference as thoughts. The effect of 0.1 M of nitric acid in cathode compartment as enhancing agent also did not speed up the cation migration. The main factor behind less cations removal efficiency was the tendency of Ni2+ ions adsorption in the form Ni (OH)2 precipitate inside the soil. However, we may get the desired results for cations under different possible conditions.
References
- Zulfiqar W, MA Iqbal, MK Butt (2017) Pb2+ ions mobility perturbation by iron particles during electro-kinetic remediation of contaminated soil. Chemosphere 169: 257-261.
- Shi WY, HB Shao, H Li, MA Shao, SD (2009) Progress in the remediation of hazardous heavy metal-polluted soils by natural zeolite. Journal of Hazardous Materials 170(1): 1-6.
- Vona Y, A Arita, M Costa (2012) Carcinogenic metals and the epigenome understanding the effect of nickel, arsenic and chromium. Metallomics 4(7): 619-627.
- Li T, S Yuan, J Wan, L Lin, H Long, X, et al. (2009) Pilot-scale electro- kinetic movement on HCB and Zn in real contaminated sediments enhanced with hydroxypropyl-cyclodextrin. Chemosphere 76(9): 1226-1232.
- Abumaizar RJ and EH Smith (1999) Heavy metals contaminants removal by soil washing. Journal of Hazardous Material 70(1-2): 7186.
- Wuana RA, FE Okieime (2011) Heavy metals in contaminated soil, a review of sources, chemistry, risk and best available strategies for remediation. International Scholarly Research Notices 647-667.
- Acar YB, AN Alshawabkeh (1993) Principles of electro-kinetic remediation. Environmental Science & Technology. Journal of Hazardous Material 27(13): 2638-2647.
- Mitchel JK, K Soga (2005) Fundamentals of soil behavior 3rd edition. John Wiley and Sons, New York, USA.
- Hamnett R (1980) A study of processes involved in electro-reclamation of contaminated soils. Dissertation, University of Manchester, UK.
- Ottosen LM, CMD Ferreira I Christensen (2010) Electro-kinetic distillation of glazed ceramic tiles. Journal of Applied Electrochemistry 40(6): 116-117.
- Kim DH, BG Ryu, S Park, WS chang, K Baek (2009) Electro-kinetic remediation of Zn and Ni-contaminated soil. Journal of Hazardous Materials 165(1-3): 501-505.
- Kim DH, CS Jeon, K Baek, SW Ko, JS Yang (2009) Electro-kinetic remediation of fluorine-contaminated soil: conditioning of anolyte. Journal of Hazardous Materials 161(1): 565-569.
- Reddy KR, CY Xu, S Chinthamreddy (2001) Assessment of electro- kinetic removal of heavy metals from soils by sequential extraction analysis. Journal of Hazardous Materials 84(2-4): 279-296.






























