Aggregation of TiO2 Nanoparticles in Aqueous Media: Effects of pH, Ferric Ion and Humic Acid
*Penghua Wang
Nanyang Environment & Water Research Institute (NEWRI), Nanyang Technological University, Singapore
Submission: February 14, 2017; Published: March 08, 2017
*Corresponding author: Penghua Wang, Nanyang Environment & Water Research Institute (NEWRI), Nanyang Technological University, 1 Cleantech Loop, Clean Tech One, Singapore 637141, Singapore
How to cite this article: Penghua W .Aggregation of Tio2 Nanoparticles in Aqueous Media: Effects of pH, Ferric Ion and Humic Acid. Int J Environ Sci Nat Res. 2017;1(5): 555575. DOI: 10.19080/IJESNR.2017.01.555575
Abstract
The aggregation behavior of TiO2 nanoparticles was investigated through the measurement of aggregate size and zeta potential under the influence of pH, ferric ion and HA. It is of great significance for assessment of the aggregate removal of TiO2 nanoparticles. The point of zero charge (PZC) for TiO2 (P25) nanoparticles in suspension was found to be at about pH 6.2, which increased to around pH 9.0 with the presence of ferric ion (1.0 mM), decreased to less than pH 3.0 with the presence of HA (5.0 mgC/L), and decreased to around pH 4.6 with the presence of both ferric ion (1.0 mM) and HA (5.0 mg C/L). At PZC with near zero charge density on the surface of the TiO2 nanoparticles, the TiO2 nanoparticles have the most tendency of aggregation which results in the maximum aggregate size of TiO2 nanoparticles in suspension.In the region with large positive and negative zeta potential values, it was unable to promote aggregation of TiO2 nanoparticles due to the high repulsion forces among the TiO2 nanoparticles. The formation of Fe(III)-hydroxy colloids could result in enhancement in the aggregation process. HA acting as an aggregation energy barrier could inhibit the aggregation of TiO2 nanoparticles. With the presence of both ferric ion and HA in suspension, the dominant role for aggregation inhibition or promotion will depends on the concentration of ferric ion and HA. Aggregation will be promoted when the bridging effect of Fe (III)-hydroxy colloids overcomes the effect of repulsion forces among nanoparticles and the aggregation energy barrier of HA, vice versa. The settling/removal of aggregates with larger size would be easier, while it is an issue when the TiO2 nanoparticles in suspension are present in aqueous media stably.
Keywords: Titanium dioxide (TiO2); Aggregation; Ferric ion; Humic acid
Introduction
As one of the most common nanomaterials, TiO2 nanoparticles have found their industrial and household applications in cosmetics, sunscreens, pharmacy, socks, paints and coatings, catalyst, even food and many other fields, which are expected to continue to grow in the next decade [1]. However, the release of TiO2 nanoparticles into the aquatic environment is inevitable during the process of manufacturing, producing or consuming TiO2 containing products. This draws concerns of potential impact of TiO2 nanoparticles exposure to human and environment, although further investigation is required to understand their potential detailed adverse effects on human beings and the aquatic ecological environment [1,2].
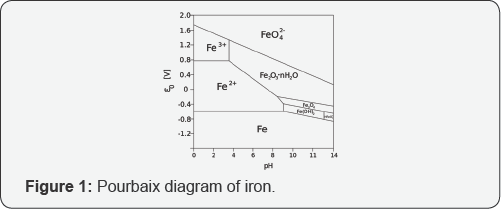
Recently, great attention has been paid to investigate the aggregation behavior of TiO2 nanoparticles in the aquatic environment [3-9]. This is important to quantify and analyze the impact of TiO2 nanoparticles. Upon releasing into the aquatic environment, TiO2 nanoparticles rarely exist as individual dispersed particles. The interactions between TiO2 nanoparticles and materials present in water under various environmental conditions can affect the aggregation fate and transport of TiO2 nanoparticles. pH is a critical parameter to determin the surface charge of TiO2 nanoparticles, followed by the aggregate size change [10-12]. The investigation of the effect of metal ions on the aggregation behavior of TiO2 nanoparticles focuses on Na and Ca ions [10,11,13,14]. Figure 1 shows Pourbaix diagram of iron. Ferric ions are the common constituents present in water. The speciation, solubility and transport of ferric ions depend on the general composition of the water as well as the presence of metal complexing ligands. However, effects of ferric ions on the surface properties and aggregation of TiO2 nanoparticles have been rarely investigated yet. Natural organic matters (NOMs) are ubiquitous in nearly all the natural waters and municipal wastewaters. With large active surface area-to-volume ratio, the TiO2 nanoparticles tend to adsorb onto NOM [11,15]. Metal ions and NOMs can present in water simultaneously. However, to the best knowledge of the author, limited and few studies were conducted on the investigation of aggregation behavior of TiO2 nanoparticles in complex aqueous conditions with the presence of metal ions and NOMs under different pH (Figure 1).
In view of above, the main objective of this study was to investigate the aggregation behavior of TiO2 nanoparticles in water with the presence of metal ions and/or Humic acid (HA, one of the most significant fractions of NOM) under different pH, in terms of aggregate size and zeta potential. It aimed to obtain insight to interprete the stability of TiO2 nanoparticles followed by developing strategies to remove them from the aquatic environment.
Experimental
Chemicals and Materials
Millipore Co. MilliQ (MQ) water with resistivity of 18.2 Ω cm was used throughout the study unless otherwise stated. Evonik P25 (hereafter referred as P25) was provided by Jebsen & Jessen Chemicals. The TiO2 powder consists of non-porous spheres with average particle size of 21 nm and specific surface area of 50 ± 15 m2/g. HA sodium salt and iron (III) chloride (FeCl3•6H2O) were purchased from Sigma-Aldrich. HA stock solution was prepared by dissolving HA in MQ water (pH 11 adjusted by 1 M NaOH solution) followed by stirring for 24 h, then filtered through a 0.45 μm cellulose acetate syringe membrane filter. The HA concentration for its stock solution was 134 mgC/L expressed in terms of total organic carbon (TOC) concentration (mgC/L) measured using a Shimadzu ASI-V TOC Analyzer. The stock solution for FeCl3•6H2O (50 mM) was prepared by dissolving FeCl3•6H2O in MQ water. Both stock solutions were stored in dark at 4°C before use.
Aggregation Experiments
50 mg/L of P25 suspension was prepared in MQ water, followed by ultrasonic treatment for 2 h. The pH value for the P25 suspension was adjusted using 1 M hydrochloric acid (HCl) and 1 M sodium hydroxide (NaOH) solutions. To investigate the effect of ferric ions and/or HA on the aggregation of TiO2 nanoparticles, different amounts of FeCl3•6H2O and/or HA stock solutions were added to P25 suspension followed by pH adjustment and magnetic stirring for 24 h before zeta potential and aggregate size measurements.
Analytical Method
Zeta potential and aggregate size for TiO2 nanoparticles in suspension were measured on a Malvern Nano-ZS Zetasizer. The zeta potential was derived from the electrophoretic mobility using Smoluchowski model [16],while the average aggregate size was measured from the diffusion coefficient through Stokes-Einstein equation [17]. The measurements were performed at 25oC, and each sample was measured in triplicate. After mixing with ferric ions and/or HA for 24 h, TiO2 nanoparticles were washed with MQ water for three times and dried for X-ray photoelectron spectroscopy (XPS) analysis on a Kratos Axis Ultra spectrometer using a monochromatized Al Kα (1486.71 eV) X-ray source to study the surface chemical compositions and bonding states of the TiO2 nanoparticles. All the spectra were calibrated with the adventitious carbon at 284.8 eV.
Results and Discussion
Effect of pH
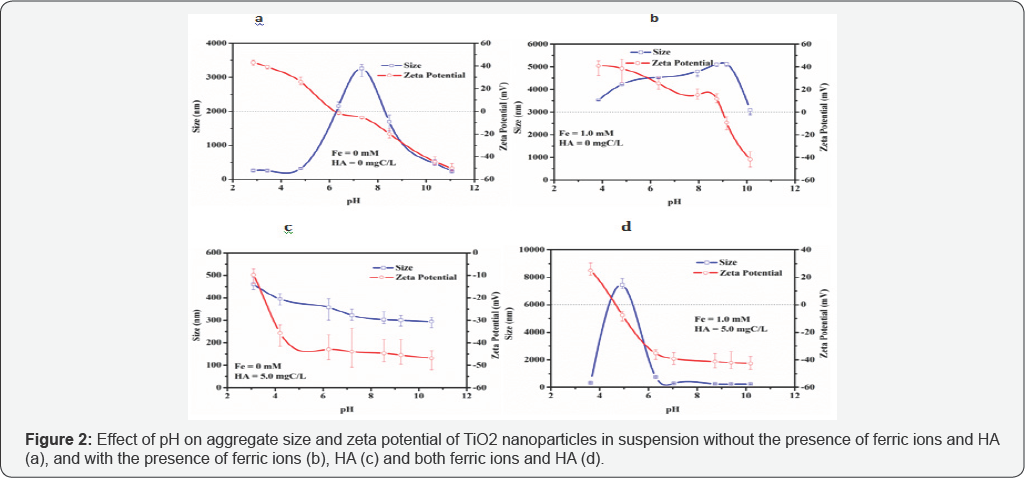
As shown in Figure 2 (a), the point of zero charge (PZC) for TiO2 nanoparticles in suspension was found to be at about pH 6.2. As an amphoteric matigerial, the TiO2 surface is positively charged at a pH lower than PZC and negatively charged at a pH higher than PZC, which can be observed in Figure 2. The decrease of zeta potential to more negative values for TiO2 nanoparticles with increasing pH could be due to the fact that the OH- concentration increased and thus bond with H+ on the surface active sites of the TiO2 nanoparticles. The charge density on the surface of the TiO2 nanoparticles is zero at PZC, thus the TiO2 nanoparticles have the most tendency of aggregation in accordance with DLVO theory [10,15], which results in the maximum aggregate size of TiO2 nanoparticles in suspension. In the region with large positive and negative zeta potential values, it was unable to promote aggregation of TiO2 nanoparticles due to the high repulsion forces among the TiO2 nanoparticles. It is concluded that the TiO2 nanoparticles in suspension were considered to be stable and settling/removal of aggregates would be an issue i n the region far away from PZC (Figure 2).
Ferric ion was added to TiO2 suspension to investigate its effect on TiO2 nanoparticles aggregation and zeta potential of TiO2 suspension. The PZC for TiO2 nanoparticles in suspension increased to around pH 9.0 with the presence of 1.0 mM ferric ion Figure 2(b). Severe TiO2 aggregation occurred at pH 8-9 due to the decrease of repulsion forces among nanoparticles in the region near to PZC with low zeta potential. At pH < 6 and > 9 with high positive and negative zeta potential respectively, strong repulsion forces among nanoparticles could prevent their aggregation. However, TiO2 nanoparticles aggregation was observed. It is noted that the hydroxyl colloids Fe(OH)3 act as the principal species of Fe(III) in the suspension [18,19]. Thus, it is assumed the formation of Fe(III)-hydroxy colloids could result in enhancement in the aggregation process. The bridging effect of Fe(III)-hydroxy colloids dominated over electrostatic repulsion existed among nanoparticles and led to the formation of large aggregates.
The PZC for TiO2 nanoparticles in suspension decreased to less than pH 3.0 with the presence of 5.0 mg/L HA, and the TiO2 nanoparticles were negatively charged at pH 3-10 due to HA adsorption on the surface of TiO2 nanoparticles (Figure 2c). The aggregate size of TiO2 nanoparticles decreased slightly with pH increasing along with the increase of negative zeta potential values due to the repulsion forces among the TiO2 nanoparticles. It can be concluded that HA could act as aggregation energy barrier to inhibit the aggregation of TiO2 nanoparticles [10].
The PZC for TiO2 nanoparticles in suspension was at around pH 6.2 (Figure 2a), which increased to around pH 9.0 with the presence of ferric ion (Figure 2b), and decreased to less than pH 3.0 with the presence of HA (Figure 2c). As shown in (Figure 2d), with the presence of both ferric ion (1.0 mM) and HA (5.0 mgC/L), the PZC of TiO2 nanoparticles in suspension was at around pH 4.6. Due to the weak repulsion forces among TiO2 nanoparticles at PZC, significant aggregation was observed. With the increase of pH from PZC onwards, aggregate size of TiO2 nanoparticles decreased rapidly. This could be attributed to the effect of HA acting as an aggregation energy barrier where it disregarded the unstable effect of ferric ions that could have posed in the suspension. This could be explanatory by the observation of reduced aggregation effect occurred at pH 6 onwards, which was contrary with that in mono-system of ferric ions (Figure 2b). These results suggested that, compared to ferric ion, HA exhibited dominant effect on aggregate size and zeta potential of TiO2 nanoparticles in suspension with the presence of both ferric ion and HA.
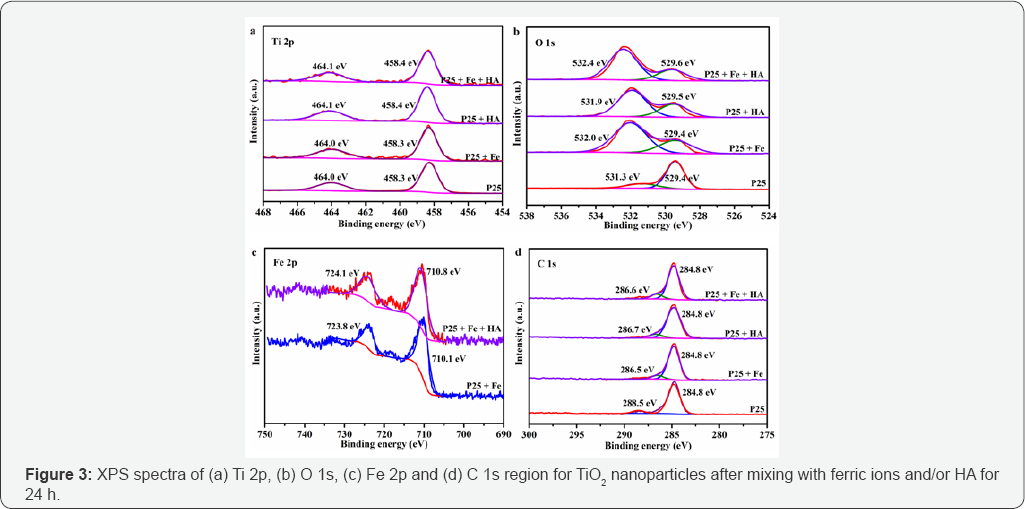
XPS spectra of Ti 2p, O 1s, Fe 2p and C 1s region for TiO2 nanoparticles after mixing with ferric ions and/or HA for 24 h were measured to investigate the surface chemical compositions and bonding states of the TiO2 nanoparticles. (Figure 3a) illustrates the presence of Ti4+ oxidation state, peaks at 458.3458.4 and 464.0-464.1 eV for Ti 2p3/2 and Ti 2p1/2, respectively [20]. The TiO2 nanoparticles mixing with ferric ions and/or HA exhibited insignificant affect to Ti 2p spectra. Figure 3b demonstrates the XPS spectra of O 1s. The peaks at 529.4-529.6 eV are ascribed to the Ti-O bonds in the TiO2 lattice [21], while the peaks at 531.3-532.4 eV are related to the oxygen in the Effect of Ferric Ions Concentration surface hydroxyl groups (H-O bonds) and/or in the carboxyl groups (C-O bonds) [21,22]. It is indicated that the mass ratio of oxygen in Ti-O bonds was higher than that in the surface hydroxyl groups (H-O bonds) and/or in the carboxyl groups (C-O bonds). After mixing with ferric ions and/or HA, the oxygen in the surface hydroxyl groups (H-O bonds) and/or in the carboxyl groups (C-O bonds) increased. Figure 3c reveals the presence of Fe 2p in TiO2 nanoparticles surface after mixing with ferric ions. The peaks at 710.1-710.8 eV and 723.8-724.1 eV correspond to Fe 2p3/2 and Fe 2p1/2, respectively [23-25] (Figure 3).
Effect of Ferric Ions Concentration

With the increase of ferric concentration in TiO2 suspension, the aggregate size increased along with increase of zeta potential positively as shown in (Figure 4a). The possible reason for TiO2 nanoparticles to continue to aggregate even with the increase of ferric concentration along with the increase of positive charges could be due to the formation of Fe(III)-hydroxy colloids which retarded the effect of high repulsion forces among nanoparticles, and the bridging effect of Fe(III)-hydroxy colloids led to the formation of large aggregates. Hence, the TiO2 nanoparticles would achieve instability and settle down from water in a real aquatic environment with pH at around 6 and certain ferric ion to form Fe(III)-hydroxy colloids (Figure 4).
Compared to the effect of ferric ion on aggregate size and zeta potential of TiO2 nanoparticles in suspension without the presence of HA (Figure 4a), the aggregate size was evidently reduced with the addition of HA along with the TiO2 nanoparticles became negatively charged when ferric concentration was less than 1.0 mM (Figure 4b). When ferric concentration was higher than 1.0 mM, both the aggregate size and zeta potential of TiO2 nanoparticles in suspension increased. Ferric ions could exhibit aggregation ability due to the formation of Fe(III)-hydroxy colloids which could eliminate the effect of repulsion forces among nanoparticles, while HA as an aggregation energy barrier could prevent aggregation. It is indicated that, with 5.0 mgC/L of HA in suspension, HA played dominant role for aggregation prevention at ferric concentration of less than 1.0 mM, while Fe(III)-hydroxy colloids played dominant role for aggregation promotion at ferric concentration of more than 1.0 mM. The TiO2 nanoparticles would be easily settled down with a high ferric concentration and a low HA concentration.
Effect of HA
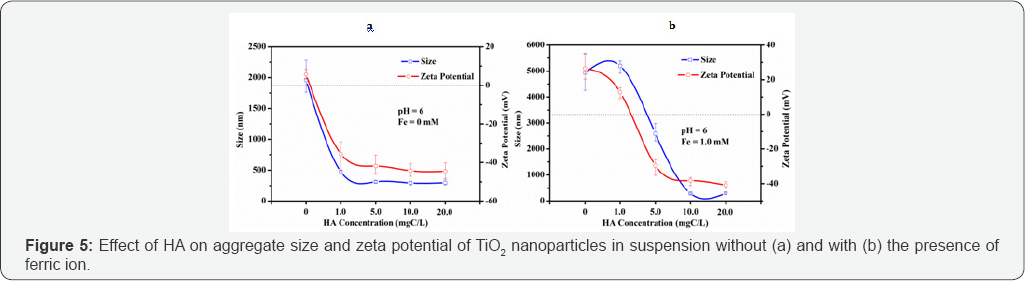
Figure 5a showed that the TiO2 nanoparticles were positively charged in absence of HA. With the increase of HA concentration in suspension, TiO2 nanoparticles formed a more compact cluster coated with the negatively-charged HA, followed by the modification of the characteristics of TiO2 nanoparticles. A sharp decrease of aggregate size was found when HA concentration increased from 0 to 1.0 mgC/L while zeta potential was found to decrease from 6 mV to -36 mV. Further increase in HA concentration resulted in insignificant change of zeta potential with the surface charges of the TiO2 nanoparticles remained in the negative range (Figure 5).
It was assumed that, under electrostatic interaction of TiO2 nanoparticles, the suspension was considered to be unstable in absence of HA which allowed the HA-coated TiO2 nanoparticles to settle down easily. As the HA concentration increased from 1.0 mgC/L onwards, negative zeta potentials of TiO2 nanoparticles continued to decline to more than -40 mV until it approached a steady state, and aggregation was prevented due to the stronger repulsion forces which inhibit aggregation, and thus TiO2 nanoparticles would have an issue in settling down and might remain in water.
Compared to the effect of HA on aggregate size and zeta potential of TiO2 naunoparticles in suspension without the presence of ferric ion (Figure 5a), TiO2 nanoparticles were positively charged due to the addition of 1.0 mM of ferric ion with 1.0 mgC/L of HA in suspension, while they were negatively charged with HA concentration increase from 1.0 mgC/L onwards (Figure 5b). With the addition of ferric ion, the aggregate size was substantially increased with HA concentration till 1.0 mgC/L. With further increase of HA concentration, the aggregation was inhibited due to the increase of electrostatic repulsion forces. Before HA was added into the suspension, ferric ions dominated in the aggregation process under positive zeta potential values. This was attributed to the bridging effect of Fe(III)-hydroxy colloids which overcame the effect of repulsion forces among nanoparticles. When 1.0 mgC/L of HA was added into the suspension of ferric ions-coated TiO2 nanoparticles, ferric ions continued to dominate as larger aggregate size was obtained. As HA concentration increased from 1.0 mgC/L onwards, the effect of Fe(III)-hydroxy colloids faded and HA became the dominant element as aggregation energy barrier to prevent aggregation due to the strong repulsion forces among nanoparticles.
Conclusion
With the widespread application of nanotechnology, TiO2 nanoparticles can find their ways entering into surface water, in which ferric ion and HA are omnipresent commonly. This draws concerns of impact and risk on human and environment. Ferric ion coated on TiO2 nanoparticles has the potential to promote the aggregation due to the formation of Fe(III)-hydroxy colloids, which could be easily removed from aqueous media. However, HA as an aggregation energy barrier could inhibit the aggregation of TiO2 nanoparticles, and thus TiO2 nanoparticles are stable in aqueous media followed by risk on human and environment. This study gains a wider insight on the influence of pH, ferric ion and HA on TiO2 nanoparticles aggregation. It is useful to predict the fate and transport of TiO2 nanoparticles, along with their risks in aqueous media.
Acknowledgement
The author is grateful for the research support from the Nanyang Environment & Water Research Institute (NEWRI).
References
- CO Robichaud, AE Uyar, MR Darby, LG Zucker, MR Wiesner (2009) Estimates of upper bounds and trends in nano-TiO2 production as a basis for exposure assessment. Environmental Science & Technology 43: 4227-4233.
- B Nowack, TD Bucheli (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environmental Pollution 150(1): 5-22.
- J Liu, S Legros, G Ma, JG Veinot, F von der Kammer, et al. (2012) Influence of surface functionalization and particle size on the aggregation kinetics of engineered nanoparticles. Chemosphere 87: 918-924.
- BJ Thio, D Zhou, AA Keller (2011) Influence of natural organic matter on the aggregation and desposition of titanium dioxide nanopartcles. Journal of Hazardous Materials 189 : 556-563.
- IG Godinez CJ Darnault (2011) Aggregation and transport of nano-TiO2 in saturated porous media: Effects of pH, surfactants and flow velocity. Water Research 45(2): 839-851.
- AA Keller, H Wang, D Zhou, HS Lenihan, G Cherr, et al. (2010) Stability and aggregation of metal oxide nanoparticles in natural aqueous matrices. Environmental Science & Technology 44(6): 1962-1967.
- JD Hu, Y Zevi, XM Kou, J Xiao, XJ Wang, et al. (2010) Effect of dissolved organic matter on the stability of magnetite nanoparticles under different pH and ionic strength conditions. Science of the Total Environment 408(16): 3477-3489.
- RF Domingos, N Tufenkji, KJ Wilkinson (2009) Aggregation of titanium dioxide nanoparticles: Role of a fulvic acid. Environmental Science Technology 43(5): 1282-1286.
- E Illes, E Tombacz (2006) The effect of Humic acid adsorption on pH-dependent surface charging and aggregation of magnetite nanoparticles. Journal of Colloid and Interface Science 295(1): 115123.
- W Liu, W Sun, AGL Borthwick, J Ni (2013) Comparison on aggregation and sedimentation of titanium dioxide, titanate nanotubes and titanate nanotubes-TiO2: Influence of pH, ionic strength and natural organic matter. Colloids and Surfaces A Physicochemical and Engineering Aspects 434: 319-328.
- MB Romanello, MM Fidalgo de Cortalezzi (2013) An experimental study on the aggregation of TiO2 nanoparticles under environmentally relevant conditions. Water Research 47(12): 3887-3898.
- J Fang, MJ Xu, DJ Wang, B Wen, JY Han (2013) Modeling the transport of TiO2 nanoparticle aggregates in saturated and unsaturated granular media: Effects of ionic strength and pH. Water Research 47(3): 13991408.
- D Dickson, G Liu, C Li, G Tachiev, Y Cai (2012) Dispersion and stability of bare hematite nanoparticles: Effect of dispersion tools, nanoparticle concentration, humic acid and ionic strength. Science of the Total Environment 419(1): 170-177.
- Yh Shih, CM Zhuang, YH Peng, CH Lin, YM Tseng (2012) The effect of inorganic ions on the aggregation kinetics of lab-made TiO2 nanoparticles in water. Science of the Total Environment 435-436(1): 446-452.
- J Liu, S Legros, F Von der Kammer, T Hofmann (2013) Natural organic matter concentration and hydrochemistry influence aggregation kinetics of functionalized engineered nanoparticles. Environmental Science & Technology 47: 4113-4120.
- KM Buettner, CI Rinciog, SE Mylon (2010) Aggregation kinetics of cerium oxide nanoparticles in monovalent and divalent electrolytes Colloids and Surfaces A: Physicochemical and Engineering Aspects 366: 74-79.
- IL Hsiao, YJ Huang (2011) Effects of various physicochemical characteristics on the toxicities of ZnO and TiO2 nanoparticles toward human lung epithelial cells. Science of the Total Environment 409: 1219-1228.
- S Li, W Sun (2011) A comparative study on aggregation/sedimentation of TiO2 nanoparticles in mono- and binary systems of fulvic acids and Fe(III). Journal of Hazardous Materials 197: 70-79.
- T Weber, T Allard, MF Benedetti (2006) Iron speciation in interaction with organic matter: Modelling and experimental approach. Journal of Geochemical Exploration 88: 166-171.
- J Yang, H Bai, Q Jiang, J Lian (2008) Visible-light photocatalysis in nitrogen-carbon-doped TiO2 films obtained by heating TiO2 gel-film in an ionized N2 gas. Thin Solid Films 516(8): 1736-1742.
- P Gorska, A Zaleska, E Kowalska, T Klimczuk, J W Sobczak, et al, (2008) TiO2 photoactivity in vis and UV light: The influence of calcination temperature and surface properties. Applied Catalysis B Environmental 84: 440-447.
- X Chen, YB Lou, ACS Samia, C Burda, J Gole (2005) Formation of oxynitride as the photocatalytic enhancing site in nitrogen-doped titania nanocatalysts: Comparison to a commercial nanopowder. Advanced Functional Materials 15(1): 41-49.
- W Qian, H Wang, J Chen, Y Kong (2015) Spherical V-Fe-MCM-48: The synthesis, characterization and hydrothermal stability. Materials 8(4): 1752-1765.
- T Yamashita, P Hayes (2008) Analysis of XPS spectra of Fe2+ and Fe3* ions in oxide materials. Applied Surface Science 254(8): 2441-2449.
- IT Kim, GA Nunnery, K Jacob, J Schwartz, X Liu, et al. (2010) Synthesis, characterization, and alignment of magnetic carbon nanotubes tethered with maghemite nanoparticles. The Journal of Physical Chemistry C 114: 6944-6951.






























