New Evaluation of Selenium: Mercury Ratios in Fish and Crabs from an Impacted Tropical Estuary, Southeastern Brazil
*Alice Bosco Santos and Wanilson Luiz Silva
Department of Geology and Natural Resources, University of Campinas, USA
Submission: May 05, 2017; Published: May 09, 2017
*Corresponding author: Alice Bosco Santos, Department of Geology and Natural Resources, University of Campinas, Institute of Geosciences, Campinas, Sao Paulo 13083870, Brazil, USA, Email: alicebosco@gmail.com
How to cite this article: Alice B S, Wanilson L S. New Evaluation of Selenium: Mercury Ratios in Fish and Crabs from an Impacted Tropical Estuary, Southeastern Brazil. Int J Environ Sci Nat Res. 2017;2(3): 555589. DOI: 10.19080/IJESNR.2017.02.555589
Abstract
Large variations in the Se:Hg molar ratios and their high elemental concentrations in different tissues from fish and crabs species was observed and deserves attention due to potential risky for wildlife. The results suggested that Se protection against Hg toxicity decrease as the size of specimen's increases and that the protective effect has a close relation with diet and grow pattern, notably for fish. This study showed that although strictly carnivorous eating habits may easily lead to Hg bioaccumulation, some species can present highest concentration of the contaminant in a less sensitive tissue, in terms of minimize adverse toxic effects.
Keywords: Crabs; Fish; Se:Hg; Molar Ratio; Tropical Estuary
Introduction
Patterns of Sight molar ratios in aquatic organisms in excess of1 are thought to counteract the adverse effects of Hg, protecting against Hg toxicity [1,2]. This protection has been observed in aquatic organisms since 1967, and one of the most observed effects is an apparent modification of the distribution pattern of Hg in different organs and tissues [1]. However, little is known about the Se:Hg molar ratio variability and its relationship with species and their eating habits in tropical estuaries, where Hg contamination is relatively common. In contrast to studies with fish, studies investigating the Se:Hg molar ratio in crabs are not well documented, although expose to Hg can be more relevant due to their diet and burrow habits. In the Santos-Cubatao Estuarine System (SE Brazil) Hg contamination was recorded in both sediment [3] and fish [4], and recently high levels of selenium were identified in its sediments [3].
The Santos-Cubatao Estuarine System in southeastern Brazil has a history of metal contamination since 1950's when the largest industrial complex in Latin America was established. This study presents the Se:Hg molar ratio in fish and crabs to understand whether there are different patterns of accumulation depending on their habits. Since investigations on the Se:Hg relationship in organisms from this area have not been reported previously, this issue is of great interest once fished constitutes one of the protein sources for 90% for the fishing community riverside [5].
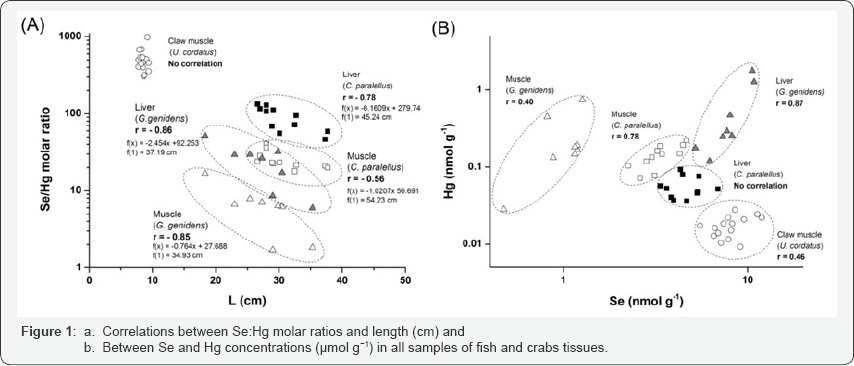
Samples of fish (Centropomus parallelus and Genidens genidens) and crabs (Ucides cordatus) were collected in Morrao river estuary from September 2010 to March 2011. This estuary is one of the most impacted by industrial activities and it was selected for this study because it is the main routes of direct discharge of industrial effluents from a large steel plant and fertilizer industries. Additional information about the specimens is shown in (Table 1). These species were chosen among the most widely consumed, representing 15% of the fishing caught in the Santos-Cubatao estuarine system [5]. For fish, muscle (fillet) and liver tissues were selected for analysis and for crabs (only male) the analysis was performed in the claw muscle.
Determination of total Hg contents was performed using the FI-CV-AAS technique, and total Se analysis was performed using ICP-MS. The results for certified reference materials DOLT-4 and DORM-3 for Se, and DORM-2 for Hg, showed good precision and accuracy for both analytical methods, with recovery between 87 and 102%. Concentrations in wet weight of Se, Hg and their molar ratio are shown in (Table 1). Selenium concentrations in both fish tissues (Table 1) were below the limit for because adverse biological affects such as 50 nmol g-1 and 38 nmol g-1 for fish and aquatic birds, respectively [6]. However, hazardous concentrations for aquatic birds were found in crab samples and deserve attention. In respect to Hg, the reference dose for chronic oral exposure for piscivorous avian is from 104 nmol kg-1 of bodyweight per day (21 μg kg-1 of bodyweight per day - USEPA 1997), which implies a maximum consumption of 900 grams of Genidens genidens for a 5 kg avian. So, selenium and Hg in species studied may represent risk of contamination for local wildlife. For human consumption, where only the concentration in muscle tissue is important, the results indicate Se as a potential hazard, if considered the reference dose for chronic oral exposure of 63.3 nmol kg-1 of bodyweight per day (0.005 mg kg-1 of bodyweight per day, USEPA [7].
Environmental Protection Agency USEPA [7] estimated a safe daily intake level of 0.1 μg kg-1 of bodyweight per day (0.498 nmol kg'1 of bodyweight per day) of methyl mercury in fish for human consumption. If considered that methyl mercury percentage in seafood is greater than 75% of the total mercury [8], this reference dose indicates a daily maximum consumption of Hg for a man with 60 kg of weight equivalent to 280 grams of Centropomus parallelus, 153 grams of Genidens genidens and 570 grams of Ucides cordatus. These amounts can easily be consumed daily by the local population and put on alert the consumption of these fish and crab species.
Higher selenium content than Hg was observed in all samples, with Se:Hg molar ratios ranging from 2 to 130 in fish, and between 307 and 984 in crabs (Table 1). These results suggested that these organisms and their predators must be protected against the adverse effects of Hg, if a 1:1 stoichiometry is considered sufficient for the protective effect of Se [2]. However, a substantial variation in the molar ratios within the individual species and between fish tissues was observed and deserves attention. The much higher molar ratios in claw muscles of Ucides cordatus specimens maybe related to the higher Se amounts and no Hg significant amounts in their diet items. Girling [9] considered the plant as the most responsible for the Se enter in the food chain, and Torres et al. [10] showed that for crabs the dietary exposure to Se played a very important role concerning the uptake of the Hg, which shows that it influences the uptake and accumulation patterns.
For omnivorous fish species (Genidens genidens) was observed that Se and Hg accumulated predominantly in the organ with metabolic activity more intense (liver), while for carnivorous species (Centropomus parallelus) higher Se and Hg contents were observed in the liver and muscle, respectively (Table 1). In fact, some studies on Hg concentrations in muscle and liver tissues of carnivorous fish and aquatic mammal’s species found higher Hg concentrations in muscles [11]. The fact that specimens of Genidens genidens (that live associated with the substrate and may ingest sediment) showed the highest concentrations of Se and Hg in liver tissue, which is the first organ that acts in the process of detoxification, corroborate the potential incorporation of these elements by the contaminated mud ingestion. However, other common diet habits that include algae, organic detritus, and benthic organisms are believed to be a more important pathway to fish contamination [12-14]. This results may indicate that strictly carnivorous eating habits can lead to a pattern of distribution of the contaminant that permit to stock the highest concentrations of Hg in a less sensitive tissue, in terms of minimize adverse toxic effects.
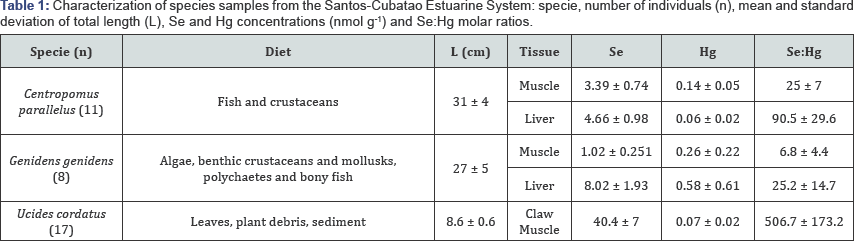
While correlation between Se:Hg and length showed that molar ratios might decrease with the increase of fish size (r = -0.56 and -0.78 for Centropomus parallelus, and -0.85 and -0.86 for Genidens genidens in muscle and liver tissues, respectively) (Figure 1A), no correlation can be observed in crabs which could be result of shell samples homogeneous width. It suggests a possible reduced Se protection in larger fish. This observation is unprecedented and seems to be consequent to their diet rich in selenium, although further study to understand the mechanism involved is necessary. To support these results in the (Figure 1A) is also being presented some prediction of the average length that each analyzed fish population should present Se:Hg molar ratio of 1 (protecting against Hg toxicity). These results show that Centropomus parallelus should present a length close to 54 cm for protective ratio effect in muscle tissue (45 cm for liver tissue) against 35 cm (37 cm for liver tissue) for Genidens genidens specimens. Between the fish species analyzed here, Centropomus parallelus raises, in average, maximum length of 72 cm while, Genidens genidens raises 42.5 cm (Fishbase, 2017). This observations point out that for Centropomus parallelus the risky of Se:Hg < 1 is bigger since the length for which ratio equal to 1 is expected to be well below the expected maximum size reached by the species.
Although strong correlation between Hg concentrations and length (range between 0.55 and 0.73) was already expected, since this metal has a good capacity of bioaccumulation [15], selenium concentrations did not suffer influence of the length (no correlation) but was strongly related to Hg concentrations in all muscle samples (Figure 1B). If Se bioaccumulation is not actually observed, these results may indicate that Se uptake should be related to dietary requirements in an attempt to reduce the toxic effects of Hg. The selenium correlations to mercury in muscle tissue is strongest in Centropomus parallelus (r = 0.78) follow for Genidens genidens (r = 0.40) and Ucides cordatus (r = 0.46). So, it is possibly that the first one ensures the better performance with higher uptake of Se when greater uptake of Hg, keeping it more protected from the adverse effects of the contaminant [16,17].
Conclusion
This study indicate that although extensive literature data presents strictly carnivorous fish species as most dangerous to consumption due to Hg bioaccumulation, some species may concentrate Hg in a less sensitive tissue with could minimize the adverse toxic effects. The same carnivorous specie presents a better performance with higher uptake of Se when greater uptake of Hg is drive. For different diet habits it was observed a similar behavior between Se:Hg molar ratio and length, with Se protection against Hg (smaller molar rations) being reduced in larger fish. Crabs samples exhibit the larger Se:Hg molar ratios (ranging between 307 and 984), being the most safe organisms analyzed due to the higher Se amounts in their diet habits.
Acknowledgement
We thank the Sao Paulo State Research Funding Agency - FAPESP (process 08-11511-8) for the financial support.
References
- Cuvin Aralar MLA, Furness RW (1991) Mercury and selenium interaction: a review. Ecotox Environ Safe 21(3): 348-364.
- Peterson SA, Ralston NVC, Whanger PD, Oldfield JE, Mosher WD (2009) Selenium and mercury interactions with emphasis on fish tissue. Environ Bioindic. 4(4): 318-334.
- Luiz-Silva W, Matos RHR, Kristosch GC, Machado W (2006) Variabilidade espacial e sazonal da concentracao de elementos-traco em sedimentos do sistema estuarino de Santos-Cubatao (SP). Quim Nova. 29: 256-263.
- Azevedo JS, Fernandez WS, Farias LA, Favaro DTI, Braga ES (2009) Use of Cathorops spixii as bioindicator of pollution of trace metals in the Santos Bay, Brazil. Ecotoxicology 18(5): 577-586.
- Gefe W, Amorim LFC, Amorim AC, Amorim AFC (2004) Aspectos socio- economicos da pesca artesanal na regiao da Baixada Santista (Socioeconomic aspects of artisanal fisheries in the Baixada Santista region). IV Congresso Brasileiro de Pesquisas Ambientais e Saude, Santos, 1321.
- Lemly AD (1996) Assessing the toxic threat of selenium to fish and aquatic birds. Environ Monit Assess 43(1): 19-35.
- USEPA (United States Environmental Protection Agency) (1991) Integrated risk information system: Selenium and compounds, USA Report, CASRN, USA, 7782: 49-2.
- Kannan K, Smith Jr RG, Lee RF, Windom HL, Heitmuller PT, et al. (1998) Distribution of total mercury and methyl mercury in water, sediment, and fish fro, South Florida estuaries. Arch Environ Contam Toxicol 34(2): 109-118.
- Girling CA (1984) Selenium in agriculture and the environment. Agr Ecosyst Environ 11: 37-65.
- Torres DP, Cadore S, Raab A, Feldmann J, Krupp EM (2014) Evaluation of dietary exposure of crabs to inorganic Mercury or methylmercury, with or without co-exposure to selenium. J Anal At Spectrom. 29: 1273-1281.
- Sorensen EM (1992) Metal poisoning in fish. (1st edn), CRC Press, Boca Raton, USA.
- Barwick M, Maher W (2003) Biotransference and biomagnification of selenium copper, cadmium, zinc, arsenic and lead in a temperate seagrass ecosystem from Lake Macquarie Estuary, NSW, Australia. Mar Environ Res 56(4): 471-502.
- Chapman PM, Adams WJ, Brooks ML, Delos CG, Luoma SN, et al. (2009) Ecological Assessment of Selenium in the Aquatic Environment. In: Summary of a SETAC Pellston Workshop; 22-28 February 2009; Pensacola. Society of Environmental Toxicology and Chemistry, Florida, USA.
- Kehrig HA, Seixas TG, Palermo E, Baeta AP, Castelo-Branco CW, et al. (2009) The relationships between mercury and selenium in plankton and fish from a tropical food web. Environ Sci Pollut Res Int 16(1): 1024.
- Harris R, Kidd K, Shanley J (2008) Bridging the knowledge gaps on the sources, speciation, fate and bioaccumulation of mercury in aquatic and terrestrial environments. Environ Pollut 154(1): 1-2.
- Kristan U, Arribere MA, Stibilj V (2013) Selenium species and their distribution in freshwater fish from Argentina. Biol Trace Elem Res. 151(2): 240-246.
- USEPA (United States Environmental Protection Agency) (1997) Mercury Study Report to Congress. Volume VII: Characterization of Human Health and Wildlife Risks from Mecury Exposure in the United States. Report: 452/R-97-009, USA.






























